Submitted:
26 September 2024
Posted:
26 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
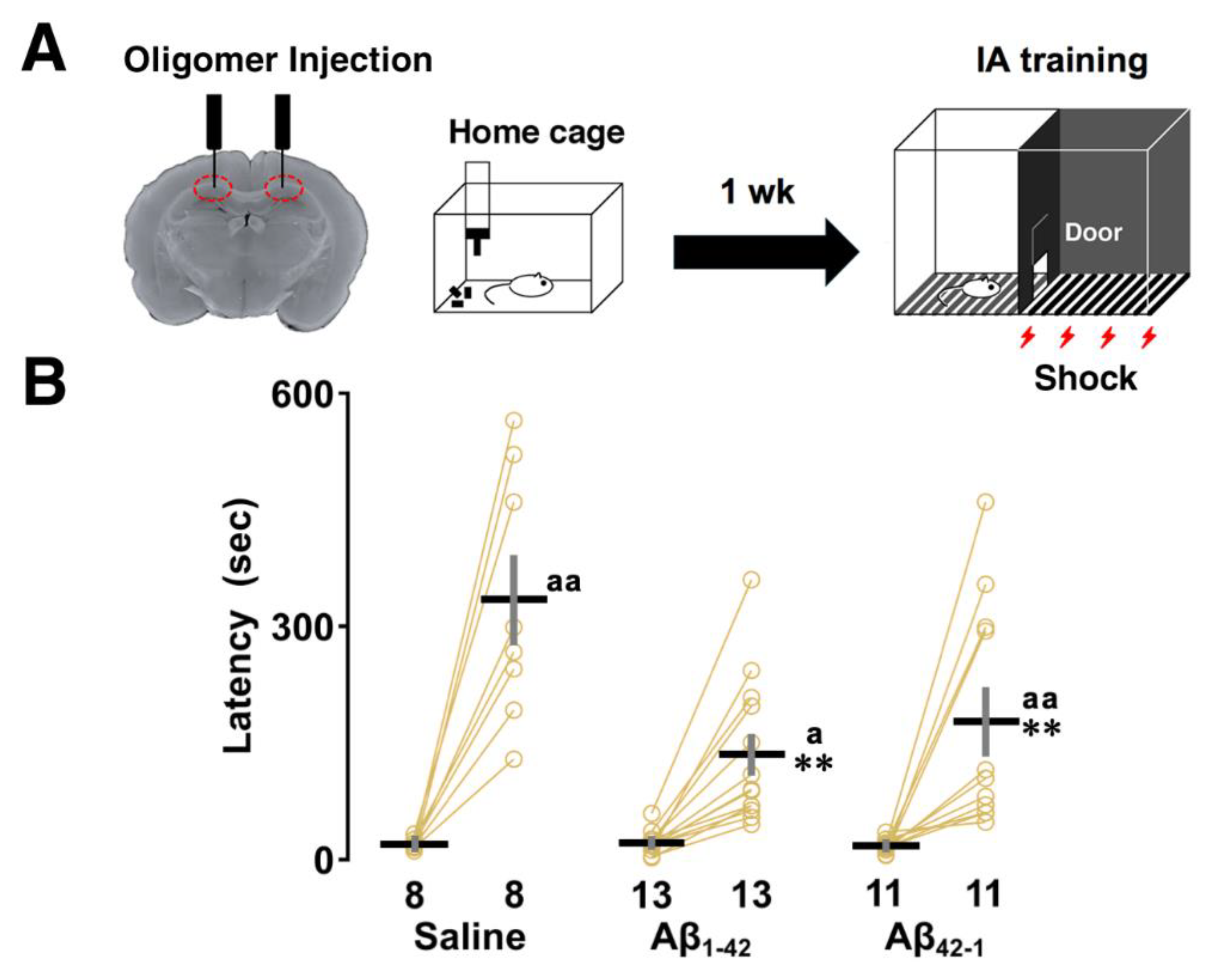
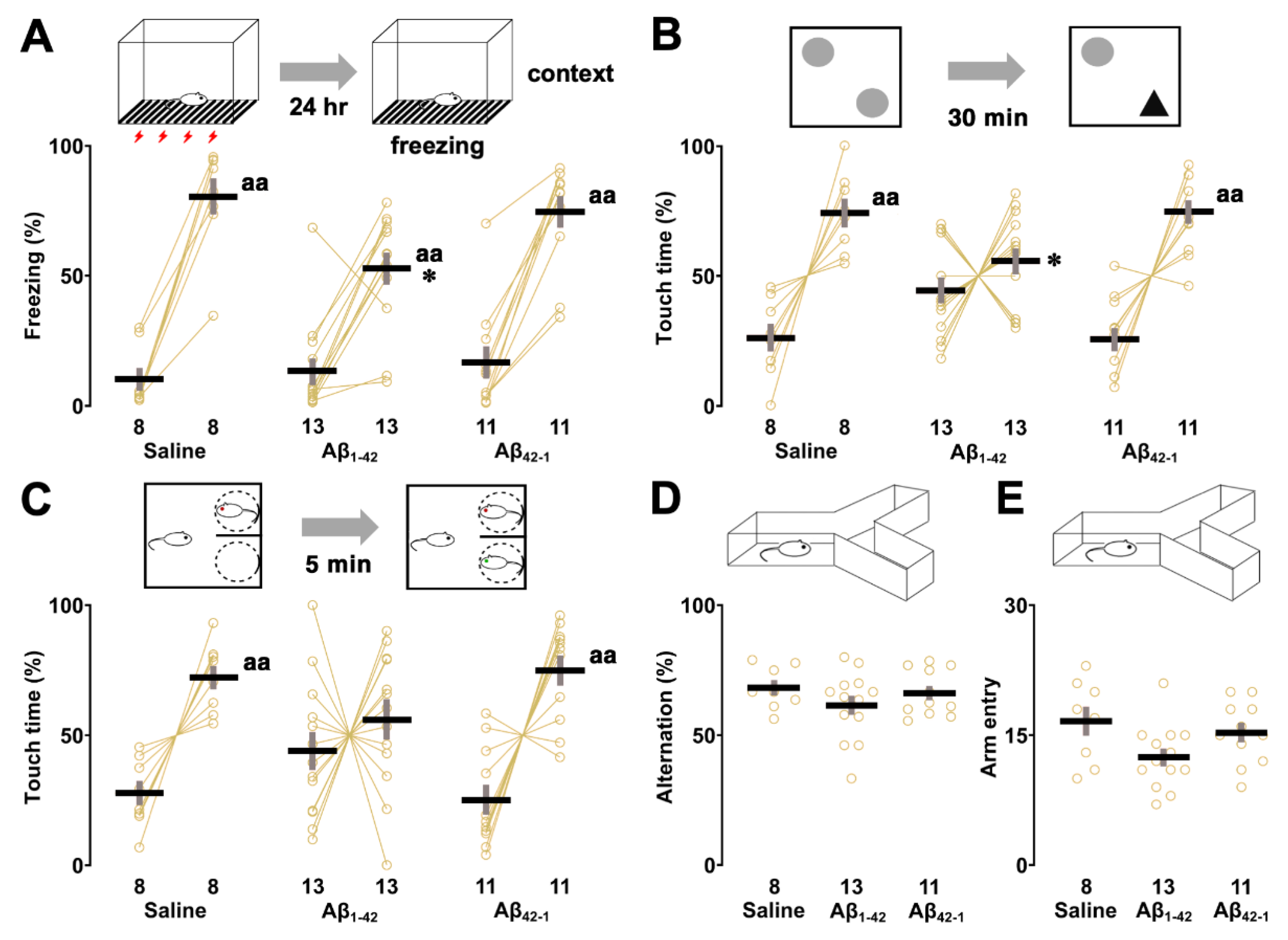
2.1. Effects of Aβ1-42 Oligomers on IA Learning
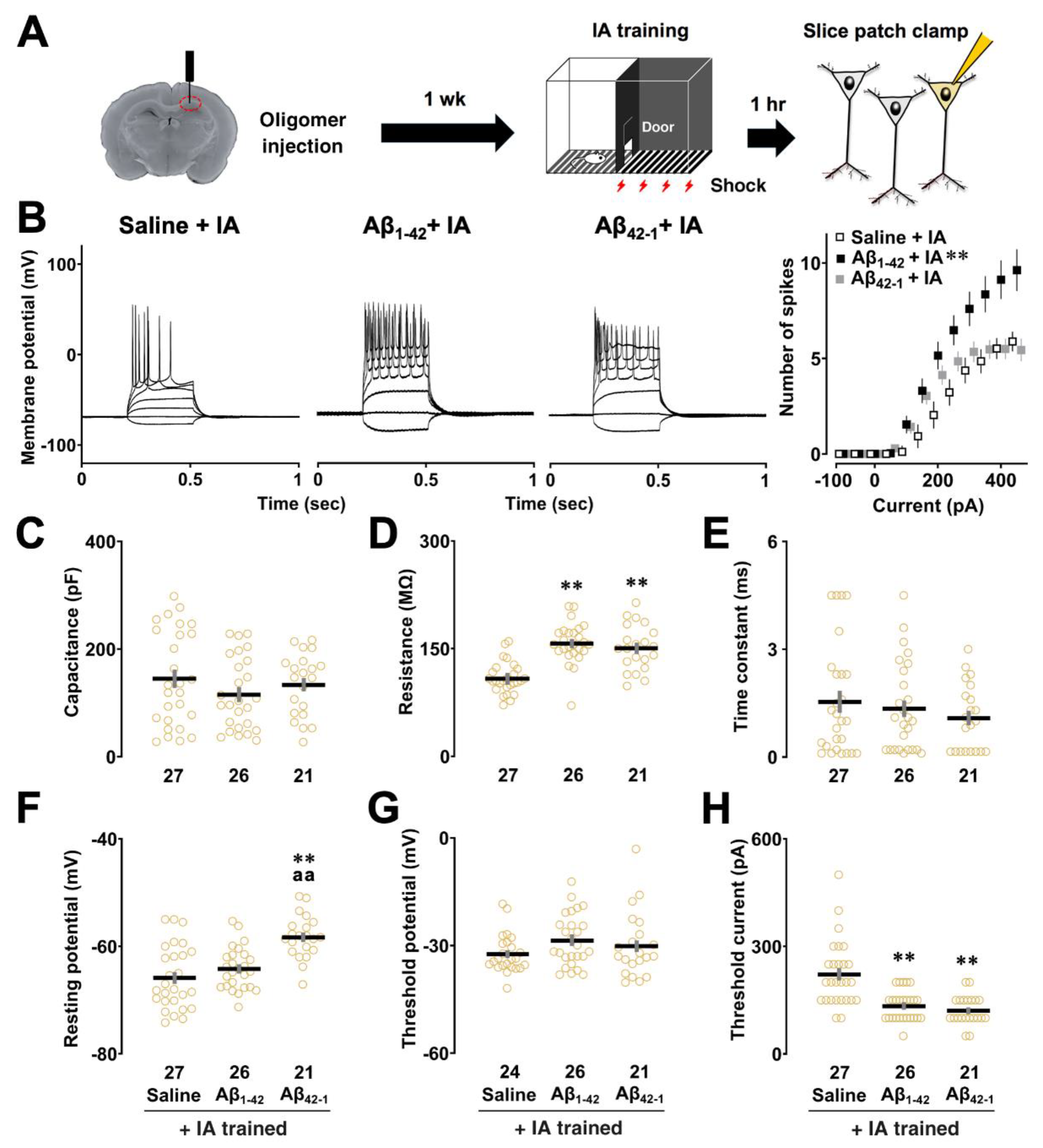
2.2. Effects of Aβ1-42 Oligomers on the Intrinsic Properties after IA Learning
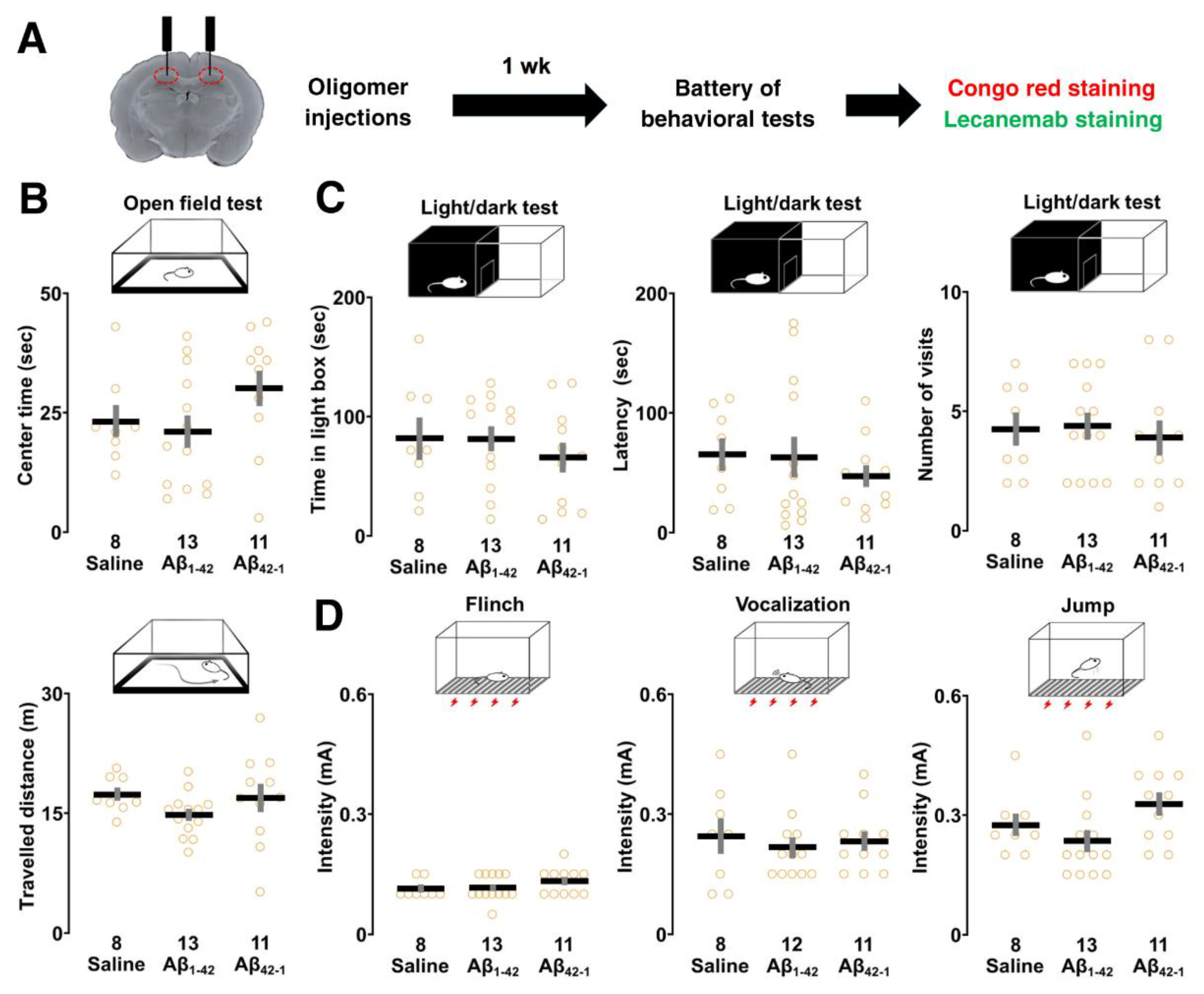
2.3. Effects of Aβ1-42 Oligomers on Sensory/Motor Functions, Pain Sensitivity and Emotional State
2.4. Effects of Aβ1-42 Oligomers on Other Hippocampus-Dependent Tasks
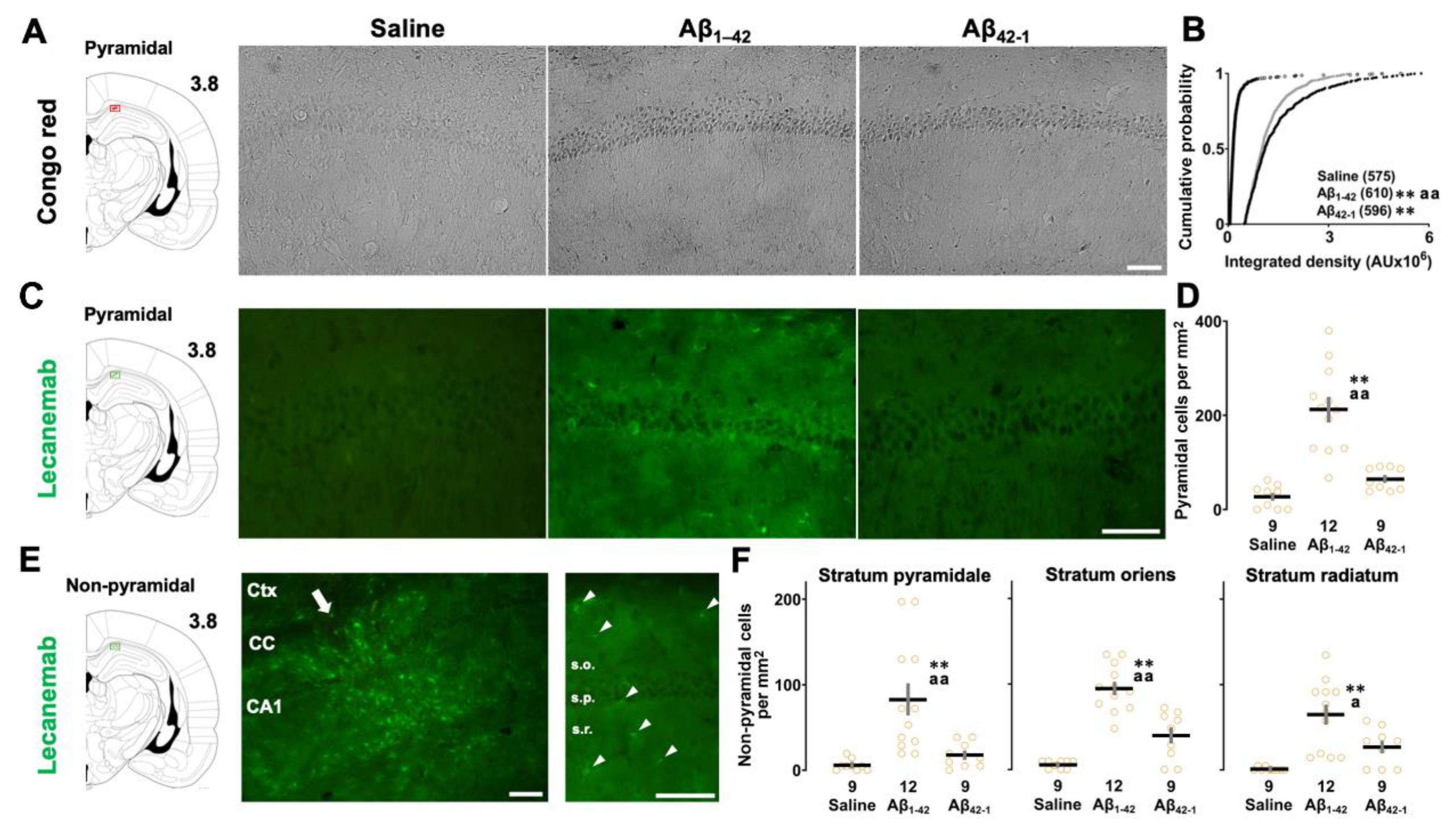
2.5. Aβ Deposition in the CA1 Region
3. Discussion
3.1. Aβ1-42 Oligomers Impaired CA1-Dependent IA Learning
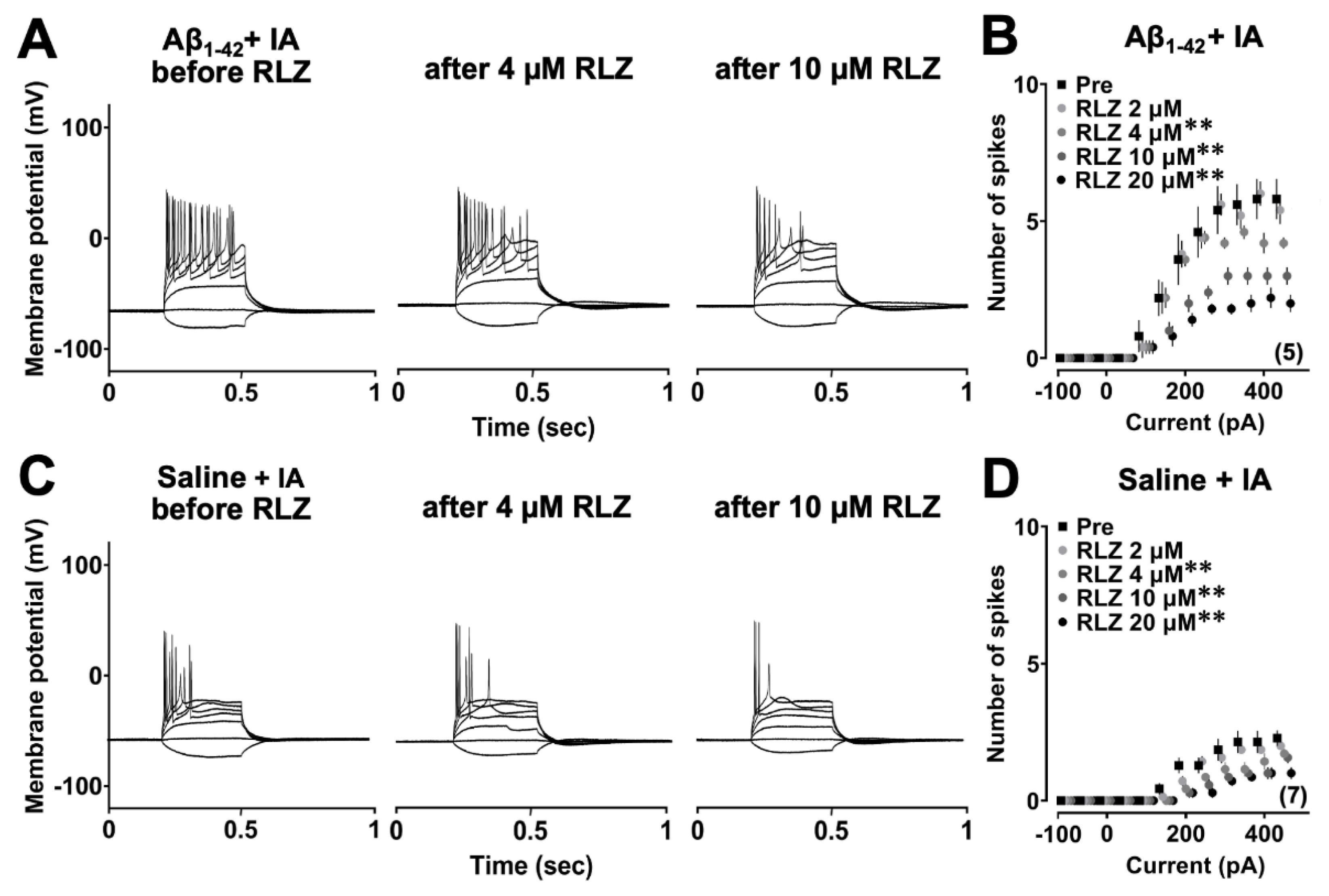
3.2. Aβ1-42 Oligomers Induced Neuronal Hyperexcitability after IA Learning
3.3. Aβ1-42 Oligomers Selectively Impaired Other Hippocampus-Dependent Tasks
3.4. Amyloid Deposition in the Target Area
4. Materials and Methods
4.1. Animals
4.2. Preparation of Aβ1-42 Oligomers
4.3. Stereotaxic Surgery and Microinjection of Oligomers
4.4. Behavioral Test Battery
4.4.1. Open Field Test
4.4.2. Object Recognition Task
4.4.3. Y-Maze Task
4.4.4. Light-Dark Box Test
4.4.5. Social Recognition Task
4.4.6. Inhibitory Avoidance (IA) Task
4.4.7. Fear Conditioning Test
4.4.8. Flinch-Jump Test
4.5. Electrophysiology
4.6. Current Clamp Recordings
4.7. Congo Red Staining
4.8. Lecanemab for Immunostaining
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hardy, J., & Selkoe, D. J. (2002). The Amyloid Hypothesis of Alzheimer’s Disease: Progress and Problems on the Road to Therapeutics. Science, 297(5580), 353–356. [CrossRef]
- Vassar, R., Bennett, B. D., Babu-Khan, S., Kahn, S., Mendiaz, E. A., Denis, P., Teplow, D. B., Ross, S., Amarante, P., Loeloff, R., Luo, Y., Fisher, S., Fuller, J., Edenson, S., Lile, J., Jarosinski, M. A., Biere, A. L., Curran, E., Burgess, T., Citron, M. (1999). β-Secretase Cleavage of Alzheimer’s Amyloid Precursor Protein by the Transmembrane Aspartic Protease BACE. Science, 286(5440), 735–741. [CrossRef]
- Xu, X. (2009). γ-Secretase Catalyzes Sequential Cleavages of the AβPP Transmembrane Domain. Journal of Alzheimer’s Disease, 16(2), 211–224. [CrossRef]
- Gravina, S. A., Ho, L., Eckman, C. B., Long, K. E., Otvos, L., Younkin, L. H., Suzuki, N., & Younkin, S. G. (1995). Amyloid β Protein (Aβ) in Alzheimeri’s Disease Brain. Journal of Biological Chemistry, 270(13), 7013–7016. [CrossRef]
- Bitan, G., Kirkitadze, M. D., Lomakin, A., Vollers, S. S., Benedek, G. B., & Teplow, D. B. (2003). Amyloid β-protein (Aβ) assembly: Aβ40 and Aβ42 oligomerize through distinct pathways. Proceedings of the National Academy of Sciences, 100(1), 330–335. [CrossRef]
- Yang, M., & Teplow, D. B. (2008). Amyloid β-Protein Monomer Folding: Free-Energy Surfaces Reveal Alloform-Specific Differences. Journal of Molecular Biology, 384(2), 450–464. [CrossRef]
- Ahmed, M., Davis, J., Aucoin, D., Sato, T., Ahuja, S., Aimoto, S., Elliott, J. I., Van Nostrand, W. E., & Smith, S. O. (2010). Structural conversion of neurotoxic amyloid-β1–42 oligomers to fibrils. Nature Structural & Molecular Biology, 17(5), 561–567. [CrossRef]
- Van Dam, D., D’Hooge, R., Staufenbiel, M., Van Ginneken, C., Van Meir, F., & De Deyn, P. P. (2003). Age-dependent cognitive decline in the APP23 model precedes amyloid deposition. European Journal of Neuroscience, 17(2), 388–396. [CrossRef]
- Mucke, L., Masliah, E., Yu, G.-Q., Mallory, M., Rockenstein, E. M., Tatsuno, G., Hu, K., Kholodenko, D., Johnson-Wood, K., & McConlogue, L. (2000). High-Level Neuronal Expression of Aβ 1–42 in Wild-Type Human Amyloid Protein Precursor Transgenic Mice: Synaptotoxicity without Plaque Formation. The Journal of Neuroscience, 20(11), 4050–4058. [CrossRef]
- Gandy, S., Simon, A. J., Steele, J. W., Lublin, A. L., Lah, J. J., Walker, L. C., Levey, A. I., Krafft, G. A., Levy, E., Checler, F., Glabe, C., Bilker, W. B., Abel, T., Schmeidler, J., & Ehrlich, M. E. (2010). Days to criterion as an indicator of toxicity associated with human Alzheimer amyloid-β oligomers. Annals of Neurology, 68(2), 220–230. [CrossRef]
- Kuo, Y.-M., Emmerling, M. R., Vigo-Pelfrey, C., Kasunic, T. C., Kirkpatrick, J. B., Murdoch, G. H., Ball, M. J., & Roher, A. E. (1996). Water-soluble Aβ (N-40, N-42) Oligomers in Normal and Alzheimer Disease Brains. Journal of Biological Chemistry, 271(8), 4077–4081. [CrossRef]
- Roher, A. E., Chaney, M. O., Kuo, Y.-M., Webster, S. D., Stine, W. B., Haverkamp, L. J., Woods, A. S., Cotter, R. J., Tuohy, J. M., Krafft, G. A., Bonnell, B. S., & Emmerling, M. R. (1996). Morphology and Toxicity of Aβ-(1-42) Dimer Derived from Neuritic and Vascular Amyloid Deposits of Alzheimer’s Disease. Journal of Biological Chemistry, 271(34), 20631–20635. [CrossRef]
- Gong, Y., Chang, L., Viola, K. L., Lacor, P. N., Lambert, M. P., Finch, C. E., Krafft, G. A., & Klein, W. L. (2003). Alzheimer’s disease-affected brain: Presence of oligomeric Aβ ligands (ADDLs) suggests a molecular basis for reversible memory loss. Proceedings of the National Academy of Sciences, 100(18), 10417–10422. [CrossRef]
- Forny-Germano, L., Lyra E Silva, N. M., Batista, A. F., Brito-Moreira, J., Gralle, M., Boehnke, S. E., Coe, B. C., Lablans, A., Marques, S. A., Martinez, A. M. B., Klein, W. L., Houzel, J.-C., Ferreira, S. T., Munoz, D. P., & De Felice, F. G. (2014). Alzheimer’s Disease-Like Pathology Induced by Amyloid-β Oligomers in Nonhuman Primates. The Journal of Neuroscience, 34(41), 13629–13643. [CrossRef]
- Haass, C., & Selkoe, D. J. (2007). Soluble protein oligomers in neurodegeneration: Lessons from the Alzheimer’s amyloid β-peptide. Nature Reviews Molecular Cell Biology, 8(2), 101–112. [CrossRef]
- Rolls ET (2018). The storage and recall of memories in the hippocampo-cortical system. Cell Tissue Res, 373(3), 577-604. [CrossRef]
- Kesner RP, Lee I, Gilbert P (2004). A behavioral assessment of hippocampal function based on a subregional analysis. Rev Neurosci, 15(5), 333-51.
- Lee I & Kesner RP (2002). Differential contribution of NMDA receptors in hippocampal subregions to spatial working memory. Nat Neurosci, 5(2), 162-8. [CrossRef]
- Remondes, M., & Schuman, E. M. (2004). Role for a cortical input to hippocampal area CA1 in the consolidation of a long-term memory. Nature, 431(7009), 699–703. [CrossRef]
- Lee I & Kesner RP (2004). Differential contributions of dorsal hippocampal subregions to memory acquisition and retrieval in contextual fear-conditioning. Hippocampus, 14(3), 301-10. [CrossRef]
- Mitsushima, D., Ishihara, K., Sano, A., Kessels, H. W., & Takahashi, T. (2011). Contextual learning requires synaptic AMPA receptor delivery in the hippocampus. Proceedings of the National Academy of Sciences, 108(30), 12503–12508. [CrossRef]
- Ásgeirsdóttir HN, Cohen SJ & Stackman RW Jr (2020). Object and place information processing by CA1 hippocampal neurons of C57BL/6J mice. J Neurophysiol, 123(3),1247-1264. [CrossRef]
- Heggland, I., Storkaas, I. S., Soligard, H. T., Kobro-Flatmoen, A., & Witter, M. P. (2015). Stereological estimation of neuron number and plaque load in the hippocampal region of a transgenic rat model of A lzheimer’s disease. European Journal of Neuroscience, 41(9), 1245–1262. [CrossRef]
- Wood ER, Dudchenko PA, Robitsek RJ & Eichenbaum H (2000). Hippocampal neurons encode information about different types of memory episodes occurring in the same location. Neuron, 27(3), 623-33. [CrossRef]
- Horsley, J. R., Jovcevski, B., Wegener, K. L., Yu, J., Pukala, T. L., & Abell, A. D. (2020). Rationally designed peptide-based inhibitor of Aβ42 fibril formation and toxicity: A potential therapeutic strategy for Alzheimer’s disease. Biochemical Journal, 477(11), 2039–2054. [CrossRef]
- Wilcock, D. M., Gordon, M. N., & Morgan, D. (2006). Quantification of cerebral amyloid angiopathy and parenchymal amyloid plaques with Congo red histochemical stain. Nature Protocols, 1(3), 1591–1595. [CrossRef]
- Arndt, J. W., Qian, F., Smith, B. A., Quan, C., Kilambi, K. P., Bush, M. W., Walz, T., Pepinsky, R. B., Bussière, T., Hamann, S., Cameron, T. O., & Weinreb, P. H. (2018). Structural and kinetic basis for the selectivity of aducanumab for aggregated forms of amyloid-β. Scientific Reports, 8(1), 6412. [CrossRef]
- Söderberg, L., Johannesson, M., Nygren, P., Laudon, H., Eriksson, F., Osswald, G., Möller, C., & Lannfelt, L. (2023). Lecanemab, Aducanumab, and Gantenerumab-Binding Profiles to Different Forms of Amyloid-Beta Might Explain Efficacy and Side Effects in Clinical Trials for Alzheimer’s Disease. Neurotherapeutics, 20(1), 195–206. [CrossRef]
- Marshall KE, Vadukul DM, Dahal L, Theisen A, Fowler MW, Al-Hilaly Y, Ford L, Kemenes G, Day IJ, Staras K & Serpell LC (2016). A Critical Role for the Self-Assembly of Amyloid-Β1-42 in Neurodegeneration. Sci Rep, 6, 30182. [CrossRef]
- Kravenska, Y., Nieznanska, H., Nieznanski, K., Lukyanetz, E., Szewczyk, A., & Koprowski, P. (2020). The monomers, oligomers, and fibrils of amyloid-β inhibit the activity of mitoBKCa channels by a membrane-mediated mechanism. Biochimica et Biophysica Acta (BBA) - Biomembranes, 1862(9), 183337. [CrossRef]
- Hirakura, Y., Lin, M. C., & Kagan, B. L. (1999). Alzheimer amyloid abeta1-42 channels: Effects of solvent, pH, and Congo Red. Journal of Neuroscience Research, 57(4), 458–466.
- Koh, M. T., Haberman, R. P., Foti, S., McCown, T. J., & Gallagher, M. (2010). Treatment Strategies Targeting Excess Hippocampal Activity Benefit Aged Rats with Cognitive Impairment. Neuropsychopharmacology, 35(4), 1016–1025. [CrossRef]
- Yassa, M. A., Stark, S. M., Bakker, A., Albert, M. S., Gallagher, M., & Stark, C. E. L. (2010). High-resolution structural and functional MRI of hippocampal CA3 and dentate gyrus in patients with amnestic Mild Cognitive Impairment. NeuroImage, 51(3), 1242–1252. [CrossRef]
- Ren, S.-Q., Yao, W., Yan, J.-Z., Jin, C., Yin, J.-J., Yuan, J., Yu, S., & Cheng, Z. (2018). Amyloid β causes excitation/inhibition imbalance through dopamine receptor 1-dependent disruption of fast-spiking GABAergic input in anterior cingulate cortex. Scientific Reports, 8(1), 302. [CrossRef]
- George, A. A., Vieira, J. M., Xavier-Jackson, C., Gee, M. T., Cirrito, J. R., Bimonte-Nelson, H. A., Picciotto, M. R., Lukas, R. J., & Whiteaker, P. (2021). Implications of Oligomeric Amyloid-Beta (oAβ42) Signaling through α7β2-Nicotinic Acetylcholine Receptors (nAChRs) on Basal Forebrain Cholinergic Neuronal Intrinsic Excitability and Cognitive Decline. The Journal of Neuroscience, 41(3), 555–575. [CrossRef]
- Taverna S, Tkatch T & Metz AE, Martina M (2005). Differential expression of TASK channels between horizontal interneurons and pyramidal cells of rat hippocampus. J Neurosci, 25(40), 9162-70. [CrossRef]
- Hu H, Vervaeke K & Storm JF. Two forms of electrical resonance at theta frequencies, generated by M-current, h-current and persistent Na+ current in rat hippocampal pyramidal cells (2002). J Physiol, 545(3), 783-805. [CrossRef]
- Matsumura, R., Yamamoto, H., Hayakawa, T., Katsurabayashi, S., Niwano, M., & Hirano-Iwata, A. (2018). Dependence and Homeostasis of Membrane Impedance on Cell Morphology in Cultured Hippocampal Neurons. Scientific Reports, 8(1), 9905. [CrossRef]
- Ungureanu, A.-A., Benilova, I., Krylychkina, O., Braeken, D., De Strooper, B., Van Haesendonck, C., Dotti, C. G., & Bartic, C. (2016). Amyloid beta oligomers induce neuronal elasticity changes in age-dependent manner: A force spectroscopy study on living hippocampal neurons. Scientific Reports, 6(1), 25841. [CrossRef]
- Demuro, A., Mina, E., Kayed, R., Milton, S. C., Parker, I., & Glabe, C. G. (2005). Calcium Dysregulation and Membrane Disruption as a Ubiquitous Neurotoxic Mechanism of Soluble Amyloid Oligomers. Journal of Biological Chemistry, 280(17), 17294–17300. [CrossRef]
- Fernandez-Perez, E. J., Muñoz, B., Bascuñan, D. A., Peters, C., Riffo-Lepe, N. O., Espinoza, M. P., Morgan, P. J., Filippi, C., Bourboulou, R., Sengupta, U., Kayed, R., Epsztein, J., & Aguayo, L. G. (2021). Synaptic dysregulation and hyperexcitability induced by intracellular amyloid beta oligomers. Aging Cell, 20(9), e13455. [CrossRef]
- Nimmrich, V., Grimm, C., Draguhn, A., Barghorn, S., Lehmann, A., Schoemaker, H., Hillen, H., Gross, G., Ebert, U., & Bruehl, C. (2008). Amyloid β Oligomers (Aβ1–42 Globulomer) Suppress Spontaneous Synaptic Activity by Inhibition of P/Q-Type Calcium Currents. The Journal of Neuroscience, 28(4), 788–797. [CrossRef]
- Tamagnini, F., Scullion, S., Brown, J. T., & Randall, A. D. (2015). Intrinsic excitability changes induced by acute treatment of hippocampal CA1 pyramidal neurons with exogenous amyloid β peptide. Hippocampus, 25(7), 786–797. [CrossRef]
- Ferrara NC, Jarome TJ, Cullen PK, Orsi SA, Kwapis JL, Trask S, Pullins SE & Helmstetter FJ (2019). GluR2 endocytosis-dependent protein degradation in the amygdala mediates memory updating. Sci Rep, 9(1), 5180. [CrossRef]
- Piromalli Girado D, Miranda M, Giachero M, Weisstaub N & Bekinschtein P (2023). Endocytosis is required for consolidation of pattern-separated memories in the perirhinal cortex. Front Syst Neurosci, 17, 1043664. [CrossRef]
- Yamamoto K, Ueta Y, Wang L, Yamamoto R, Inoue N, Inokuchi K, Aiba A, Yonekura H & Kato N (2011). Suppression of a neocortical potassium channel activity by intracellular amyloid-β and its rescue with Homer1a. J Neurosci, 31(31), 11100-9. [CrossRef]
- Ciccone, R., Franco, C., Piccialli, I., Boscia, F., Casamassa, A., De Rosa, V., Cepparulo, P., Cataldi, M., Annunziato, L., & Pannaccione, A. (2019). Amyloid β-Induced Upregulation of Nav1.6 Underlies Neuronal Hyperactivity in Tg2576 Alzheimer’s Disease Mouse Model. Scientific Reports, 9(1), 13592. [CrossRef]
- Jin S, Kedia N, Illes-Toth E, Haralampiev I, Prisner S, Herrmann A, Wanker EE & Bieschke J (2016). Amyloid-β(1-42) Aggregation Initiates Its Cellular Uptake and Cytotoxicity. J Biol Chem, 291(37), 19590-606. [CrossRef]
- Chatelier, A., Zhao, J., Bois, P., & Chahine, M. (2010). Biophysical characterisation of the persistent sodium current of the Nav1.6 neuronal sodium channel: A single-channel analysis. Pflügers Archiv - European Journal of Physiology, 460(1), 77–86. [CrossRef]
- Vervaeke, K., Hu, H., Graham, L. J., & Storm, J. F. (2006). Contrasting Effects of the Persistent Na+ Current on Neuronal Excitability and Spike Timing. Neuron, 49(2), 257–270. [CrossRef]
- Ren, S., Chen, P., Jiang, H., Mi, Z., Xu, F., Hu, B., Zhang, J., & Zhu, Z. (2014). Persistent sodium currents contribute to Aβ1-42-induced hyperexcitation of hippocampal CA1 pyramidal neurons. Neuroscience Letters, 580, 62–67. [CrossRef]
- Sipos, E., Kurunczi, A., Kasza, Á., Horváth, J., Felszeghy, K., Laroche, S., Toldi, J., Párducz, Á., Penke, B., & Penke, Z. (2007). β-Amyloid pathology in the entorhinal cortex of rats induces memory deficits: Implications for Alzheimer’s disease. Neuroscience, 147(1), 28–36. [CrossRef]
- Chang, K.-W., Zong, H.-F., Ma, K.-G., Zhai, W.-Y., Yang, W.-N., Hu, X.-D., Xu, J.-H., Chen, X.-L., Ji, S.-F., & Qian, Y.-H. (2008). Activation of α7 nicotinic acetylcholine receptor alleviates Aβ1-42-induced neurotoxicity via downregulation of p38 and JNK MAPK signaling pathways. Neurochemistry International, 120, 238–250. [CrossRef]
- Koppensteiner, P., Trinchese, F., Fà, M., Puzzo, D., Gulisano, W., Yan, S., Poussin, A., Liu, S., Orozco, I., Dale, E., Teich, A. F., Palmeri, A., Ninan, I., Boehm, S., & Arancio, O. (2016). Time-dependent reversal of synaptic plasticity induced by physiological concentrations of oligomeric Aβ42: An early index of Alzheimer’s disease. Scientific Reports, 6(1), 32553. [CrossRef]
- Christensen, R., Marcussen, A. B., Wörtwein, G., Knudsen, G. M., & Aznar, S. (2008). Aβ (1–42) injection causes memory impairment, lowered cortical and serum BDNF levels, and decreased hippocampal 5-HT2A levels. Experimental Neurology, 210(1), 164–171. [CrossRef]
- Brouillette, J., Caillierez, R., Zommer, N., Alves-Pires, C., Benilova, I., Blum, D., De Strooper, B., & Buee, L. (2012). Neurotoxicity and Memory Deficits Induced by Soluble Low-Molecular-Weight Amyloid- 1-42 Oligomers Are Revealed In Vivo by Using a Novel Animal Model. Journal of Neuroscience, 32(23), 7852–7861. [CrossRef]
- Yang, Y., Ji, W., Zhu, Z., Wu, Y., Zhang, Z., & Qu, S. (2018). Rhynchophylline suppresses soluble Aβ1-42-induced impairment of spatial cognition function via inhibiting excessive activation of extrasynaptic NR2B-containing NMDA receptors. Neuropharmacology, 135, 100–112. [CrossRef]
- Zheng, M., Liu, J., Ruan, Z., Tian, S., Ma, Y., Zhu, J., & Li, G. (2013). Intrahippocampal injection of Aβ 1-42 inhibits neurogenesis and down-regulates IFN-γ and NF-κB expression in hippocampus of adult mouse brain. Amyloid, 20(1), 13–20. [CrossRef]
- Cerbai, F., Lana, D., Nosi, D., Petkova-Kirova, P., Zecchi, S., Brothers, H. M., Wenk, G. L., & Giovannini, M. G. (2012). The Neuron-Astrocyte-Microglia Triad in Normal Brain Ageing and in a Model of Neuroinflammation in the Rat Hippocampus. PLoS ONE, 7(9), e45250. [CrossRef]
- Lana, D., Ugolini, F., Nosi, D., Wenk, G. L., & Giovannini, M. G. (2017). Alterations in the Interplay between Neurons, Astrocytes and Microglia in the Rat Dentate Gyrus in Experimental Models of Neurodegeneration. Frontiers in Aging Neuroscience, 9, 296. [CrossRef]
- O’Hare, E., Weldon, D. T., Mantyh, P. W., Ghilardi, J. R., Finke, M. P., Kuskowski, M. A., Maggio, J. E., Shephard, R. A., & Cleary, J. (1999). Delayed behavioral effects following intrahippocampal injection of aggregated Aβ (1–42). Brain Research, 815(1), 1–10. [CrossRef]
- Sakimoto, Y., Shintani, A., Yoshiura, D., Goshima, M., Kida, H., & Mitsushima, D. (2022). A critical period for learning and plastic changes at hippocampal CA1 synapses. Scientific Reports, 12(1), 7199. [CrossRef]
- Beninger, R. J., Jhamandas, K., Boegman, R. J., & El-Defrawy, S. R. (1986). Effects of scopolamine and unilateral lesions of the basal forebrain on T-maze spatial discrimination and alternation in rats. Pharmacology Biochemistry and Behavior, 24(5), 1353–1360. [CrossRef]
- Yamada, K., Hiramatsu, M., Noda, Y., Mamiya, T., Murai, M., Kameyama, T., Komori, Y., Nikai, T., Sugihara, H., & Nabeshima, T. (1996). Role of nitric oxide and cyclic GMP in the dizocilpine-induced impairment of spontaneous alternation behavior in mice. Neuroscience, 74(2), 365–374. [CrossRef]
- Arrant, A. E., Schramm-Sapyta, N. L., & Kuhn, C. M. (2013). Use of the light/dark test for anxiety in adult and adolescent male rats. Behavioural Brain Research, 256, 119–127. [CrossRef]
- Lee, Y., Kim, H., & Han, P.-L. (2018). Striatal Inhibition of MeCP2 or TSC1 Produces Sociability Deficits and Repetitive Behaviors. Experimental Neurobiology, 27(6), 539–549. [CrossRef]
- Takase, K., Sakimoto, Y., Kimura, F., & Mitsushima, D. (2014). Developmental trajectory of contextual learning and 24-h acetylcholine release in the hippocampus. Scientific Reports, 4(1), 3738. [CrossRef]
- Kulesskaya, N., & Voikar, V. (2014). Assessment of mouse anxiety-like behavior in the light–dark box and open-field arena: Role of equipment and procedure. Physiology & Behavior, 133, 30–38. [CrossRef]
- Lehner M, Wisłowska-Stanek A, Maciejak P, Szyndler J, Sobolewska A, Krzaścik P & Płaźnik A. (2010). The relationship between pain sensitivity and conditioned fear response in rats. Acta Neurobiologiae Experimentalis, 70(1), 56–66. [CrossRef]
- Kida, H., Sakimoto, Y., & Mitsushima, D. (2017). Slice Patch Clamp Technique for Analyzing Learning-Induced Plasticity. Journal of Visualized Experiments, 129, 55876. [CrossRef]
- Paxinos, G. & Watson, C, The Rat Bain in Stereotaxic Coordinates, 6th ed. Academic Press: San Diego, CA, USA; 2006.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



