Submitted:
25 September 2024
Posted:
26 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
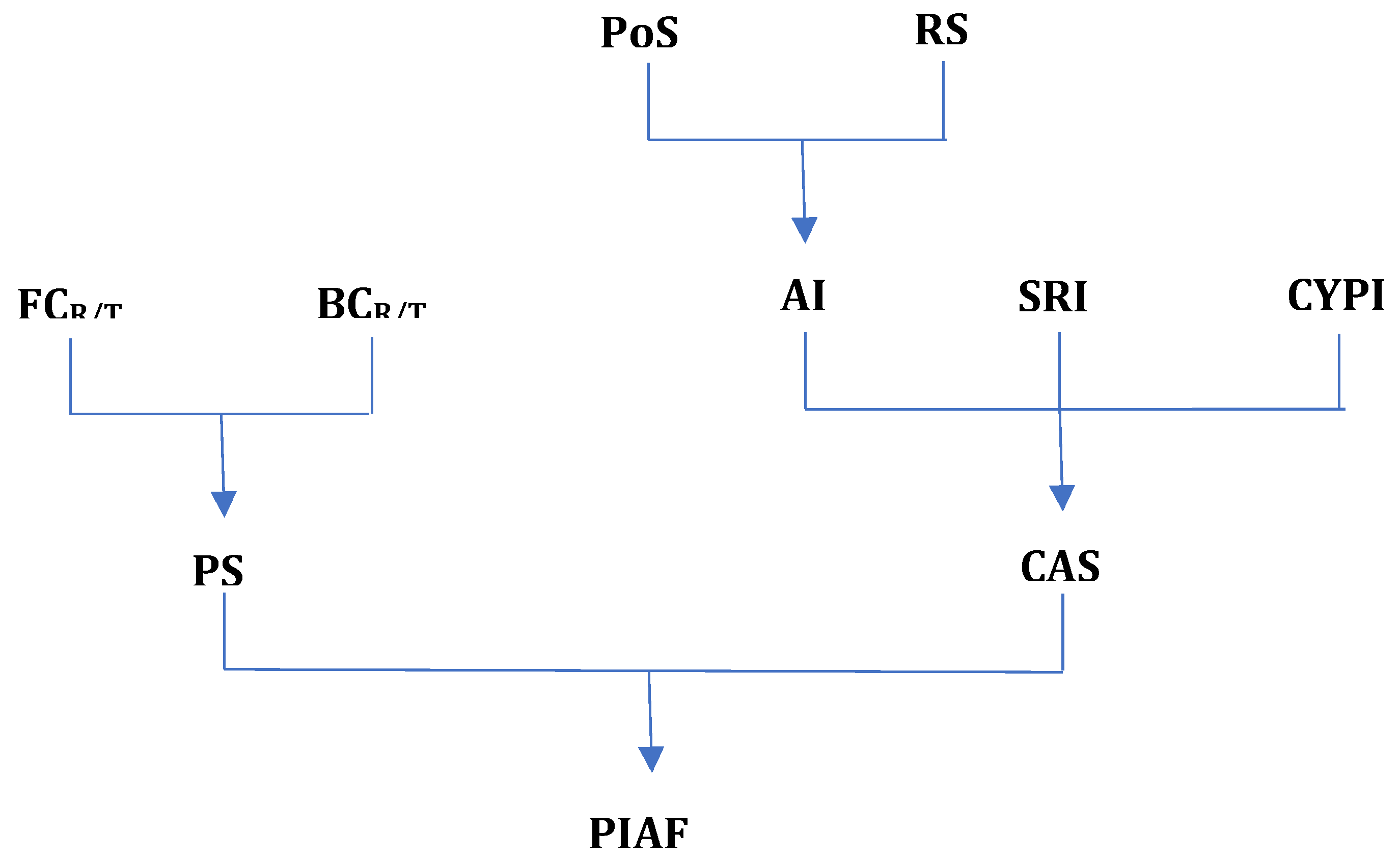
2. Methodology
- A)
- Pharmacodynamics: To assess the effects on functionality from a pharmacodynamic perspective, three parameters have been defined:
- 1.
- Functionality coefficient (FCR/T) represents the score associated with each receptor or reuptake pump (receptor/transporter) regarding functionality according to the formula:
- 2.
- Binding coefficient (BCR/T) is a parameter that expresses the affinity of an antipsychotic molecule for the receptor (R) and transporter (T). This parameter was necessary because antipsychotic molecules have different affinities for these substrates, thus leading to different amplitudes of effects.
- 3.
- Pharmacodynamic Score (PS) is a parameter that quantifies how functionality is influenced through the pharmacodynamic mechanisms of antipsychotics. For each pharmacodynamic mechanism (action on receptor or transporter), the influence on functionality is calculated by the product . For this reason, an antipsychotic that acts through multiple mechanisms pharmacodynamic has PS as the sum of products according to the formula:
- B)
- Comfort of Administration. To assess the effects on functionality from the perspective of administration comfort, the parameter Comfort of Administration Score (CAS) has been defined. This parameter takes into account the following criteria:
- 4.
- Special Requests Index (SRI) which quantifies the special requirements related to the administration of the antipsychotic.
- 5.
- CYP Index (CYPI) which expresses the potential for drug interactions generated at the level of cytochrome P450 (CYP450) enzymes.
3. Discussions
Limitations
4. Conclusions
Supplementary Materials
References
- Patel KR, Cherian J, Gohil K, and Atkinson D. Schizophrenia: Overview and Treatment Options. P&T September 2014 Vol. 39 No. 9; 638-645.
- Correll CU, Solmi M, Croatto G, Schneider LK, Rohani-Montez SC, Fairley L, Smith N, Bitter I, Gorwood P, Taipale H, Tiihonen J. Mortality in people with schizophrenia: a systematic review and meta-analysis of relative risk and aggravating or attenuating factors. World Psychiatry 2022;21:248–271.
- Pillinger T, McCutheon RA, Vano L, Mizuno Y, Arumuham A, Hindley G, Beck K, Natesan S, Efthimiu O, Cipriani A, Howes OD. Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia, predictors of metabolic dysregulation, and association with psychopathology: a systematic review and network meta-analysis. Lancet Psychiatry 2020; 7: 64–77.
- Scorza FA, de Almeida A-CG, Scorza CA, Cysneiros RM and Finsterer J. Sudden death in schizophrenia: pay special attention and develop preventive strategies. Curr Med Res Opin. 2021;37(1):109–121.
- Hjorthøj C, Stürup AE, McGrath JJ, Nordentoft M. Years of potential life lost and life expectancy in schizophrenia: a systematic review and meta-analysis. Lancet Psychiatry 2017; 4: 295–301.
- Boland R, Verduin ML, Ruiz P. 2022. Kaplan & Sadock's Synopsis of Psychiatry 12th edition. Wolters Kluwer.
- Switaj P, Anczewska M, Chrostek A, Sabariego C, Cieza A, Bickenbach J, and Chatterji S. Disability and schizophrenia: a systematic review of experienced psychosocial difficulties. BMC Psychiatry. (2012) 12:193. [CrossRef]
- Correll CU. Using Patient-Centered Assessment in Schizophrenia Care: Defining Recovery and Discussing Concerns and Preferences. Journal of Clinical Psychiatry Volume: 81 Issue: 3 Pages: MS19053BR2C. [CrossRef]
- Ifteni P, Petric P-S and Teodorescu A (2021) Rating Opportunity for Long-Acting Injectable Antipsychotic Initiation Index (ROLIN). Front. Psychiatry 12:767756. [CrossRef]
- Stahl SM. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications - 5th ed. 2021 ISBN 978-1-108-83857-3.
- Ishigooka J, Nakagome K, Ohmori T, Iwata N, Inada K, Iga J-I, Kishi T, Fujita K, Kikuchi Y, Shichijo T, Tabuse H, Koretsune S, Terada H, Terada H, Kishimoto T, Tsutsumi Y, Kanda Y, Ohi K and Sekiyama K. Discontinuation and remission rates and social functioning in patients with schizophrenia receiving second-generation antipsychotics: 52-week evaluation of JUMPs, a randomized, open-label study. Psychiatry and Clinical Neurosciences 76: 22–31, 2022.
- Leucht S, Leucht C, Huhn M, Chaimani A, Mavridis D, Helfer B, Samara M, Rabaioli M, Bächer S, Cipriani A, Geddes JR, Salanti G, Davis JM. Sixty Years of Placebo-Controlled Antipsychotic Drug Trials in Acute Schizophrenia: Systematic Review, Bayesian Meta-Analysis, and Meta-Regression of Efficacy Predictors. [CrossRef]
- Huhn M, Nikolakopoulou A, Schneider-Thoma J, Krause M, Samara M, Peter N, Arndt T, Bäckers L, Rothe P, Cipriani A, Davis J, Salanti G, Leucht S. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet 2019; 394: 939–51. [CrossRef]
- Stahl SM, Djokic G. Comparing the pharmacology and pharmacokinetics of antipsychotics: Choosing an antipsychotic and dosing a long-acting injectable. European Neuropsychopharmacology 73 (2023) 108–118.
- de Filippis R, De Fazio P, Gaetano R, Steardo L, Cedro C, Bruno A, Zoccali RA and Muscatello MRA (2021) Current and emerging long-acting antipsychotics for the treatment of schizophrenia, Expert Opinion on Drug Safety, 20:7, 771-790. [CrossRef]
- https://www.msdmanuals.com/home/drugs/adverse-drug-reactions/severity-of-adverse-drug-reactions.
- Procyshyn RM, Bezchlibnyk-Butler KZ, Kim DD. Clinical Handbook of Psychotropic Drugs 25th edition. 2023 Hogrefe Publishing ISBN 978-0-88937-474-4.
- Procyshyn RM, Bezchlibnyk-Butler KZ, Jeffries JJ. Clinical Handbook of Psychotropic Drugs 23rd edition. 2019 Hogrefe Publishing ISBN 978-0-88937-474-4.
- PDSP ki Database https://pdsp.unc.edu/databases/kidb.php.
- Correll CU, Kim E, Sliwa JK, Hamm W, Gopal S, Mathews M, Venkatasubramanian R, Saklad SR. Pharmacokinetic Characteristics of Long-Acting Injectable Antipsychotics for Schizophrenia: An Overview. CNS Drugs (2021) 35:39–59. [CrossRef]
- https://www.medscape.com.
- https://ec.europa.eu/health/documents/community-register/html/h479.htm.
- https://nomenclator.anm.ro/medicamente.
- Kerwin R, Millet B, Herman E, Banki CM, Lublin H, Pans M, Hanssens L, L’Italien G, McQuade RD, Beuzen J-N. A multicentre, randomized, naturalistic, open-label study between aripiprazole and standard of care in the management of community-treated schizophrenic patients Schizophrenia Trial of Aripiprazole: (STAR) study. European Psychiatry 22 (2007) 433-443.
- Naber D, Hansen K, Forray C, Baker RA, Sapin C, Beillat M, Peters-Strickland T, Nylander A-G, Hertel P, Andersen HS, Eramo A, Loze J-Y, Potkin SG. Qualify: a randomized head-to-head study of aripiprazole once-monthly and paliperidone palmitate in the treatment of schizophrenia. Schizophrenia Research 168 (2015) 498-504.
- Naber D, Baker RA, Eramo A, Forray C, Hansen K, Sapin C, Peters-Strickland T, Nylander A-G, Hertel P, Schmidt SN, Loze J-Y, Potkin SG. Long-term effectiveness of aripiprazole once-monthly for schizophrenia is maintained in the QUALIFY extension study. Schizophrenia Research 192 (2018) 205-210.
| Pro-functionality action on receptor/transporter | Score |
|---|---|
| Reduce positive symptoms | 3 |
| Reduce negative symptoms | 3 |
| Anti-aggressiveness effect | 3 |
| Pro-cognitive effect | 2 |
| Sleep improvement | 2 |
| Motivation improvement | 2 |
| Antidepressant effect | 2 |
| Antimanic effect | 2 |
| Anxiolytic effect | 2 |
| Any antipsychotic’s adverse event improvement | 1 (for each) |
| Any other pro-functionality effect | 1 (for each) |
| Adverse events severity | Score |
|---|---|
| Mild | −0.25 |
| Moderate | −0.5 |
| Severe | −0.75 |
| Lethal | −1 |
| Receptor/neurotransmitter pump action | Pro-functionality effects | Pro-functionality score | Anti-functionality effects | Anti-functionality score | Functionality coefficient of receptor/transporter (FCR/T) |
|---|---|---|---|---|---|
| Antagonism of postsynaptic D2 receptors | In mesolimbic/mesostriatal tract – reduction in positive symptoms, antimanic | 5 | – In nigrostriatal tract – EPS (acute dystonia, pseudoparkinsonism, akathisia, tardive dyskinesia), Neuroleptic malignant syndrome – In tuberoinfundibular tract – prolactin elevation leading to galactorrhoea, sexual dysfunction, infertility especially in woman, demineralisation of bones, weight gain – In mesocortical/mesostriatal tract – may exacerbate negative symptoms, affective symptoms, cognitive symptoms |
-6.25 | -1.25 |
| Partial agonism of postsynaptic D2 receptors | reduction in positive symptoms, improve negative symptoms, reduction of hyperprolactinemia | 7 | Some akathisia | -0.5 | 6.5 |
| Partial agonism/antagonism of D3 receptors | antidepressant effect, improve negative symptoms, pro-cognitive, motivation improvement | 9 | NA | 0 | 9 |
| Antagonism of H1 receptors | Anti-emetic effect, anxiolytic effects | 3 | Sedation, drowsiness, appetite increase, weight gain, postural hypotension | -1.75 | 1.25 |
| Antagonism of M1 receptors | Mitigation of extrapyramidal adverse effects | 1 | Dry mouth, dry eyes, blurred vision, constipation, urinary retention, sinus tachycardia, QRS changes, confusion, worsening cognition, delirium, sedation, exacerbation/attack of narrow-angle glaucoma Potentiation of effects of drugs with anticholinergic properties |
-6 | -5 |
| Antagonism of M3 receptors | NA | 0 | Beta cell failure, reduced insulin release, glucose intolerance, type 2 diabetes mellitus | -2 | -2 |
| Antagonism of ⍺1 receptors | NA | 0 | Postural hypotension, dizziness, reflex tachycardia, sedation | 1.5 | -1.5 |
| Antagonism of ⍺2 receptors | – May improve cognitive deficits and have – Antagonism of presynaptic α2-adrenergic receptors enhances serotonergic and noradrenergic transmission |
4 | Sexual dysfunction, priapism | -1.25 | 2.75 |
| Antagonism/partial agonism of 5-HT1A receptors | Procognitive, anxiolytic, and antidepressant effects, antiaggresive action | 9 | NA | 0 | 9 |
| Antagonism of 5-HT1B receptors | antidepressant effect, procognitive | 4 | NA | 0 | 4 |
| Antagonism of 5-HT1D receptors | antidepressant effect | 2 | NA | 0 | 2 |
| Antagonism of 5-HT2A receptors | Ameliorate EPS, improve negative, cognitive, and mood symptoms, ameliorate hyperprolactinemia, improve positive symptoms, anxiolytic, antimigraine effect, and improved sleep | 16 | Sedation, hypotension, ejaculatory problems | -1 | 15 |
| Antagonism of 5-HT2C receptors | Procognitive and antidepressant effects, anxiolytic | 6 | Increased appetite, weight gain | -0.75 | 5.25 |
| Antagonism of 5-HT3 receptors | antidepressant, procognitive, improve nausea and vomiting | 6 | NA | 0 | 6 |
| Antagonism of 5-HT6 receptors | antidepressant | 2 | NA | 0 | 2 |
| Antagonism of 5-HT7 receptors | procognitive, anxiolytic, and antidepressant effects | 6 | NA | 0 | 6 |
| Norepinephrine transporter (NET) inhibition | antidepressant actions | 2 | Tremors, tachycardia, hypertension, sweating, insomnia, erectile and ejaculation problems | -2.5 | -0.5 |
| Serotonin transporter (SERT) inhibition | antidepressant actions, anti-anxiety, anti-panic, anti-obsessional | 4 | Dyspepsia, nausea, headache, nervousness, akathisia, extrapyramidal effects, anorexia, sexual side effects | -3 | 1 |
| Binding coefficient of receptor/transporter (BCR/T) | Ki(nM) |
|---|---|
| 1 | 0.001-1 |
| 0.8 | 1-10 |
| 0.6 | 10-100 |
| 0.4 | 100-1000 |
| 0.2 | 1000-10.000 |
| 0 | >10.000 |
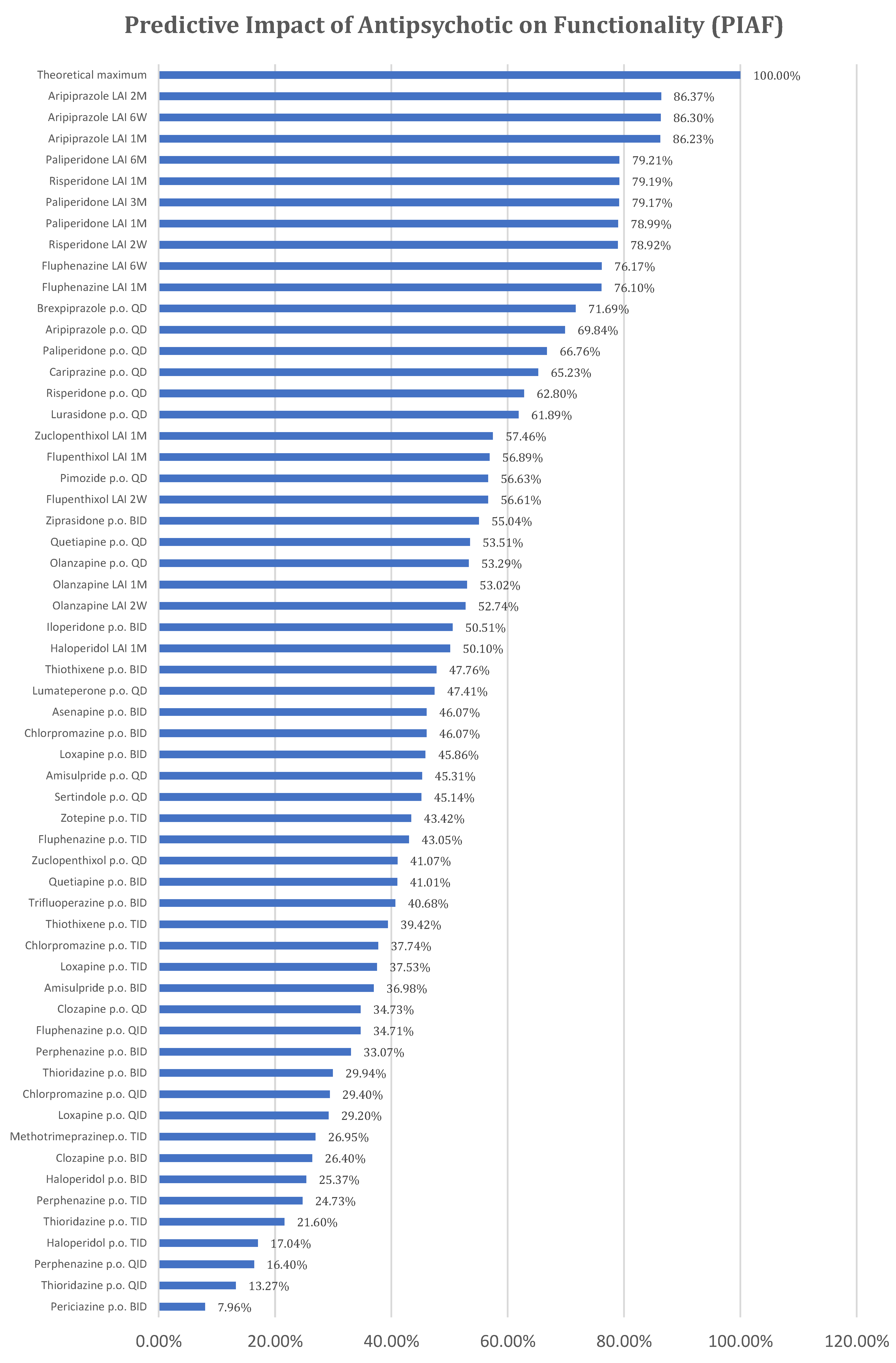
| Antipsychotic | PS | PS% |
|---|---|---|
| Chlorpromazine | 25.6 | 42.14% |
| Flupenthixol | 8.7 | 14.32% |
| Fluphenazine | 32.05 | 52.76% |
| Haloperidol | 20.7 | 34.07% |
| Loxapine | 25.35 | 41.73% |
| Methotrimeprazine | 12.5 | 20.58% |
| Periciazine | -0.45 | -0.74% |
| Perphenazine | 30.05 | 49.47% |
| Pimozide | 28.3 | 46.58% |
| Thioridazine | 26.25 | 43.21% |
| Thiothixene | 27.65 | 45.51% |
| Trifluoperazine | 19.05 | 31.36% |
| Zuclopenthixol | 9.4 | 15.47% |
| Asenapine | 45.85 | 75.47% |
| Clozapine | 21.95 | 36.13% |
| Iloperidone | 31 | 51.03% |
| Sertindole | 34.6 | 56.95% |
| Lumateperone | 17.1 | 28.15% |
| Lurasidone | 34.7 | 57.12% |
| Olanzapine | 24.25 | 39.92% |
| Zotepine | 32.5 | 53.50% |
| Paliperidone | 35.55 | 58.52% |
| Quetiapine | 19.45 | 32.02% |
| Amisulpride | 14.55 | 23.95% |
| Risperidone | 35.8 | 58.93% |
| Ziprasidone | 36.5 | 60.08% |
| Aripiprazole(a) | 44.35 | 73.00% |
| Brexpiprazole(a) | 46.6 | 76.71% |
| Cariprazine(a) | 38.75 | 63.79% |
| Theoretical maximum | 60.75 | 100.00% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




