Submitted:
25 September 2024
Posted:
25 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Ustekinumab and Proposed Biosimilars
2.1. Ustekinumab
2.1.1. Generation
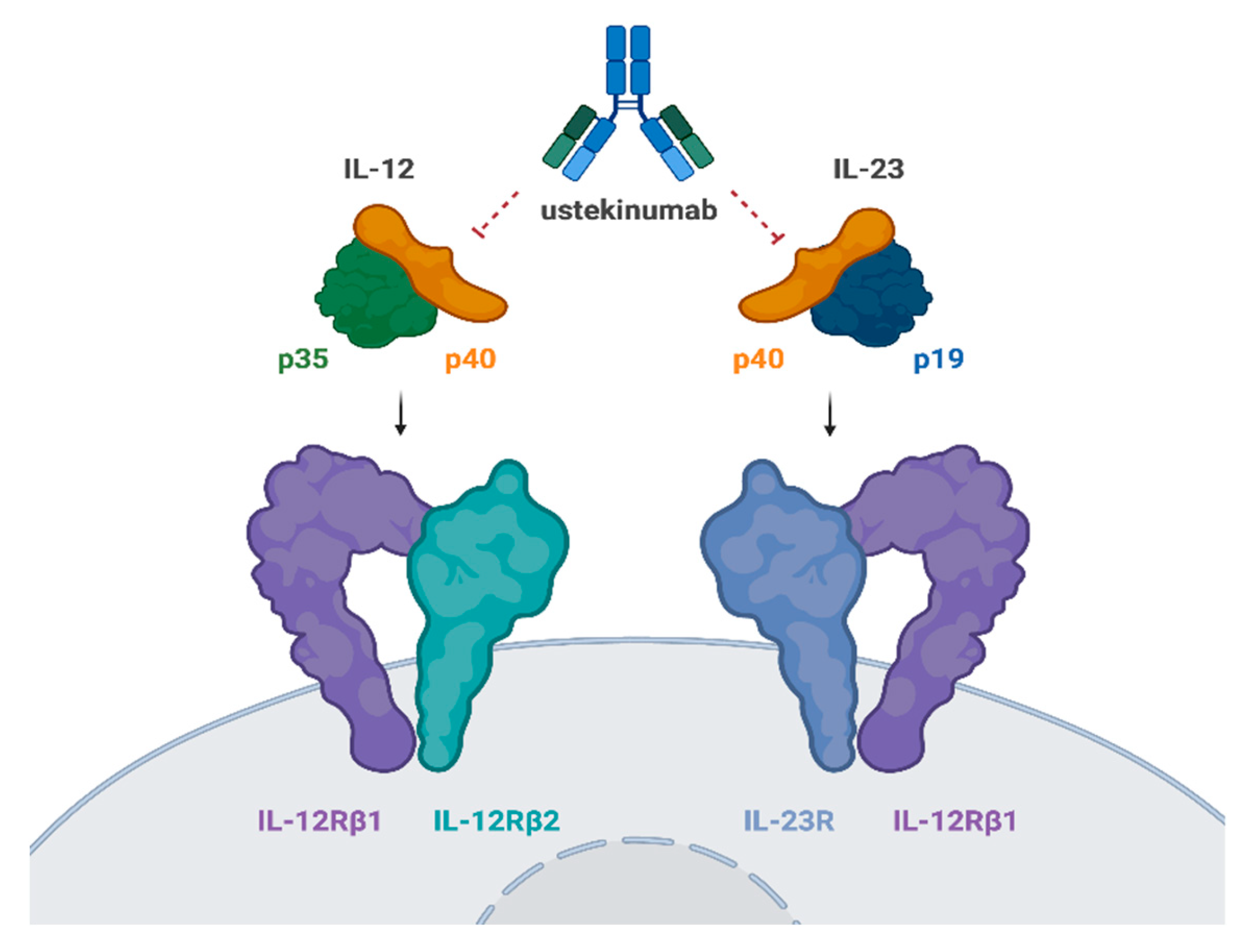
2.1.2. Mechanism of Action
2.1.3. Indications and Efficacy Data
| Ustekinumab approved indications | Adults and children 6 years and older with moderate to severe psoriasis who may benefit from taking injections or pills (systemic therapy) or phototherapy (treatment using ultraviolet light alone or with pills). Adults and children 6 years and older with active psoriatic arthritis. Adults 18 years and older with moderately to severely active Crohn’s disease. Adults 18 years and older with moderately to severely active ulcerative colitis. |
| Off-label reported uses of ustekinumab | Hidradenitis suppurativa Takayasu arteritis Giant cell arteritis Behçet disease Myelodysplastic syndrome Pyoderma grangrenosum Pityriasis rubra pilaris Synovitis, acne, pustulosis, hyperostosis, and osteitis (SAPHO) syndrome Lichen planus Atopic dermatitis Systemic lupus erythematosus |
2.2. ABP 654 (Wezlana® or Wezenla®)
2.2.1. Pharmacology
2.2.2. Clinical Efficacy and Safety
2.2.3. Switching from ustekinumab RP
2.3. AVT04 (Uzpruvo® or Selarsdi®)
2.3.1. Pharmacology
2.3.2. Clinical Efficacy and Safety
2.4. SB17 (Pyzchiva®, Eksunbi®)
2.4.1. Pharmacology
2.4.2. Clinical Efficacy and Safety
2.5. CT-P43 (SteQeyma®)
2.6. BAT2206
2.7. DMB-3115
2.8. FYB202 (Fymskina®, Otulfi®)
2.9. QX001S
2.10. BFI-751
2.11. Other Proposed Ustekinumab Biosimilars
3. Discussion
4. Conclusions
Authorship declaration
Conflicts of interest
References
- Michalek, I.M.; Loring, B.; John, S.M. A Systematic Review of Worldwide Epidemiology of Psoriasis. Journal of the European Academy of Dermatology and Venereology 2017, 31, 205–212. [CrossRef]
- Rendon, A.; Schäkel, K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci 2019, 20, 1475. [CrossRef]
- Blauvelt, A.; Chiricozzi, A. The Immunologic Role of IL-17 in Psoriasis and Psoriatic Arthritis Pathogenesis. Clin Rev Allergy Immunol 2018, 55, 379–390. [CrossRef]
- Girolomoni, G.; Strohal, R.; Puig, L.; Bachelez, H.; Barker, J.; Boehncke, W.H.; Prinz, J.C. The Role of IL-23 and the IL-23/TH17 Immune Axis in the Pathogenesis and Treatment of Psoriasis. Journal of the European Academy of Dermatology and Venereology 2017, 31, 1616–1626. [CrossRef]
- Nussbaum, L.; Chen, Y.L.; Ogg, G.S. Role of Regulatory T Cells in Psoriasis Pathogenesis and Treatment. British Journal of Dermatology 2021, 184, 14–24. [CrossRef]
- Baumgart, D.C.; Misery, L.; Naeyaert, S.; Taylor, P.C. Biological Therapies in Immune-Mediated Inflammatory Diseases: Can Biosimilars Reduce Access Inequities? Front Pharmacol 2019, 10, 279. [CrossRef]
- Kabir, E.R.; Moreino, S.S.; Sharif Siam, M.K. The Breakthrough of Biosimilars: A Twist in the Narrative of Biological Therapy. Biomolecules 2019, 9. [CrossRef]
- Markus, R.; Liu, J.; Ramchandani, M.; Landa, D.; Born, T.; Kaur, P. Developing the Totality of Evidence for Biosimilars: Regulatory Considerations and Building Confidence for the Healthcare Community. BioDrugs 2017, 31, 175–187,. [CrossRef]
- EMA. Guideline on Similar Biological Medicinal Products Containing Monoclonal AntibodiesdNonclinical and Clinical Issues. EMA; 2012.
- FDA. Scientific Considerations in Demonstrating Biosimilarity to a Reference Product. FDA; 2015.
- Griffiths, C.E.M.; Thaçi, D.; Gerdes, S.; Arenberger, P.; Pulka, G.; Kingo, K.; Weglowska, J.; EGALITY study group; Hattebuhr, N.; Poetzl, J.; et al. The EGALITY Study: A Confirmatory, Randomized, Double-Blind Study Comparing the Efficacy, Safety and Immunogenicity of GP2015, a Proposed Etanercept Biosimilar, vs. the Originator Product in Patients with Moderate-to-Severe Chronic Plaque-Type Psoriasis. Br J Dermatol 2017, 176, 928–938. [CrossRef]
- Hercogová, J.; Papp, K.A.; Chyrok, V.; Ullmann, M.; Vlachos, P.; Edwards, C.J. AURIEL-PsO: A Randomized, Double-Blind Phase III Equivalence Trial to Demonstrate the Clinical Similarity of the Proposed Biosimilar MSB11022 to Reference Adalimumab in Patients with Moderate-to-Severe Chronic Plaque-Type Psoriasis. Br J Dermatol 2020, 182, 316–326. [CrossRef]
- Papp, K.; Bachelez, H.; Costanzo, A.; Foley, P.; Gooderham, M.; Kaur, P.; Narbutt, J.; Philipp, S.; Spelman, L.; Weglowska, J.; et al. Clinical Similarity of Biosimilar ABP 501 to Adalimumab in the Treatment of Patients with Moderate to Severe Plaque Psoriasis: A Randomized, Double-Blind, Multicenter, Phase III Study. J Am Acad Dermatol 2017, 76, 1093–1102. [CrossRef]
- EMA. Stelara®: EPARdProduct Information; Annex I, Summary of Product Characteristics. EMA; 2023.
- FDA. Stelara®: Prescribing Information. FDA; 2023.
- Kalb, R.E.; Fiorentino, D.F.; Lebwohl, M.G.; Toole, J.; Poulin, Y.; Cohen, A.D.; Goyal, K.; Fakharzadeh, S.; Calabro, S.; Chevrier, M.; et al. Risk of Serious Infection With Biologic and Systemic Treatment of Psoriasis: Results From the Psoriasis Longitudinal Assessment and Registry (PSOLAR). JAMA Dermatol 2015, 151, 961–969. [CrossRef]
- Fishwild, D.M.; O’Donnell, S.L.; Bengoechea, T.; Hudson, D. V; Harding, F.; Bernhard, S.L.; Jones, D.; Kay, R.M.; Higgins, K.M.; Schramm, S.R.; et al. High-Avidity Human IgG Kappa Monoclonal Antibodies from a Novel Strain of Minilocus Transgenic Mice. Nat Biotechnol 1996, 14, 845–851. [CrossRef]
- Lonberg, N. Human Antibodies from Transgenic Animals. Nat Biotechnol 2005, 23, 1117–1125. [CrossRef]
- Benson, J.M.; Peritt, D.; Scallon, B.J.; Heavner, G.A.; Shealy, D.J.; Giles-Komar, J.M.; Mascelli, M.A. Discovery and Mechanism of Ustekinumab: A Human Monoclonal Antibody Targeting Interleukin-12 and Interleukin-23 for Treatment of Immune-Mediated Disorders. MAbs 2011, 3.
- Luo, J.; Wu, S.-J.; Lacy, E.R.; Orlovsky, Y.; Baker, A.; Teplyakov, A.; Obmolova, G.; Heavner, G.A.; Richter, H.-T.; Benson, J. Structural Basis for the Dual Recognition of IL-12 and IL-23 by Ustekinumab. J Mol Biol 2010, 402, 797–812. [CrossRef]
- Presky, D.H.; Yang, H.; Minetti, L.J.; Chua, A.O.; Nabavi, N.; Wu, C.Y.; Gately, M.K.; Gubler, U. A Functional Interleukin 12 Receptor Complex Is Composed of Two Beta-Type Cytokine Receptor Subunits. Proc Natl Acad Sci U S A 1996, 93, 14002–14007. [CrossRef]
- Oppmann, B.; Lesley, R.; Blom, B.; Timans, J.C.; Xu, Y.; Hunte, B.; Vega, F.; Yu, N.; Wang, J.; Singh, K.; et al. Novel P19 Protein Engages IL-12p40 to Form a Cytokine, IL-23, with Biological Activities Similar as Well as Distinct from IL-12. Immunity 2000, 13, 715–725. [CrossRef]
- Parham, C.; Chirica, M.; Timans, J.; Vaisberg, E.; Travis, M.; Cheung, J.; Pflanz, S.; Zhang, R.; Singh, K.P.; Vega, F.; et al. A Receptor for the Heterodimeric Cytokine IL-23 Is Composed of IL-12Rbeta1 and a Novel Cytokine Receptor Subunit, IL-23R. J Immunol 2002, 168, 5699–5708. [CrossRef]
- Leonardi, C.L.; Kimball, A.B.; Papp, K.A.; Yeilding, N.; Guzzo, C.; Wang, Y.; Li, S.; Dooley, L.T.; Gordon, K.B.; PHOENIX 1 study investigators Efficacy and Safety of Ustekinumab, a Human Interleukin-12/23 Monoclonal Antibody, in Patients with Psoriasis: 76-Week Results from a Randomised, Double-Blind, Placebo-Controlled Trial (PHOENIX 1). Lancet 2008, 371, 1665–1674. [CrossRef]
- Papp, K.A.; Langley, R.G.; Lebwohl, M.; Krueger, G.G.; Szapary, P.; Yeilding, N.; Guzzo, C.; Hsu, M.-C.; Wang, Y.; Li, S.; et al. Efficacy and Safety of Ustekinumab, a Human Interleukin-12/23 Monoclonal Antibody, in Patients with Psoriasis: 52-Week Results from a Randomised, Double-Blind, Placebo-Controlled Trial (PHOENIX 2). Lancet 2008, 371, 1675–1684. [CrossRef]
- Kimball, A.B.; Papp, K.A.; Wasfi, Y.; Chan, D.; Bissonnette, R.; Sofen, H.; Yeilding, N.; Li, S.; Szapary, P.; Gordon, K.B.; et al. Long-Term Efficacy of Ustekinumab in Patients with Moderate-to-Severe Psoriasis Treated for up to 5 Years in the PHOENIX 1 Study. J Eur Acad Dermatol Venereol 2013, 27, 1535–1545. [CrossRef]
- Gerdes, S.; Hoffmann, M.; Asadullah, K.; Korge, B.; Mortazawi, D.; Krüger, N.; Personke, Y.; Tabori, S.; Gomez, M.; Wegner, S.; et al. Effectiveness, Safety and Quality-of-Life Effects of Guselkumab and Ustekinumab in Patients with Psoriasis: Week 104 Results from the Non-Interventional, Prospective, German Multicentre PERSIST Study. J Eur Acad Dermatol Venereol 2023. [CrossRef]
- Yiu, Z.Z.N.; Becher, G.; Kirby, B.; Laws, P.; Reynolds, N.J.; Smith, C.H.; Warren, R.B.; Griffiths, C.E.M.; BADBIR Study Group Drug Survival Associated With Effectiveness and Safety of Treatment With Guselkumab, Ixekizumab, Secukinumab, Ustekinumab, and Adalimumab in Patients With Psoriasis. JAMA Dermatol 2022, 158, 1131–1141. [CrossRef]
- Griffiths, C.E.M.; Strober, B.E.; van de Kerkhof, P.; Ho, V.; Fidelus-Gort, R.; Yeilding, N.; Guzzo, C.; Xia, Y.; Zhou, B.; Li, S.; et al. Comparison of Ustekinumab and Etanercept for Moderate-to-Severe Psoriasis. N Engl J Med 2010, 362, 118–128. [CrossRef]
- Armstrong, A.W.; Soliman, A.M.; Betts, K.A.; Wang, Y.; Gao, Y.; Puig, L.; Augustin, M. Comparative Efficacy and Relative Ranking of Biologics and Oral Therapies for Moderate-to-Severe Plaque Psoriasis: A Network Meta-Analysis. Dermatol Ther (Heidelb) 2021, 11, 885–905. [CrossRef]
- Sbidian, E.; Chaimani, A.; Garcia-Doval, I.; Doney, L.; Dressler, C.; Hua, C.; Hughes, C.; Naldi, L.; Afach, S.; Le Cleach, L. Systemic Pharmacological Treatments for Chronic Plaque Psoriasis: A Network Meta-Analysis. Cochrane Database Syst Rev 2022, 5, CD011535. [CrossRef]
- Cantin, G.; Liu, Q.; Shah, B.; Kuhns, S.; Wikström, M.; Cao, S.; Liu, J. Analytical and Functional Similarity of the Biosimilar Candidate ABP 654 to Ustekinumab Reference Product. Drugs in R and D 2023, 23, 421–438. [CrossRef]
- Chow, V.; Mytych, D.T.; Das, S.; Franklin, J. Pharmacokinetic Similarity of ABP 654, an Ustekinumab Biosimilar Candidate: Results from a Randomized, Double-Blind Study in Healthy Subjects. Clin Pharmacol Drug Dev 2023, 12, 863–873. [CrossRef]
- Zhu, Y.; Wang, Q.; Frederick, B.; Bouman-Thio, E.; Marini, J.C.; Keen, M.; Petty, K.J.; Davis, H.M.; Zhou, H. Comparison of the Pharmacokinetics of Subcutaneous Ustekinumab between Chinese and Non-Chinese Healthy Male Subjects across Two Phase 1 Studies. Clin Drug Investig 2013, 33, 291–301. [CrossRef]
- Wynne, C.; Hamilton, P.; McLendon, K.; Stroissnig, H.; Smith, M.; Duijzings, P.; Ruffieux, R.; Otto, H.; Sattar, A.; Haliduola, H.N.; et al. A Randomized, Double-Blind, 3-Arm, Parallel Study Assessing the Pharmacokinetics, Safety, Tolerability and Immunogenicity of AVT04, an Ustekinumab Candidate Biosimilar, in Healthy Adults. Expert Opin Investig Drugs 2023, 32, 417–427. [CrossRef]
- Feldman, S.R.; Reznichenko, N.; Berti, F.; Duijzings, P.; Ruffieux, R.; Otto, H.; Haliduola, H.N.; Leutz, S.; Stroissnig, H. Randomized, Double-Blind, Multicenter Study to Evaluate Efficacy, Safety, Tolerability, and Immunogenicity between AVT04 and the Reference Product Ustekinumab in Patients with Moderate-to-Severe Chronic Plaque Psoriasis. Expert Opin Biol Ther 2023, 23, 759–771,. [CrossRef]
- Jeong, H.; Kang, T.; Lee, J.; Im, S. Comparison of SB17 and Reference Ustekinumab in Healthy Adults: A Randomized, Double-Blind, Single-Dose, Phase I Study. Int J Clin Pharmacol Ther 2024, 62, 231–240. [CrossRef]
- Feldman, S.R.; Narbutt, J.; Girolomoni, G.; Brzezicki, J.; Reznichenko, N.; Zegadło-Mylik, M.A.; Pulka, G.; Dmowska-Stecewicz, M.; Kłujszo, E.; Rekalov, D.; et al. A Randomized, Double-Blind, Phase III Study Assessing Clinical Similarity of SB17 (Proposed Ustekinumab Biosimilar) to Reference Ustekinumab in Subjects with Moderate-to-Severe Plaque Psoriasis. J Am Acad Dermatol 2024. [CrossRef]
- Papp, K.A.; Lebwohl, M.G.; Thaçi, D.; Jaworski, J.; Kwiek, B.; Trefler, J.; Dudek, A.; Szepietowski, J.C.; Reznichenko, N.; Narbutt, J.; et al. Efficacy and Safety of Candidate Biosimilar CT-P43 Versus Originator Ustekinumab in Moderate to Severe Plaque Psoriasis: 28-Week Results of a Randomised, Active-Controlled, Double-Blind, Phase III Study. BioDrugs 2024, 38, 121–131. [CrossRef]
- Wu, M.; Li, X.; Yang, D.; Wang, M.; Zhang, H.; Li, C.; Mai, J.; Yang, L.; Qi, Y.; Yu, J.C.; et al. Comparison of Pharmacokinetic Similarity, Immunogenicity, and Safety of Ustekinumab and BAT2206 in Healthy Chinese Male Subjects in a Double-Blind, Randomized, Single-Dose, Parallel-Group Phase I Trial. BioDrugs 2023, 37, 89–98. [CrossRef]
- Gao, L.; Li, Q.; Zhang, H.; Wu, M.; Fang, M.; Yang, L.; Li, X.; Liu, J.; Li, C.; Chen, H.; et al. A Biosimilarity Study Between QX001S and Ustekinumab in Healthy Chinese Male Subjects. Front Pharmacol 2021, 12. [CrossRef]
- Hausfeld, J.N.; Challand, R.; McLendon, K.; Macapagal, N.; Bruce-Staskal, P.; Fiaschetti, C.; Sampey, D.B. Pharmacokinetic Profiles of a Proposed Biosimilar Ustekinumab (BFI-751): Results From a Randomized Phase 1 Trial. Clin Pharmacol Drug Dev 2023, 12, 1001–1012,. [CrossRef]
- Estes, S.; Melville, M. Mammalian Cell Line Developments in Speed and Efficiency. In; 2013; pp. 11–33.
- Dumont, J.; Euwart, D.; Mei, B.; Estes, S.; Kshirsagar, R. Human Cell Lines for Biopharmaceutical Manufacturing: History, Status, and Future Perspectives. Crit Rev Biotechnol 2016, 36, 1110–1122. [CrossRef]
- Sun, X.; Cui, Z.; Wang, Q.; Liu, L.; Ding, X.; Wang, J.; Cai, X.; Li, B.; Li, X. Formation and Clinical Effects of Anti-Drug Antibodies against Biologics in Psoriasis Treatment: An Analysis of Current Evidence. Autoimmun Rev 2024, 23, 103530. [CrossRef]
- Loeff, F.C.; Tsakok, T.; Dijk, L.; Hart, M.H.; Duckworth, M.; Baudry, D.; Russell, A.; Dand, N.; van Leeuwen, A.; Griffiths, C.E.M.; et al. Clinical Impact of Antibodies against Ustekinumab in Psoriasis: An Observational, Cross-Sectional, Multicenter Study. J Invest Dermatol 2020, 140, 2129–2137. [CrossRef]
- Tsai, T.-F.; Ho, J.-C.; Song, M.; Szapary, P.; Guzzo, C.; Shen, Y.-K.; Li, S.; Kim, K.-J.; Kim, T.-Y.; Choi, J.-H.; et al. Efficacy and Safety of Ustekinumab for the Treatment of Moderate-to-Severe Psoriasis: A Phase III, Randomized, Placebo-Controlled Trial in Taiwanese and Korean Patients (PEARL). J Dermatol Sci 2011, 63, 154–163,. [CrossRef]
- Chiu, H.-Y.; Chu, T.W.; Cheng, Y.-P.; Tsai, T.-F. The Association between Clinical Response to Ustekinumab and Immunogenicity to Ustekinumab and Prior Adalimumab. PLoS One 2015, 10, e0142930. [CrossRef]
- Mojtahed Poor, S.; Henke, M.; Ulshöfer, T.; Köhm, M.; Behrens, F.; Burkhardt, H.; Schiffmann, S. The Role of Antidrug Antibodies in Ustekinumab Therapy and the Impact of Methotrexate. Rheumatology (Oxford) 2023, 62, 3993–3999. [CrossRef]
- Roblin, X.; Duru, G.; Papamichael, K.; Cheifetz, A.S.; Kwiatek, S.; Berger, A.-E.; Barrau, M.; Waeckel, L.; Nancey, S.; Paul, S. Development of Antibodies to Ustekinumab Is Associated with Loss of Response in Patients with Inflammatory Bowel Disease. J Clin Med 2023, 12. [CrossRef]
- Phan, D.B.; Elyoussfi, S.; Stevenson, M.; Lunt, M.; Warren, R.B.; Yiu, Z.Z.N. Biosimilars for the Treatment of Psoriasis. JAMA Dermatol 2023, 159, 763. [CrossRef]
- Naldi, L.; Addis, A. Biosimilars for the Treatment of Psoriasis-A Systematic Review of Clinical Trials and Observational Studies Looking Beyond Single-Trial Evidence for a Valuable Choice. Photography in Clinical Medicine 2014, 2020, 491–499,.
- Ustekinumab Prescribing Information. Www.Stelarainfo.Com. August 2024.
- Colquhoun M, Kemp AK. Ustekinumab. [Updated 2023 Mar 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: Https://Www.Ncbi.Nlm.Nih.Gov/Books/NBK570645/.
- Masson, R.; Seivright, J.; Grogan, T.; Atluri, S.; Hamzavi, I.; Hogeling, M.; Shi, V.Y.; Hsiao, J.L. Ustekinumab in Hidradenitis Suppurativa: A Systematic Review and Meta-Analysis. Dermatol Ther (Heidelb) 2024, 14, 1901–1916. [CrossRef]
- Ye, L.; Wu, Z.; Li, C.; Zhao, X.; Wan, M.; Wang, L. Off-Label Uses of Ustekinumab. Dermatol Ther 2022, 35, e15910. [CrossRef]
| Product name | Company name | Production system | Stage of development |
|---|---|---|---|
| Ustekinumab (Stelara®) | Janssen Biotech | Sp2/0 cell line | Reference product |
| ABP 654 (Wezlana® in US and Canada; Wezenla® in Europe) | Amgen | CHO cell line | FDA-approved* in October 2023 Australian Therapeutic Goods Administration approval in January 2024 Health Canada approval in March 2024 EMA-approved in April 2024 |
| AVT04 (Uzpruvo® in Europe and Japan; Serlarsdi® in US; Jamteki™ in Canada) | Alvotech / Stada / Teva | Sp2/0 cell line | Japan approval in September 2023 Health Canada approval in November 2023 EMA-approved in January 2024 FDA-approved** in April 2024 |
| SB17 (Pyzchiva® in US and Europe; Eksunbi® in Europe; EpyztekTM in Korea) | Samsung Bioepis | CHO cell line |
EMA-approved in April 2024 FDA-approved* in July 2024 Korea approval in April 2024 |
| CT-P43 (SteQeyma®) | Celltrion Healthcare | CHO cell line |
Approved In South Korea (South Korean Ministry of Food and Drug Safety) in June-2024 Approved In Canada in July-2024 EMA-approved in August 2024 BLA submitted to FDA, pending acceptance |
| RT-111 (oral CT-P43; RaniPill®) | Rani Therapeutics | Unknown | Phase I study ongoing (NCT05890118) |
| FYB202 (Fymskina®, Otulfi®) | Formycon – Fresenius Kabi | CHO cell line |
Phase I and phase III trials completed with positive results BLA accepted by FDA MAA accepted by EMA with positive opinion from the CHMP in July 2024 |
| BAT2206 | Bio-Thera / Hikma | CHO cell line |
Phase I and phase III trials completed with positive results BLA accepted by FDA MAA accepted by EMA Approved in Brazil for psoriatic arthritis |
| DMB-3115 |
Dong-A ST / Accord BioPharma |
Sp2/0 cells |
Phase I and phase III trials completed with positive results BLA accepted by FDA MAA accepted by EMA |
| QX001S | Qyuns Therapeutics | CHO cell line |
Phase I and phase III trials completed with positive results BLA accepted by China National Medical Products Administration |
| BFI-751 | BioFactura / CuraTeQ | Murine myeloma, NS0 | Phase 1 completed with positive results (NCT04843631) |
| NeuLara | Neuclone / Serum Institute of India | Unknown | Phase 1 completed in April 2020 with positive results |
| ONS3040 | Oncobiologics | Unknown | Pre-clinical development |
| BOW090 | Epirus Biopharmaceuticals / Bioceros Holding | Unknown | Pre-clinical development |
| Product | ADA incidence in healthy individuals | ADA incidence in psoriasis patients* | NADA incidence in psoriasis patients** |
|---|---|---|---|
| UST RP [15] | 5.6% | 4.1% | 67% |
| ABP 654 [32,33] | ABP 654: 15.4% EU UST: 36.3% US UST: 38.0% |
ABP 654: 18.6% UST: 37.1% |
ABP 654: 8.6% UST: 17.9% |
| AVT04 [35,36] | AVT04: 36.7% EU UST: 59.6% US UST: 53.6% |
AVT04: 21.2% UST: 26.2%% |
AVT04: 33.3% UST: 22.9% |
| SB17 [37,38] | SB17: 26.9% EU UST: 34.3% US UST: 34.3% |
SB17: 13.3% UST: 39.4% |
SB17: 13.7% UST: 35.4% |
| CT-P43 [39] | N/A | CT-P43: 10.2% UST: 17.0% |
CT-P43: 5.9% UST: 7.9% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




