Submitted:
22 September 2024
Posted:
24 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhadoria, P., G. Gupta, and A. Agarwal, Viral Pandemics in the Past Two Decades: An Overview. J Family Med Prim Care, 2021. 10(8): p. 2745-2750. [CrossRef]
- Organization, W.H. Coronavirus disease (COVID-19). 2023 9 August 2023; Available from: https://www.who.int/news-room/fact-sheets/detail/coronavirus-disease-(covid-19).
- Li, X., et al., Transmission dynamics and evolutionary history of 2019-nCoV. J Med Virol, 2020. 92(5): p. 501-511.
- Lamptey, E., et al., COVID-19 vaccines development in Africa: a review of current situation and existing challenges of vaccine production. Clin Exp Vaccine Res, 2022. 11(1): p. 82-88. [CrossRef]
- Al Kaabi, N., et al., Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults: A Randomized Clinical Trial. Jama, 2021. 326(1): p. 35-45. [CrossRef]
- Palacios, R., et al., Efficacy and Safety of a COVID-19 Inactivated Vaccine in Healthcare Professionals in Brazil: The PROFISCOV Study. SSRN Electronic Journal, 2021. [CrossRef]
- Tanriover, M.D., et al., Efficacy and safety of an inactivated whole-virion SARS-CoV-2 vaccine (CoronaVac): interim results of a double-blind, randomised, placebo-controlled, phase 3 trial in Turkey. Lancet, 2021. 398(10296): p. 213-222. [CrossRef]
- Fadlyana, E., et al., A phase III, observer-blind, randomized, placebo-controlled study of the efficacy, safety, and immunogenicity of SARS-CoV-2 inactivated vaccine in healthy adults aged 18-59 years: An interim analysis in Indonesia. Vaccine, 2021. 39(44): p. 6520-6528. [CrossRef]
- Mengstu, S. and A. Beyene Berha, Safety and Efficacy of COVID-19 Vaccine in Africa: Systematic Review. Infect Drug Resist, 2023. 16: p. 3085-3100. [CrossRef]
- Ashmawy, R., et al., Effectiveness and Safety of Inactivated SARS-CoV-2 Vaccine (BBIBP-CorV) among Healthcare Workers: A Seven-Month Follow-Up Study at Fifteen Central Hospitals. Vaccines (Basel), 2023. 11(5). [CrossRef]
- Belayachi, J., et al., Long term effectiveness of inactivated vaccine BBIBP-CorV (Vero Cells) against COVID-19 associated severe and critical hospitalization in Morocco. PLoS One, 2022. 17(12): p. e0278546. [CrossRef]
- Zhang, Y., et al., Real-world study of the effectiveness of BBIBP-CorV (Sinopharm) COVID-19 vaccine in the Kingdom of Morocco. BMC Public Health, 2022. 22(1): p. 1584. [CrossRef]
- Control, A.C.f.D. Proportion of Vaccine Type Acquired by Member States in Africa. 2021 [cited 2024; Available from: https://africacdc.org/covid-19/covid-19-vaccination/.
- Flores-Vega, V.R., et al., SARS-CoV-2: Evolution and Emergence of New Viral Variants. Viruses, 2022. 14(4). [CrossRef]
- Dean, N.E., J.W. Hogan, and M.E. Schnitzer, Covid-19 Vaccine Effectiveness and the Test-Negative Design. New England Journal of Medicine, 2021. 385(15): p. 1431-1433. [CrossRef]
- Organization, W.H. Evaluation of COVID-19 vaccine effectiveness. 2021; Available from: https://www.who.int/publications/i/item/WHO-2019-nCoV-vaccine_effectiveness-measurement-2021.1.
- Viana, R., et al., Rapid epidemic expansion of the SARS-CoV-2 Omicron variant in southern Africa. Nature, 2022. 603(7902): p. 679-686. [CrossRef]
- Organziation, W.H. WHO COVID-19 Case definition. 2022 [cited 2024; Available from: https://www.who.int/publications/i/item/WHO-2019-nCoV-Surveillance_Case_Definition-2022.1.
- Huang, Z., et al., Effectiveness of inactivated and Ad5-nCoV COVID-19 vaccines against SARS-CoV-2 Omicron BA. 2 variant infection, severe illness, and death. BMC Med, 2022. 20(1): p. 400. [CrossRef]
- Rana, R., et al., Omicron variant: Current insights and future directions. Microbiol Res, 2022. 265: p. 127204. [CrossRef]
- Xu, S., et al., Real-world effectiveness and factors associated with effectiveness of inactivated SARS-CoV-2 vaccines: a systematic review and meta-regression analysis. BMC Med, 2023. 21(1): p. 160. [CrossRef]
- Madhi, S.A., et al., Population Immunity and Covid-19 Severity with Omicron Variant in South Africa. N Engl J Med, 2022. 386(14): p. 1314-1326. [CrossRef]
- Madhi, S.A., et al., Durability of ChAdOx1 nCoV-19 (AZD1222) vaccine and hybrid humoral immunity against variants including omicron BA.1 and BA.4 6 months after vaccination (COV005): a post-hoc analysis of a randomised, phase 1b-2a trial. Lancet Infect Dis, 2023. 23(3): p. 295-306. [CrossRef]
- Davies, M.-A., et al., Outcomes of laboratory-confirmed SARS-CoV-2 infection during resurgence driven by Omicron lineages BA.4 and BA.5 compared with previous waves in the Western Cape Province, South Africa. International Journal of Infectious Diseases, 2023. 127: p. 63-68. [CrossRef]
- Abdullah, F., et al., Decreased severity of disease during the first global omicron variant covid-19 outbreak in a large hospital in tshwane, south africa. Int J Infect Dis, 2022. 116: p. 38-42. [CrossRef]
- Tang, L., et al., Relative vaccine effectiveness against Delta and Omicron COVID-19 after homologous inactivated vaccine boosting: a retrospective cohort study. BMJ Open, 2022. 12(11): p. e063919. [CrossRef]
- Menni, C., et al., COVID-19 vaccine waning and effectiveness and side-effects of boosters: a prospective community study from the ZOE COVID Study. Lancet Infect Dis, 2022. 22(7): p. 1002-1010. [CrossRef]
- Ferdinands, J.M., et al., Waning of vaccine effectiveness against moderate and severe covid-19 among adults in the US from the VISION network: test negative, case-control study. Bmj, 2022. 379: p. e072141. [CrossRef]
- Trust, F. FinScope Consumer Survey Report. 2022 [cited 2024; Available from: https://www.rbz.co.zw/documents/BLSS/2022/Zimbabwe_FinScope_Consumer_2022_Survey_Report.pdf.
- Hamisu, M., et al., Microplanning verification and 2017/2018 measles vaccination campaign in Nigeria: Lessons learnt. Vaccine, 2021. 39 Suppl 3: p. C46-c53. [CrossRef]
- Yeatman, S., S. Chamberlin, and K. Dovel, Women's (health) work: A population-based, cross-sectional study of gender differences in time spent seeking health care in Malawi. PLoS One, 2018. 13(12): p. e0209586. [CrossRef]
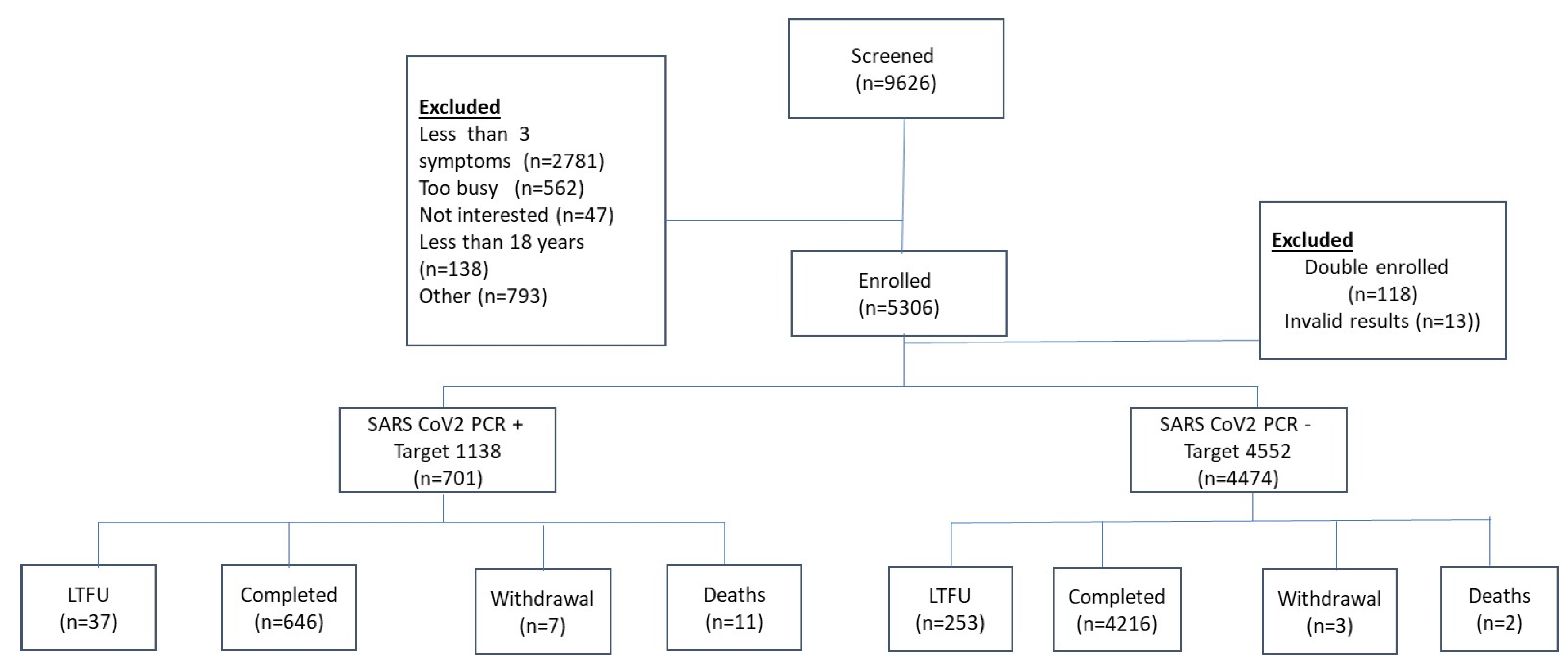
| Variable | Controls | Cases | Total | p-value |
|---|---|---|---|---|
| Age (median, IQR) | 36 (26-44) | 37 (27-48) | 36 (27-45) | 0.001 |
| Age-group | ||||
| 18-49 | 3805/4474 (85.05) | 547/701 (78.03) | 4352/5175 (84.10) | <0.001 |
| 50-64 | 555/4474 (12.41) | 105/701 (14.98) | 660/5175 (12.75) | |
| 65+ | 114/4474 (2.55) | 49/701 (6.99) | 163/5175 (3.15) | |
| Gender | ||||
| Male | 1437/4474 (32.12) | 200/701 (28.53) | 1637/5175 (31.63) | 0.057 |
| Female | 3037/4474 (67.88) | 501/701 (71.47) | 3538/5175 (68.37) | |
| Enrolment site | ||||
| Hospital outpatient or ER | 2955/4474 (66.05) | 498/701 (71.04) | 3453/5175 (66.72) | <0.001 |
| Hospitalized In-patient | 17/4474 (0.38) | 54/701 (7.70) | 71/5175 (1.37) | |
| Community Outpatient | 1502/4474 (33.57) | 149/701 (21.26) | 1651/5175 (31.90) | |
| Socio-economic status | ||||
| Low | 2378/4458 (53.34) | 391/700 (55.86) | 2769/5158 (53.68) | 0.366 |
| Middle | 1972/4458 (44.24) | 290/700 (41.43) | 2262/5158 (43.85) | |
| High | 108/4458 (2.42) | 19/700 (2.71) | 127/5158 (2.46) | |
| Ethnicity | ||||
| Asian, not Indian | 7/4463 (0.16) | 1/699 (0.14) | 8/5162 (0.15) | 0.007 |
| Black | 4444/4463 (99.57) | 689/699 (98.57) | 5133/5162 (99.44) | |
| Indian | 2/4463 (0.04) | 2/699 (0.29) | 4/5162 (0.08) | |
| Mixed | 3/4463 (0.07) | 3/699 (0.43) | 6/5162 (0.12) | |
| White | 5/4463 (0.11) | 3/699 (0.43) | 8/5162 (0.15) | |
| Other | 2/4463 (0.04) | 1/699 (0.14) | 3/5162 (0.06) | |
| Variable | Controls (%) | Cases (%) | Total (%) | p-value |
|---|---|---|---|---|
| BMI (median, IQR) | 25 (22-29) | 26 (23-30) | 25 (22-30) | 0.005 |
| BMI range | ||||
| <18 | 103/4474 (2.30) | 21/701 (3.00) | 124/5175 (2.40) | 0.013 |
| 18-24.9 | 1899/4474 (42.45) | 249/701 (35.52) | 2148/5175 (41.51) | |
| 25.29.9 | 1285/4474 (28.72) | 226/701 (32.24) | 1511/5175 (29.20) | |
| ≥30 | 1094/4474 (24.45) | 191/701 (27.25) | 1285/5175 (24.83) | |
| Missing | 93/4474 (2.08) | 14/701 (2.00) | 107/5175 (2.07) | |
| Medical comorbidities | ||||
| Hypertension | 631/4458 (14.15) | 133/694 (19.16) | 764/5152 (14.83) | 0.001 |
| Diabetes | 128/4457 (2.87) | 26/694 (3.75) | 154/5151 (2.99) | 0.208 |
| Dementia | 5/4457 (0.11) | 1/694 (0.14) | 6/5151 (0.12) | 0.819 |
| Chronic kidney disease | 10/4457 (0.22) | 3/694 (0.43) | 13/5151 (0.25) | 0.309 |
| Asthma | 112/4467 (2.51) | 27/700 (3.86) | 139/5167 (2.69) | 0.039 |
| Tuberculosis | 29/3719 (0.78) | 10/602 (1.66) | 39/4321 (0.9) | 0.034 |
| HIV status | 992/4361 (22.75) | 149/675 (22.07) | 1141/5036 (22.66) | 0.698 |
| On ART | 831/992 (83.8) | 121/149 (81.2) | 952/1141 (83.4) | 0.424 |
| Currently smoking | 287/4424 (6.49) | 25/693 (3.61) | 312/5117 (6.1) | 0.003 |
| Cancer | 20/4458 (0.45) | 7/694 (1.01) | 27/5152 (0.52) | 0.057 |
| Pregnancy | 218/2511 (8.68) | 68/420 (16.19) | 286/2931 (9.76) | <0.001 |
| High risk occupation | 1986/4462 (44.51) | 251/701(35.81) | 2237/5163 (43.33) | <0.001 |
| Previous COVID diagnosis | 1436/4408 (32.58) | 254/691 (36.76) | 1690/5099 (33.14) | 0.03 |
| Variable | Controls | Cases | Total | p-value |
| Vaccinated | 3720/4474 (83.1) | 572/701 (81.6) | 4292/5175 (82.9) | 0.276 |
| Verified | 2879/3618 (79.6) | 418/544 (76.8) | 3297/4162 (79.2) | 0.143 |
| Vaccine Brand | ||||
| Sinopharm | 2128/3720 (57.2) | 337/572 (58.9) | 2465/4292 (57.4) | 0.601 |
| Sinovac | 1483/3720 (39.9) | 212/572 (37.1) | 1695/4292 (39.5) | |
| Covaxin | 16/3720 (0.4) | 3/572 (0.5) | 19/4292 (0.4) | |
| Other* | 16/3720 ( 0.4) | 2/572 (0.3) | 18/4292 (0.4) | |
| Unknown/not sure** | 77/3720 (1.8) | 18/572 (2.8) | 95/4292 (2.2) | |
| Vaccination status*** | ||||
| Unvaccinated | 748/4372 (17.1) | 129/675 (19.1) | 877/5047(17.4) | 0.057 |
| Partial vaccination | 309/4372 (7.1) | 55/675 (8.2) | 364/5047 (7.2) | |
| Fully vaccinated | 1827/4372 (41.8) | 296/675 (43.9) | 2123/5047 (42.1) | |
| Boosted | 1488/4372 (34.0) | 195/675 (28.9) | 1683/5047 (33.4) | |
| Total (%) | Age (18-49 y) | Age (50-64 y) | Age ≥65 years | |
|---|---|---|---|---|
| Partial Vaccination | 364/5047 (7.2) | 338/4244 (8.0) | 23/645 (3.6) | 3/158 (1.9) |
| Full Vaccination | 2123/5047 (42.1) | 1827/4244 (43.1) | 246/645 (38.1) | 50/158 (31.7) |
| Booster Vaccination | 1683/5047 (33.4) | 1279/4244 (30.1) | 322/645 (49.9) | 82/158 (51.9) |
| List of vaccine brands | ||||
| Sinopharm | 2465/4292 (57.4) | 2054/3547 (57.9) | 338/605 (55.9) | 73/140 (52.1) |
| Sinovac | 1695/4292 (39.5) | 1382/3547 (39.0) | 251/605 (41.5) | 62/140 (44.7) |
| Covaxin | 19/4292 (0.4) | 15/3547 (0.4) | 4/605 (0.7) | 0 (0.0) |
| Other* | 18/4292 (0.4%) | 16/3547 (0.5) | 2/605 (0.3) | 0 |
| Unknown/not sure** | 95/4292 (2.2%) | 80/3547 (2.3) | 10/605 (1.7) | 5/140 (3.6) |
| Time since vaccination | ||||
| Days since last Vaccine dose (Median, IQR) | 434 (266-616) | 433 (260-620.5) | 440 (294-602) | 403 (312-543) |
| Days since Booster Vaccine dose (Median, IQR) | 310(205-409) | 300 (193-399) | 335 (244-438) | 368 (307-467) |
| Total10F[1] | Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | p-value | aOR | 95% CI | p-value | |
| Sex (Ref Female) | 1630 | 0.99 | 0.850-1.162 | 0.937 | |||
| BMI (Ref 18-24.9) | |||||||
| <18 | 124 | 0.61 | 0.409-0.907 | 0.015 | 0.50 | 0.312-0.814 | 0.006 |
| 25-29.9 | 1506 | 1.31 | 1.101-1.552 | 0.002 | 1.19 | 0.965-1.455 | 0.112 |
| ≥30 | 1281 | 2.00 | 1.634-2.443 | <0.001 | 1.44 | 1.127-1.835 | 0.004 |
| Clinic Type (Ref outpatient) | |||||||
| Hospital | 3448 | 2.42 | 2.088-2.817 | <0.001 | 1.49 | 1.231-1.791 | <0.001 |
| In-patient | 71 | 0.78 | 0.463-1.297 | 0.333 | 0.58 | 0.316-1.051 | 0.065 |
| Previous COVID diagnosis | 1684 | 1.49 | 1.265-1.756 | <0.001 | 2.85 | 2.051-3.952 | <0.001 |
| HIV Positive | 1136 | 0.89 | 0.745-1.049 | 0.159 | |||
| Active TB | 39 | 0.37 | 0.187-0.717 | 0.003 | 0.44 | 0.211-0.904 | 0.026 |
| Asthma | 139 | 1.59 | 0.942-2.699 | 0.082 | |||
| Hypertension | 762 | 1.96 | 1.532-2.509 | <0.001 | 1.64 | 1.225-2.206 | 0.001 |
| Diabetes Mellitus | 153 | 2.08 | 1.195-3.623 | 0.010 | |||
| PCR positive | 699 | 0.89 | 0.725-1.097 | 0.280 | |||
| High risk occupation | 2234 | 3.83 | 3.20+-4.574 | <0.001 | 2.98 | 2.430-3.643 | <0.001 |
| Age group | |||||||
| 50-64 years | 658 | 2.53 | 1.895-3.377 | <0.001 | |||
| 65+ years | 163 | 1.38 | 0.881-2.158 | 0.159 | |||
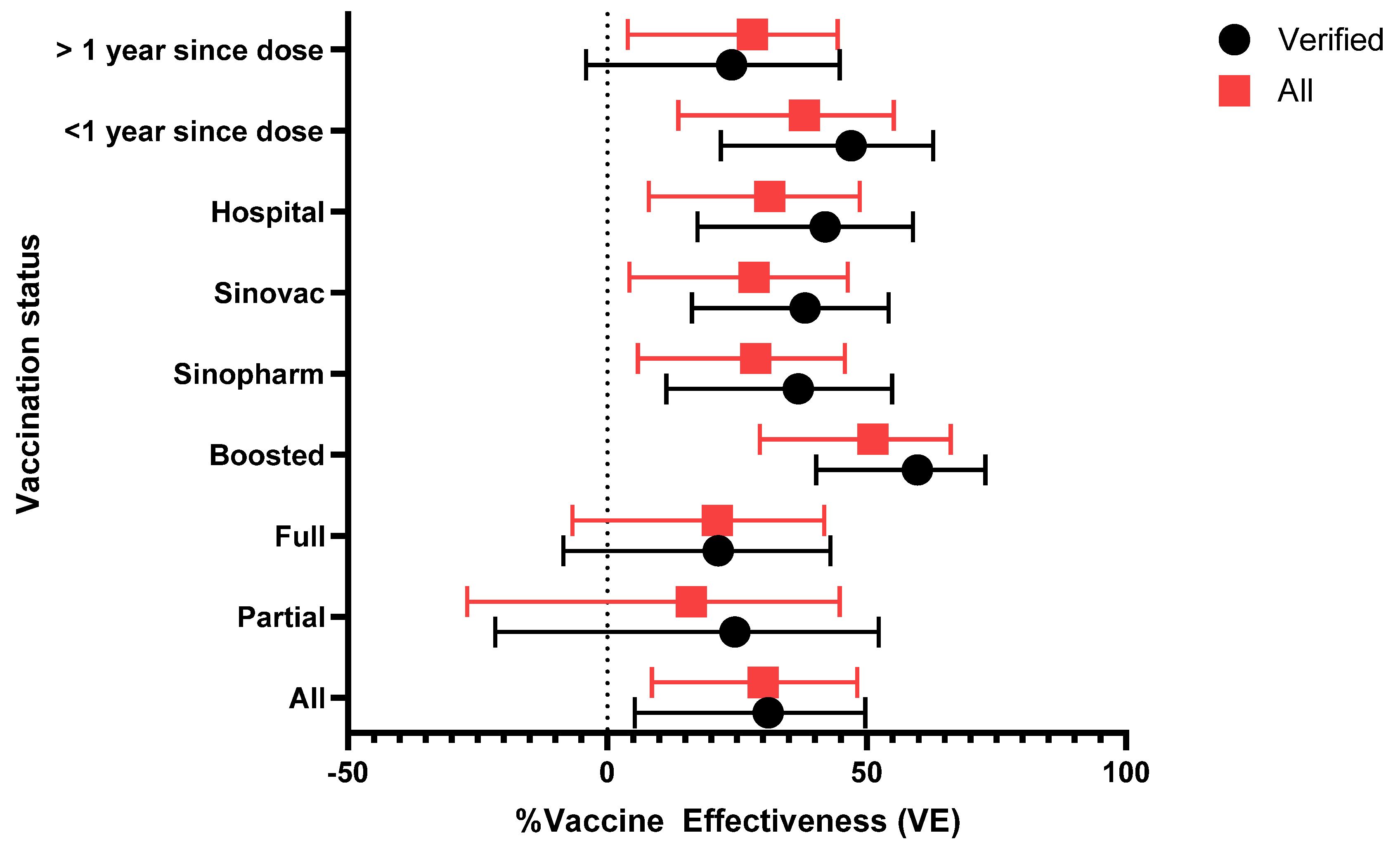
| Verified participants | All participants | |||||
|---|---|---|---|---|---|---|
| Subgroup | Total (N) | Case (N, %) | Adjusted VE, % (95% CI) |
Total (N) | Case (N, %) | Adjusted VE, % (95% CI) |
| Overall | 4184 | 547 (13.1) | 31 (5.3%, 49.7%) | 5175 | 701 (13.5) | 30 (8.6%, 48.2%) |
| Hospital enrolled | 2848 | 429 (15.1) | 42 (17.4%, 58.9%) | 3524 | 552 (15.7) | 31.3 (8.0%, 48.7%) |
| Vaccine Brand | ||||||
| Sinopharm | 1877 | 240 (12.8) | 36.8 (11.4%, 54.9%) | 2469 | 338 (13.7) | 28.6 (5.9%, 45.8%) |
| Sinovac | 1392 | 175 (12.6) | 38.1 (16.3%, 54.2%) | 1702 | 213 (12.5) | 28.3 (4.3%, 46.3%) |
| Vaccination status | ||||||
| Partially | 220 | 28 (12.7) | 24.6 (-21.6%, 52.3%) | 364 | 55 (15.1) | 16.2 (-27%, 44.8%) |
| Fully | 1627 | 225 (13.8) | 21.4 (-8.5%, 43%) | 2123 | 296 (13.9) | 21.2 (-6.7%, 41.8%) |
| Boosted | 1446 | 165 (11.4) | 59.8 (40.3%, 72.9%) | 1683 | 195 (11.6) | 51.2 (29.4%, 66.2%) |
| Time since vaccination | ||||||
| < 1 year | 1346 | 148 (11.0) | 47 (21.9%, 62.8%) | 1718 | 208 (12.1) | 38 (13.7%, 55.2%) |
| >1 year | 1947 | 270 (13.9) | 24 (-4.1%, 44.8%) | 2452 | 338 (13.8) | 28 (4%, 44.4%) |
| 1 | Total represents that total number with reference indication included in the model. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




