Submitted:
17 September 2024
Posted:
18 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
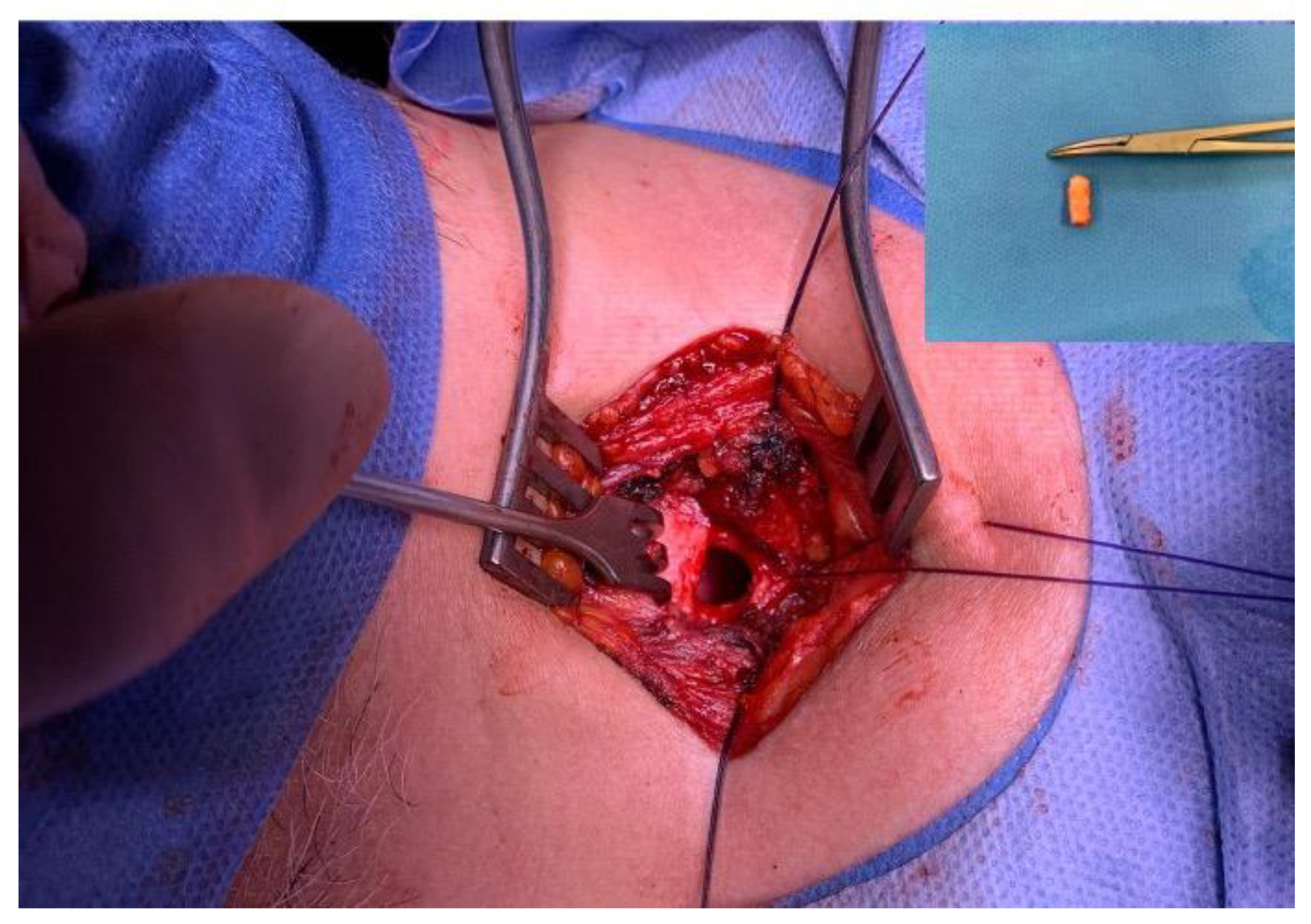
2. Materials and Methods
3. Results
3.1. Characteristics and Differences between Patients with and without SARS-CoV-2 Infection
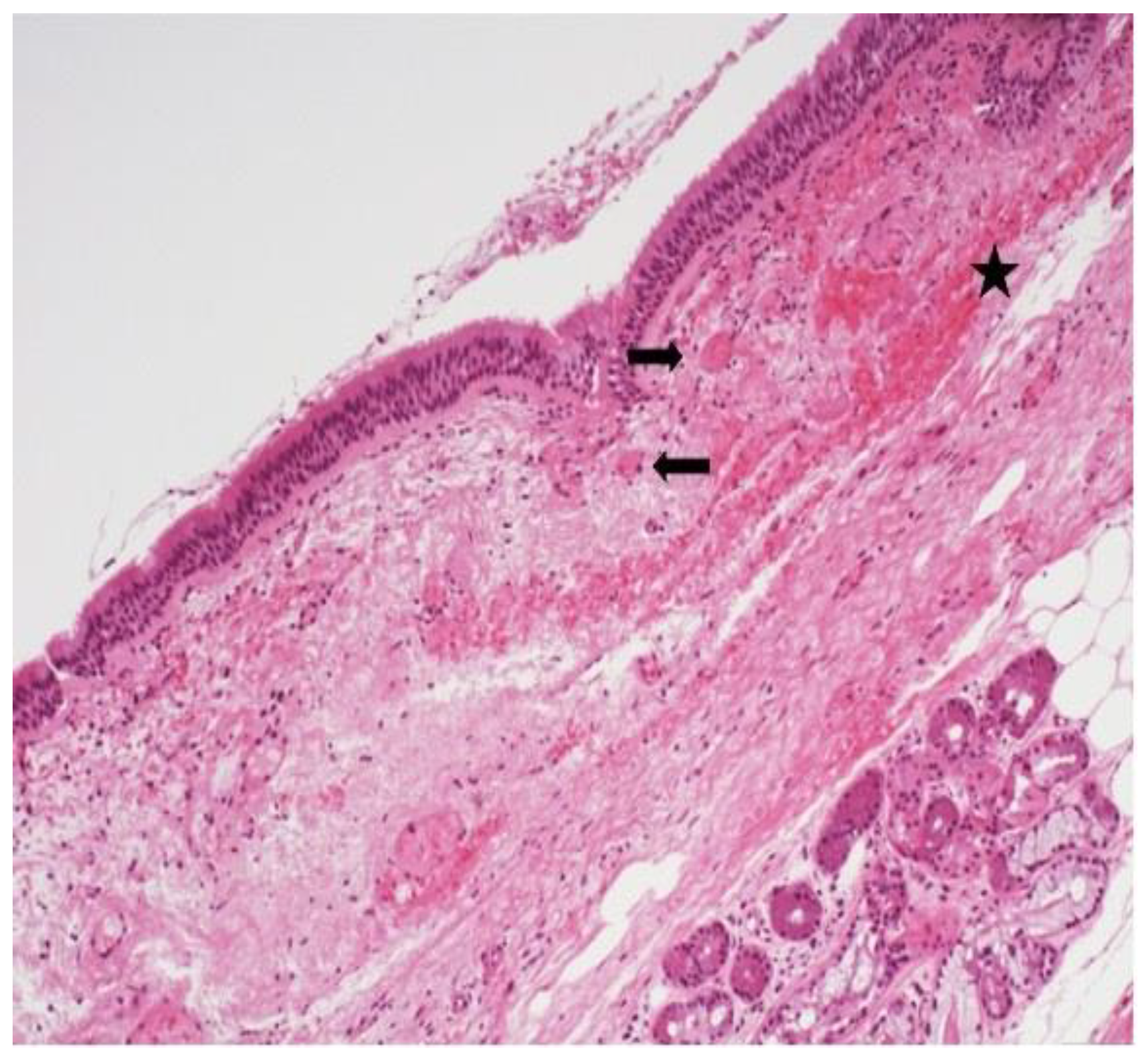
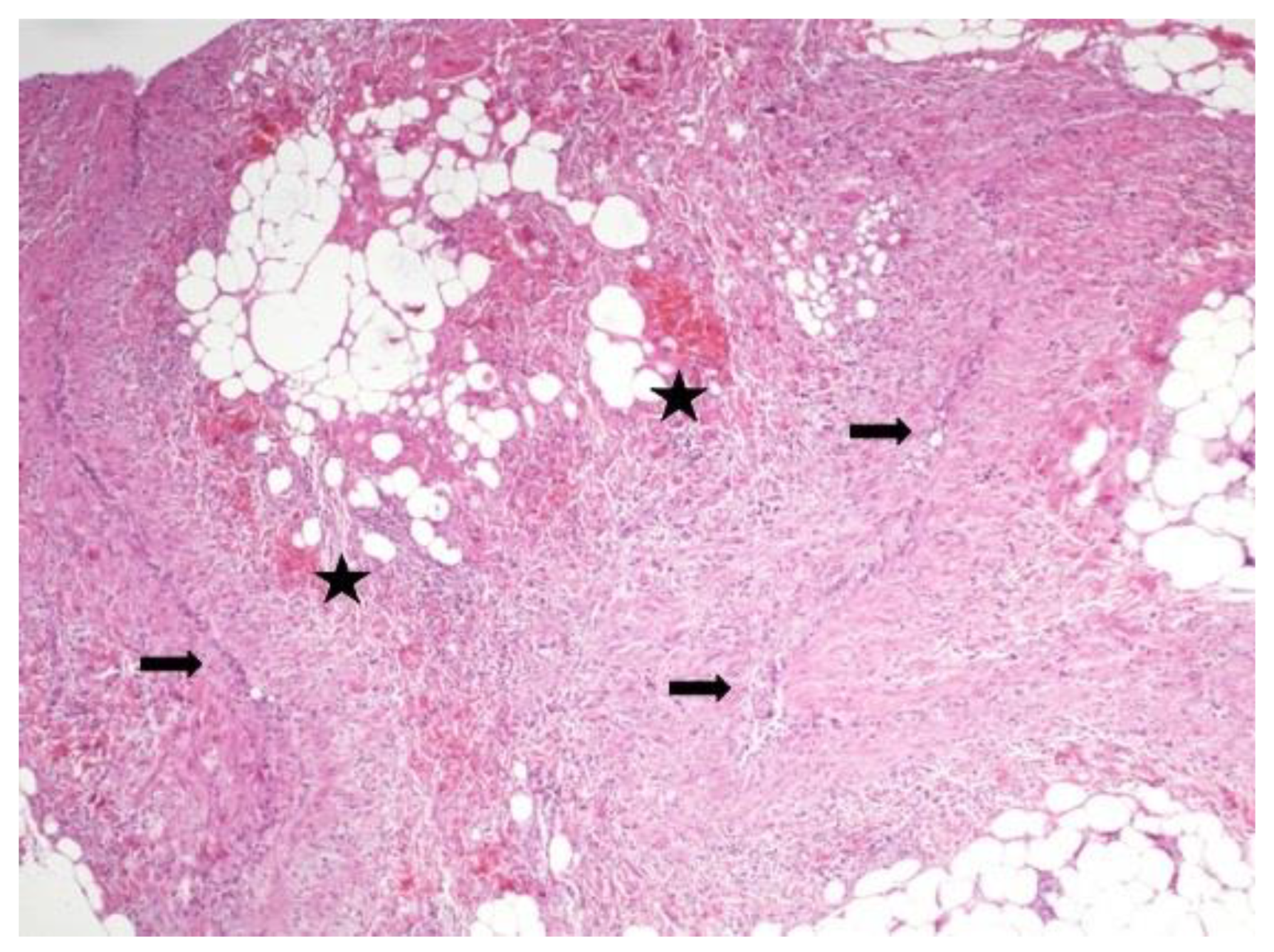
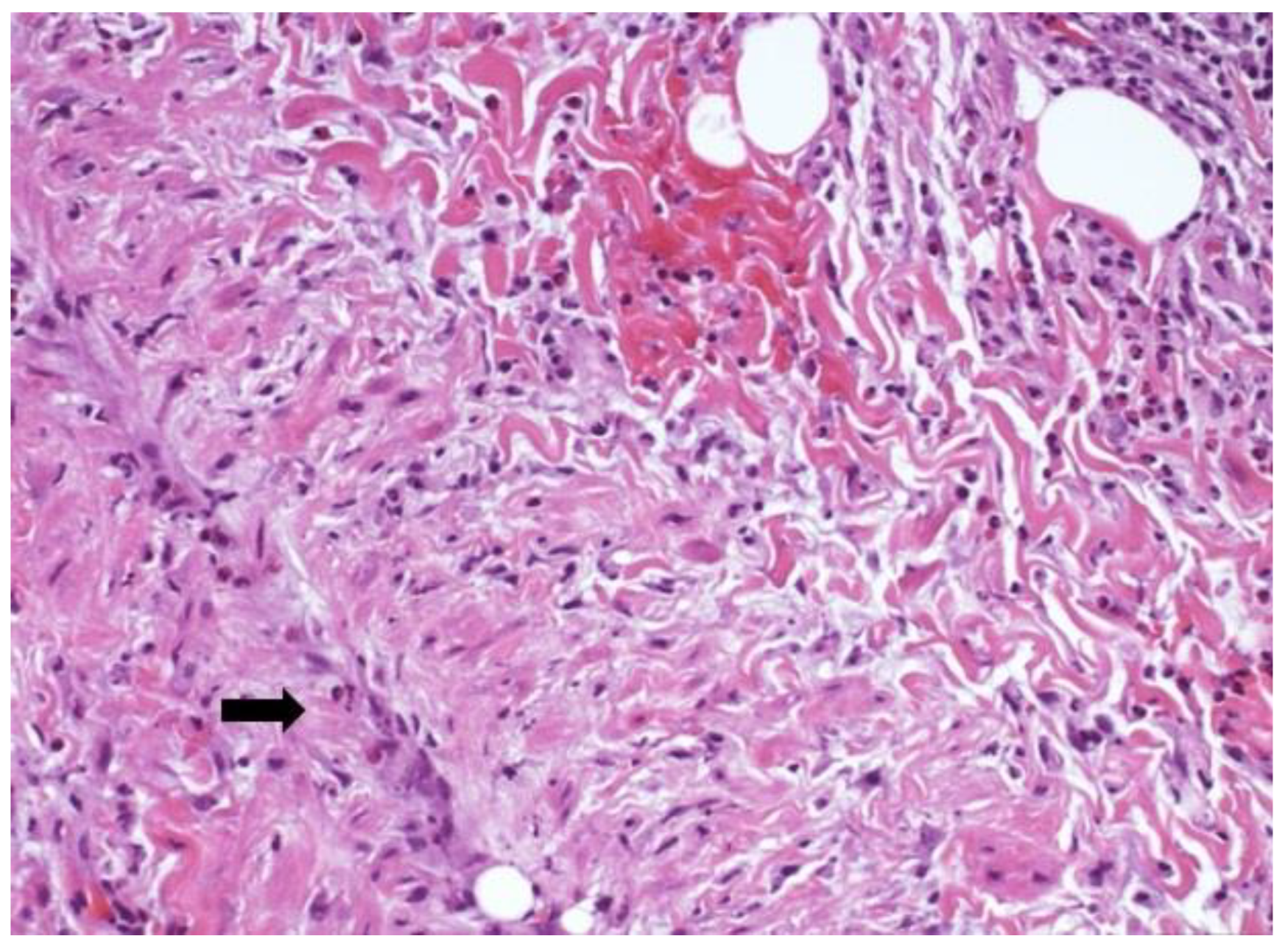
3.2. Histological Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Menter, T.; Haslbauer, J.D.; Nienhold, R.; Savic, S.; Hopfer, H.; Deigendesch, N.; et al. Postmortem examination of COVID-19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings in lungs and other organs suggesting vascular dysfunction. Histopathology 2020, 77, 198–209. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bradley, B.T.; Maioli, H.; Johnston, R.; Chaudhry, I.; Fink, S.L.; Xu, H.; et al. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet 2020, 396, 320–332, Erratum in: Lancet. 2020 Aug 1;396(10247):312. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vasquez-Bonilla, W.O.; Orozco, R.; Argueta, V.; Sierra, M.; Zambrano, L.I.; Muñoz-Lara, F.; et al. A review of the main histopathological findings in coronavirus disease 2019. Hum Pathol. 2020, 105, 74–83. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Polak, S.B.; Van Gool, I.C.; Cohen, D.; von der Thüsen, J.H.; van Paassen, J. A systematic review of pathological findings in COVID-19: a pathophysiological timeline and possible mechanisms of disease progression. Mod Pathol. 2020, 33, 2128–2138. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N Engl J Med. 2020, 383, 120–128. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Deshmukh, V.; Motwani, R.; Kumar, A.; Kumari, C.; Raza, K. Histopathological observations in COVID-19: a systematic review. J Clin Pathol. 2020, Epub ahead of print. jclinpath-2020-206995. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, I.M.; Padera, R.F.; Solomon, I.H.; Kanjilal, S.; Hammer, M.M.; Hornick, J.L.; et al. In situ detection of SARS-CoV-2 in lungs and airways of patients with COVID-19. 2020 Nov;33(11):2104-2114. Mod Pathol. 2020, 33, 2104–2114. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Borczuk, A.C.; Salvatore, S.P.; Seshan, S.V.; Patel, S.S.; Bussel, J.B.; Mostyka, M.; et al. COVID-19 pulmonary pathology: a multi-institutional autopsy cohort from Italy and New York City. Mod Pathol. 2020, 33, 2156–2168. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; https://www.R-project.
- Mata-Castro, N.; Sanz López, L.; Pinacho-Martínez, P.; Varillas-Delgado, V.; Miró-Murillo, M.; Martín-Delgado, M.C. Tracheostomy in patients with SARS-CoV-2 reduces time on mechanical ventilation but not intensive care unit stay. Am J Otolaryngol. 2021, 42, 102867. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Oliver, C.M.; Campbell, M.; Dulan, O.; Hamilton, N.; Birchall, M. Appearance and management of COVID-19 laryngo-tracheitis: two case reports. F1000Res. 2020, 9, 310. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lucchi, M.; Ambrogi, M.; Aprile, V.; Ribechini, A.; Fontanini, G. Laryngotracheal resection for a post-tracheotomy stenosis in a patient with coronavirus disease 2019 (COVID-19). JTCVS Tech. 2020, 4, 360–364. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bassi, M.; Anile, M.; Pecoraro, Y.; Ruberto, F.; Martelli, S.; Piazzolla, M.; et al. Bedside transcervical transtracheal post-intubation injury repair in a Covid-19 patient. Ann Thorac Surg.
- Roncati, L.; Bergonzini, G.; Lusenti, B.; Nasillo, V.; Paolini, A.; Zanelli, G.; et al. High density of IgG4-secreting plasma cells in the fibrotic tissue from a surgically resected tracheal ring impaired by complex subglottic stenosis post-tracheostomy as immune expression of a Th2 response due to severe COVID-19. Ann Hematol. 2021, 100, 2659–2660. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ward, P.A.; Collier, J.M.; Weir, J.; Osborn, M.; Osborn, B.; Smellie, W.J.B. Chelwest COVID-19 Tracheostomy Group. Histological findings of tracheal samples from COVID-19 positive critically ill mechanically ventilated patients. Clin Otolaryngol. 2022, 47, 131–137. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
|
Overall, N = 801 |
SARS-COV-2 negative, N = 401 |
SARS-COV-2 positive, N = 401 |
p-value | Effect size | |
| SEX | 0.52 | 0.055 | |||
| Male | 57 (71.3%) | 27 (67.5%) | 30 (75.0%) | ||
| Female | 23 (28.8%) | 13 (32.5%) | 10 (25.0%) | ||
| AGE | 0.33 | 0.116 | |||
| Mean (SD) | 67.4 (8.3) | 67.8 (9.6) | 67.1 (6.9) | ||
| Median [25%–75%] | 69.0 [63.0–73.0] | 71.0 [59.8–75.3] | 68.0 [63.0–70.5] | ||
| Days until tracheostomy | 0.0913 | 0.190 | |||
| Mean (SD) | 17.2 (4.7) | 18.5 (4.2) | 16.0 (4.9) | ||
| Median [25%–75%] | 17.0 [15.0–20.0] | 18.0 [15.8–20.3] | 16.0 [12.5–19.0] | ||
| PEEP at intubation | <0.0013 | 0.671 | |||
| Mean (SD) | 10.4 (2.6) | 8.8 (1.8) | 12.2 (2.1) | ||
| Median [25%–75%] | 10.0 [8.0–12.0] | 8.0 [7.0–10.3] | 12.0 [10.0–14.0] | ||
| PEEP at tracheostomy | 0.53 | 0.084 | |||
| Mean (SD) | 9.6 (2.1) | 9.4 (2.0) | 9.8 (2.2) | ||
| Median [25%–75%] | 10.0 [8.0–11.0] | 9.5 [8.0–11.0] | 10.0 [8.0–12.0] | ||
| PAFI at intubation | 0.0413 | 0.318 | |||
| Mean (SD) | 156.1 (86.7) | 226.0 (97.9) | 146.6 (82.0) | ||
| Median [25%–75%] | 136.0 [100.0–183.3] | 180.0 [180.0–200.0] | 120.0 [100.0–181.0] | ||
| PAFI at tracheostomy | |||||
| Mean (SD) | 187.5 (47.5) | 187.5 (49.3) | 187.5 (46.1) | 0.93 | 0.019 |
| Median [25%–75%] | 190.0 [157.3–209.3] | 190.0 [160.0–200.0] | 190.0 [150.0–213.0] | ||
| COMORBITIES | <0.0013 | 0.501 | |||
| Mean (SD) | 6.0 (5.0) | 8.3 (4.4) | 3.6 (4.5) | ||
| Median [25%–75%] | 7.0 [0.0–11.0] | 10.0 [2.5–11.0] | 1.0 [0.0–8.5] | ||
| Unknown | 3 | 1 | 2 | ||
| HISTOLOGICAL FINDINGS | |||||
| normal | 65 (82.3%) | 35 (87.5%) | 30 (76.9%) | <0.0012 | 0.427 |
| subepitelial chronic inflammation moderate | 6 (7.6%) | 0 (0.0%) | 6 (15.4%) | ||
| low subepitelial chronic inflammation | 5 (6.3%) | 5 (12.5%) | 0 (0.0%) | ||
| subepitelial chronic inflammation + vasculitis |
2 (2.5%) | 0 (0.0%) | 2 (5.1%) | ||
| subepitelial chronic inflammation + microangiopathy trombotic |
1 (1.3%) | 0 (0.0%) | 1 (2.6%) | ||
| Without histological alterations | Chronic subepithelial inflammation | Vasculitis | Thrombotic microangiopathy | Total | |
|---|---|---|---|---|---|
| Negative SARS-CoV-2 group | 35 (53.0%) | 5 (45.5%) | 0 (0.0%) | 0 (0.0%) | 40 (100%) |
| Positive SARS-CoV-2 group | 31 (47.0%) | 6 (54.5%) | 1 (100.0%) | 2 (100.0%) | 40 (100%) |
| TOTAL | 66 (82.5 %) | 11 (13.75%) | 1 (1.25%) | 2 (2.5%) | 80 (100%) |
| Relevant findings | No relevant findings | Total | Prevalence x100 | Odds | |
|---|---|---|---|---|---|
| Negative SARS-CoV-2 group | 5 | 35 | 40 | 12.5 | 0.143 |
| Positive SARS-CoV-2 group | 9 | 31 | 40 | 22.5 | 0.290 |
| TOTAL | 14 | 66 | 80 | 17.5 | 0.212 |
| Overall, N = 791 | No relevant findings, N = 651 | Relevant findings, N = 141 | p-value | Effect size | |
| SAR-Cov-2 +1/-0 | 0.22 | 0.105 | |||
| Negative | 40 (50.6%) | 35 (53.8%) | 5 (35.7%) | ||
| Positive | 39 (49.4%) | 30 (46.2%) | 9 (64.3%) | ||
| SEX | >0.93 | 0.000 | |||
| Male | 56 (70.9%) | 46 (70.8%) | 10 (71.4%) | ||
| Female | 23 (29.1%) | 19 (29.2%) | 4 (28.6%) | ||
| AGE | 0.84 | 0.027 | |||
| Mean (SD) | 67.4 (8.4) | 67.6 (7.9) | 66.8 (10.7) | ||
| Median [25%–75%] | 69.0 [63.0–73.0] | 69.0 [63.0–72.0] | 70.5 [57.8–75.0] | ||
| Day until tracheostomy | >0.94 | 0.001 | |||
| Mean (SD) | 17.3 (4.7) | 17.2 (4.3) | 17.9 (6.4) | ||
| Median [25%–75%] | 17.0 [15.0–20.0] | 17.0 [15.0–20.0] | 18.0 [15.0–19.0] | ||
| PEEP at intubation | >0.94 | 0.002 | |||
| Mean (SD) | 10.4 (2.6) | 10.4 (2.7) | 10.4 (1.7) | ||
| Median [25%–75%] | 10.0 [8.0–12.0] | 11.0 [8.0–12.0] | 10.0 [9.0–12.0] | ||
| Unknown | 3 | 2 | 1 | ||
| PEEP at tracheostomy | 0.94 | 0.018 | |||
| Mean (SD) | 9.5 (2.1) | 9.5 (2.2) | 9.6 (1.8) | ||
| Median [25%–75%] | 10.0 [8.0–11.0] | 10.0 [8.0–11.0] | 10.0 [8.3–11.0] | ||
| PAFI at intubation | >0.94 | 0.003 | |||
| Mean (SD) | 155.1 (87.6) | 157.6 (92.6) | 144.9 (66.7) | ||
| Median [25%–75%] | 132.0 [100.0–181.0] | 132.0 [100.0–180.0] | 130.0 [100.0–202.0] | ||
| Unknown | 38 | 32 | 6 | ||
| PAFI at tracheosotomy | 0.0484 | 0.224 | |||
| Mean (SD) | 187.2 (47.7) | 193.0 (45.5) | 160.2 (50.2) | ||
| Median [25%–75%] | 190.0 [156.5–206.5] | 190.0 [160.0–216.0] | 178.0 [144.8–189.5] | ||
| COMORBITIES | 0.74 | 0.042 | |||
| Mean (SD) | 5.9 (5.1) | 5.9 (5.1) | 6.1 (5.3) | ||
| Median [25%–75%] | 5.5 [0.0–11.0] | 6.5 [0.0–11.0] | 5.5 [0.5–11.0] | ||
| Unknown | 3 | 3 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




