Submitted:
05 September 2024
Posted:
06 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
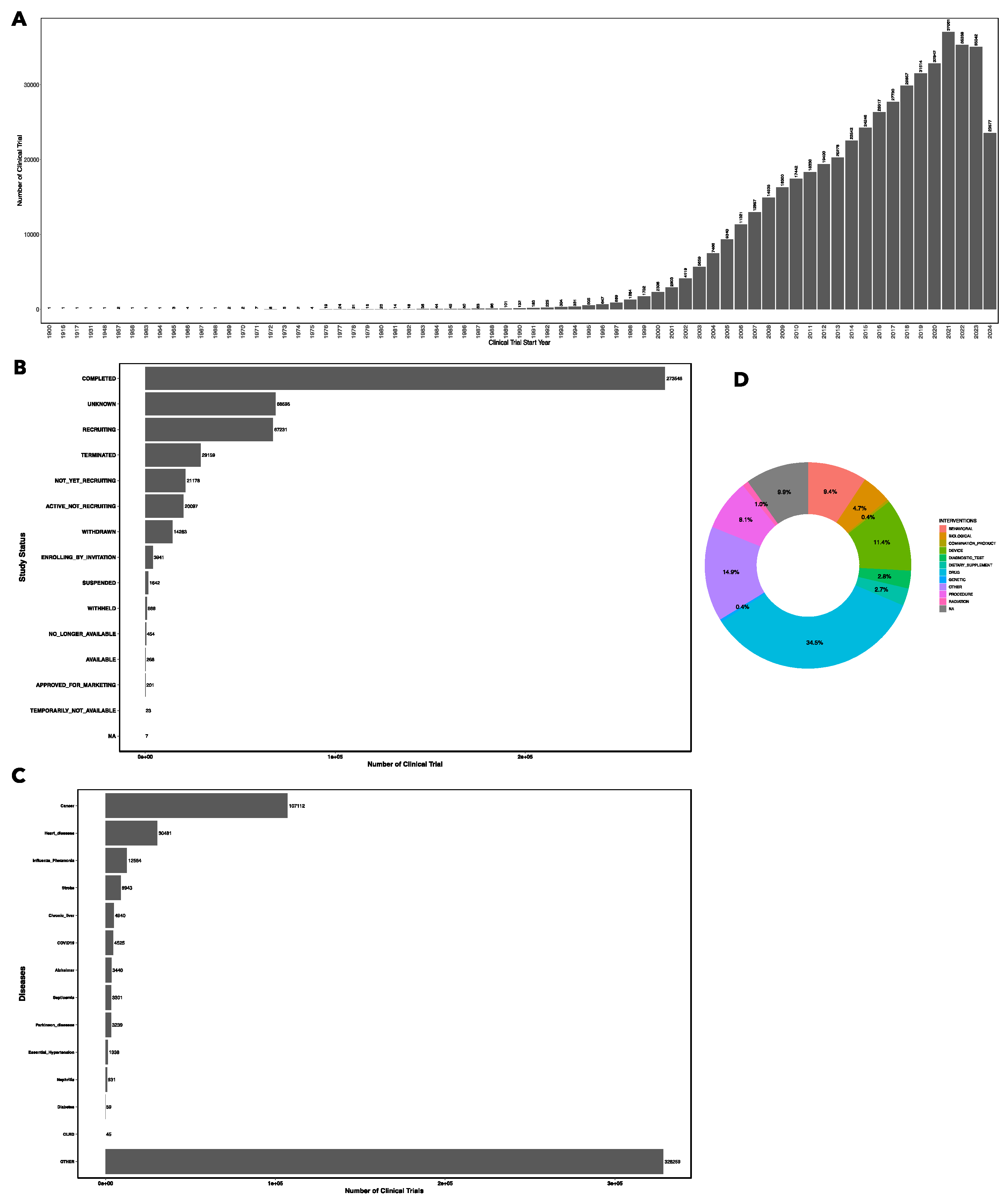
3.1. Overall Trends
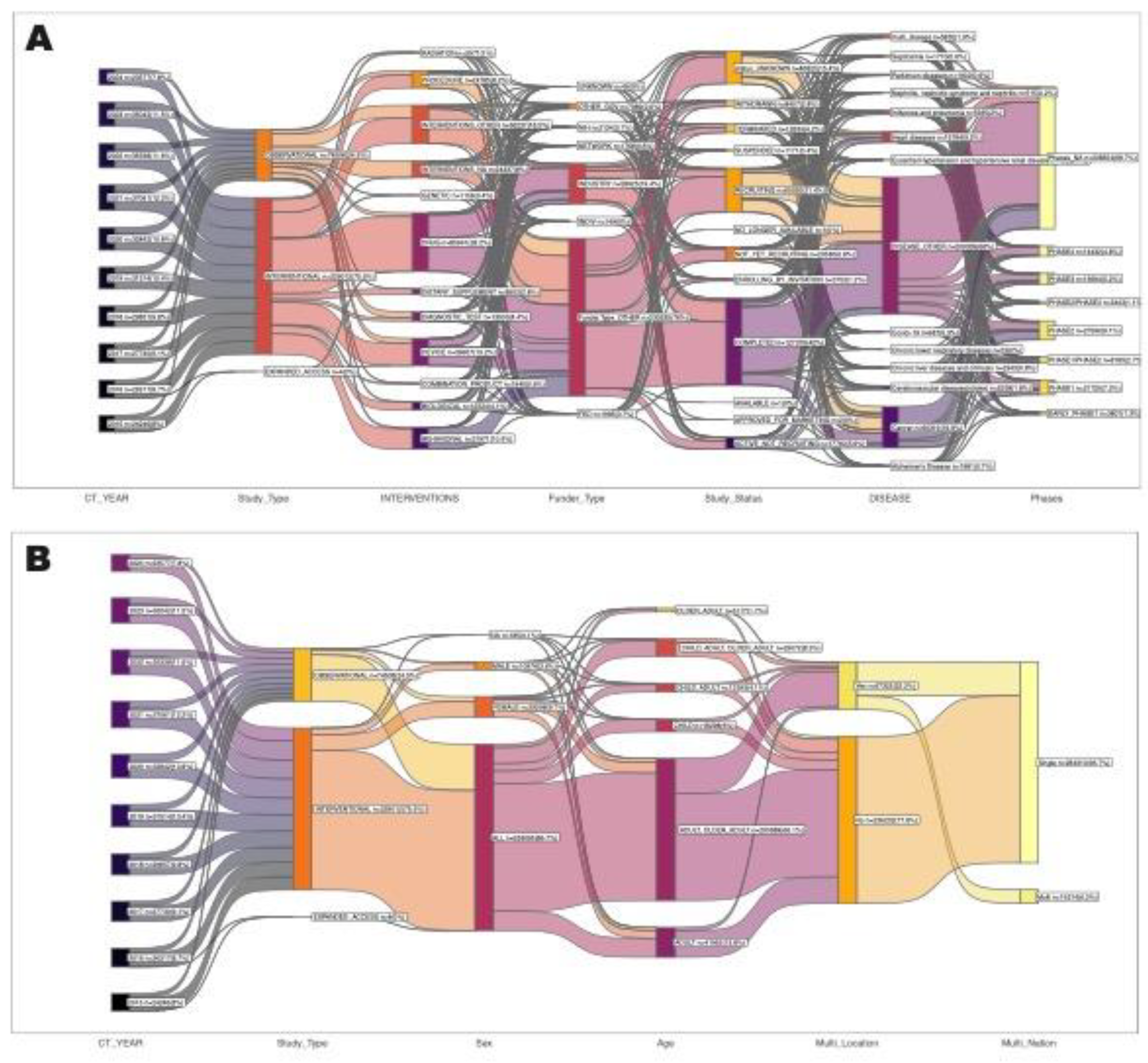
3.2. Study Status of Clinical Trials
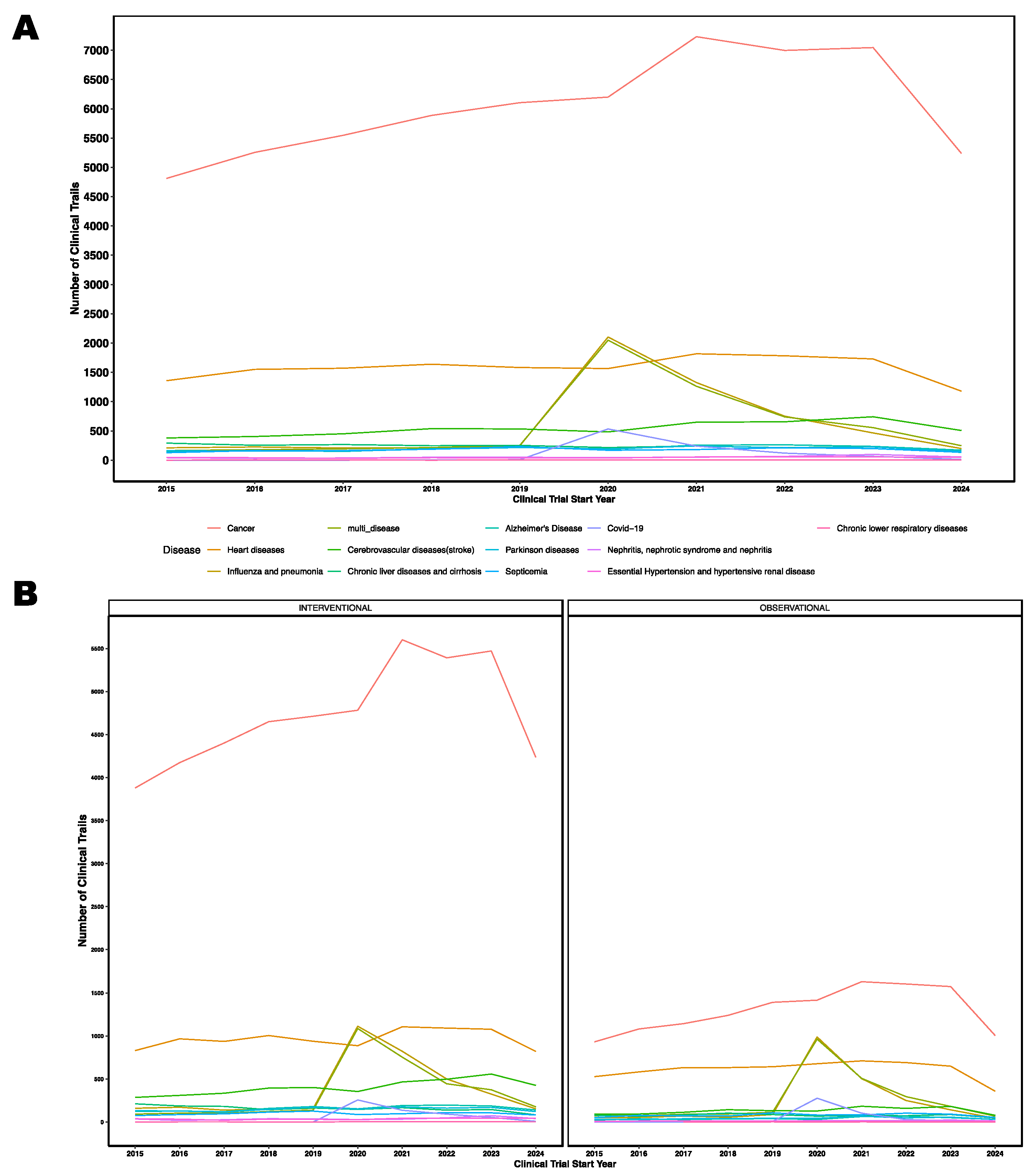
3.3. Clinical Trials by Disease/Condition
3.4. Types of Intervention/Treatment
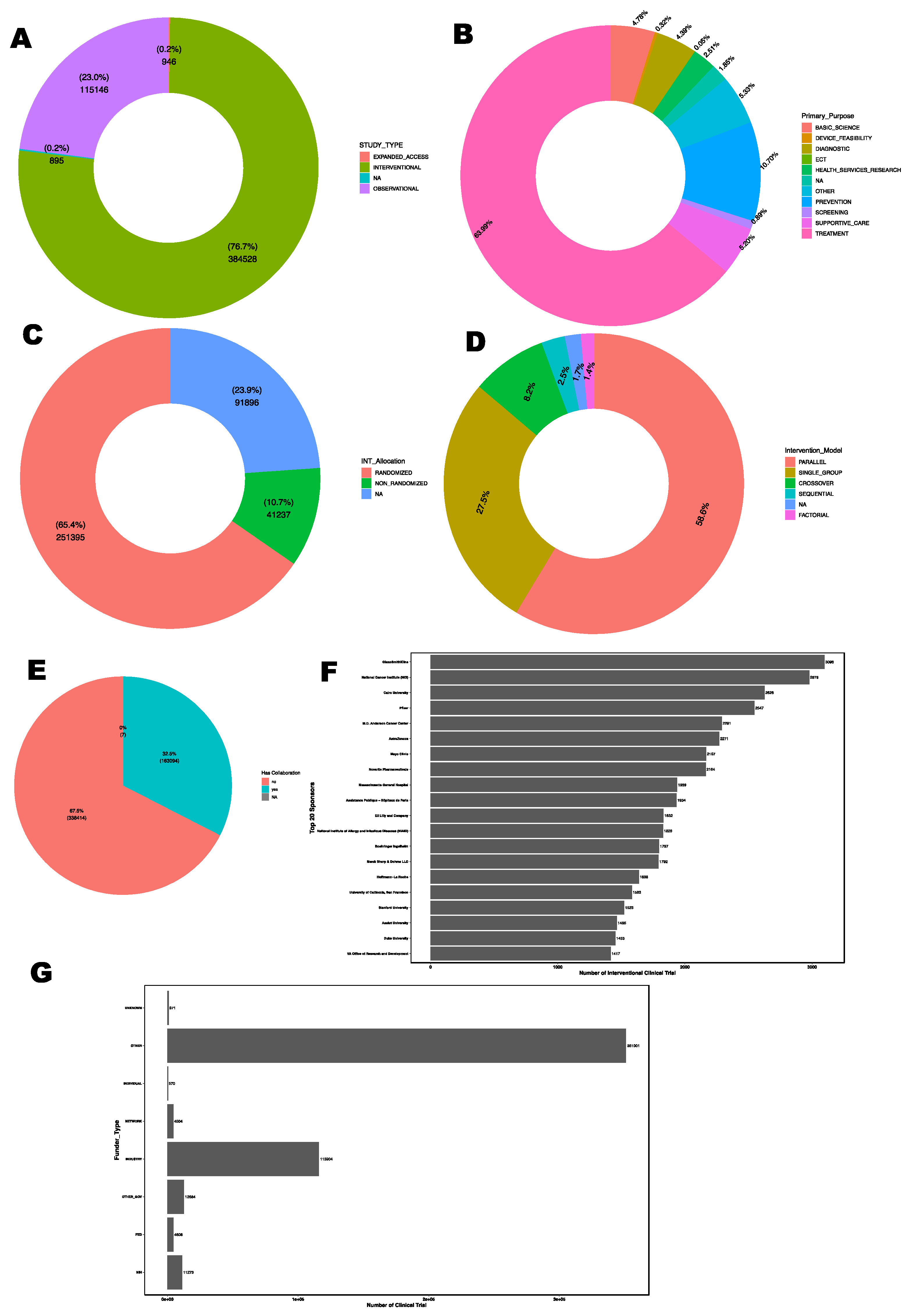
3.5. Nature of Clinical Trials
3.6. Purpose of Interventional Trials
3.7. Randomization and Interventional Model
3.8. Collaborations, Sponsorship and Funder Type
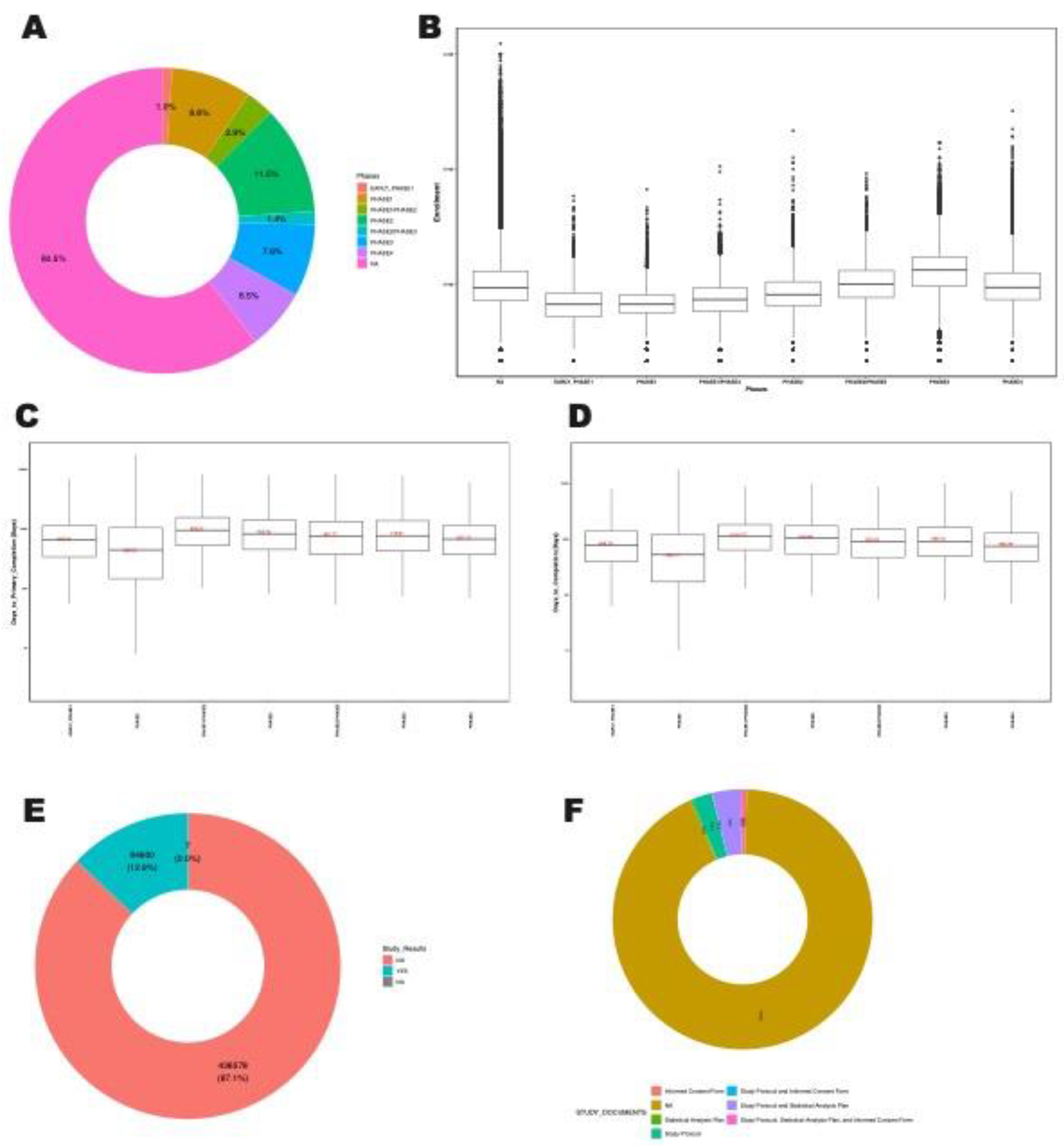
3.9. Trial Phases and Enrollments
3.10. Clinical Trial Duration, Reporting and Documentation
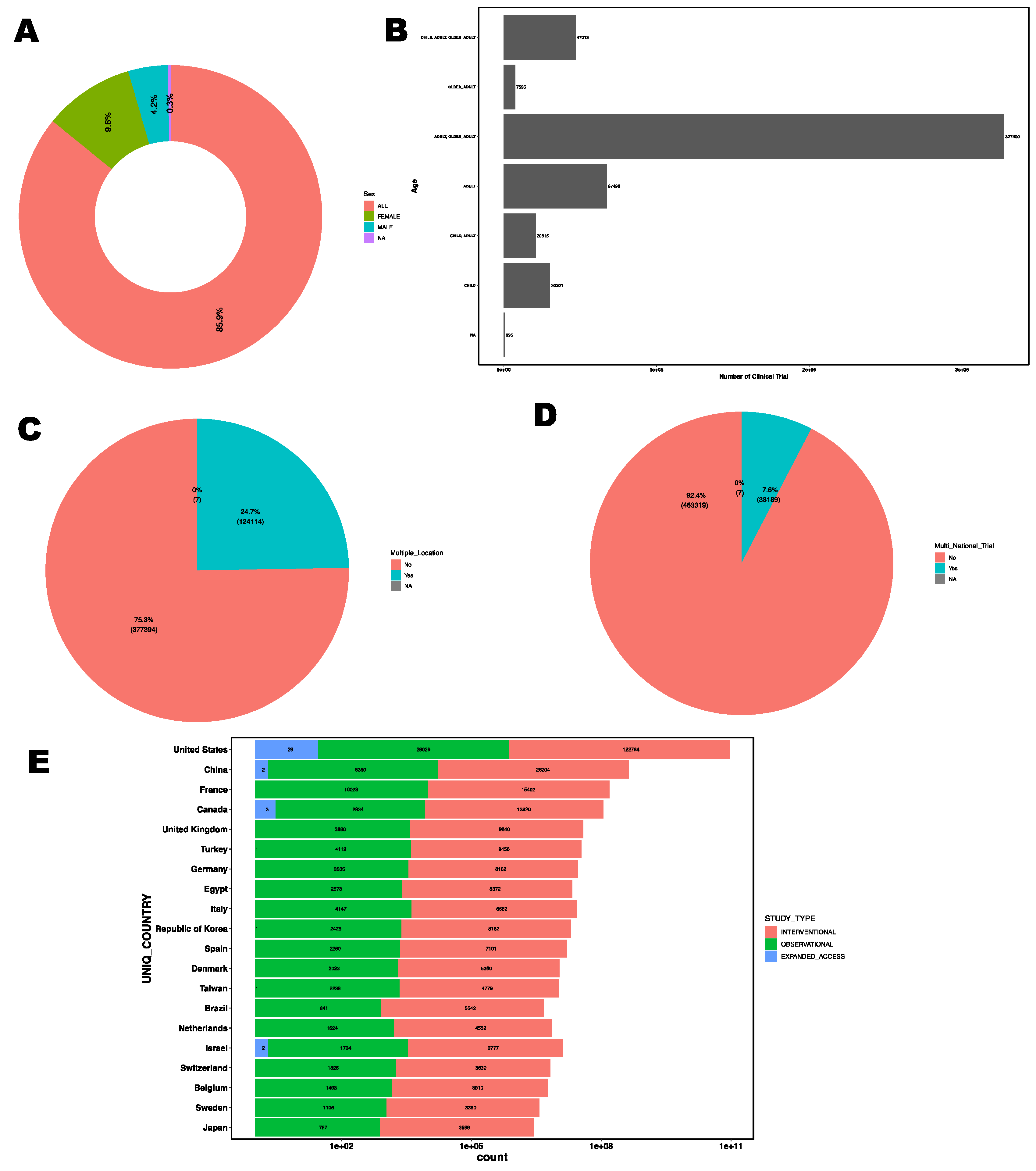
3.11. Participant Demographics and Geographical Distribution
3.12 Trends in Last Decade
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Learn About Studies | ClinicalTrials.gov. https://clinicaltrials.gov/study-basics/learn-about-studies#q1.
- Schultz, A., Saville, B. R., Marsh, J. A. & Snelling, T. L. An introduction to clinical trial design. Paediatric Respiratory Reviews 32, 30–35 (2019). [CrossRef]
- Introduction and History of Clinical Trial Research. in Introduction to Adaptive Trial Designs and Master Protocols (eds. Mills, E. J., Wathen, J. K. & Park, J. J. H.) 1–20 (Cambridge University Press, Cambridge, 2023).
- Ballman, K. V. Introduction to Clinical Trials, Clinical Trial Designs, and Statistical Terminology Used for Predictive Biomarker Research and Validation. in Predictive Biomarkers in Oncology: Applications in Precision Medicine (eds. Badve, S. & Kumar, G. L.) 19–36 (Springer International Publishing, Cham, 2019). [CrossRef]
- What Are Clinical Trials? - NCI. https://www.cancer.gov/research/participate/clinical-trials/what-are-clinical-trials (2023).
- Adesoye, T., Katz, M. H. G. & Offodile, A. C. Meeting Trial Participants Where They Are: Decentralized Clinical Trials as a Patient-Centered Paradigm for Enhancing Accrual and Diversity in Surgical and Multidisciplinary Trials in Oncology. JCO Oncol Pract 19, 317–321 (2023). [CrossRef]
- Tong, A. et al. Patient-centred clinical trial design. Nat Rev Nephrol 18, 514–523 (2022). [CrossRef]
- Ghadessi, M. et al. Decentralized clinical trials and rare diseases: a Drug Information Association Innovative Design Scientific Working Group (DIA-IDSWG) perspective. Orphanet Journal of Rare Diseases 18, 79 (2023). [CrossRef]
- Jacobson, R. M., Pignolo, R. J. & Lazaridis, K. N. Clinical Trials for Special Populations: Children, Older Adults, and Rare Diseases. Mayo Clinic Proceedings 99, 318–335 (2024). [CrossRef]
- Examination of Clinical Trial Costs and Barriers for Drug Development. ASPE https://aspe.hhs.gov/reports/examination-clinical-trial-costs-barriers-drug-development-0 (2014).
- Mohan, S. V. & Freedman, J. A Review of the Evolving Landscape of Inclusive Research and Improved Clinical Trial Access. Clinical Pharmacology & Therapeutics 113, 518–527 (2023). [CrossRef]
- Home | ClinicalTrials.gov. https://clinicaltrials.gov/.
- CDC WONDER. https://wonder.cdc.gov/.
- R: The R Project for Statistical Computing. https://www.r-project.org/.
- Mullard, A. Shifts in the clinical trial landscape. Nature Reviews Drug Discovery 23, 239–239 (2024). [CrossRef]
- Briel, M. et al. Exploring reasons for recruitment failure in clinical trials: a qualitative study with clinical trial stakeholders in Switzerland, Germany, and Canada. Trials 22, 844 (2021). [CrossRef]
- Hilgers, R.-D., FranzKönig & Senn, G. M. and S. Design and analysis of clinical trials for small rare disease populations. Journal of Rare Diseases Research & Treatment 1, (2016).
- May, M. Rare-disease researchers pioneer a unique approach to clinical trials. Nature Medicine 29, 1884–1886 (2023). [CrossRef]
- Sessler, D. I., Alman, B. A., Treggiari, M. M. & Mont, M. A. Pro-Con Debate: Interdisciplinary Perspectives on Industry-Sponsored Research. The Journal of Arthroplasty 38, 986–991 (2023). [CrossRef]
- Shyr, C., Sulieman, L. & Harris, P. A. Illuminating the landscape of high-level clinical trial opportunities in the All of Us Research Program. Journal of the American Medical Informatics Association ocae062 (2024). [CrossRef]
- Yin, S. et al. Disparities in COVID-19 clinical studies from high-income and low-and middle-income countries. International Journal of Infectious Diseases 132, 9–16 (2023). [CrossRef]
- Ramanan, M., Tong, S. Y. C., Kumar, A. & Venkatesh, B. Geographical Representation of Low- and Middle-Income Countries in Randomized Clinical Trials for COVID-19. JAMA Network Open 5, e220444 (2022). [CrossRef]
- Payedimarri, A. B., Mouhssine, S., Aljadeeah, S., Gaidano, G. & Ravinetto, R. Globalisation of industry-sponsored clinical trials for breast, lung and colon cancer research: trends, threats and opportunities. bmjonc 2, (2023). [CrossRef]
- Sheng, J., Feldhake, E., Zarin, D. A. & Kimmelman, J. Completeness of clinical evidence citation in trial protocols: A cross-sectional analysis. Med 3, 335-343.e6 (2022). [CrossRef]
- Subbiah, V. The next generation of evidence-based medicine. Nat Med 29, 49–58 (2023). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



