Submitted:
02 September 2024
Posted:
04 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Statistical Methods
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moran A, Pekow P, Grover P, Zorn M, Slovis B, Pilewski J, Tullis et al. Cystic Fibrosis Related Diabetes Therapy Study Group. Insulin therapy to improve BMI in cystic fibrosis-related diabetes without fasting hyperglycemia: results of the cystic fibrosis related diabetes therapy trial. Diabetes Care. 1: 2009 Oct;32(10), 2009.
- Granados, A.; Chan, C.L.; Ode, K.L.; Moheet, A.; Moran, A.; Holl, R. Cystic fibrosis related diabetes: Pathophysiology, screening and diagnosis. J. Cyst. Fibros. 2019, 18, S3–S9. [Google Scholar] [CrossRef]
- Gottlieb, P.A.; Yu, L.; Babu, S.; Wenzlau, J.; Bellin, M.; Frohnert, B.I.; Moran, A. No Relation Between Cystic Fibrosis–Related Diabetes and Type 1 Diabetes Autoimmunity. Diabetes Care 2012, 35, e57–e57. [Google Scholar] [CrossRef] [PubMed]
- Ntimbane, T.; Mailhot, G.; Spahis, S.; Rabasa-Lhoret, R.; Kleme, M.-L.; Melloul, D.; Brochiero, E.; Berthiaume, Y.; Levy, E. CFTR silencing in pancreatic β-cells reveals a functional impact on glucose-stimulated insulin secretion and oxidative stress response. Am. J. Physiol. Metab. 2016, 310, E200–E212. [Google Scholar] [CrossRef]
- Lanng, S.; Thorsteinsson, B.; Nerup, J.; Koch, C. Influence of the development of diabetes mellitus on clinical status in patients with cystic fibrosis. Eur. J. Pediatr. 1992, 151, 684–687. [Google Scholar] [CrossRef]
- Burgess, J.C.; Bridges, N.; Banya, W.; Gyi, K.M.; Hodson, M.E.; Bilton, D.; Simmonds, N.J. HbA1c as a screening tool for cystic fibrosis related diabetes. J. Cyst. Fibros. 2015, 15, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Scully, K.J.; Marchetti, P.; Sawicki, G.S.; Uluer, A.; Cernadas, M.; Cagnina, R.E.; Kennedy, J.C.; Putman, M.S. The effect of elexacaftor/tezacaftor/ivacaftor (ETI) on glycemia in adults with cystic fibrosis. J. Cyst. Fibros. 2021, 21, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Norris, A.W.; Ode, K.L.; Merjaneh, L.; Sanda, S.; Yi, Y.; Sun, X.; Engelhardt, J.F.; Hull, R.L. Survival in a bad neighborhood: pancreatic islets in cystic fibrosis. J. Endocrinol. 2019, 241, R35–R50. [Google Scholar] [CrossRef]
- Grancini V, Gramegna A, Zazzeron L, Alicandro G, Porcaro LL, Piedepalumbo F et al. Effects of elexacaftor / tezacaftor / ivacaftor triple combination therapy on glycaemic control and body composition in patients with cystic fibrosis-related diabetes. Diabetes Metab. 2023 Sep;49(5):101466.
- Taylor-Cousar JL, Mall MA, Ramsey BW, McKone EF, Tullis E, Marigowda G et al. Clinical development of triple-combination CFTR modulators for cystic fibrosis patients with one or two F508delalleles. ERJ Open Res. 2019 Jun 17;5(2), 2019.
- Nielsen BU, Olsen MF, Mabuza Mathiesen IH, Pressler T, Ritz C, Katzenstein TL at al. Decline in HbA1c during the first year of elexacaftor/tezacaftor/ivacaftor treatment in the Danish cystic fibrosis cohort: Short title: Decline in HbA1c after elexacaftor/tezacaftor/ivacaftor treatment. J Cyst Fibros. 2024 Jan;23(1):103-108. [CrossRef]
- Lurquin, F.; Gohy, S.; Hermans, M.P.; Preumont, V. Combined CFTR modulator therapies are linked with anabolic benefits and insulin-sparing in cystic fibrosis-related diabetes. J. Clin. Transl. Endocrinol. 2023, 33, 100320. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.; Mass, A.; Reiter, J.; Zangen, D.H.; Cohen-Cymberknoh, M. Long-Term therapy with CFTR modulators consistently improves glucose metabolism in adolescents and adults with Cystic Fibrosis. Respir. Med. 2024, 228, 107664. [Google Scholar] [CrossRef]
- Salazar-Barragan, M.; Taub, D.R. The Effects of Elexacaftor, Tezacaftor, and Ivacaftor (ETI) on Blood Glucose in Patients With Cystic Fibrosis: A Systematic Review. Cureus 2023, 15, e41697. [Google Scholar] [CrossRef]
- Volkova, N.; Moy, K.; Evans, J.; Campbell, D.; Tian, S.; Simard, C.; Higgins, M.; Konstan, M.W.; Sawicki, G.S.; Elbert, A.; et al. Disease progression in patients with cystic fibrosis treated with ivacaftor: Data from national US and UK registries. J. Cyst. Fibros. 2019, 19, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Tsabari, R.; Elyashar, H.I.; Cymberknowh, M.C.; Breuer, O.; Armoni, S.; Livnat, G.; Kerem, E.; Zangen, D.H. CFTR potentiator therapy ameliorates impaired insulin secretion in CF patients with a gating mutation. J. Cyst. Fibros. 2015, 15, e25–e27. [Google Scholar] [CrossRef] [PubMed]
- Colombo, C.; Foppiani, A.; Bisogno, A.; Gambazza, S.; Daccò, V.; Nazzari, E.; Leone, A.; Giana, A.; Mari, A.; Battezzati, A. Lumacaftor/ivacaftor in cystic fibrosis: effects on glucose metabolism and insulin secretion. J. Endocrinol. Investig. 2021, 44, 2213–2218. [Google Scholar] [CrossRef] [PubMed]
- Dawood, S.N.; Rabih, A.M.; Niaj, A.; Raman, A.; Uprety, M.; Calero, M.J.; Villanueva, M.R.B.; Joshaghani, N.; Villa, N.; Badla, O.; et al. Newly Discovered Cutting-Edge Triple Combination Cystic Fibrosis Therapy: A Systematic Review. Cureus 2022, 14, e29359. [Google Scholar] [CrossRef]
- Zaher, A.; ElSaygh, J.; Elsori, D.; ElSaygh, H.; Sanni, A. A Review of Trikafta: Triple Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Modulator Therapy. Cureus 2021, 13, e16144. [Google Scholar] [CrossRef] [PubMed]
- Crow H, Bengtson C, Shi X, Graves L 3rd, Anabtawi A. CGM patterns in adults with cystic fibrosis-related diabetes before and after elexacaftor-tezacaftor-ivacaftor therapy. J Clin Transl Endocrinol. 1: 2022 Oct 1;30, 2022.
- Carnovale V, Iacotucci P, Terlizzi V, Colangelo C, Ferrillo L, Pepe A et al. Elexacaftor/Tezacaftor/Ivacaftor in Patients with Cystic Fibrosis Homozygous for the F508delMutation and Advanced Lung Disease: A 48-Week Observational Study. J Clin Med. 2022 Feb 16;11(4):1021.
- Middleton, P.G.; Mall, M.A.; Dřevínek, P.; Lands, L.C.; McKone, E.F.; Polineni, D.; Ramsey, B.W.; Taylor-Cousar, J.L.; Tullis, E.; Vermeulen, F.; et al. Elexacaftor–Tezacaftor–Ivacaftor for Cystic Fibrosis with a Single Phe508del Allele. N. Engl. J. Med. 2019, 381, 1809–1819. [Google Scholar] [CrossRef]
- Regard, L.; Martin, C.; Burnet, E.; Da Silva, J.; Burgel, P.-R. CFTR Modulators in People with Cystic Fibrosis: Real-World Evidence in France. Cells 2022, 11, 1769. [Google Scholar] [CrossRef]
- Walter, E.; Bass, J.L. The Effect of Elexacaftor/Tezacaftor/Ivacaftor on Hospitalizations and Intravenous Antibiotic Use. Perm. J. 2022, 26, 73–79. [Google Scholar] [CrossRef]
- Caley, L.; Jarosz-Griffiths, H.; Smith, L.; Gale, L.; Barrett, J.; Kinsey, L.; Davey, V.; Nash, M.; Jones, A.; Whitehouse, J.; et al. Body mass index and nutritional intake following Elexacaftor/Tezacaftor/Ivacaftor modulator therapy in adults with cystic fibrosis. J. Cyst. Fibros. 2023, 22, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Taelman, V.; Declercq, D.; Van Biervliet, S.; Weygaerde, Y.V.; Lapauw, B.; Van Braeckel, E. Effect of 18 months elexacaftor-tezacaftor-ivacaftor on body mass index and glycemic control in adults with cystic fibrosis. Clin. Nutr. ESPEN 2023, 58, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Tervo, J.P.; DiMango, E.; Gudis, D.A.; Keating, C.; Zhang, Y.; Leu, C.; Altman, K.; Vilarello, B.; Jacobson, P.; Overdevest, J.B. Olfaction, body mass index, and quality of life with cystic fibrosis combination therapy. Int. Forum Allergy Rhinol. 2023, 13, 2165–2171. [Google Scholar] [CrossRef] [PubMed]
- Piehler, L.; Thalemann, R.; Lehmann, C.; Thee, S.; Röhmel, J.; Syunyaeva, Z.; Stahl, M.; Mall, M.A.; Graeber, S.Y. Effects of elexacaftor/tezacaftor/ivacaftor therapy on mental health of patients with cystic fibrosis. Front. Pharmacol. 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Kos, R.; Neerincx, A.H.; Fenn, D.W.; Brinkman, P.; Lub, R.; Vonk, S.E.M.; Roukema, J.; Reijers, M.H.; Terheggen-Lagro, S.W.J.; Altenburg, J.; et al. Real-life efficacy and safety of elexacaftor/tezacaftor/ivacaftor on severe cystic fibrosis lung disease patients. Pharmacol. Res. Perspect. 2022, 10, e01015. [Google Scholar] [CrossRef] [PubMed]
- Griese, M.; Costa, S.; Linnemann, R.W.; Mall, M.A.; McKone, E.F.; Polineni, D.; Quon, B.S.; Ringshausen, F.C.; Taylor-Cousar, J.L.; Withers, N.J.; et al. Safety and Efficacy of Elexacaftor/Tezacaftor/Ivacaftor for 24 Weeks or Longer in People with Cystic Fibrosis and One or More F508del Alleles: Interim Results of an Open-Label Phase 3 Clinical Trial. Am. J. Respir. Crit. Care Med. 2021, 203, 381–385. [Google Scholar] [CrossRef] [PubMed]
| Age (years) | ||
| Mean | 33.5 | |
| Median | 33.0 | |
| Standard Deviation | ± 13.26 | |
| Frequency (N) | Percentage (%) | |
| Sex | Total = 28 | |
| Female | 12 | 42.9 |
| Male | 16 | 57.1 |
| Genotype | Total = 28 | |
| F508del/F508del (homozygosis) | 16 | 57.1 |
| F508del/other (heteroyzygosis) | 12 | 42.9 |
| Previous use of CFTR-modulators | Total = 28 | |
| None | 14 | 50.0 |
| Lumacaftor/Ivacaftor | 12 | 42.9 |
| Tezacaftor/Ivacaftor | 2 | 7.1 |
| Insulin Regimen | Total = 28 | |
| Once Daily Basal Insulin | 16 | 57.1 |
| Basal-Bolus | 12 | 42.9 |
| N | Pre-T0 | N | T0 | T1 | T0 vs T1 | N | T2 | Pre-T0 vs T2 | N | T3 | T0 vs T3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hospitalizations | 28 | 0.86±0.89 | 28 | 0.32±0.48 | 0.003 | |||||||
| Pulmonary exacerbations | 28 | 1.64±1.59 | 28 | 0.46±0.79 | 0.002 | |||||||
| Six minute walking test | 19 | 574.18±68.04 | 19 | 572.75±92.99 | 0.71 | |||||||
| Chloride sweat test | 15 | 118.53±22.21 | 15 | 49.93±17.81 | 0.001 | |||||||
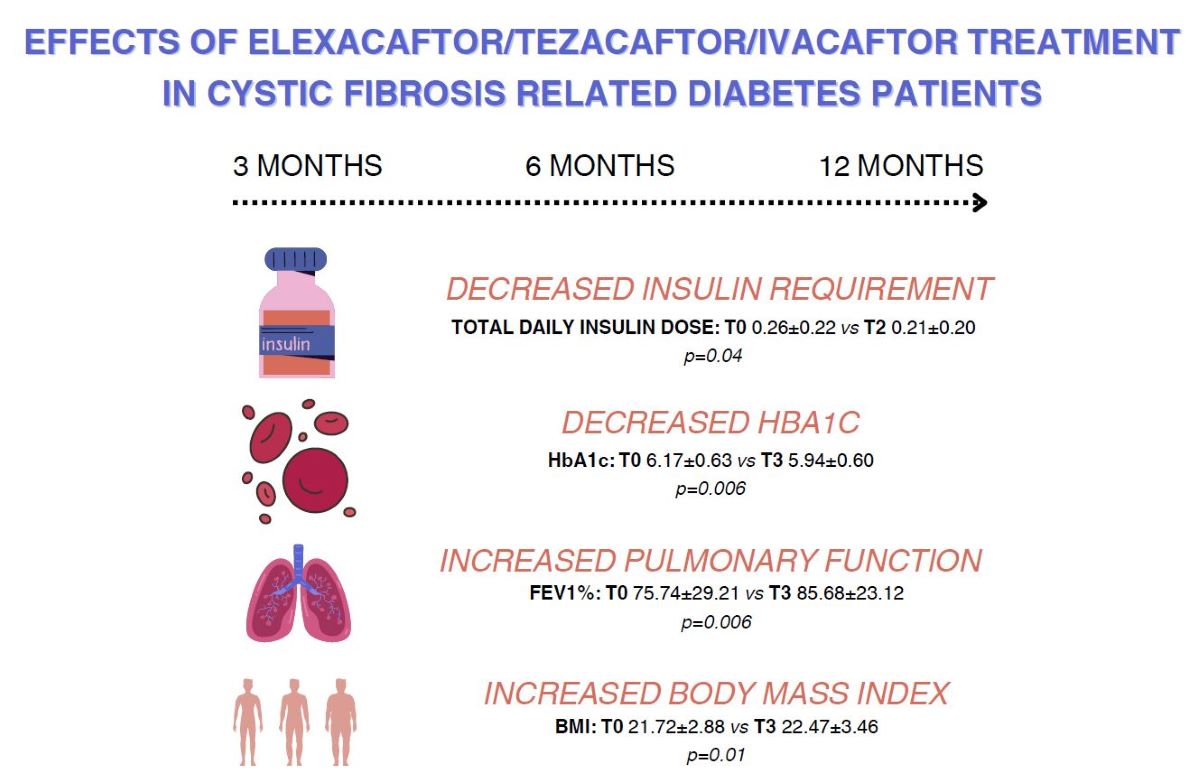
| Body mass index | 26 | 21.72±2.88 | 22.53±3.11 | 0.0001 | 26 | 22.48±3.02 | 0.0001 | 25 | 22.47±3.46 | 0.006 | ||
| Fev1 | 27 | 2420.0±1038.4 | 2782.2±946.6 | 0.0001 | 27 | 2774.8±991.7 | 0.0001 | 25 | 2771.6±967.1 | 0.001 | ||
| Fvc | 27 | 3267.4±1043.3 | 3640.0±899.1 | 0.001 | 27 | 3694.8±959.8 | 0.0001 | 25 | 3671.5±938.7 | 0.001 | ||
| Fev1% | 27 | 75.74±29.21 | 85.44±23.70 | 0.001 | 27 | 85.37±25.40 | 0.002 | 25 | 85.68±23.12 | 0.01 | ||
| Total daily insulin dose | 27 | 0.26±0.22 | 0.18±0.18 | 0.01 | 25 | 0.21±0.20 | 0.04 | 23 | 0.21±0.22 | 0.10 | ||
| Hba1c | 23 | 6.17±0.63 | 5.94±0.58 | 0.001 | 26 | 5.89±0.59 | 0.004 | 23 | 5.94±0.60 | 0.006 | ||
| Cfqr | 26 | 64.63±7.59 | 26 | 71.74±4.12 | 0.0001 | 15 | 70.13±4.84 | 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




