Submitted:
03 September 2024
Posted:
04 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Ketogenic diet as regenerative immunotherapy
1.2. Carnivore Diet as a Regenerative Immunotherapy
2. Hypothesized Mechanisms of the Carnivore Diet
- 1.
- 2.
- 3.
- Reduced Omega-6 (Linoleic Acid) consumption: Linoleic Acid may directly induce inflammation in the intestinal epithelium via formation of oxidative linoleic acid metabolites (OXLAMs) and consequent dysregulation of the Endocannabinoid System [23]. Carnivore diets more closely resemble the pre-modern consumption of <2g/day linoleic acid vs. the modern consumption of 29g/day [24];
- 4.
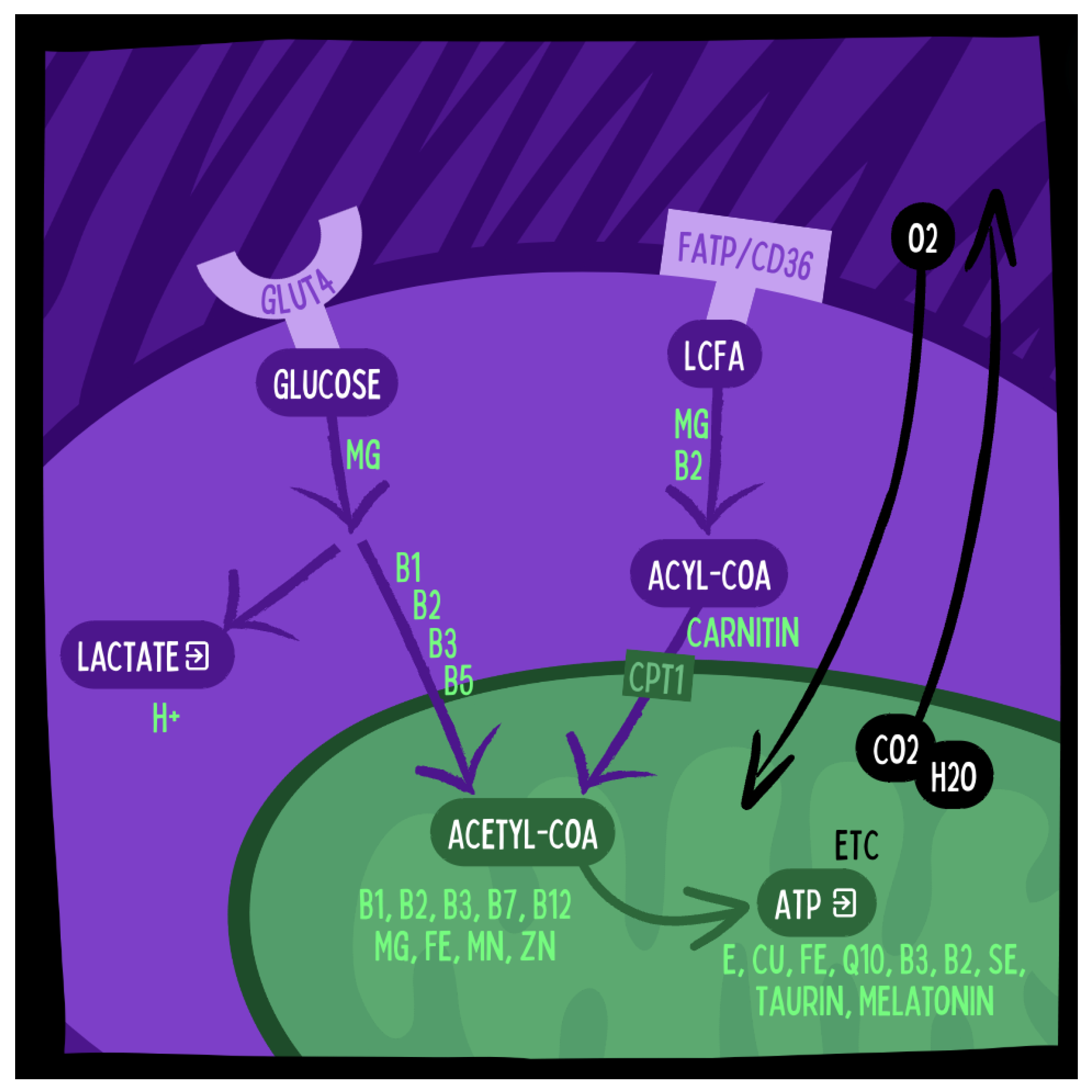
- Higher Micronutrient Density: Animal Foods are more dense in most micronutrients (vitamins and minerals) relevant to ATP synthesis (see Figure 4) compared with plant foods and lack anti-nutrients such as phytates [16,17], which may improve immune regulation and regenerative capacity of intestinal epithelial cells;
- 5.
3. Possible Concerns
- 1.
- Gout Risks: The supersaturation of uric acid can -under the wrong circumstances- lead to the deposition of monosodium urate monohydrate crystals in the tissues, with resultant gout arthritis. Gout can be manifested by the elevation of serum urate, acute gouty arthritic attacks, the formation of tophi, gouty nephropathy, and uric acid stones. Meat itself has not been established as a causative agent, but the high amount of purine within it can serve as a triggering factor in causing episodes of gout arthritis in a pre-existing metabolic dysregulation. Our own clinical experience shows that a ketogenic/carnivore diet can even alleviate gout medium term. Hypothetically, this could be due to reduced oxidative stress since uric acid acts as an antioxidant, reduced availability of dietary monosodium (glutamate), or perhaps increased exercise in our patient population since muscle activity induces myokine secretion, hence helping in the conversion of uric acid to allantoin for excretion through the kidneys [25]. Indeed, recent reviews have confirmed our observation of reduced uric acid in very low carbohydrate ketogenic diets [26].
- 2.
- Carcinogenicity: The World Healths Organizations (WHO) International Agency for the Research of Cancer (IARC) has classified Processed Meat as carcinogenic (Class I), and unprocessed Red Meat as possibly carcinogenic (Class IIa). No causal relationships have been established and no causal agents in red meat have been identified to date. By the classification standards of IARC, the classification is to be based on an associative relationship and does not establish the magnitude of risk. Recent systematic reviews have argued that evidence even for the proposed associative relationship between unprocessed red meat and negative health outcomes (including cancer) is lacking, and recommendations for reduced consumption of unprocessed red meat are currently not backed by scientific data [14,27].
- 3.
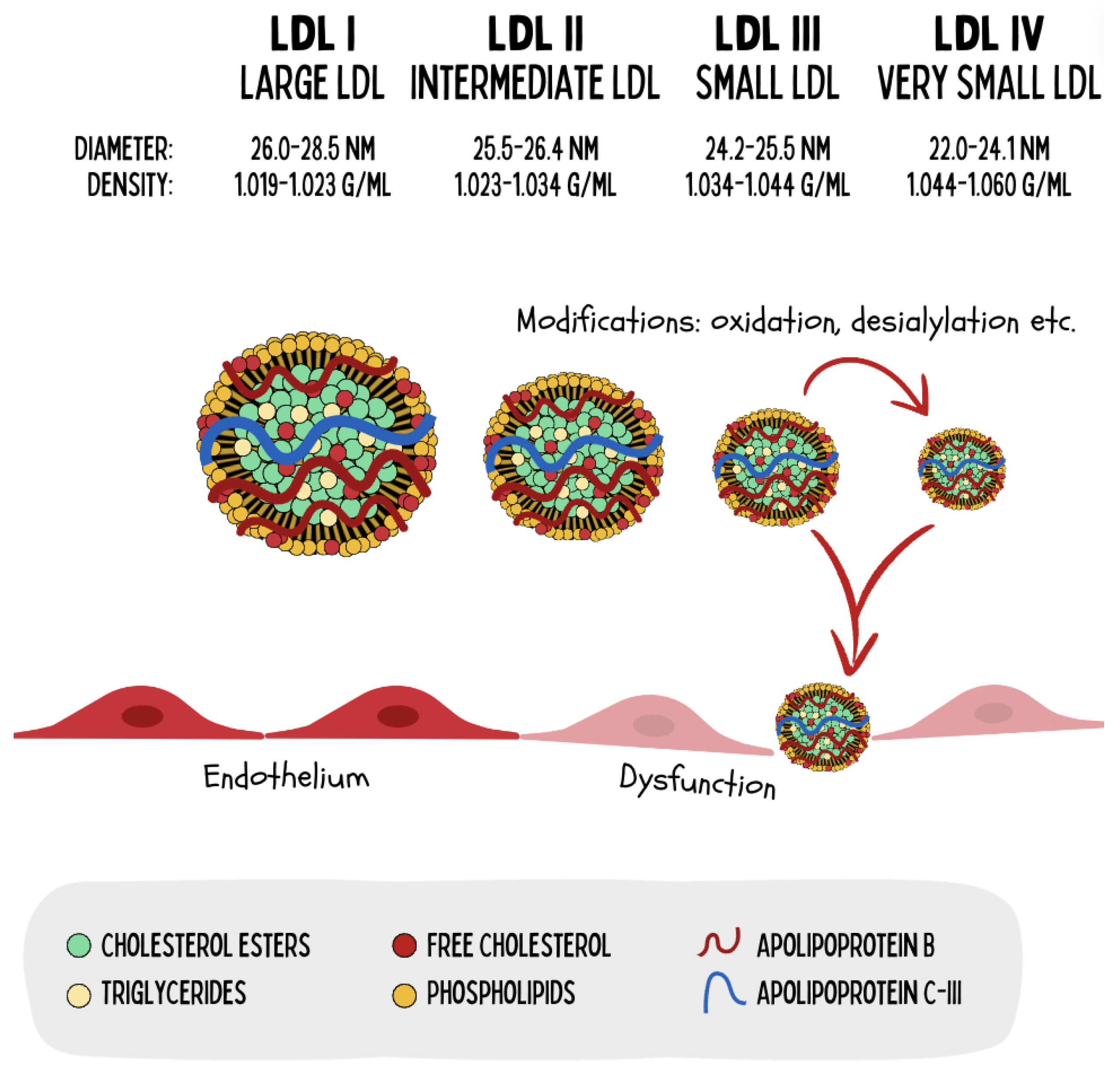
- Dyslipidemia and Cardiovascular Risks: For a given population on a standard diet, increased serum total LDL Lipoprotein molecular mass (measured in mg/dl) has been considered causal in the progression of atherosclerosis (Libby 2021). On ketogenic diets, total serum LDL lipoprotein mass can and most likely will increase; however, the size of the LDL particle becomes larger, thereby reducing the number of atherogenic particles [28,29,30,31]. A reduction in the number of the atherogenic small and dense LDL lipoproteins and concurrent increase in the lipoprotein size is associated with improved cardiovascular risk markers such as reduced BMI, body weight, inflammatory markers, sdLDL, Triglycerides, Lipoprotein A, Apolipoprotein B, Blood Glucose, HbA1c, Insulin, and Blood Pressure, and increased HDL. During ketogenic diets LDL Lipoproteins serve other functions as in standard diets and are not to be interpreted as signs of metabolic dysfunction [32]. Therefore, increased serum LDL on a ketogenic diet has to be evaluated differently than increased serum LDL on a standard diet and statin therapy is usually not warranted in a low-carbohydrate ketogenic diet [33].
4. Testing the Hypothesis – Study Design and Methodology
4.1. Study Objectives
4.2. Study Design
4.3. Participant Criteria
4.3.1. Inclusion Criteria
- Diagnosis of IBD as per Montreal classification,
- Aged 18-70 years.
4.3.2. Exclusion Criteria: General
- Pregnant or intending to become pregnant within the next 3 months,
- Currently abusing substances,
- On ketogenic or carnivore diet in last 6 months,
- Currently Vegan or vegetarian diet and unwilling to switch to carnivore diet,
- Hospitalization during the last 3 months,
- Participation in another research project,
- Inability to fill out the initial questionnaires,
- Active liver, kidney, or cardiovascular diseases, kidney stones, severe hyperlipidemia.
4.3.3. Exclusion Criteria: Metabolic Disorders
- Glycogen storage disease type 1 (von Gierke disease),
- Carnitine palmitoyltransferase deficiencies (CPT I/II),
- Primary carnitine deficiency,
- Carnitine-acylcarnitine translocase deficiency,
- Pyruvate carboxylase deficiency,
- Succinyl-CoA acetoacetate transferase deficiency,
- Various fatty acid oxidation disorders,
- Acute intermittent porphyria.
4.4. Implementation and Follow-Up
4.4.1. Participant Training
- Nutritional science behind the ketogenic and carnivore diets,
- Appropriate foods and sample recipes,
- Targeted ketone and glucose levels,
- Food measurement-grams,
- Preparation for diet initiation across environments,
- Overcoming obstacles-quality, procurement, and preparation,
- Dining out, traveling, and illness guidelines,
- Medication guidelines,
- Prevention/management of potential side effects; for example hypoglycemia or hyperketosis,
- Why diets may fail,
- Modifications for illness-more water, no concern for ketone level,
- Fitting the diet into larger ecological, spiritual, and economic contexts.
4.4.2. Ketone Monitoring
5. Discussion
- for gout risk, check uric acid levels on a regular basis.
- for cancer risk, perform long-term monitoring of biomarkers for cancer.
- for cardiovascular risk, evaluate lipid profiles including Apolipoprotein B as a measure of particle number, blood pressure, visceral body fat and markers of systemic inflammation such as hsCRP.
6. Conclusions
Funding
Acknowledgments
Appendix A. Suggested Lab Analysis
Appendix A.1. Standard Labs
Appendix A.2. Specialized Labs
Appendix A.3. Stool and Saliva Labs
References
- Martin-McGill, K.J.; Bresnahan, R.; Levy, R.G.; Cooper, P.N. Ketogenic diets for drug-resistant epilepsy. Cochrane Database of Systematic Reviews 2020, 2020, Not available. [Google Scholar] [CrossRef]
- Sourbron, J.; Klinkenberg, S.; van Kuijk, S.M.J.; Lagae, L.; Lambrechts, D.; Braakman, H.M.H.; Majoie, M. Ketogenic diet for the treatment of pediatric epilepsy: Rview and meta-analysis. Child’s Nervous System 2020, 36, 1099–1109. [Google Scholar] [CrossRef]
- Kossoff, E.H.; Turner, Z.; Cervenka, M.C.; Barron, B.J. Ketogenic Diet Therapies For Epilepsy and Other Conditions. Not available 2020, Not available, Not available. [Google Scholar] [CrossRef]
- Needham, N.; Campbell, I.; Grossi, H.; Kamenska, I.; Rigby, B.; Simpson, S.; McIntosh, E.; Bahuguna, P.; Meadowcroft, B.; Creasy, F.; Mitchell-Grigorjeva, M.; Norrie, J.; Thompson, G.; Gibbs, M.; McLellan, A.; Fisher, C.; Moses, T.; Burgess, K.; Brown, R.; Smith, D. Pilot study of a ketogenic diet in bipolar disorder. BJPsych Open 2023, 9. [Google Scholar] [CrossRef]
- Bohnen, J.L.B.; Albin, R.L.; Bohnen, N.I. Ketogenic interventions in mild cognitive impairment, Alzheimer’s disease, and Parkinson’s disease: A systematic review and critical appraisal. Frontiers in Neurology 2023, 14, Not available. [Google Scholar] [CrossRef]
- Tóth, C.; Dabóczi, A.; Howard, M.; Miller, N.J.; Clemens, Z. Crohn’s disease successfully treated with the paleolithic ketogenic diet. International Journal of Case Reports and Images 2016, 7, 570. [Google Scholar] [CrossRef]
- Srivastava, S.; Pawar, V.; Tyagi, A.; Sharma, K.; Kumar, V.; Shukla, S. Immune Modulatory Effects of Ketogenic Diet in Different Disease Conditions. Immuno 2022, 3, 1–15. [Google Scholar] [CrossRef]
- Kaur, N.; Chen, C.C.; Luther, J.; Kao, J.Y. Intestinal dysbiosis in inflammatory bowel disease. Gut Microbes 2011, 2, 211–216. [Google Scholar] [CrossRef]
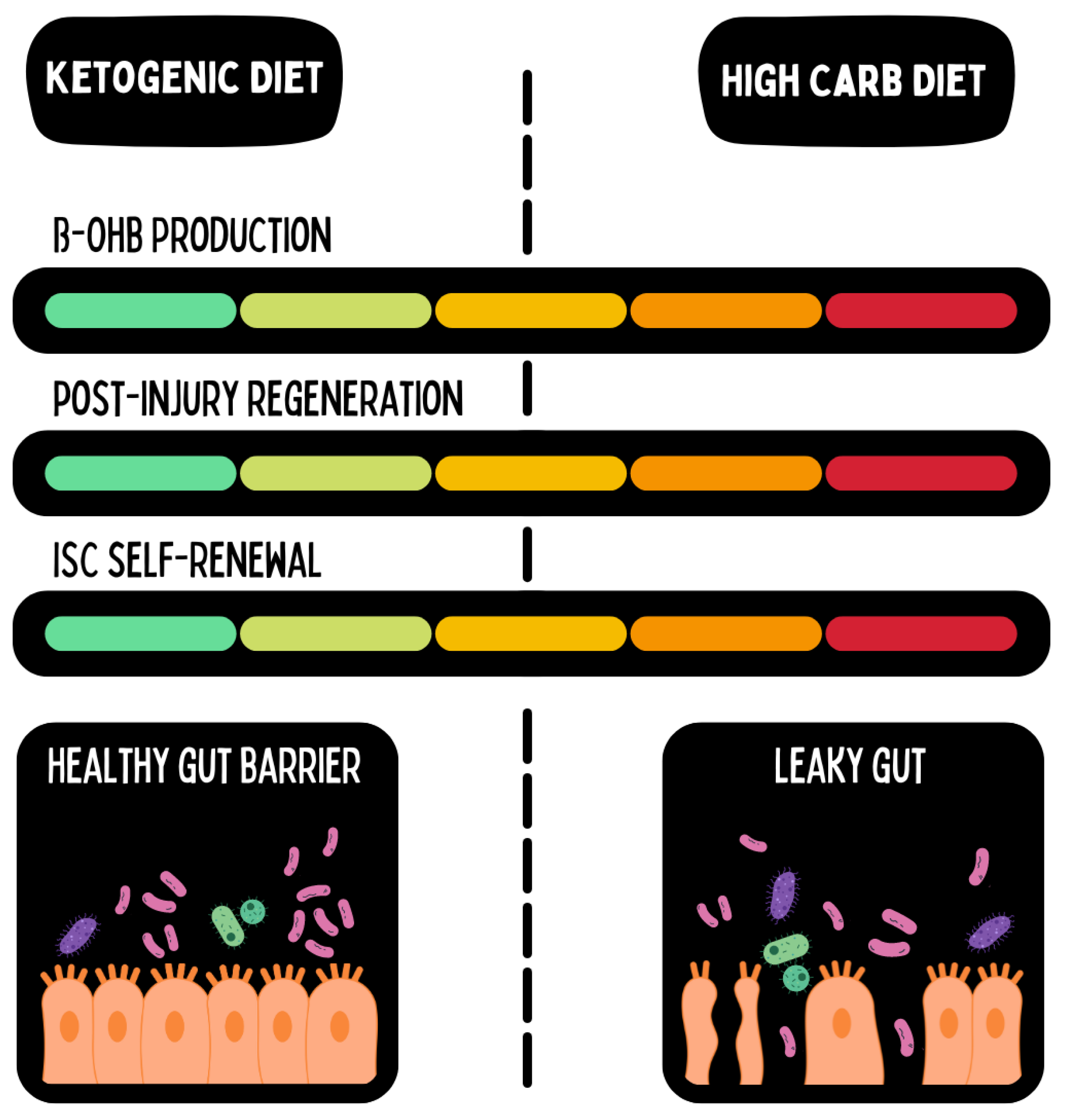
- Andersen, O.E.; Poulsen, J.V.; Farup, J.; de Morree, A. Regulation of adult stem cell function by ketone bodies. Frontiers in Cell and Developmental Biology 2023, 11, Not available. [Google Scholar] [CrossRef]
- Cheng, C.W.; Biton, M.; Haber, A.L.; Gunduz, N.; Eng, G.; Gaynor, L.T.; Tripathi, S.; Calibasi-Kocal, G.; Rickelt, S.; Butty, V.L.; Moreno-Serrano, M.; Iqbal, A.M.; Bauer-Rowe, K.E.; Imada, S.; Ulutas, M.S.; Mylonas, C.; Whary, M.T.; Levine, S.S.; Basbinar, Y.; Hynes, R.O.; Mino-Kenudson, M.; Deshpande, V.; Boyer, L.A.; Fox, J.G.; Terranova, C.; Rai, K.; Piwnica-Worms, H.; Mihaylova, M.M.; Regev, A.; Ömer, H. Yilmaz. Ketone Body Signaling Mediates Intestinal Stem Cell Homeostasis and Adaptation to Diet. Cell 2019, 178, 1115–1131.e15. [Google Scholar] [CrossRef]
- Ben-Dor, M.; Sirtoli, R.; Barkai, R. The evolution of the human trophic level during the Pleistocene. American Journal of Physical Anthropology 2021, 175, 27–56. [Google Scholar] [CrossRef]
- Kuang, R.; Levinthal, D.J.; Ghaffari, A.A.; del Aguila de Rivers, C.R.; Tansel, A.; Binion, D.G. Nightshade Vegetables: A Dietary Trigger for Worsening Inflammatory Bowel Disease and Irritable Bowel Syndrome? Digestive Diseases and Sciences 2023, 68, 2853–2860. [Google Scholar] [CrossRef]
- Konijeti, G.G.; Kim, N.; Lewis, J.D.; Groven, S.; Chandrasekaran, A.; Grandhe, S.; Diamant, C.; Singh, E.; Oliveira, G.; Wang, X.; Molparia, B.; Torkamani, A. Efficacy of the Autoimmune Protocol Diet for Inflammatory Bowel Disease. Inflammatory Bowel Diseases 2017, 23, 2054–2060. [Google Scholar] [CrossRef]
- Lescinsky, H.; Afshin, A.; Ashbaugh, C.; Bisignano, C.; Brauer, M.; Ferrara, G.; Hay, S.; He, J.; Iannucci, V.; Marczak, L.; McLaughlin, S.; Mullany, E.; Parent, M.; Serfes, A.; Sorensen, R.; Aravkin, A.; Zheng, P.; Murray, C. Health effects associated with consumption of unprocessed red meat: A Burden of Proof study. Nature Medicine 2022, 28, 1–8. [Google Scholar] [CrossRef]
- Iablokov, V.; Sydora, B.C.; Foshaug, R.; Meddings, J.; Driedger, D.; Churchill, T.; Fedorak, R.N. Naturally Occurring Glycoalkaloids in Potatoes Aggravate Intestinal Inflammation in Two Mouse Models of Inflammatory Bowel Disease. Digestive Diseases and Sciences 2010, 55, 3078–3085. [Google Scholar] [CrossRef]
- O’Hearn, A. Can a carnivore diet provide all essential nutrients? Current Opinion in Endocrinology, Diabetes and Obesity 2020, 27, 312–316. [Google Scholar] [CrossRef]
- Beal, T.; Ortenzi, F. Priority Micronutrient Density in Foods. Frontiers in Nutrition 2022, 9, Not available. [Google Scholar] [CrossRef]
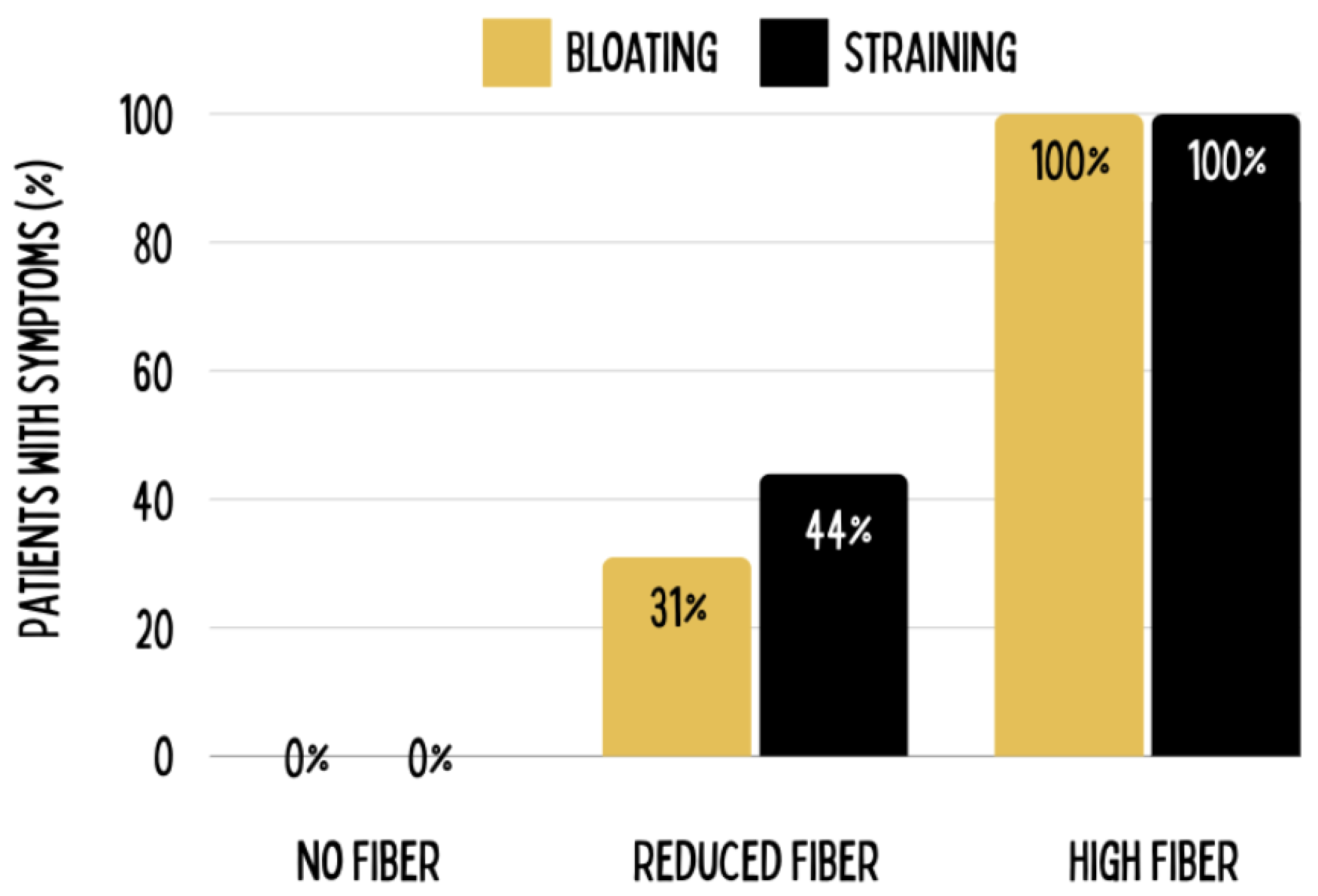
- Tan, K.Y. Fiber and colorectal diseases: Separating fact from fiction. World Journal of Gastroenterology 2007, 13, 4161. [Google Scholar] [CrossRef]
- Ho, K.S. Stopping or reducing dietary fiber intake reduces constipation and its associated symptoms. World Journal of Gastroenterology 2012, 18, 4593. [Google Scholar] [CrossRef]
- Lennerz, B.S.; Mey, J.T.; Henn, O.H.; Ludwig, D.S. Behavioral Characteristics and Self-Reported Health Status among 2029 Adults Consuming a “Carnivore Diet”. Current Developments in Nutrition 2021, 5, nzab133. [Google Scholar] [CrossRef]
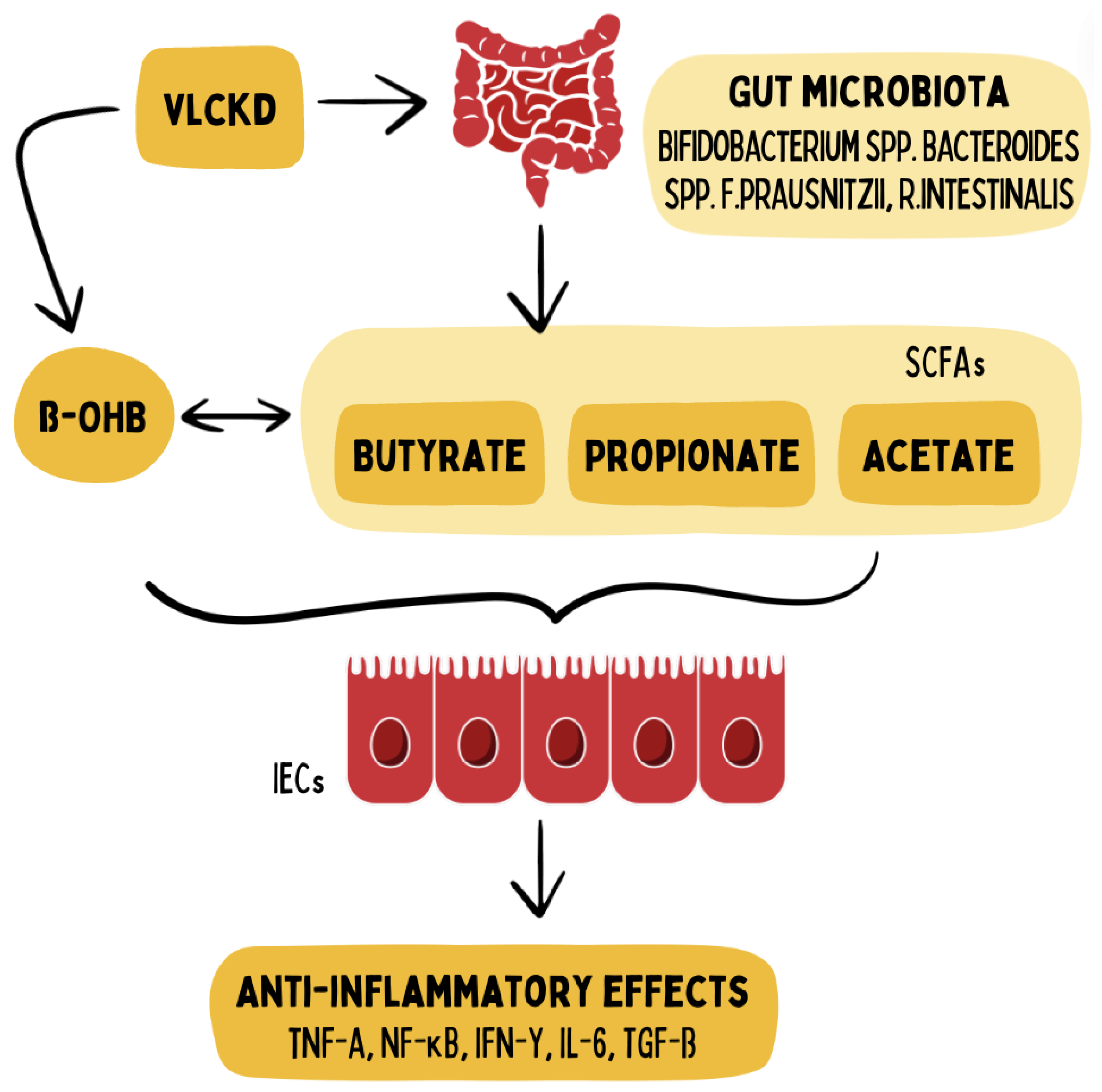
- Alsharairi, N.A. The Role of Short-Chain Fatty Acids in Mediating Very Low-Calorie Ketogenic Diet-Infant Gut Microbiota Relationships and Its Therapeutic Potential in Obesity. Nutrients 2021, 13, 3702. [Google Scholar] [CrossRef]
- Venegas, D.P.; la Fuente, M.K.D.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Frontiers in Immunology 2019, 10, Not available. [Google Scholar] [CrossRef]
- Deol, P.; Ruegger, P.; Logan, G.D.; Shawki, A.; Li, J.; Mitchell, J.D.; Yu, J.; Piamthai, V.; Radi, S.H.; Hasnain, S.; Borkowski, K.; Newman, J.W.; McCole, D.F.; Nair, M.G.; Hsiao, A.; Borneman, J.; Sladek, F.M. Diet high in linoleic acid dysregulates the intestinal endocannabinoid system and increases susceptibility to colitis in Mice. Gut Microbes 2023, 15, Not. [Google Scholar] [CrossRef]
- Mercola, J.; D’Adamo, C.R. Linoleic Acid: A Narrative Review of the Effects of Increased Intake in the Standard American Diet and Associations with Chronic Disease. Nutrients 2023, 15, 3129. [Google Scholar] [CrossRef]
- Roman, Y.M. The Role of Uric Acid in Human Health: Insights from the Uricase Gene. Journal of Personalized Medicine 2023, 13, 1409. [Google Scholar] [CrossRef]
- Gohari, S.; Ghobadi, S.; Jafari, A.; Ahangar, H.; Gohari, S.; Mahjani, M. The effect of dietary approaches to stop hypertension and ketogenic diets intervention on serum uric acid concentration: a systematic review and meta-analysis of randomized controlled trials. Scientific Reports 2023, 13, Not available. [Google Scholar] [CrossRef]
- Unprocessed Red Meat and Processed Meat Consumption: Dietary Guideline Recommendations From the Nutritional Recommendations (NutriRECS) Consortium. Annals of Internal Medicine 2019, 171, 756–764. [CrossRef] [PubMed]
- Westman, E.C.; Yancy, W.S.; Olsen, M.K.; Dudley, T.; Guyton, J.R. Effect of a low-carbohydrate, ketogenic diet program compared to a low-fat diet on fasting lipoprotein subclasses. International Journal of Cardiology 2006, 110, 212–216. [Google Scholar] [CrossRef]
- Falkenhain, K.; Roach, L.A.; McCreary, S.; McArthur, E.; Weiss, E.J.; Francois, M.E.; Little, J.P. Effect of carbohydrate-restricted dietary interventions on LDL particle size and number in adults in the context of weight loss or weight maintenance: A systematic review and meta-analysis. The American Journal of Clinical Nutrition 2021, 114, 1455–1466. [Google Scholar] [CrossRef]
- Froyen, E. The effects of fat consumption on low-density lipoprotein particle size in healthy individuals: a narrative review. Lipids in Health and Disease 2021, 20. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.N.; Zou, Y.L.; Guo, S.D. Low-density lipoprotein particles in atherosclerosis. Frontiers in Physiology 2022, 13, Not available. [Google Scholar] [CrossRef]
- Norwitz, N.G.; Soto-Mota, A.; Kaplan, B.; Ludwig, D.S.; Budoff, M.; Kontush, A.; Feldman, D. The Lipid Energy Model: Reimagining Lipoprotein Function in the Context of Carbohydrate-Restricted Diets. Metabolites 2022, 12, 460. [Google Scholar] [CrossRef] [PubMed]
- Diamond, D.M.; Bikman, B.T.; Mason, P. Statin therapy is not warranted for a person with high LDL-cholesterol on a low-carbohydrate diet. Current Opinion in Endocrinology, Diabetes and Obesity 2022, 29, 497–511. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



