Submitted:
28 August 2024
Posted:
29 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cortical Defect Animal Model
2.2. Hypoxyprobe Treatments
2.3. Tissue Harvesting and Staining Analysis
2.4. Femoral Fracture Animal Model
2.5. Treatments and Cell Study
2.6. Statistical Design and Analysis
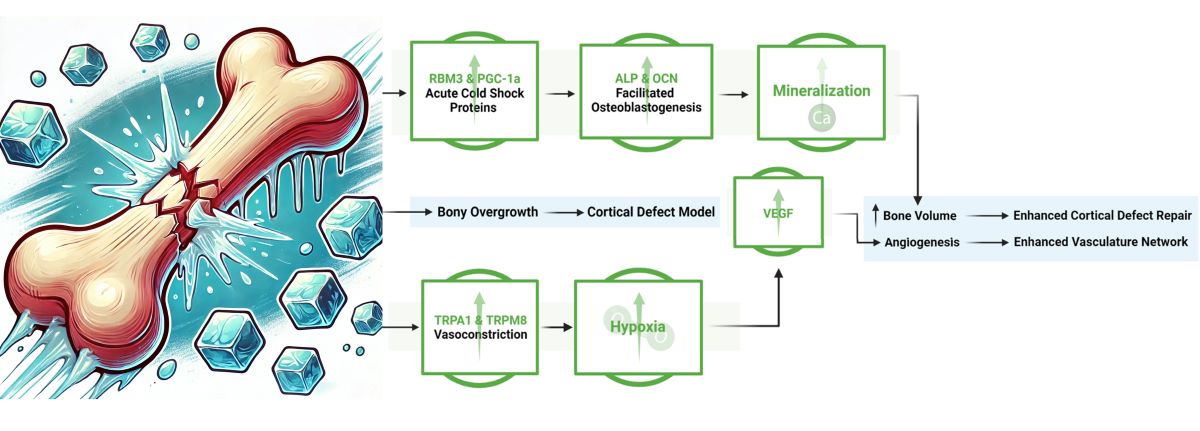
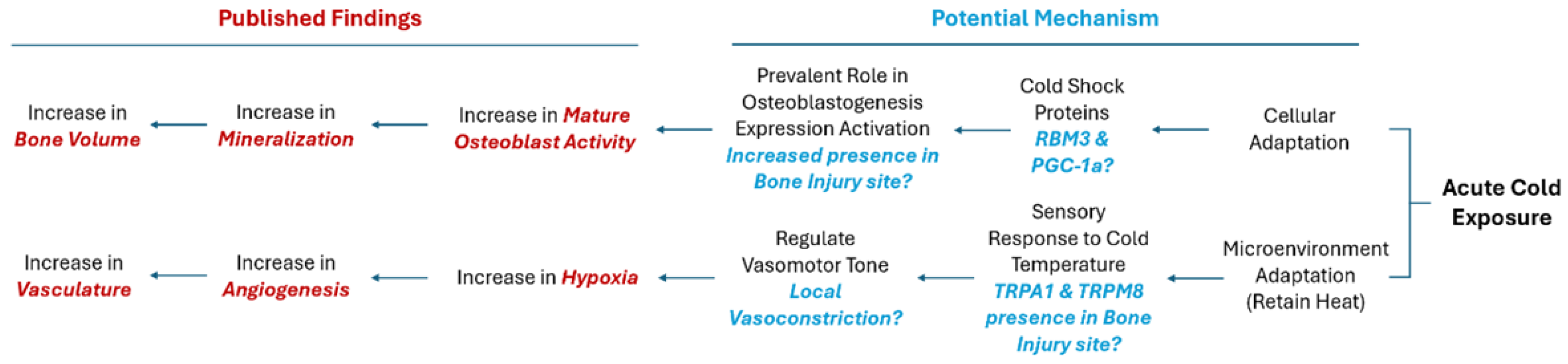
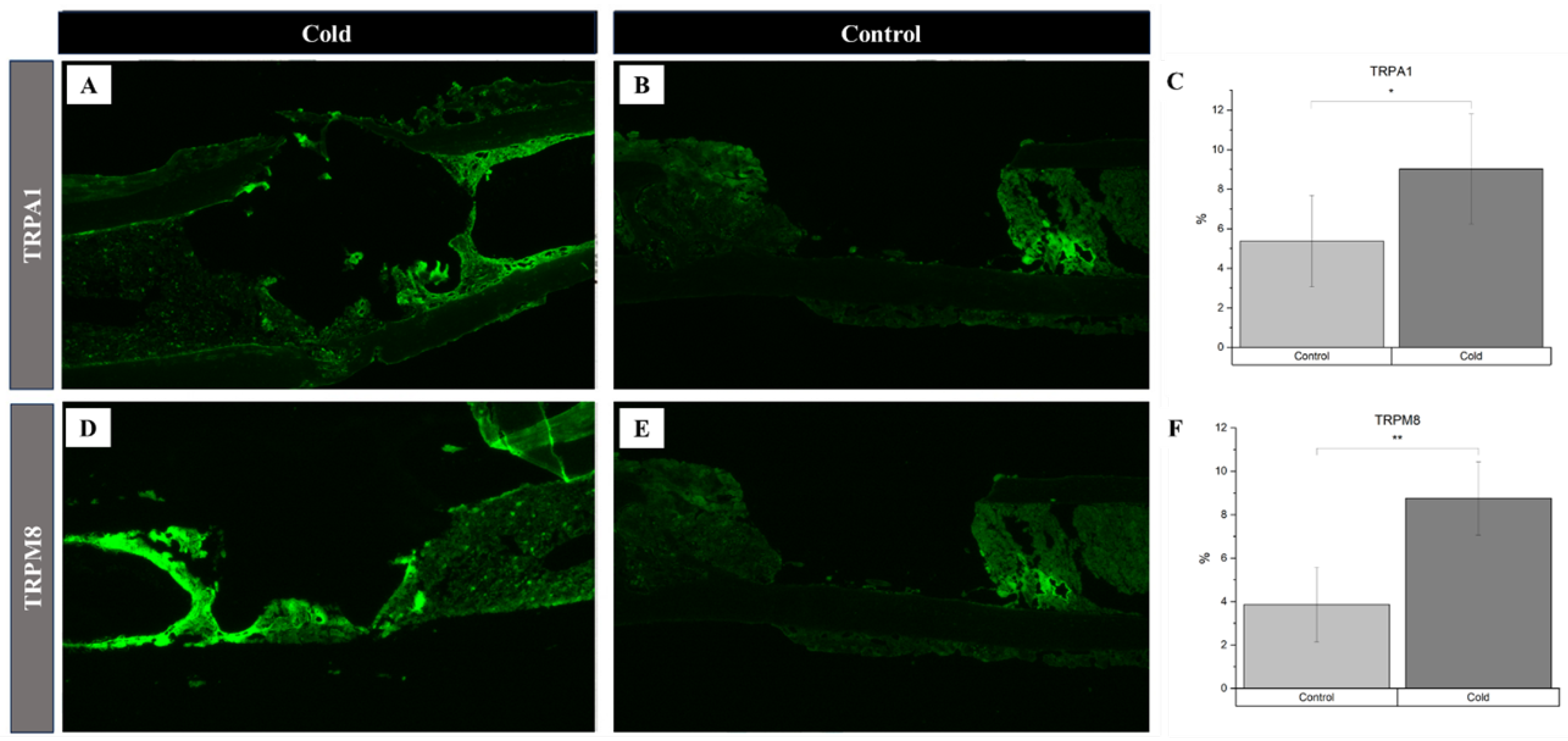
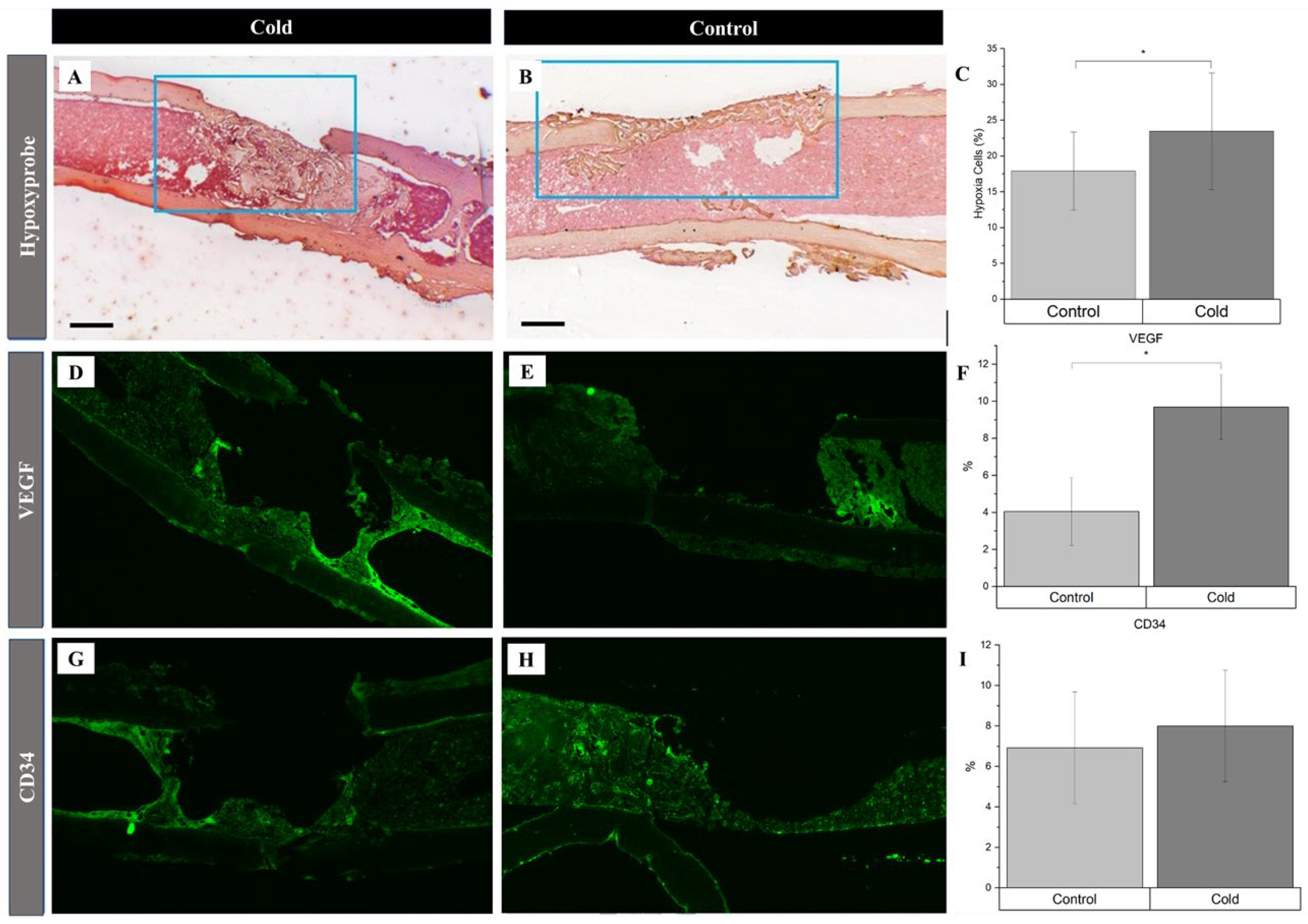
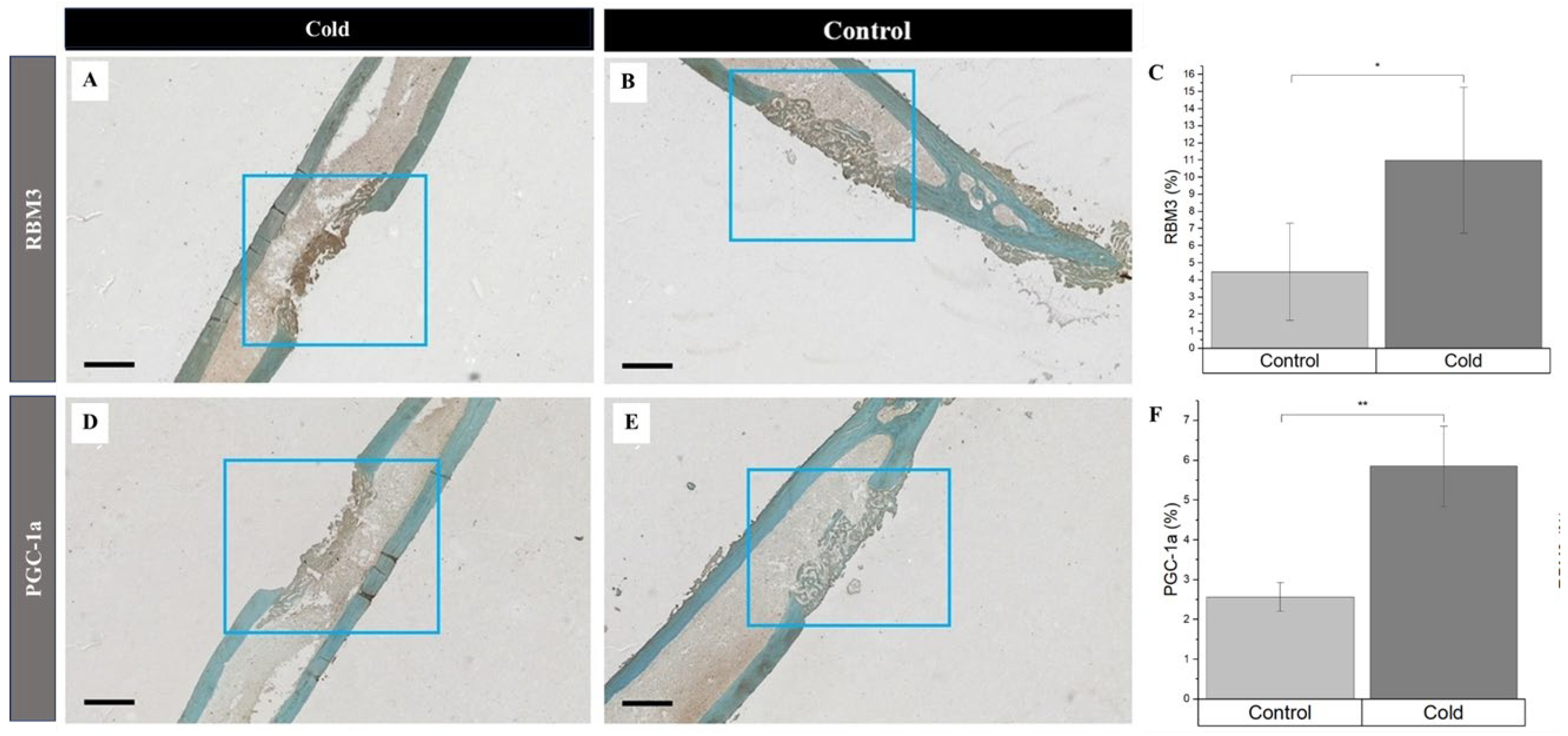
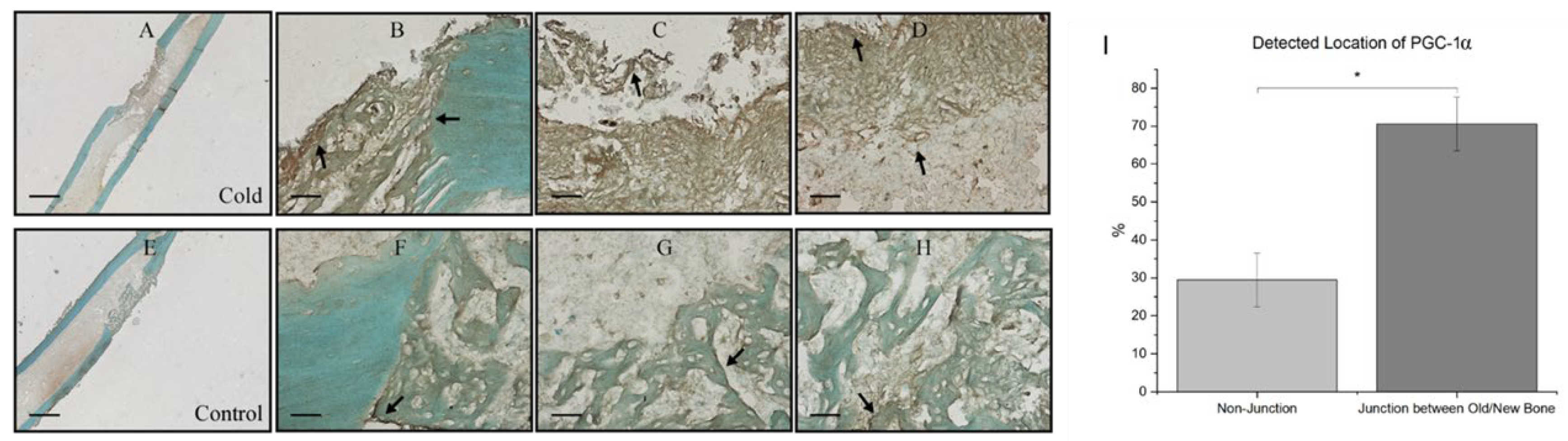
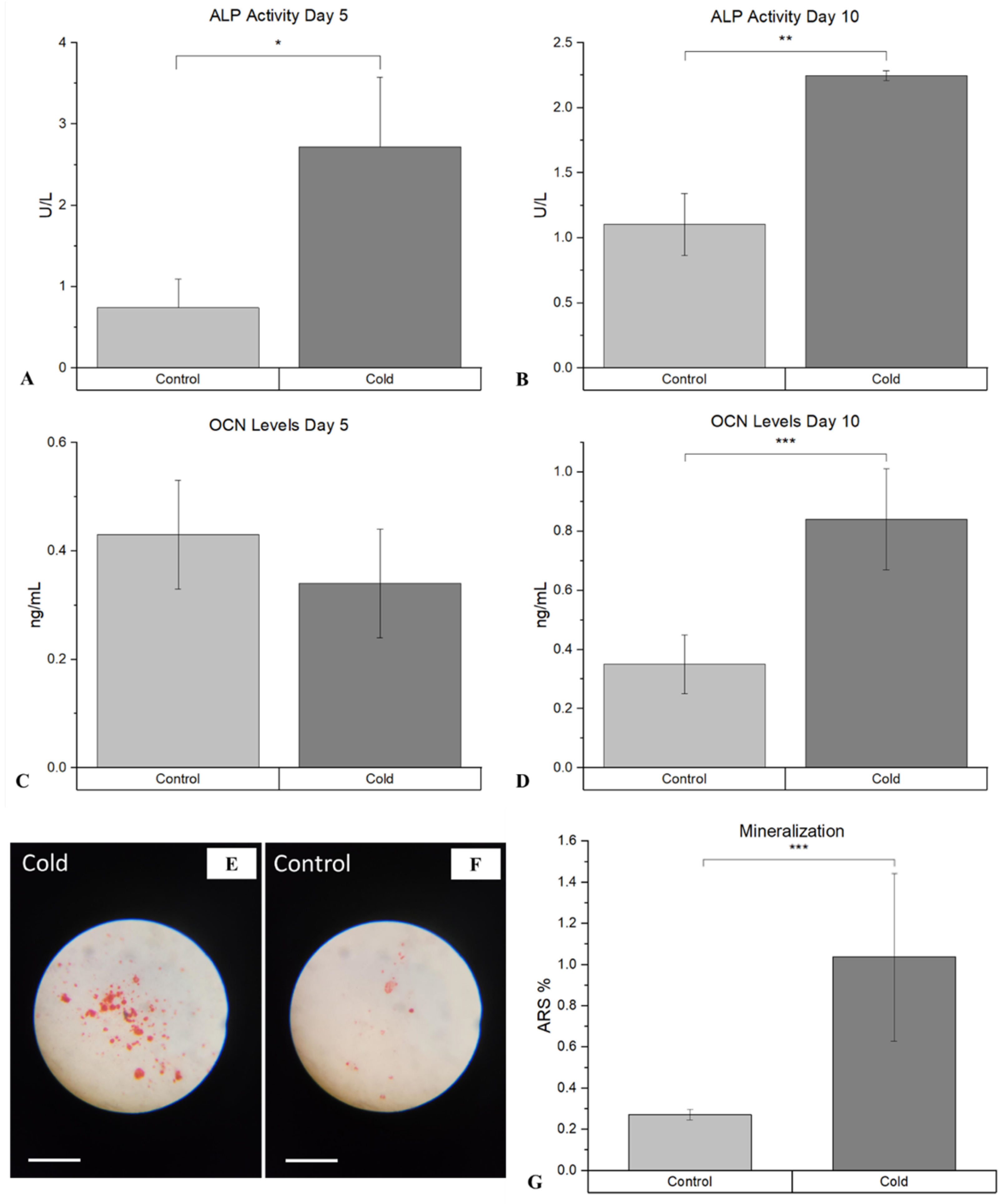
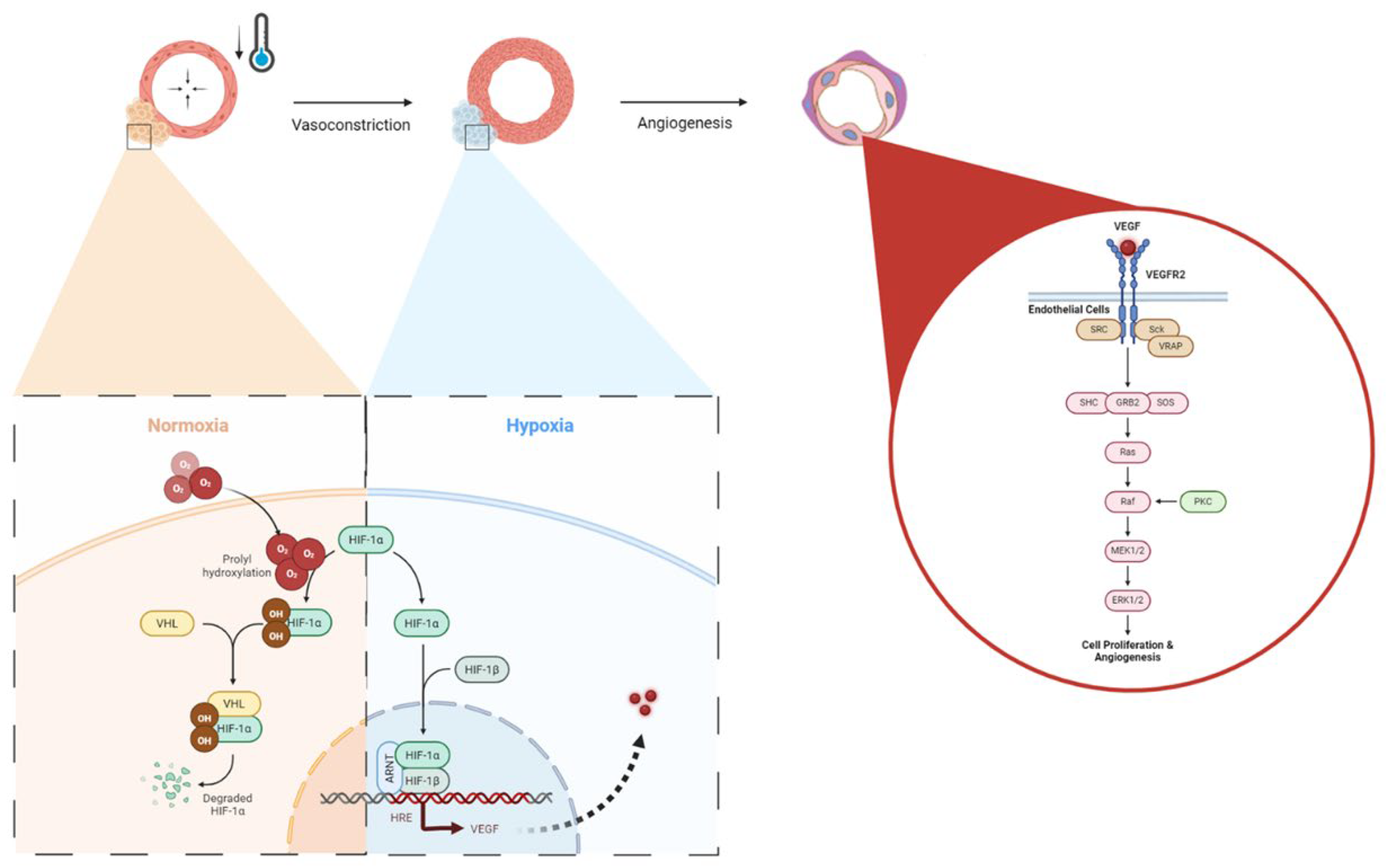
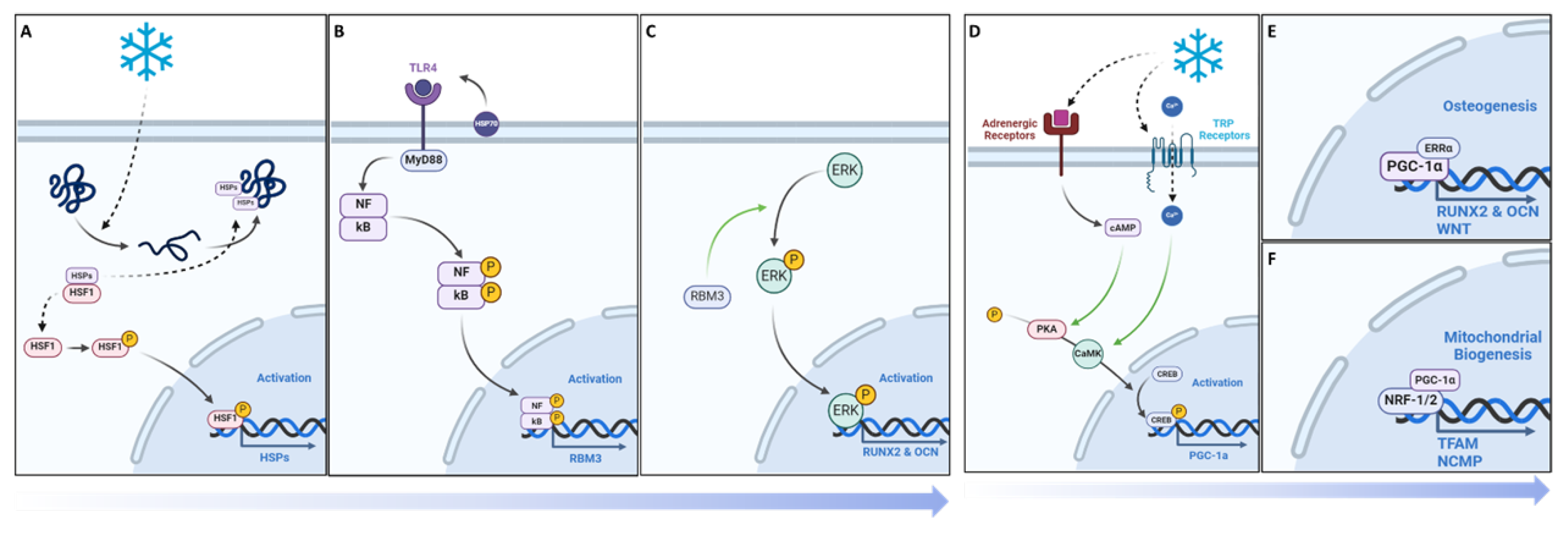
3. Results
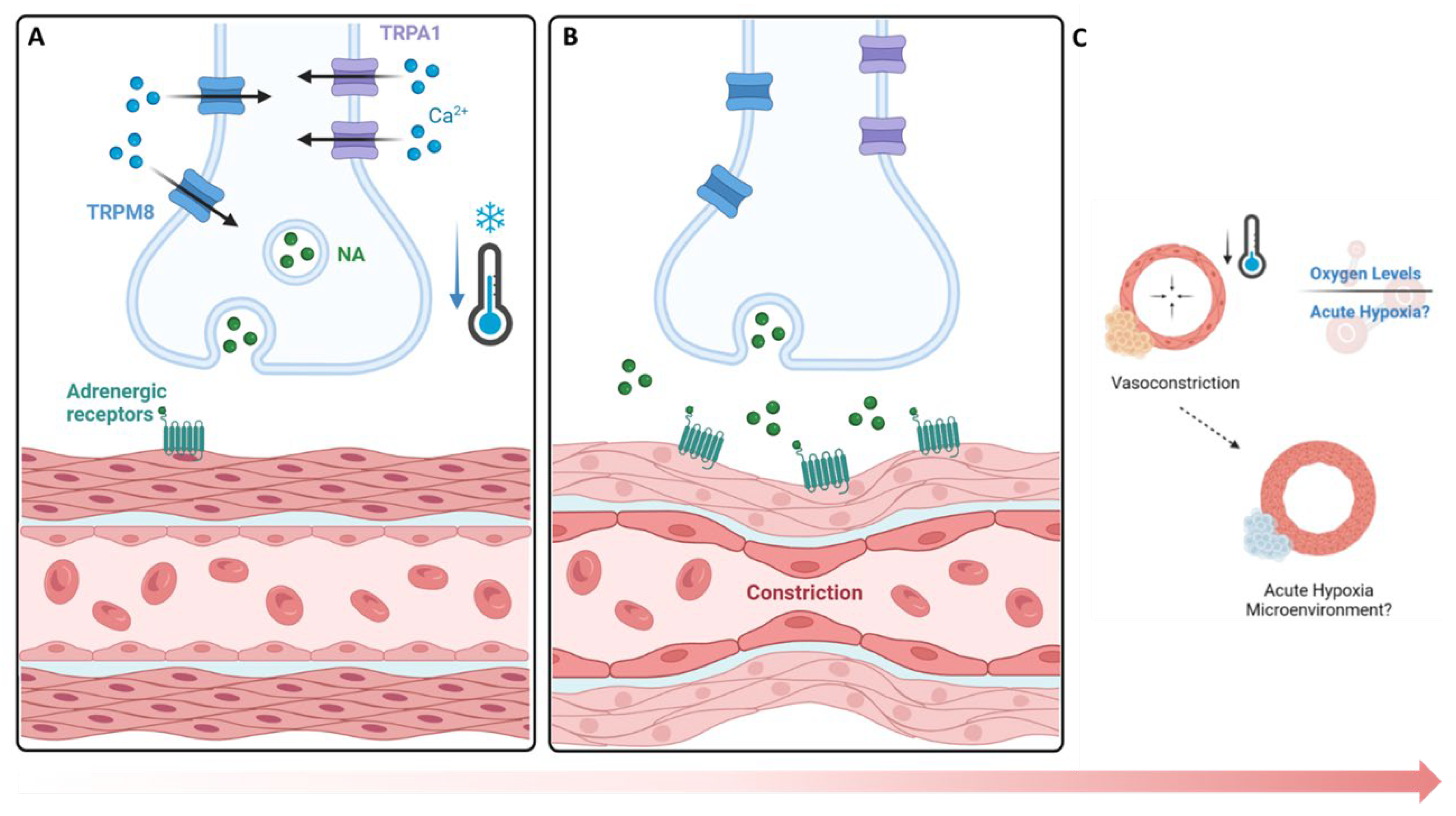
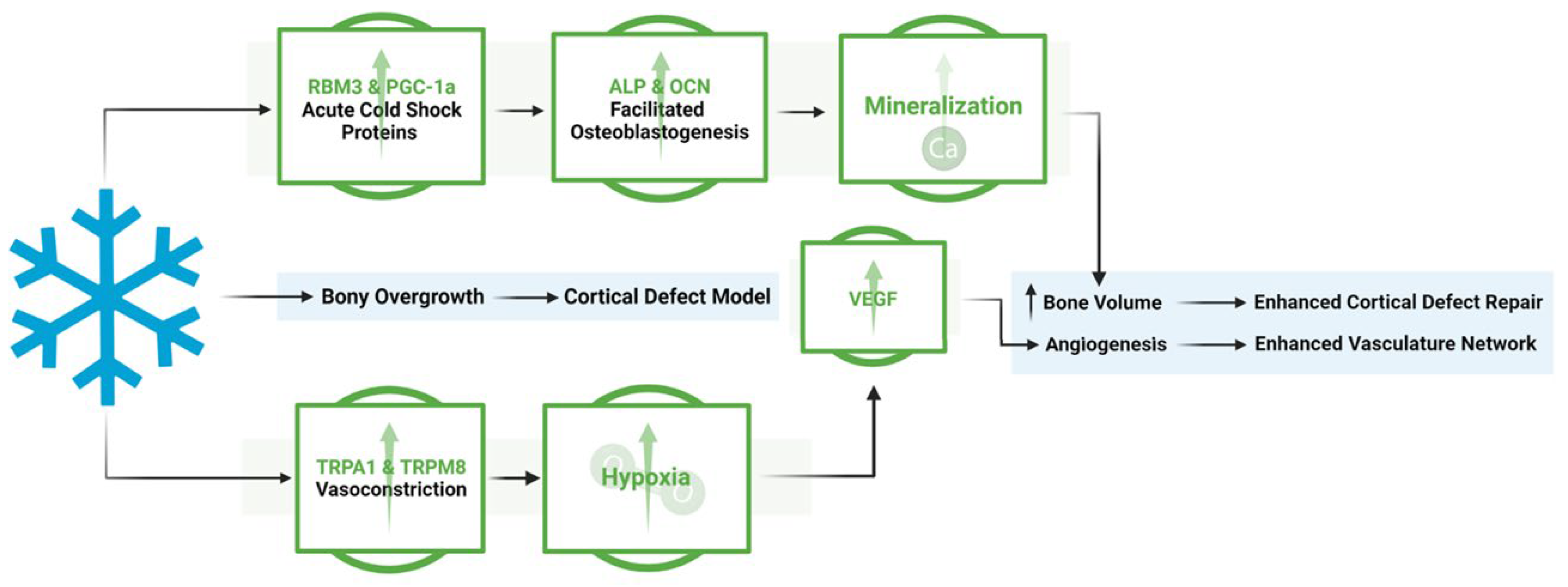
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morgan R, Kalbarczyk A, Mohan D, Jacobs C, Mishra M, Tyagi P, Cox-Roman C, Williamson C. Counting older women: Measuring the health and wellbeing of older women in LMICs. Cell Rep Med. 2024 Jun 18;5(6):101607. [CrossRef]
- Zakaria M, Allard J, Garcia J, Matta J, Honjol Y, Schupbach D, Grant M, Mwale F, Harvey E, Merle G. Enhancing Bone Healing Through Localized Cold Therapy in a Murine Femoral Fracture Model. Tissue Eng Part A. 2024 Aug 7; [CrossRef]
- Castano D, Comeau-Gauthier M, Ramirez-GarciaLuna JL, Drager J, Harvey E, Merle G. Noninvasive Localized Cold Therapy: A New Mode of Bone Repair Enhancement. Tissue Eng Part A. 2019 Apr;25(7–8):554–62. [CrossRef]
- Du J, He Z, Cui J, Li H, Xu M, Zhang S, Zhang S, Yan M, Qu X, Yu Z. Osteocyte Apoptosis Contributes to Cold Exposure-induced Bone Loss. Front Bioeng Biotechnol. 2021;9:733582. [CrossRef]
- Tanaka T, Wakamatsu T, Daijo H, Oda S, Kai S, Adachi T, Kizaka-Kondoh S, Fukuda K, Hirota K. Persisting mild hypothermia suppresses hypoxia-inducible factor-1alpha protein synthesis and hypoxia-inducible factor-1-mediated gene expression. Am J Physiol Regul Integr Comp Physiol. 2010 Mar;298(3):R661-671. [CrossRef]
- Janský L, Pospíšilová D, Honzová S, Uličný B, Šrámek P, Zeman V, Kamínková J. Immune system of cold-exposed and cold-adapted humans. European Journal of Applied Physiology and Occupational Physiology. 1996 Mar 1;72(5):445–50.
- Gregson W, Black MA, Jones H, Milson J, Morton J, Dawson B, Atkinson G, Green DJ. Influence of cold water immersion on limb and cutaneous blood flow at rest. Am J Sports Med. 2011 Jun;39(6):1316–23. [CrossRef]
- FOWLER EP JR, OSMUN PM. NEW BONE GROWTH DUE TO COLD WATER IN THE EARS. Archives of Otolaryngology. 1942 Oct 1;36(4):455–66.
- White GE, Wells GD. Cold-water immersion and other forms of cryotherapy: physiological changes potentially affecting recovery from high-intensity exercise. Extrem Physiol Med. 2013 Sep 1;2(1):26. [CrossRef]
- Iommarini L, Porcelli AM, Gasparre G, Kurelac I. Non-Canonical Mechanisms Regulating Hypoxia-Inducible Factor 1 Alpha in Cancer. Frontiers in Oncology. 2017;7:286. [CrossRef]
- Duvall CL, Taylor WR, Weiss D, Wojtowicz AM, Guldberg RE. Impaired angiogenesis, early callus formation, and late stage remodeling in fracture healing of osteopontin-deficient mice. J Bone Miner Res. 2007 Feb;22(2):286–97. [CrossRef]
- Coassin M, Duncan KG, Bailey KR, Singh A, Schwartz DM. Hypothermia reduces secretion of vascular endothelial growth factor by cultured retinal pigment epithelial cells. Br J Ophthalmol. 2010 Dec;94(12):1678–83. [CrossRef]
- Leegwater NC, Bakker AD, Hogervorst JMA, Nolte PA, Klein-Nulend J. Hypothermia reduces VEGF-165 expression, but not osteogenic differentiation of human adipose stem cells under hypoxia. PLOS ONE. 2017 Feb 6;12(2):e0171492. [CrossRef]
- Mohd Din A, Nor-Ashikin MNK, Ab. Rahim S, Nawawi H, Kapitonova M, Froemming G. Short-term moderate hypothermia stimulates alkaline phosphatase activity and osteocalcin expression in osteoblasts by upregulating Runx2 and osterix in vitro. Experimental Cell Research. 2014 Aug 1;326.
- Kim JC, Yi HK, Hwang PH, Yoon JS, Kim HJ, Kawano F, Ohira Y, Kim CK. Effects of cold-water immersion on VEGF mRNA and protein expression in heart and skeletal muscles of rats. Acta Physiol Scand. 2005 Apr;183(4):389–97. [CrossRef]
- Xue Y, Petrovic N, Cao R, Larsson O, Lim S, Chen S, Feldmann HM, Liang Z, Zhu Z, Nedergaard J, Cannon B, Cao Y. Hypoxia-Independent Angiogenesis in Adipose Tissues during Cold Acclimation. Cell Metabolism. 2009 Jan 7;9(1):99–109. [CrossRef]
- Shakurov AV, Lukina YuS, Skriabin AS, Bionyshev-Abramov LL, Serejnikova NB, Smolencev DV. Enhanced bone healing using local cryostimulation: In vivo rat study. Journal of Thermal Biology. 2023 Apr 1;113:103501. [CrossRef]
- Ihsan M, Abbiss CR, Allan R. Adaptations to Post-exercise Cold Water Immersion: Friend, Foe, or Futile? Front Sports Act Living. 2021;3:714148. [CrossRef]
- Lindquist JA, Mertens PR. Cold shock proteins: from cellular mechanisms to pathophysiology and disease. Cell Communication and Signaling. 2018 Sep 26;16(1):63. [CrossRef]
- Muhlig Nielsen M, Overgaard J, Sørensen JG, Holmstrup M, Justesen J, Loeschcke V. Role of HSF activation for resistance to heat, cold and high-temperature knock-down. Journal of Insect Physiology. 2005 Dec 1;51(12):1320–9. [CrossRef]
- Chen H, Fan W, He H, Huang F. PGC-1: a key regulator in bone homeostasis. J Bone Miner Metab. 2022 Jan;40(1):1–8. [CrossRef]
- Hu Y, Liu Y, Quan X, Fan W, Xu B, Li S. RBM3 is an outstanding cold shock protein with multiple physiological functions beyond hypothermia. J Cell Physiol. 2022 Oct;237(10):3788–802. [CrossRef]
- Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM. A Cold-Inducible Coactivator of Nuclear Receptors Linked to Adaptive Thermogenesis. Cell. 1998 Mar 20;92(6):829–39. [CrossRef]
- Oryan A, Monazzah S, Bigham-Sadegh A. Bone Injury and Fracture Healing Biology. Biomedical and Environmental Sciences. 2015 Jan 1;28(1):57–71. [CrossRef]
- Grzegorz Szczęsny ED1 - Ozgur Karcioglu ED2 - Hakan Topacoglu. Fracture Repair: Its Pathomechanism and Disturbances. In: Trauma Surgery [Internet]. Rijeka: IntechOpen; 2018 [cited 2021 Feb 12]. p. Ch. 1. Available from: . [CrossRef]
- Schoutens A, Bergmann P, Verhas M. Bone blood flow measured by 85 Sr microspheres and bone seeker clearances in the rat. Am J Physiol. 1979 Jan;236(1):H1-6. [CrossRef]
- Pan Y, Thapa D, Baldissera L, Argunhan F, Aubdool AA, Brain SD. Relevance of TRPA1 and TRPM8 channels as vascular sensors of cold in the cutaneous microvasculature. Pflügers Archiv - European Journal of Physiology. 2018 May 1;470(5):779–86. [CrossRef]
- Lieben L, Carmeliet G. The Involvement of TRP Channels in Bone Homeostasis. Frontiers in Endocrinology. 2012;3:99. [CrossRef]
- Earley S, Brayden JE. Transient receptor potential channels in the vasculature. Physiol Rev. 2015 Apr;95(2):645–90. [CrossRef]
- McKemy DD. The molecular and cellular basis of cold sensation. ACS Chem Neurosci. 2013 Feb 20;4(2):238–47. [CrossRef]
- Aubdool AA, Graepel R, Kodji X, Alawi KM, Bodkin JV, Srivastava S, Gentry C, Heads R, Grant AD, Fernandes ES, Bevan S, Brain SD. TRPA1 is essential for the vascular response to environmental cold exposure. Nature Communications. 2014 Dec 11;5(1):5732. [CrossRef]
- Ali E, Birch M, Hopper N, Rushton N, McCaskie AW, Brooks RA. Human osteoblasts obtained from distinct periarticular sites demonstrate differences in biological function in vitro. Bone Joint Res. 2021 Sep;10(9):611–8. [CrossRef]
- Dey P, Rajalaxmi S, Saha P, Thakur PS, Hashmi MA, Lal H, Saini N, Singh N, Ramanathan A. Cold-shock proteome of myoblasts reveals role of RBM3 in promotion of mitochondrial metabolism and myoblast differentiation. Communications Biology. 2024 Apr 30;7(1):515. [CrossRef]
- Xu L, Ma X, Bagattin A, Mueller E. The transcriptional coactivator PGC1α protects against hyperthermic stress via cooperation with the heat shock factor HSF1. Cell Death & Disease. 2016 Feb 1;7(2):e2102–e2102. [CrossRef]
- Krock BL, Skuli N, Simon MC. Hypoxia-induced angiogenesis: good and evil. Genes Cancer. 2011 Dec;2(12):1117–33. [CrossRef]
- Thapa D, Valente J de S, Barrett B, Smith MJ, Argunhan F, Lee SY, Nikitochkina S, Kodji X, Brain SD. Dysfunctional TRPM8 signalling in the vascular response to environmental cold in ageing. Mangoni AA, Barton M, Garami A, editors. eLife. 2021 Nov 2;10:e70153. [CrossRef]
- Arteel GE, Thurman RG, Raleigh JA. Reductive metabolism of the hypoxia marker pimonidazole is regulated by oxygen tension independent of the pyridine nucleotide redox state. Eur J Biochem. 1998 May 1;253(3):743–50. [CrossRef]
- Abhinand CS, Raju R, Soumya SJ, Arya PS, Sudhakaran PR. VEGF-A/VEGFR2 signaling network in endothelial cells relevant to angiogenesis. J Cell Commun Signal. 2016 Dec;10(4):347–54.
- Wang X, Bove AM, Simone G, Ma B. Molecular Bases of VEGFR-2-Mediated Physiological Function and Pathological Role. Frontiers in Cell and Developmental Biology [Internet]. 2020;8. Available from: https://www.frontiersin.org/articles/10.3389/fcell.2020.599281. [CrossRef]
- Negri S, Faris P, Berra-Romani R, Guerra G, Moccia F. Endothelial Transient Receptor Potential Channels and Vascular Remodeling: Extracellular Ca2 + Entry for Angiogenesis, Arteriogenesis and Vasculogenesis. Frontiers in Physiology [Internet]. 2020;10. Available from: https://www.frontiersin.org/journals/physiology/articles/10.3389/fphys.2019.01618. [CrossRef]
- Vrenken KS, Jalink K, Leeuwen FN van, Middelbeek J. Beyond ion-conduction: Channel-dependent and -independent roles of TRP channels during development and tissue homeostasis. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 2016;1863(6, Part B):1436–46. [CrossRef]
- Kim DY, Kim KM, Kim EJ, Jang WG. Hypothermia-induced RNA-binding motif protein 3 (RBM3) stimulates osteoblast differentiation via the ERK signaling pathway. Biochemical and Biophysical Research Communications. 2018 Apr 6;498(3):459–65. [CrossRef]
- Buccoliero C, Dicarlo M, Pignataro P, Gaccione F, Colucci S, Colaianni G, Grano M. The Novel Role of PGC1α in Bone Metabolism. Int J Mol Sci. 2021 Apr 28;22(9). [CrossRef]
- Shi H, Yao R, Lian S, Liu P, Liu Y, Yang YY, Yang H, Li S. Regulating glycolysis, the TLR4 signal pathway and expression of RBM3 in mouse liver in response to acute cold exposure. Stress. 2019 May;22(3):366–76. [CrossRef]
- Hang K, Ye C, Chen E, Zhang W, Xue D, Pan Z. Role of the heat shock protein family in bone metabolism. Cell Stress Chaperones. 2018 Nov;23(6):1153–64. [CrossRef]
- McGee-Lawrence ME, Carpio LR, Bradley EW, Dudakovic A, Lian JB, van Wijnen AJ, Kakar S, Hsu W, Westendorf JJ. Runx2 is required for early stages of endochondral bone formation but delays final stages of bone repair in Axin2-deficient mice. Bone. 2014 Sep;66:277–86. [CrossRef]
- Einhorn TA, Gundberg CM, Devlin VJ, Warman J. Fracture healing and osteocalcin metabolism in vitamin K deficiency. Clin Orthop Relat Res. 1988 Dec;(237):219–25. [CrossRef]
- Nie Y, Yan Z, Yan W, Xia Q, Zhang Y. Cold exposure stimulates lipid metabolism, induces inflammatory response in the adipose tissue of mice and promotes the osteogenic differentiation of BMMSCs via the p38 MAPK pathway in vitro. Int J Clin Exp Pathol. 2015;8(9):10875–86.
- Cheng Z, Li A, Tu CL, Maria CS, Szeto N, Herberger A, Chen TH, Song F, Wang J, Liu X, Shoback DM, Chang W. Calcium-Sensing Receptors in Chondrocytes and Osteoblasts Are Required for Callus Maturation and Fracture Healing in Mice. J Bone Miner Res. 2020 Jan;35(1):143–54. [CrossRef]
- Larson C, Opichka M, McGlynn ML, Collins CW, Slivka D. Exercise- and Cold-Induced Human PGC-1α mRNA Isoform Specific Responses. Int J Environ Res Public Health. 2020 Aug 8;17(16). [CrossRef]
- Zhang Z, Yang D, Xiang J, Zhou J, Cao H, Che Q, Bai Y, Guo J, Su Z. Non-shivering Thermogenesis Signalling Regulation and Potential Therapeutic Applications of Brown Adipose Tissue. Int J Biol Sci. 2021;17(11):2853–70. [CrossRef]
- Wang H, Wang J. Estrogen-related receptor alpha interacts cooperatively with peroxisome proliferator-activated receptor-gamma coactivator-1alpha to regulate osteocalcin gene expression. Cell Biol Int. 2013 Nov;37(11):1259–65. [CrossRef]
- McKee MD, Pedraza CE, Kaartinen MT. Osteopontin and wound healing in bone. Cells Tissues Organs. 2011;194(2–4):313–9. [CrossRef]
- Boonrungsiman S, Gentleman E, Carzaniga R, Evans ND, McComb DW, Porter AE, Stevens MM. The role of intracellular calcium phosphate in osteoblast-mediated bone apatite formation. Proceedings of the National Academy of Sciences. 2012 Aug 28;109(35):14170–5. [CrossRef]
- Abe Y, Fujiwara Y, Takahashi H, Matsumura Y, Sawada T, Jiang S, Nakaki R, Uchida A, Nagao N, Naito M, Kajimura S, Kimura H, Osborne TF, Aburatani H, Kodama T, Inagaki T, Sakai J. Histone demethylase JMJD1A coordinates acute and chronic adaptation to cold stress via thermogenic phospho-switch. Nature Communications. 2018 Apr 19;9(1):1566. [CrossRef]
- Casali C, Galgano L, Zannino L, Siciliani S, Cavallo M, Mazzini G, Biggiogera M. Impact of heat and cold shock on epigenetics and chromatin structure. European Journal of Cell Biology. 2024 Mar 1;103(1):151373. [CrossRef]
- Li JY, Wang TT, Ma L, Zhang Y, Zhu D. Silencing of Jumonji domain-containing 1C inhibits the osteogenic differentiation of bone marrow mesenchymal stem cells via nuclear factor-κB signaling. World J Stem Cells. 2024 Feb 26;16(2):151–62.
- Zeng ZL, Xie H. Mesenchymal stem cell-derived extracellular vesicles: a possible therapeutic strategy for orthopaedic diseases: a narrative review. Biomater Transl. 2022;3(3):175–87. [CrossRef]
- Wang Q. Biomaterials Translational - The New Vehicle for Translational Medicine. Biomater Transl. 2020;1(1):1–2.
- Zhang C, Cai D, Liao P, Su JW, Deng H, Vardhanabhuti B, Ulery BD, Chen SY, Lin J. 4D Printing of shape-memory polymeric scaffolds for adaptive biomedical implantation. Acta Biomaterialia. 2021 Mar 1;122:101–10. [CrossRef]
- El-Husseiny HM, Mady EA, Hamabe L, Abugomaa A, Shimada K, Yoshida T, Tanaka T, Yokoi A, Elbadawy M, Tanaka R. Smart/stimuli-responsive hydrogels: Cutting-edge platforms for tissue engineering and other biomedical applications. Materials Today Bio. 2022 Jan 1;13:100186. [CrossRef]
- Cheng Y, Yu Y, Zhang Y, Zhao G, Zhao Y. Cold-Responsive Nanocapsules Enable the Sole-Cryoprotectant-Trehalose Cryopreservation of β Cell-Laden Hydrogels for Diabetes Treatment. Small. 2019 Dec;15(50):e1904290.
- Molkenova A, Choi HE, Lee G, Baek H, Kwon M, Lee SB, Park JM, Kim JH, Han DW, Park J, Hahn SK, Kim KS. Cold-Responsive Hyaluronated Upconversion Nanoplatform for Transdermal Cryo-Photodynamic Cancer Therapy. Advanced Science. 2024 May 1;11(19):2306684. [CrossRef]
- Kleiter MM, Thrall DE, Malarkey DE, Ji X, Lee DYW, Chou SC, Raleigh JA. A comparison of oral and intravenous pimonidazole in canine tumors using intravenous CCI-103F as a control hypoxia marker. Int J Radiat Oncol Biol Phys. 2006 Feb 1;64(2):592–602. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



