Submitted:
29 August 2024
Posted:
29 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Brief Review of the Mechanisms of 3D Printing
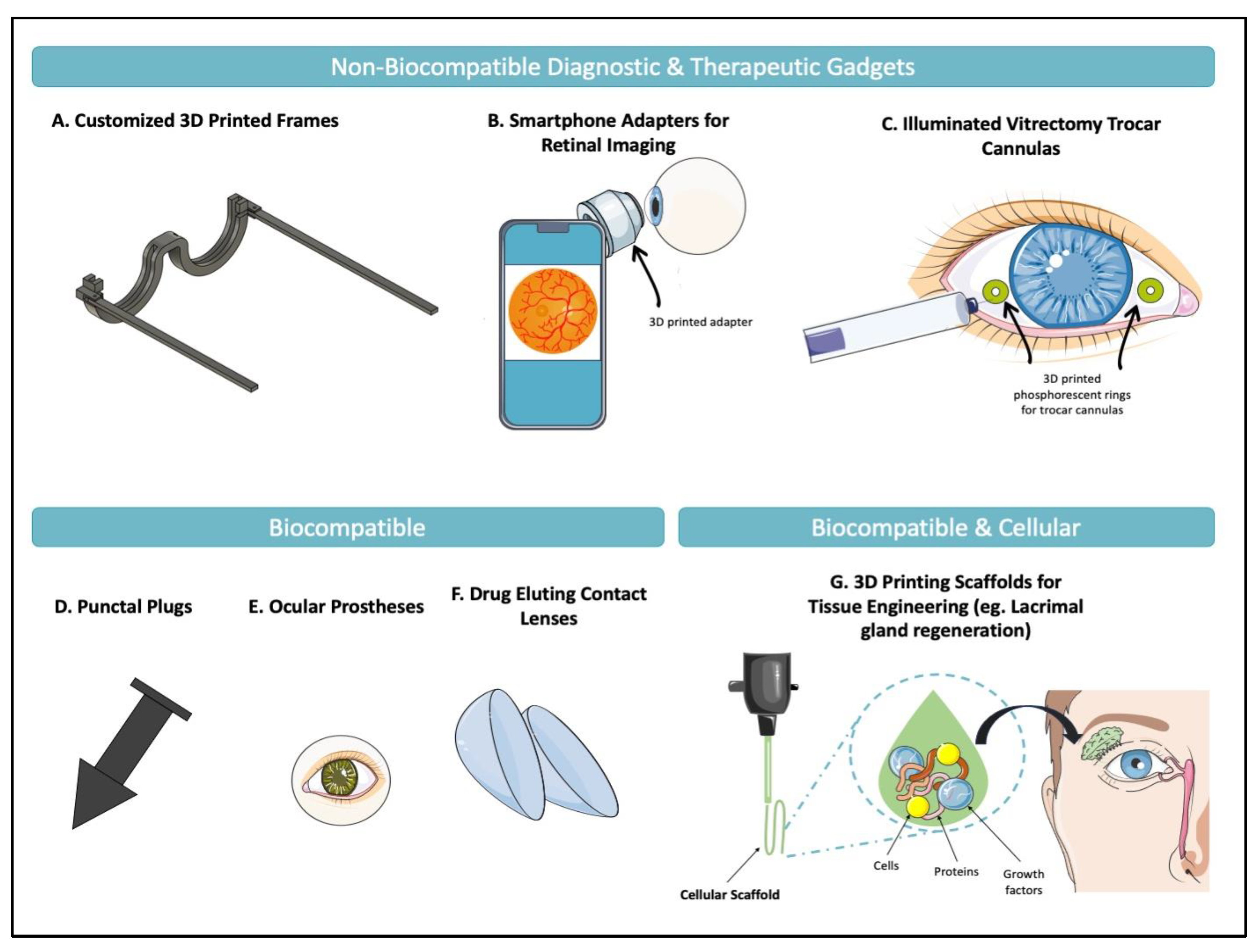
3. 3D Printing in Ophthalmology
3.1. Cornea
3.1.1. Bioink in Corneal Applications
3.1.2. Challenges and Future Prospects in Corneal Bioprinting
3.2. Oculoplastics
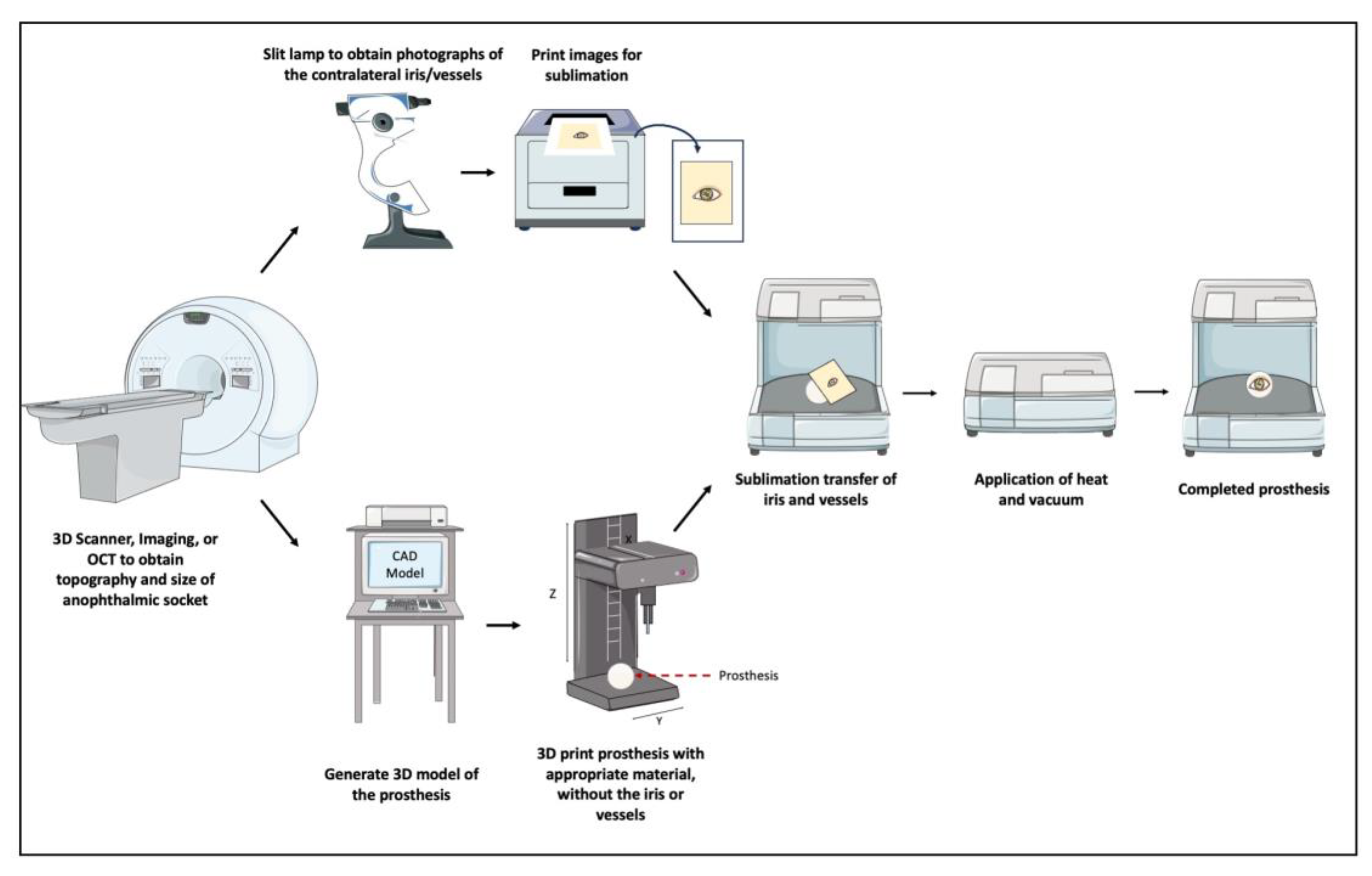
3.2.1. Ocular Prosthetics
3.2.2. Facial & Orbital Implants
3.2.3. Eyelid Crutches
3.2.4. Dry Eye Syndrome: Lacrimal Gland Regeneration & Punctal Plugs
3.3. Drug Delivery Systems – Glaucoma, Retina, & Uveal Melanoma
4. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- D. A. Zopf, S. J. Hollister, M. E. Nelson, R. G. Ohye, and G. E. Green, “Bioresorbable Airway Splint Created with a Three-Dimensional Printer,” N Engl J Med, vol. 368, no. 21, pp. 2043–2045, May 2013. [CrossRef]
- W. Huang and X. Zhang, “3D Printing: Print the Future of Ophthalmology,” Invest. Ophthalmol. Vis. Sci., vol. 55, no. 8, p. 5380, Aug. 2014. [CrossRef]
- J. Reinhard, P. Urban, S. Bell, D. Carpenter, and M. S. Sagoo, “Automatic data-driven design and 3D printing of custom ocular prostheses,” Nat Commun, vol. 15, no. 1, p. 1360, Feb. 2024. [CrossRef]
- A. Isaacson, S. Swioklo, and C. J. Connon, “3D bioprinting of a corneal stroma equivalent,” Experimental Eye Research, vol. 173, pp. 188–193, Aug. 2018. [CrossRef]
- L. Lee et al., “3-D printed spectacles: potential, challenges and the future,” Clinical and Experimental Optometry, vol. 103, no. 5, pp. 590–596, Sep. 2020. [CrossRef]
- Y. Tian, L. Li, and R. Ball, “A Qualitative Study for Parametric Designed Custom-Fit Eyewear Frames: Fit Test Evaluation and User Insights,” in Design, User Experience, and Usability, vol. 14712, A. Marcus, E. Rosenzweig, and M. M. Soares, Eds., in Lecture Notes in Computer Science, vol. 14712. , Cham: Springer Nature Switzerland, 2024, pp. 354–370. [CrossRef]
- A. A. A. Latip et al., “Development of 3D-printed universal adapter in enhancing retinal imaging accessibility,” 3D Print Med, vol. 10, no. 1, p. 23, Jul. 2024,. [CrossRef]
- G. Rubegni et al., “Design of a new 3D printed all-in-one magnetic smartphone adapter for fundus and anterior segment imaging,” European Journal of Ophthalmology, p. 11206721241246187, Apr. 2024. [CrossRef]
- A. Ruzza et al., “Preloaded donor corneal lenticules in a new validated 3D printed smart storage glide for Descemet stripping automated endothelial keratoplasty,” Br J Ophthalmol, vol. 99, no. 10, pp. 1388–1395, Oct. 2015. [CrossRef]
- P. Chandrakanth, S. Verghese, K. Chandrakanth, P. Basaiawmoit, and V. Joseph, “The Glowport – Illuminated vitrectomy trocar cannulas,” Indian Journal of Ophthalmology, Jul. 2024. [CrossRef]
- E. V. Navajas and M. Ten Hove, “Three-Dimensional Printing of a Transconjunctival Vitrectomy Trocar-Cannula System,” Ophthalmologica, vol. 237, no. 2, pp. 119–122, 2017. [CrossRef]
- H. Gómez-Fernández et al., “Comprehensive review of the state-of-the-art in corneal 3D bioprinting, including regulatory aspects,” International Journal of Pharmaceutics, vol. 662, p. 124510, Sep. 2024. [CrossRef]
- A. Hu and K. F. Damji, “New open source 3-dimensional printed smartphone fundus imaging adaptor,” Canadian Journal of Ophthalmology, vol. 54, no. 3, pp. 399–400, Jun. 2019. [CrossRef]
- P. Gain et al., “Global Survey of Corneal Transplantation and Eye Banking,” JAMA Ophthalmol, vol. 134, no. 2, p. 167, Feb. 2016. [CrossRef]
- B. H. Jeng and S. Ahmad, “In Pursuit of the Elimination of Corneal Blindness: Is Establishing Eye Banks and Training Surgeons Enough?,” Ophthalmology, vol. 128, no. 6, pp. 813–815, Jun. 2021. [CrossRef]
- L. Balters and S. Reichl, “3D bioprinting of corneal models: A review of the current state and future outlook,” J Tissue Eng, vol. 14, p. 20417314231197793, Jan. 2023. [CrossRef]
- P. M. Mathews, K. Lindsley, A. J. Aldave, and E. K. Akpek, “Etiology of Global Corneal Blindness and Current Practices of Corneal Transplantation: A Focused Review,” Cornea, vol. 37, no. 9, pp. 1198–1203, Sep. 2018. [CrossRef]
- M. Oliva, T. Schottman, and M. Gulati, “Turning the tide of corneal blindness,” Indian J Ophthalmol, vol. 60, no. 5, p. 423, 2012. [CrossRef]
- E. Moraru, G. O. Dontu, S. Cananau, and V.-A. Stanescu, “Approaches and Processing Technologies for Medical Devices: Considerations from Micro- and Macroscale Perspectives,” in International Conference on Reliable Systems Engineering (ICoRSE) - 2023, vol. 762, D. D. Cioboată, Ed., in Lecture Notes in Networks and Systems, vol. 762. , Cham: Springer Nature Switzerland, 2023, pp. 345–362. [CrossRef]
- M. H. Mobarak et al., “Recent advances of additive manufacturing in implant fabrication – A review,” Applied Surface Science Advances, vol. 18, p. 100462, Dec. 2023. [CrossRef]
- A. Haleem and M. Javaid, “Role of CT and MRI in the design and development of orthopaedic model using additive manufacturing,” Journal of Clinical Orthopaedics and Trauma, vol. 9, no. 3, pp. 213–217, Jul. 2018. [CrossRef]
- C. Dong, M. Petrovic, and I. J. Davies, “Applications of 3D printing in medicine: A review,” Annals of 3D Printed Medicine, vol. 14, p. 100149, May 2024. [CrossRef]
- N. Lin, M. Gagnon, and K. Y. Wu, “The Third Dimension of Eye Care: A Comprehensive Review of 3D Printing in Ophthalmology,” Hardware, vol. 2, no. 1, pp. 1–32, Jan. 2024. [CrossRef]
- X. Nie et al., “3D printing sequentially strengthening high-strength natural polymer hydrogel bilayer scaffold for cornea regeneration,” Regenerative Biomaterials, vol. 11, p. rbae012, Jan. 2024. [CrossRef]
- M. Sridhar, “Anatomy of cornea and ocular surface,” Indian J Ophthalmol, vol. 66, no. 2, p. 190, 2018. [CrossRef]
- H. S. Dua, L. A. Faraj, D. G. Said, T. Gray, and J. Lowe, “Human Corneal Anatomy Redefined,” Ophthalmology, vol. 120, no. 9, pp. 1778–1785, Sep. 2013. [CrossRef]
- L. E. Downie et al., “BCLA CLEAR - Anatomy and physiology of the anterior eye,” Contact Lens and Anterior Eye, vol. 44, no. 2, pp. 132–156, Apr. 2021. [CrossRef]
- J. W. Ruberti and J. D. Zieske, “Prelude to corneal tissue engineering – Gaining control of collagen organization,” Progress in Retinal and Eye Research, vol. 27, no. 5, pp. 549–577, Sep. 2008. [CrossRef]
- L. J. Muller, “The specific architecture of the anterior stroma accounts for maintenance of corneal curvature,” British Journal of Ophthalmology, vol. 85, no. 4, pp. 437–443, Apr. 2001. [CrossRef]
- P. S. Gungor-Ozkerim, I. Inci, Y. S. Zhang, A. Khademhosseini, and M. R. Dokmeci, “Bioinks for 3D bioprinting: an overview,” Biomater. Sci., vol. 6, no. 5, pp. 915–946, 2018. [CrossRef]
- A. Sorkio et al., “Human stem cell based corneal tissue mimicking structures using laser-assisted 3D bioprinting and functional bioinks,” Biomaterials, vol. 171, pp. 57–71, Jul. 2018. [CrossRef]
- D. F. Duarte Campos et al., “Hand-held bioprinting for de novo vascular formation applicable to dental pulp regeneration,” Connective Tissue Research, vol. 61, no. 2, pp. 205–215, Mar. 2020. [CrossRef]
- Y. Liu, L. Ren, and Y. Wang, “Crosslinked collagen–gelatin–hyaluronic acid biomimetic film for cornea tissue engineering applications,” Materials Science and Engineering: C, vol. 33, no. 1, pp. 196–201, Jan. 2013. [CrossRef]
- S. Kutlehria, T. C. Dinh, A. Bagde, N. Patel, A. Gebeyehu, and M. Singh, “High-throughput 3D bioprinting of corneal stromal equivalents,” J Biomed Mater Res, vol. 108, no. 7, pp. 2981–2994, Oct. 2020. [CrossRef]
- Z. Wu, X. Su, Y. Xu, B. Kong, W. Sun, and S. Mi, “Bioprinting three-dimensional cell-laden tissue constructs with controllable degradation,” Sci Rep, vol. 6, no. 1, p. 24474, Apr. 2016. [CrossRef]
- M. Nikkhah, M. Akbari, A. Paul, A. Memic, A. Dolatshahi-Pirouz, and A. Khademhosseini, “Gelatin-Based Biomaterials For Tissue Engineering And Stem Cell Bioengineering,” in Biomaterials from Nature for Advanced Devices and Therapies, 1st ed., N. M. Neves and R. L. Reis, Eds., Wiley, 2016, pp. 37–62. [CrossRef]
- K. Tonsomboon and M. L. Oyen, “Composite electrospun gelatin fiber-alginate gel scaffolds for mechanically robust tissue engineered cornea,” Journal of the Mechanical Behavior of Biomedical Materials, vol. 21, pp. 185–194, May 2013. [CrossRef]
- C. Kilic Bektas and V. Hasirci, “Cell loaded 3D bioprinted GelMA hydrogels for corneal stroma engineering,” Biomater. Sci., vol. 8, no. 1, pp. 438–449, 2020. [CrossRef]
- S. S. Mahdavi, M. J. Abdekhodaie, H. Kumar, S. Mashayekhan, A. Baradaran-Rafii, and K. Kim, “Stereolithography 3D Bioprinting Method for Fabrication of Human Corneal Stroma Equivalent,” Ann Biomed Eng, vol. 48, no. 7, pp. 1955–1970, Jul. 2020. [CrossRef]
- R. Vijayaraghavan, S. Loganathan, and R. B. Valapa, “Fabrication of GelMA – Agarose Based 3D Bioprinted Photocurable Hydrogel with In Vitro Cytocompatibility and Cells Mirroring Natural Keratocytes for Corneal Stromal Regeneration,” Macromolecular Bioscience, p. 2400136, Aug. 2024. [CrossRef]
- U. Bhutani et al., “Biopolymeric corneal lenticules by digital light processing based bioprinting: a dynamic substitute for corneal transplant,” Biomed. Mater., vol. 19, no. 3, p. 035017, May 2024. [CrossRef]
- S. Ulag et al., “3D printed artificial cornea for corneal stromal transplantation,” European Polymer Journal, vol. 133, p. 109744, Jun. 2020. [CrossRef]
- H. Kim, M.-N. Park, J. Kim, J. Jang, H.-K. Kim, and D.-W. Cho, “Characterization of cornea-specific bioink: high transparency, improved in vivo safety,” J Tissue Eng, vol. 10, p. 204173141882338, Jan. 2019. [CrossRef]
- M. Zhang et al., “3D bioprinting of corneal decellularized extracellular matrix: GelMA composite hydrogel for corneal stroma engineering,” IJB, vol. 9, no. 5, p. 774, Jun. 2023. [CrossRef]
- M. Uyanıklar, G. Günal, A. Tevlek, P. Hosseinian, and H. M. Aydin, “Hybrid Cornea: Cell Laden Hydrogel Incorporated Decellularized Matrix,” ACS Biomater. Sci. Eng., vol. 6, no. 1, pp. 122–133, Jan. 2020. [CrossRef]
- A. Kostenko, S. Swioklo, and C. J. Connon, “Alginate in corneal tissue engineering,” Biomed. Mater., Jan. 2022. [CrossRef]
- B. Zhang et al., “Integrated 3D bioprinting-based geometry-control strategy for fabricating corneal substitutes,” J. Zhejiang Univ. Sci. B, vol. 20, no. 12, pp. 945–959, Dec. 2019. [CrossRef]
- M. Sun et al., “Hyaluronan Derived From the Limbus is a Key Regulator of Corneal Lymphangiogenesis,” Invest. Ophthalmol. Vis. Sci., vol. 60, no. 4, p. 1050, Mar. 2019. [CrossRef]
- A. Mörö et al., “Hyaluronic acid based next generation bioink for 3D bioprinting of human stem cell derived corneal stromal model with innervation,” Biofabrication, vol. 15, no. 1, p. 015020, Jan. 2023. [CrossRef]
- Z. Zhong et al., “Bioprinting of dual ECM scaffolds encapsulating limbal stem/progenitor cells in active and quiescent statuses,” Biofabrication, vol. 13, no. 4, p. 044101, Oct. 2021. [CrossRef]
- M. Kumar Singh, “Textiles Functionalization - A Review of Materials, Processes, and Assessment,” in Textiles for Functional Applications, B. Kumar, Ed., IntechOpen, 2021. [CrossRef]
- P. Grönroos et al., “Bioprinting of human pluripotent stem cell derived corneal endothelial cells with hydrazone crosslinked hyaluronic acid bioink,” Stem Cell Res Ther, vol. 15, no. 1, p. 81, Mar. 2024,. [CrossRef]
- K. W. Kim, S. J. Lee, S. H. Park, and J. C. Kim, “Ex Vivo Functionality of 3D Bioprinted Corneal Endothelium Engineered with Ribonuclease 5-Overexpressing Human Corneal Endothelial Cells,” Adv Healthcare Materials, vol. 7, no. 18, p. 1800398, Sep. 2018. [CrossRef]
- G. L. Duffy, H. Liang, R. L. Williams, D. A. Wellings, and K. Black, “3D reactive inkjet printing of poly-ɛ-lysine/gellan gum hydrogels for potential corneal constructs,” Materials Science and Engineering: C, vol. 131, p. 112476, Dec. 2021. [CrossRef]
- B. He et al., “3D printed biomimetic epithelium/stroma bilayer hydrogel implant for corneal regeneration,” Bioactive Materials, vol. 17, pp. 234–247, Nov. 2022. [CrossRef]
- A. L. De Araujo, “Corneal stem cells and tissue engineering: Current advances and future perspectives,” WJSC, vol. 7, no. 5, p. 806, 2015. [CrossRef]
- D. Karamichos et al., “A Role for Topographic Cues in the Organization of Collagenous Matrix by Corneal Fibroblasts and Stem Cells,” PLoS ONE, vol. 9, no. 1, p. e86260, Jan. 2014. [CrossRef]
- R. M. Gouveia, G. Lepert, S. Gupta, R. R. Mohan, C. Paterson, and C. J. Connon, “Assessment of corneal substrate biomechanics and its effect on epithelial stem cell maintenance and differentiation,” Nat Commun, vol. 10, no. 1, p. 1496, Apr. 2019. [CrossRef]
- A. Modugno, F. Mantelli, S. Sposato, C. Moretti, A. Lambiase, and S. Bonini, “Ocular prostheses in the last century: a retrospective analysis of 8018 patients,” Eye, vol. 27, no. 7, pp. 865–870, Jul. 2013. [CrossRef]
- A. L. W. Groot, J. S. Remmers, and D. T. Hartong, “Three-Dimensional Computer-Aided Design of a Full-Color Ocular Prosthesis with Textured Iris and Sclera Manufactured in One Single Print Job,” 3D Printing and Additive Manufacturing, vol. 8, no. 6, pp. 343–348, Dec. 2021. [CrossRef]
- R. Gunaseelaraj, S. Karthikeyan, M. Kumar, T. Balamurugan, and A. Jagadeeshwaran, “Custom-made ocular prosthesis,” J Pharm Bioall Sci, vol. 4, no. 6, p. 177, 2012,. [CrossRef]
- M. C. Goiato, L. C. Bannwart, M. F. Haddad, D. M. Dos Santos, A. A. Pesqueira, and G. I. Miyahara, “Fabrication Techniques for Ocular Prostheses – An Overview,” Orbit, vol. 33, no. 3, pp. 229–233, Jun. 2014. [CrossRef]
- M. Van Der Stelt et al., “Improving Lives in Three Dimensions: The Feasibility of 3D Printing for Creating Personalized Medical Aids in a Rural Area of Sierra Leone,” The American Journal of Tropical Medicine and Hygiene, vol. 102, no. 4, pp. 905–909, Apr. 2020. [CrossRef]
- A. J. Sterkenburg et al., “Quality of life of patients with 3D-printed arm prostheses in a rural area of Sierra Leone,” Heliyon, vol. 7, no. 7, p. e07447, Jul. 2021. [CrossRef]
- S. Ruiters, Y. Sun, S. De Jong, C. Politis, and I. Mombaerts, “Computer-aided design and three-dimensional printing in the manufacturing of an ocular prosthesis,” Br J Ophthalmol, vol. 100, no. 7, pp. 879–881, Jul. 2016. [CrossRef]
- N. Puls, D. Carluccio, M. D. Batstone, and J. I. Novak, “The rise of additive manufacturing for ocular and orbital prostheses: A systematic literature review,” Annals of 3D Printed Medicine, vol. 4, p. 100036, Dec. 2021. [CrossRef]
- B. R. Kim et al., “A Pilot Clinical Study of Ocular Prosthesis Fabricated by Three-dimensional Printing and Sublimation Technique,” Korean J Ophthalmol, vol. 35, no. 1, pp. 37–43, Feb. 2021. [CrossRef]
- S.-Y. Park et al., “Custom-made artificial eyes using 3D printing for dogs: A preliminary study,” PLoS ONE, vol. 15, no. 11, p. e0242274, Nov. 2020. [CrossRef]
- Md. S. Alam, M. Sugavaneswaran, G. Arumaikkannu, and B. Mukherjee, “An innovative method of ocular prosthesis fabrication by bio-CAD and rapid 3-D printing technology: A pilot study,” Orbit, vol. 36, no. 4, pp. 223–227, Jul. 2017. [CrossRef]
- J. Ko, S. H. Kim, S. W. Baek, M. K. Chae, and J. S. Yoon, “Semi-automated fabrication of customized ocular prosthesis with three–dimensional printing and sublimation transfer printing technology,” Sci Rep, vol. 9, no. 1, p. 2968, Feb. 2019. [CrossRef]
- R. B. Kormann, R. Mörschbächer, H. Moreira, and P. Akaishi, “A three-dimensional printed photopolymer resin implant for orbital rehabilitation for evisceration,” Arquivos Brasileiros de Oftalmologia, vol. 82, no. 6, 2019. [CrossRef]
- Y. C. Kim, W. S. Jeong, T. Park, J. W. Choi, K. S. Koh, and T. S. Oh, “The accuracy of patient specific implant prebented with 3D-printed rapid prototype model for orbital wall reconstruction,” Journal of Cranio-Maxillofacial Surgery, vol. 45, no. 6, pp. 928–936, Jun. 2017,. [CrossRef]
- S. Kang et al., “Generation of customized orbital implant templates using 3-dimensional printing for orbital wall reconstruction,” Eye, vol. 32, no. 12, pp. 1864–1870, Dec. 2018. [CrossRef]
- A. Murray-Douglass, C. Snoswell, C. Winter, and R. Harris, “Three-dimensional (3D) printing for post-traumatic orbital reconstruction, a systematic review and meta-analysis,” British Journal of Oral and Maxillofacial Surgery, vol. 60, no. 9, pp. 1176–1183, Nov. 2022. [CrossRef]
- S. Mukai, T. Tsuge, S. Akaishi, R. Ogawa, and H. Kuwahara, “Utilizing 3D Printing for the Surgical Management of Orbital Floor Fractures,” Plastic and Reconstructive Surgery - Global Open, vol. 11, no. 11, p. e5433, Nov. 202. [CrossRef]
- A. B. Callahan, A. A. Campbell, C. Petris, and M. Kazim, “Low-Cost 3D Printing Orbital Implant Templates in Secondary Orbital Reconstructions,” Ophthalmic Plastic & Reconstructive Surgery, vol. 33, no. 5, pp. 376–380, Sep. 2017. [CrossRef]
- E. H. Weisson, M. Fittipaldi, C. A. Concepcion, D. Pelaez, L. Grace, and D. T. Tse, “Automated Noncontact Facial Topography Mapping, 3-Dimensional Printing, and Silicone Casting of Orbital Prosthesis,” American Journal of Ophthalmology, vol. 220, pp. 27–36, Dec. 2020. [CrossRef]
- T. S. Oh, W. S. Jeong, T. J. Chang, K. S. Koh, and J.-W. Choi, “Customized Orbital Wall Reconstruction Using Three-Dimensionally Printed Rapid Prototype Model in Patients With Orbital Wall Fracture,” Journal of Craniofacial Surgery, vol. 27, no. 8, pp. 2020–2024, Nov. 2016. [CrossRef]
- D. L. Mourits et al., “3D Orbital Reconstruction in a Patient with Microphthalmos and a Large Orbital Cyst—A Case Report,” Ophthalmic Genetics, vol. 37, no. 2, pp. 233–237, Apr. 2016. [CrossRef]
- M. Vehmeijer, M. Van Eijnatten, N. Liberton, and J. Wolff, “A Novel Method of Orbital Floor Reconstruction Using Virtual Planning, 3-Dimensional Printing, and Autologous Bone,” Journal of Oral and Maxillofacial Surgery, vol. 74, no. 8, pp. 1608–1612, Aug. 2016. [CrossRef]
- D. Amin, N. Nguyen, A. J. Manhan, J. H. Kim, S. M. Roser, and G. F. Bouloux, “Does a Point-of-Care 3-Dimensional Printer Result in a Decreased Length of Surgery for Orbital Fractures?,” Journal of Oral and Maxillofacial Surgery, p. S0278239124005974, Jul. 2024. [CrossRef]
- A. Tel et al., “Endoscopically assisted computer-guided repair of internal orbital floor fractures: an updated protocol for minimally invasive management,” Journal of Cranio-Maxillofacial Surgery, vol. 47, no. 12, pp. 1943–1951, Dec. 2019. [CrossRef]
- N. B Jamayet, Y. J Abdullah, Z. A Rajion, A. Husein, and M. K Alam, “New Approach to 3D Printing of Facial Prostheses Using Combination of Open Source Software and Conventional Techniques: A Case Report,” Bull. Tokyo Dent. Coll., vol. 58, no. 2, pp. 117–124, 2017. [CrossRef]
- M. G. Sun, D. Rojdamrongratana, M. I. Rosenblatt, V. K. Aakalu, and C. Q. Yu, “3D printing for low cost, rapid prototyping of eyelid crutches,” Orbit, vol. 38, no. 4, pp. 342–346, Jul. 2019. [CrossRef]
- O. Lapid, “Eyelid Crutches for Ptosis: A Forgotten Solution,” Plastic and Reconstructive Surgery, vol. 106, no. 5, pp. 1213–1214, Oct. 2000.
- D. Wróbel-Dudzińska, N. Osial, P. W. Stępień, A. Gorecka, and T. Żarnowski, “Prevalence of Dry Eye Symptoms and Associated Risk Factors among University Students in Poland,” IJERPH, vol. 20, no. 2, p. 1313, Jan. 2023. [CrossRef]
- C. Chan, S. Ziai, V. Myageri, J. G. Burns, and C. L. Prokopich, “Economic burden and loss of quality of life from dry eye disease in Canada,” BMJ Open Ophth, vol. 6, no. 1, p. e000709, Sep. 2021. [CrossRef]
- A. C. Lieu, M. K. Shoji, V. K. Aakalu, and C. Y. Liu, “Approaches to Restoring Lacrimal Gland Function: From stem Cells to Tissue Engineering,” Curr Ophthalmol Rep, Jul. 2024. [CrossRef]
- C. Adine, K. K. Ng, S. Rungarunlert, G. R. Souza, and J. N. Ferreira, “Engineering innervated secretory epithelial organoids by magnetic three-dimensional bioprinting for stimulating epithelial growth in salivary glands,” Biomaterials, vol. 180, pp. 52–66, Oct. 2018. [CrossRef]
- X. Xu et al., “3D Printed Punctal Plugs for Controlled Ocular Drug Delivery,” Pharmaceutics, vol. 13, no. 9, p. 1421, Sep. 2021. [CrossRef]
- T. Khanna, J. Akkara, V. Bawa, and E. Sargunam, “Designing and making an open source, 3D-printed, punctal plug with drug delivery system,” Indian J Ophthalmol, vol. 71, no. 1, p. 297, 2023. [CrossRef]
- M. M. Marcet et al., “Safety and Efficacy of Lacrimal Drainage System Plugs for Dry Eye Syndrome,” Ophthalmology, vol. 122, no. 8, pp. 1681–1687, Aug. 2015. [CrossRef]
- J. Gayton, “Etiology, prevalence, and treatment of dry eye disease,” OPTH, p. 405, Jul. 2009. [CrossRef]
- R. B. Singh, P. Ichhpujani, S. Thakur, and S. Jindal, “Promising therapeutic drug delivery systems for glaucoma: a comprehensive review,” Ophthalmol Eye Dis, vol. 12, p. 251584142090574, Jan. 2020. [CrossRef]
- L. Quaranta, A. Novella, M. Tettamanti, L. Pasina, R. N. Weinreb, and A. Nobili, “Adherence and Persistence to Medical Therapy in Glaucoma: An Overview,” Ophthalmol Ther, vol. 12, no. 5, pp. 2227–2240, Oct. 2023. [CrossRef]
- L. Tamrat, G. Gessesse, and Y. Gelaw, “Adherence to topical glaucoma medications in Ethiopian patients,” Middle East Afr J Ophthalmol, vol. 22, no. 1, p. 59, 2015. [CrossRef]
- F. M. Wagner, A. K. Schuster, K. Kianusch, J. Stingl, N. Pfeiffer, and E. M. Hoffmann, “Long-term success after trabeculectomy in open-angle glaucoma: results of a retrospective cohort study,” BMJ Open, vol. 13, no. 2, p. e068403, Feb. 2023. [CrossRef]
- S. J. Gedde, J. C. Schiffman, W. J. Feuer, L. W. Herndon, J. D. Brandt, and D. L. Budenz, “Treatment Outcomes in the Tube Versus Trabeculectomy (TVT) Study After Five Years of Follow-up,” American Journal of Ophthalmology, vol. 153, no. 5, pp. 789-803.e2, May 2012. [CrossRef]
- N. Ioannou et al., “3D-printed long-acting 5-fluorouracil implant to prevent conjunctival fibrosis in glaucoma,” Journal of Pharmacy and Pharmacology, vol. 75, no. 2, pp. 276–286, Feb. 2023. [CrossRef]
- Y. M. G. Mohamdeen et al., “Development of 3D printed drug-eluting contact lenses,” Journal of Pharmacy and Pharmacology, vol. 74, no. 10, pp. 1467–1476, Oct. 2022,. [CrossRef]
- F. Alam et al., “3D Printed Contact Lenses,” ACS Biomater. Sci. Eng., vol. 7, no. 2, pp. 794–803, Feb. 2021. [CrossRef]
- M. Hisham, A. E. Salih, and H. Butt, “3D Printing of Multimaterial Contact Lenses,” ACS Biomater. Sci. Eng., vol. 9, no. 7, pp. 4381–4391, Jul. 2023. [CrossRef]
- F. Zhao, J. Wang, L. Wang, and L. Chen, “An approach for simulating the fitting of rigid gas-permeable contact lenses using 3D printing technology,” Contact Lens and Anterior Eye, vol. 42, no. 2, pp. 165–169, Apr. 2019. [CrossRef]
- S. Hittini et al., “Fabrication of 3D-Printed Contact Lenses and Their Potential as Color Blindness Ocular Aids,” Macro Materials & Eng, vol. 308, no. 5, p. 2200601, May 2023. [CrossRef]
- F. Alam et al., “Prospects for Additive Manufacturing in Contact Lens Devices,” Adv Eng Mater, vol. 23, no. 1, p. 2000941, Jan. 2021,. [CrossRef]
- J. Y. Won et al., “3D printing of drug-loaded multi-shell rods for local delivery of bevacizumab and dexamethasone: A synergetic therapy for retinal vascular diseases,” Acta Biomaterialia, vol. 116, pp. 174–185, Oct. 2020. [CrossRef]
- T. Lescot et al., “Tumor Shape-Specific Brachytherapy Implants by 3D-Printing, Precision Radioactivity Painting, and Biomedical Imaging,” Adv Healthcare Materials, vol. 12, no. 25, p. 2300528, Oct. 2023. [CrossRef]
- F. Alam, M. Ali, M. Elsherif, A. E. Salih, N. El-Atab, and H. Butt, “3D printed intraocular lens for managing the color blindness,” Additive Manufacturing Letters, vol. 5, p. 100129, Apr. 2023,. [CrossRef]
- R. Raveendran et al., “Current Innovations in Intraocular Pressure Monitoring Biosensors for Diagnosis and Treatment of Glaucoma—Novel Strategies and Future Perspectives,” Biosensors, vol. 13, no. 6, p. 663, Jun. 2023. [CrossRef]
- K. A. Ramirez, L. E. Drew-Bear, M. Vega-Garces, H. Betancourt-Belandria, and J. F. Arevalo, “An update on visual prosthesis,” Int J Retin Vitr, vol. 9, no. 1, p. 73, Nov. 2023. [CrossRef]
| Biomaterial | Advantages | Disadvantages | References |
|---|---|---|---|
| Collagen |
|
|
[28]: collagen-alginate[31]: collagen-laminin[32]: collagen-agarose[33]: collagen–gelatin-hyaluronic acid[34,35]: collagen-alginate-gelatin |
| Gelatin |
|
|
[37]: Electrospun gelatin nanofibers + infiltrated alginate [38,39]: GelMA[40]: GelMA-agarose[41]: GelMA-HAMA* |
| Chitosan |
|
|
[42]: Chitosan + PVA* |
| dECM* |
|
|
[43]: dECM[44,45]: dECM+GelMA |
| Alginate |
|
[28]: collagen-alginate[34,35]: collagen-alginate-gelatin[47]: alginate-gelatin | |
| HA* |
|
|
[49]: HA-carbodihydrazide + HA-aldehyde + collagen & HA-carbodihydrazide-dopamine + HA-aldehyde + collagen[50]: HA glycidyl methacrylate + GelMA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




