Submitted:
27 August 2024
Posted:
28 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Theoretical Background
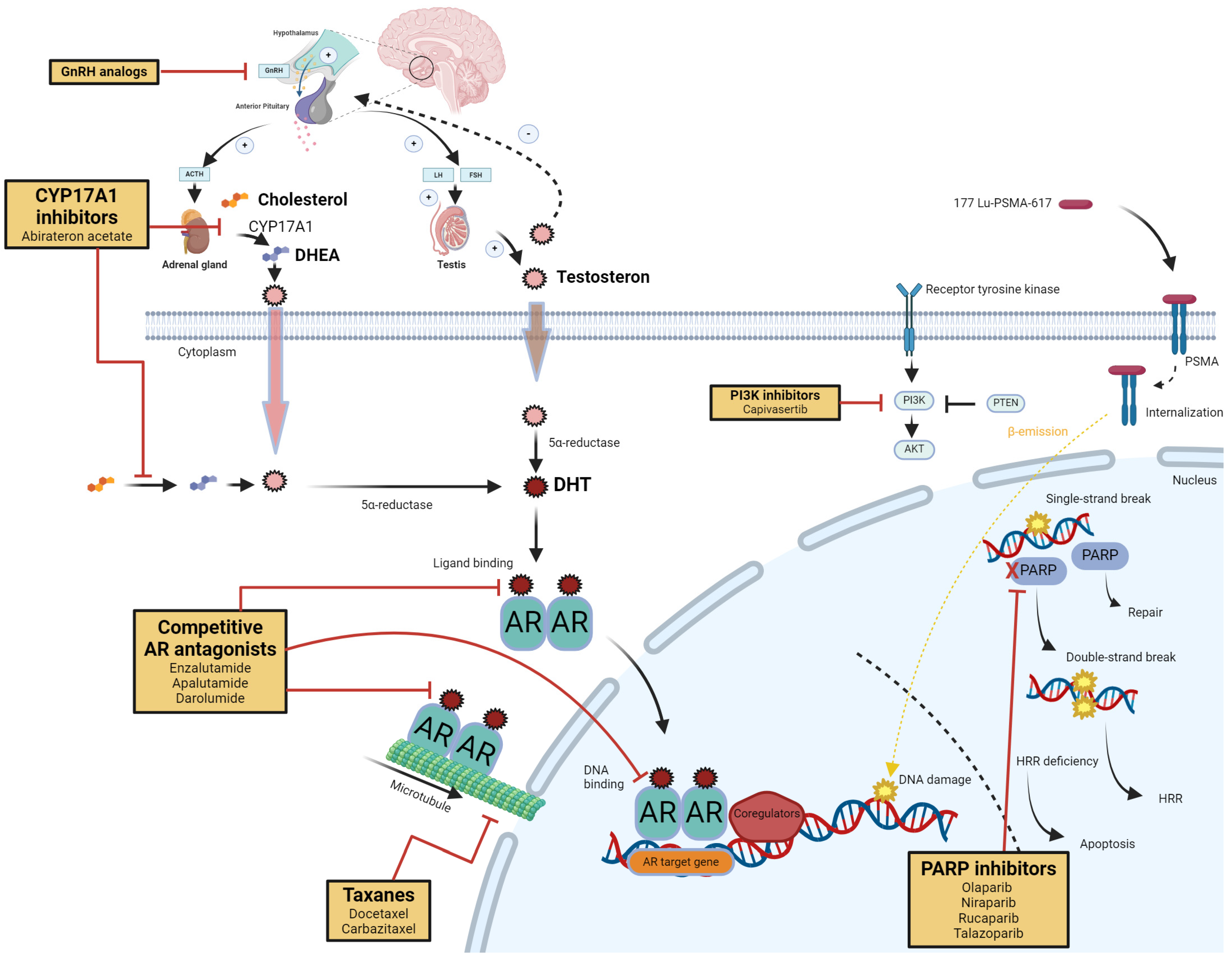
2.1. Targeting the Androgen Signaling Axis (Figure 1)
2.2. Combination Therapies
3. Clinical Development for Combination Therapy
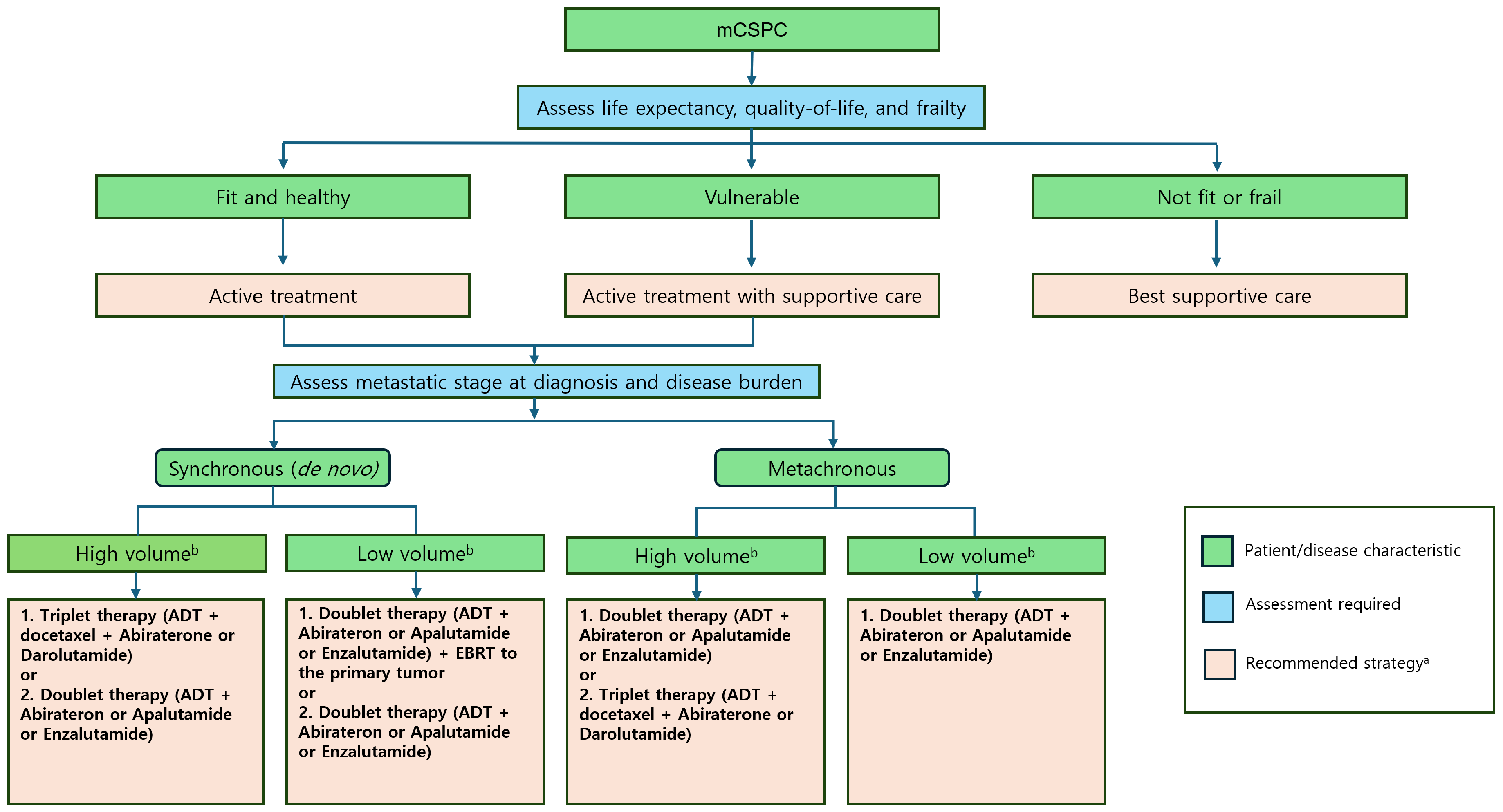
3.1. Systemic Treatment in mCSPC
3.1.1. Androgen-Deprivation Therapy
3.1.2. Treatment Intensification-Doublet Therapies (Table 1)
Apalutamide
Enzalutamide
Docetaxel
| Trial | Patients enrolled | Intervention arm | Control arm | Previous/concurrent docetaxel | Median follow-up (months) | Median OS in the intervention arm (months) | Median OS in the control arm (months) | Hazard ratio (HR) (95% CI) | P-value | Most common AEs (any grade; ≥15% of the investigation group) vs.. the comparator group | Key findings |
|---|---|---|---|---|---|---|---|---|---|---|---|
| STAMPEDE [45] | ≈1,000 | ADT + abiraterone | ADT | Not allowed | 40 | 79.0 | 46.0 | HR: 0.61 (0.49–0.75) | <0.001 | Hypertension (17% vs.. 7%), hypokalemia (15% vs.. 7%), fatigue (20% vs.. 15%) | Significant OS improvement with abiraterone; benefits seen in both low- and high-risk groups |
| LATITUDE [47] | 1,199 | ADT + abiraterone + prednisone | ADT + placebo | Not allowed | 51.8 | 53.3 | 36.5 | HR: 0.66 (0.56–0.78) | <0.001 | Hypertension (20% vs. 10%), hypokalemia (18% vs. 9%), edema (16% vs. 10%) | Significant OS and rPFS improvement; notable but generally manageable AEs |
| TITAN [50] | 1,052 | ADT + apalutamide | ADT + placebo | Allowed (11%) | 44 | Not reached | 52.2 | HR: 0.65 (0.53–0.79) | <0.001 | Rash (27% vs. 8%), hypothyroidism (16% vs. 6%), ischemic heart disease (15% vs. 7%) | Substantial OS and rPFS benefit; consistent benefits across subgroups |
| ENZAMET [55] | 1,125 | ADT + enzalutamide | ADT + first-generation antiandrogen | Allowed (concurrent 45%) | 68 | Not reported | Not reported | HR: 0.70 (0.58–0.84) | <0.001 | Fatigue (22% vs. 14%), hypertension (16% vs. 10%), seizures (2% vs. 1%) | Significant OS and PFS improvements, particularly in synchronous disease |
| ARCHES [58] | 1,150 | ADT + enzalutamide | ADT + placebo | Allowed (previous 18%) | 44.6 | Not reported | Not reported | HR: 0.66 (0.53–0.81) | <0.001 | Fatigue (21% vs. 15%), hypertension (15% vs. 9%), seizures (1.5% vs. 0.5%) | Robust improvement in rPFS and OS; manageable safety profile |
| CHAARTED [63] | 790 | ADT + docetaxel | ADT | Not allowed | 53.7 | 57.6 | 47.2 | HR: 0.72 (0.59–0.89) | 0.002 | Neutropenia (40% vs. 10%), febrile neutropenia (16% vs. 5%), fatigue (18% vs. 10%) | Greater OS benefit in HV disease; limited benefit in LV disease |
| GETUG-AFU-15 [64] | 385 | ADT + docetaxel | ADT | Not allowed | 83.9 | Not significant | Not significant | HR: 1.01 (0.75–1.36) | 0.14 | Neutropenia (35% vs. 13%), febrile neutropenia (17% vs. 5%), fatigue (20% vs. 10%) | Trend toward OS benefit in HV disease; no significant OS benefit overall |
3.1.3. Treatment Intensification-Triplet Therapies (Table 2)
Rationale
| Trial name | Patients enrolled | Intervention arm | control arm | % Synchronous | % High volume | Median follow-up (months) | Median OS in the intervention arm (months) | Median OS in the control arm (months) | Group: HR: (95% CI) | Key adverse events | Key findings |
|---|---|---|---|---|---|---|---|---|---|---|---|
| PEACE-1 [76] | 1173 | SOC + abiraterone (with or without RT) | SOC (with or without RT) | 100% | 64% | 45.6 | NR | 53.2 | 0.75 (0.59–0.95) | Hypertension, transaminase increase | Significant improvement in OS and rPFS, particularly in HV disease. Higher incidence of grade ≥3 AEs in triplet therapy. |
| ARASENS [77] | 1306 | ADT + docetaxel + darolutamide | ADT + docetaxel + placebo | 86% | 77% | 43.7 | NR | 48.9 | 0.68 (0.57–0.80) | Rash, hypertension | Improved OS and secondary endpoints, consistent benefit across subgroups. Less clear benefit in LV disease. |
| ENZAMET [56] | 1125 | ADT + docetaxel + enzalutamide | ADT + docetaxel + first-generation antiandrogen | 72% | 71% | 68 | Not reported | Not reported | 0.73 (0.55–0.99) | Similar to previous trials with enzalutamide | Significant OS improvement in synchronous mCSPC, not in metachronous disease. Early chemotherapy beneficial in high-risk patients. |
Docetaxel Plus Abiraterone
Docetaxel Plus Darolutamide
Docetaxel Plus Enzalutamide
3.1.4. Network Meta-Analysis (Table 3)
| Study | Focus | Treatment comparisons | Key findings (LV mCSPC) | Key findings (HV mCSPC) | Other notes |
|---|---|---|---|---|---|
| Hoeh et al. (2023) [79] | Comparative efficacy of triplet vs. doublet therapies in mCSPC stratified by disease volume | ARPI + ADT or docetaxel + ADT vs. ARPI + docetaxel + ADT | No significant OS differences between triplet and doublet therapies; other combinations did not show benefits over ADT alone | All combinations improved OS compared to ADT alone; darolutamide + docetaxel + ADT ranked the highest in OS | Emphasized the importance of stratifying patients by disease volume for treatment decisions |
| Jian et al. (2023) [127] | Systematic review and NMA comparing the efficacy of combination therapies in mCSPC | Various combination therapies, including triplet and doublet regimens | ADT + ARAT ranked the highest in OS and rPFS; triplet therapies showed no OS or rPFS improvements and a higher risk of AEs | Triplet therapy ranked first in OS and rPFS; ADT + rezvilutamide and ADT + docetaxel were also effective | Highlighted the need for careful consideration of disease volume due to the increased risk of AEs with triplet therapies |
| Riaz et al. (2023) [80] | Evaluation of systemic treatment options for mCSPC | Triplet therapies vs. doublet therapies | Triplet therapies did not significantly outperform ARPIs doublets or docetaxel + ADT; higher risk of AEs | Triplet therapies showed OS advantage; darolutamide and abiraterone triplets significantly improved OS over docetaxel + ADT | Subgroup analyses showed that triplet therapies provided OS advantage for HV disease but not LV; emphasized safety vs. efficacy balance |
3.2. Radiotherapy in mCSPC
4. Guidelines
5. Considerations
Real-World Evidence and Patterns
Factors Influencing the Low Uptake of Combination Therapy
6. Ongoing Issues and Trials in mCSPC Treatment (Table 4)
What Is the Optimal Agent to Use Following Triplet Therapy?
Post-Triplet Therapy Strategy for Patients with HRRm
Comparative Efficacy of Triplet Therapy vs. PARPis in the HRR-Mutated Population
The Need for Head-to-Head Trials
Novel Emerging Therapeutic Agents
Treatment Intensification and De-Escalation
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yamada, Y.; Beltran, H. The treatment landscape of metastatic prostate cancer. Cancer Lett 2021, 519, 20–29. [Google Scholar] [CrossRef]
- Zhao, S.; Yu, E.Y. Castrate-resistant prostate cancer: postdocetaxel management. Curr Opin Urol 2013, 23, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Turco, F.; Tucci, M.; Delcuratolo, M.D.; Di Stefano, R.F.; Pisano, C.; Audisio, A.; Audisio, M.; Ungaro, A.; Ortega, C.; Di Maio, M.; et al. Treatment intensification for metastatic prostate cancer: New treatment landscapes in androgen deprivation-based therapy. Cancer Commun (Lond) 2022, 42, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Riaz, I.B.; Sweeney, C.J. The role of chemotherapy in metastatic prostate cancer. Curr Opin Urol 2022, 32, 292–301. [Google Scholar] [CrossRef]
- Desai, M.M.; Cacciamani, G.E.; Gill, K.; Zhang, J.; Liu, L.; Abreu, A.; Gill, I.S. Trends in Incidence of Metastatic Prostate Cancer in the US. JAMA Netw Open 2022, 5, e222246. [Google Scholar] [CrossRef]
- Dong, L.; Zieren, R.C.; Xue, W.; de Reijke, T.M.; Pienta, K.J. Metastatic prostate cancer remains incurable, why? Asian J Urol 2019, 6, 26–41. [Google Scholar] [CrossRef]
- Hamid, A.A.; Sayegh, N.; Tombal, B.; Hussain, M.; Sweeney, C.J.; Graff, J.N.; Agarwal, N. Metastatic hormone-sensitive prostate cancer: toward an era of adaptive and personalized treatment. American Society of Clinical Oncology Educational Book 2023, 43, e390166. [Google Scholar] [CrossRef] [PubMed]
- Mittal, A.; Sridhar, S.S.; Ong, M.; Jiang, D.M. Triplet Therapy in Metastatic Castrate Sensitive Prostate Cancer (mCSPC)-A Potential New Standard of Care. Curr Oncol 2023, 30, 4365–4378. [Google Scholar] [CrossRef] [PubMed]
- Ehsani, M.; David, F.O.; Baniahmad, A. Androgen receptor-dependent mechanisms mediating drug resistance in prostate cancer. Cancers 2021, 13, 1534. [Google Scholar] [CrossRef]
- Saranyutanon, S.; Srivastava, S.K.; Pai, S.; Singh, S.; Singh, A.P. Therapies targeted to androgen receptor signaling axis in prostate cancer: progress, challenges, and hope. Cancers 2019, 12, 51. [Google Scholar] [CrossRef]
- Chen, Q.-H. Crosstalk between microtubule stabilizing agents and prostate cancer. Cancers 2023, 15, 3308. [Google Scholar] [CrossRef]
- Plana, D.; Palmer, A.C.; Sorger, P.K. Independent drug action in combination therapy: implications for precision oncology. Cancer discovery 2022, 12, 606–624. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; O’Brien, J.; McVey, A.; Jenjitranant, P.; Kelly, B.D.; Kasivisvanathan, V.; Lawrentschuk, N.; Murphy, D.G.; Azad, A.A. Combination treatment in metastatic prostate cancer: is the bar too high or have we fallen short? Nature Reviews Urology 2023, 20, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Westaby, D.; Fenor de La Maza, M.d.L.D.; Paschalis, A.; Jimenez-Vacas, J.M.; Welti, J.; de Bono, J.; Sharp, A. A new old target: androgen receptor signaling and advanced prostate cancer. Annual review of pharmacology and toxicology 2022, 62, 131–153. [Google Scholar] [CrossRef] [PubMed]
- Davies, A.H.; Zoubeidi, A. Targeting androgen receptor signaling: a historical perspective. Endocrine-Related Cancer 2021, 28, T11–T18. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Dehm, S.M.; Sharifi, N. Targeting the androgen signaling axis in prostate cancer. Journal of Clinical Oncology 2023, 41, 4267–4278. [Google Scholar] [CrossRef]
- Davey, R.A.; Grossmann, M. Androgen Receptor Structure, Function and Biology: From Bench to Bedside. Clin Biochem Rev 2016, 37, 3–15. [Google Scholar]
- Sharifi, N.; Gulley, J.L.; Dahut, W.L. Androgen deprivation therapy for prostate cancer. Jama 2005, 294, 238–244. [Google Scholar] [CrossRef]
- Ku, S.-Y.; Gleave, M.E.; Beltran, H. Towards precision oncology in advanced prostate cancer. Nature Reviews Urology 2019, 16, 645–654. [Google Scholar] [CrossRef]
- Boudadi, K.; Antonarakis, E.S. Resistance to Novel Antiandrogen Therapies in Metastatic Castration-Resistant Prostate Cancer. Clin Med Insights Oncol 2016, 10, 1–9. [Google Scholar] [CrossRef]
- Cai, C.; Balk, S.P. Intratumoral androgen biosynthesis in prostate cancer pathogenesis and response to therapy. Endocrine-related cancer 2011, 18, R175–R182. [Google Scholar] [CrossRef] [PubMed]
- Adeniji, A.O.; Chen, M.; Penning, T.M. AKR1C3 as a target in castrate resistant prostate cancer. J Steroid Biochem Mol Biol 2013, 137, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Glaviano, A.; Foo, A.S.C.; Lam, H.Y.; Yap, K.C.H.; Jacot, W.; Jones, R.H.; Eng, H.; Nair, M.G.; Makvandi, P.; Geoerger, B.; et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol Cancer 2023, 22, 138. [Google Scholar] [CrossRef] [PubMed]
- Teo, M.Y.; Rathkopf, D.E.; Kantoff, P. Treatment of advanced prostate cancer. Annual review of medicine 2019, 70, 479–499. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.C.; James, N.D.; Brawley, C.D.; Clarke, N.W.; Hoyle, A.P.; Ali, A.; Ritchie, A.W.S.; Attard, G.; Chowdhury, S.; Cross, W.; et al. Radiotherapy to the primary tumour for newly diagnosed, metastatic prostate cancer (STAMPEDE): a randomised controlled phase 3 trial. Lancet 2018, 392, 2353–2366. [Google Scholar] [CrossRef]
- Rebello, R.J.; Oing, C.; Knudsen, K.E.; Loeb, S.; Johnson, D.C.; Reiter, R.E.; Gillessen, S.; Van der Kwast, T.; Bristow, R.G. Prostate cancer. Nature Reviews Disease Primers 2021, 7, 9. [Google Scholar] [CrossRef]
- Armenia, J.; Wankowicz, S.A.M.; Liu, D.; Gao, J.; Kundra, R.; Reznik, E.; Chatila, W.K.; Chakravarty, D.; Han, G.C.; Coleman, I.; et al. The long tail of oncogenic drivers in prostate cancer. Nature Genetics 2018, 50, 645–651. [Google Scholar] [CrossRef]
- Crawford, E.D.; Schellhammer, P.F.; McLeod, D.G.; Moul, J.W.; Higano, C.S.; Shore, N.; Denis, L.; Iversen, P.; Eisenberger, M.A.; Labrie, F. Androgen Receptor Targeted Treatments of Prostate Cancer: 35 Years of Progress with Antiandrogens. J Urol 2018, 200, 956–966. [Google Scholar] [CrossRef]
- Labrecque, M.P.; Coleman, I.M.; Brown, L.G.; True, L.D.; Kollath, L.; Lakely, B.; Nguyen, H.M.; Yang, Y.C.; da Costa, R.M.G.; Kaipainen, A.; et al. Molecular profiling stratifies diverse phenotypes of treatment-refractory metastatic castration-resistant prostate cancer. J Clin Invest 2019, 129, 4492–4505. [Google Scholar] [CrossRef]
- Bluemn, E.G.; Coleman, I.M.; Lucas, J.M.; Coleman, R.T.; Hernandez-Lopez, S.; Tharakan, R.; Bianchi-Frias, D.; Dumpit, R.F.; Kaipainen, A.; Corella, A.N.; et al. Androgen Receptor Pathway-Independent Prostate Cancer Is Sustained through FGF Signaling. Cancer Cell 2017, 32, 474–489.e476. [Google Scholar] [CrossRef]
- Watson, P.A.; Arora, V.K.; Sawyers, C.L. Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat Rev Cancer 2015, 15, 701–711. [Google Scholar] [CrossRef]
- Mohler, M.L.; Sikdar, A.; Ponnusamy, S.; Hwang, D.J.; He, Y.; Miller, D.D.; Narayanan, R. An Overview of Next-Generation Androgen Receptor-Targeted Therapeutics in Development for the Treatment of Prostate Cancer. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Mateo, J.; Fizazi, K.; Saad, F.; Shore, N.; Sandhu, S.; Chi, K.N.; Sartor, O.; Agarwal, N.; Olmos, D.; et al. Survival with Olaparib in Metastatic Castration-Resistant Prostate Cancer. N Engl J Med 2020, 383, 2345–2357. [Google Scholar] [CrossRef] [PubMed]
- Damiana, T.S.T.; Dalm, S.U. Combination Therapy, a Promising Approach to Enhance the Efficacy of Radionuclide and Targeted Radionuclide Therapy of Prostate and Breast Cancer. Pharmaceutics 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Hussein, M.A.; Munirathinam, G. Androgen Receptor Signaling in Prostate Cancer Genomic Subtypes. Cancers 2023, 15, 4969. [Google Scholar] [CrossRef]
- He, Y.; Xu, W.; Xiao, Y.-T.; Huang, H.; Gu, D.; Ren, S. Targeting signaling pathways in prostate cancer: mechanisms and clinical trials. Signal Transduction and Targeted Therapy 2022, 7, 198. [Google Scholar] [CrossRef]
- Kumar, R.; Sena, L.A.; Denmeade, S.R.; Kachhap, S. The testosterone paradox of advanced prostate cancer: mechanistic insights and clinical implications. Nat Rev Urol 2023, 20, 265–278. [Google Scholar] [CrossRef]
- Gomella, L.G. Effective testosterone suppression for prostate cancer: is there a best castration therapy? Rev Urol 2009, 11, 52–60. [Google Scholar]
- Pagliarulo, V. Androgen deprivation therapy for prostate cancer. Molecular & Diagnostic Imaging in Prostate Cancer: Clinical Applications and Treatment Strategies 2018, 1-30.
- Hamid, A.A.; Sayegh, N.; Tombal, B.; Hussain, M.; Sweeney, C.J.; Graff, J.N.; Agarwal, N. Metastatic Hormone-Sensitive Prostate Cancer: Toward an Era of Adaptive and Personalized Treatment. American Society of Clinical Oncology Educational Book 2023, e390166. [Google Scholar] [CrossRef]
- Shore, N.D.; Saad, F.; Cookson, M.S.; George, D.J.; Saltzstein, D.R.; Tutrone, R.; Akaza, H.; Bossi, A.; Veenhuyzen, D.F.v.; Selby, B.; et al. Oral Relugolix for Androgen-Deprivation Therapy in Advanced Prostate Cancer. New England Journal of Medicine 2020, 382, 2187–2196. [Google Scholar] [CrossRef]
- Sweeney, C.J.; Chen, Y.-H.; Carducci, M.; Liu, G.; Jarrard, D.F.; Eisenberger, M.; Wong, Y.-N.; Hahn, N.; Kohli, M.; Cooney, M.M.; et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. New England Journal of Medicine 2015, 373, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer—2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. European Urology 2021, 79, 243–262. [Google Scholar] [CrossRef]
- Sountoulides, P.; Rountos, T. Adverse effects of androgen deprivation therapy for prostate cancer: prevention and management. ISRN Urol 2013, 2013, 240108. [Google Scholar] [CrossRef]
- Fizazi, K.; Tran, N.; Fein, L.; Matsubara, N.; Rodriguez-Antolin, A.; Alekseev, B.Y.; Özgüroğlu, M.; Ye, D.; Feyerabend, S.; Protheroe, A.; et al. Abiraterone plus Prednisone in Metastatic, Castration-Sensitive Prostate Cancer. New England Journal of Medicine 2017, 377, 352–360. [Google Scholar] [CrossRef] [PubMed]
- James, N.D.; Clarke, N.W.; Cook, A.; Ali, A.; Hoyle, A.P.; Attard, G.; Brawley, C.D.; Chowdhury, S.; Cross, W.R.; Dearnaley, D.P.; et al. Abiraterone acetate plus prednisolone for metastatic patients starting hormone therapy: 5-year follow-up results from the STAMPEDE randomised trial (NCT00268476). Int J Cancer 2022, 151, 422–434. [Google Scholar] [CrossRef] [PubMed]
- Fizazi, K.; Tran, N.; Fein, L.; Matsubara, N.; Rodriguez-Antolin, A.; Alekseev, B.Y.; Özgüroğlu, M.; Ye, D.; Feyerabend, S.; Protheroe, A.; et al. Abiraterone acetate plus prednisone in patients with newly diagnosed high-risk metastatic castration-sensitive prostate cancer (LATITUDE): final overall survival analysis of a randomised, double-blind, phase 3 trial. Lancet Oncol 2019, 20, 686–700. [Google Scholar] [CrossRef]
- Chi, K.N.; Protheroe, A.; Rodríguez-Antolín, A.; Facchini, G.; Suttman, H.; Matsubara, N.; Ye, Z.; Keam, B.; Damião, R.; Li, T.; et al. Patient-reported outcomes following abiraterone acetate plus prednisone added to androgen deprivation therapy in patients with newly diagnosed metastatic castration-naive prostate cancer (LATITUDE): an international, randomised phase 3 trial. Lancet Oncol 2018, 19, 194–206. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network (NCCN) Guidelines Version 4.2024, “Prostate Cancer.”. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1459.
- Chi, K.N.; Agarwal, N.; Bjartell, A.; Chung, B.H.; Pereira de Santana Gomes, A.J.; Given, R.; Juárez Soto, Á.; Merseburger, A.S.; Özgüroğlu, M.; Uemura, H.; et al. Apalutamide for Metastatic, Castration-Sensitive Prostate Cancer. N Engl J Med 2019, 381, 13–24. [Google Scholar] [CrossRef]
- Chi, K.N.; Chowdhury, S.; Bjartell, A.; Chung, B.H.; Pereira de Santana Gomes, A.J.; Given, R.; Juárez, A.; Merseburger, A.S.; Özgüroğlu, M.; Uemura, H.; et al. Apalutamide in Patients With Metastatic Castration-Sensitive Prostate Cancer: Final Survival Analysis of the Randomized, Double-Blind, Phase III TITAN Study. J Clin Oncol 2021, 39, 2294–2303. [Google Scholar] [CrossRef]
- Agarwal, N.; McQuarrie, K.; Bjartell, A.; Chowdhury, S.; Pereira de Santana Gomes, A.J.; Chung, B.H.; Özgüroğlu, M.; Juárez Soto, Á.; Merseburger, A.S.; Uemura, H.; et al. Health-related quality of life after apalutamide treatment in patients with metastatic castration-sensitive prostate cancer (TITAN): a randomised, placebo-controlled, phase 3 study. Lancet Oncol 2019, 20, 1518–1530. [Google Scholar] [CrossRef]
- Cattrini, C.; España, R.; Mennitto, A.; Bersanelli, M.; Castro, E.; Olmos, D.; Lorente, D.; Gennari, A. Optimal sequencing and predictive biomarkers in patients with advanced prostate cancer. Cancers 2021, 13, 4522. [Google Scholar] [CrossRef]
- Shen, J.; Chowdhury, S.; Agarwal, N.; Karsh, L.I.; Oudard, S.; Gartrell, B.A.; Feyerabend, S.; Saad, F.; Pieczonka, C.M.; Chi, K.N.; et al. Apalutamide efficacy, safety and wellbeing in older patients with advanced prostate cancer from Phase 3 randomised clinical studies TITAN and SPARTAN. Br J Cancer 2024, 130, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Davis, I.D.; Martin, A.J.; Stockler, M.R.; Begbie, S.; Chi, K.N.; Chowdhury, S.; Coskinas, X.; Frydenberg, M.; Hague, W.E.; Horvath, L.G.; et al. Enzalutamide with Standard First-Line Therapy in Metastatic Prostate Cancer. N Engl J Med 2019, 381, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, C.J.; Martin, A.J.; Stockler, M.R.; Begbie, S.; Cheung, L.; Chi, K.N.; Chowdhury, S.; Frydenberg, M.; Horvath, L.G.; Joshua, A.M.; et al. Testosterone suppression plus enzalutamide versus testosterone suppression plus standard antiandrogen therapy for metastatic hormone-sensitive prostate cancer (ENZAMET): an international, open-label, randomised, phase 3 trial. Lancet Oncol 2023, 24, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.J.; Szmulewitz, R.Z.; Petrylak, D.P.; Holzbeierlein, J.; Villers, A.; Azad, A.; Alcaraz, A.; Alekseev, B.; Iguchi, T.; Shore, N.D.; et al. ARCHES: A Randomized, Phase III Study of Androgen Deprivation Therapy With Enzalutamide or Placebo in Men With Metastatic Hormone-Sensitive Prostate Cancer. J Clin Oncol 2019, 37, 2974–2986. [Google Scholar] [CrossRef]
- Armstrong, A.J.; Azad, A.A.; Iguchi, T.; Szmulewitz, R.Z.; Petrylak, D.P.; Holzbeierlein, J.; Villers, A.; Alcaraz, A.; Alekseev, B.; Shore, N.D.; et al. Improved Survival With Enzalutamide in Patients With Metastatic Hormone-Sensitive Prostate Cancer. J Clin Oncol 2022, 40, 1616–1622. [Google Scholar] [CrossRef]
- Shelan, M.; Achard, V.; Appiagyei, F.; Mose, L.; Zilli, T.; Fankhauser, C.D.; Zamboglou, C.; Mohamad, O.; Aebersold, D.M.; Cathomas, R. Role of enzalutamide in primary and recurrent non-metastatic hormone sensitive prostate cancer: a systematic review of prospective clinical trials. Prostate Cancer and Prostatic Diseases 2024. [CrossRef]
- Sweeney, C.J.; Chen, Y.H.; Carducci, M.; Liu, G.; Jarrard, D.F.; Eisenberger, M.; Wong, Y.N.; Hahn, N.; Kohli, M.; Cooney, M.M.; et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. N Engl J Med 2015, 373, 737–746. [Google Scholar] [CrossRef]
- James, N.D.; Sydes, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Spears, M.R.; Ritchie, A.W.; Parker, C.C.; Russell, J.M.; Attard, G.; et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 2016, 387, 1163–1177. [Google Scholar] [CrossRef]
- Gravis, G.; Boher, J.M.; Joly, F.; Soulié, M.; Albiges, L.; Priou, F.; Latorzeff, I.; Delva, R.; Krakowski, I.; Laguerre, B.; et al. Androgen Deprivation Therapy (ADT) Plus Docetaxel Versus ADT Alone in Metastatic Non castrate Prostate Cancer: Impact of Metastatic Burden and Long-term Survival Analysis of the Randomized Phase 3 GETUG-AFU15 Trial. Eur Urol 2016, 70, 256–262. [Google Scholar] [CrossRef]
- Kyriakopoulos, C.E.; Chen, Y.H.; Carducci, M.A.; Liu, G.; Jarrard, D.F.; Hahn, N.M.; Shevrin, D.H.; Dreicer, R.; Hussain, M.; Eisenberger, M.; et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer: Long-Term Survival Analysis of the Randomized Phase III E3805 CHAARTED Trial. J Clin Oncol 2018, 36, 1080–1087. [Google Scholar] [CrossRef]
- Gravis, G.; Fizazi, K.; Joly, F.; Oudard, S.; Priou, F.; Esterni, B.; Latorzeff, I.; Delva, R.; Krakowski, I.; Laguerre, B.; et al. Androgen-deprivation therapy alone or with docetaxel in non-castrate metastatic prostate cancer (GETUG-AFU 15): a randomised, open-label, phase 3 trial. Lancet Oncol 2013, 14, 149–158. [Google Scholar] [CrossRef]
- Oing, C.; Bristow, R.G. Systemic treatment of metastatic hormone-sensitive prostate cancer-upfront triplet versus doublet combination therapy. ESMO Open 2023, 8, 101194. [Google Scholar] [CrossRef] [PubMed]
- Wala, J.; Nguyen, P.; Pomerantz, M. Early Treatment Intensification in Metastatic Hormone-Sensitive Prostate Cancer. Journal of Clinical Oncology 2023, 41, 3584–3590. [Google Scholar] [CrossRef] [PubMed]
- Gebrael, G.; Fortuna, G.G.; Sayegh, N.; Swami, U.; Agarwal, N. Advances in the treatment of metastatic prostate cancer. Trends in Cancer 2023. [CrossRef] [PubMed]
- Sayegh, N.; Swami, U.; Agarwal, N. Recent advances in the management of metastatic prostate cancer. JCO Oncology Practice 2022, 18, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Bungaro, M.; Buttigliero, C.; Tucci, M. Overcoming the mechanisms of primary and acquired resistance to new generation hormonal therapies in advanced prostate cancer: Focus on androgen receptor independent pathways. Cancer Drug Resistance 2020, 3, 726. [Google Scholar] [CrossRef]
- Leith, A.; Kim, J.; Ribbands, A.; Clayton, E.; Yang, L.; Ghate, S.R. Real-world treatment patterns in metastatic castration-resistant prostate cancer across Europe (France, Germany, Italy, Spain, and the United Kingdom) and Japan. Advances in Therapy 2022, 39, 2236–2255. [Google Scholar] [CrossRef]
- Jian, T.; Zhan, Y.; Hu, K.; He, L.; Chen, S.; Hu, R.; Lu, J. Systemic triplet therapy for metastatic hormone-sensitive prostate cancer: A systematic review and network meta-analysis. Front Pharmacol 2022, 13, 955925. [Google Scholar] [CrossRef]
- Sousa-Pimenta, M.; Estevinho, L.M.; Szopa, A.; Basit, M.; Khan, K.; Armaghan, M.; Ibrayeva, M.; Sönmez Gürer, E.; Calina, D.; Hano, C.; et al. Chemotherapeutic properties and side-effects associated with the clinical practice of terpene alkaloids: paclitaxel, docetaxel, and cabazitaxel. Front Pharmacol 2023, 14, 1157306. [Google Scholar] [CrossRef]
- Ehsani, M.; David, F.O.; Baniahmad, A. Androgen Receptor-Dependent Mechanisms Mediating Drug Resistance in Prostate Cancer. Cancers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Damodaran, S.; Lang, J.M.; Jarrard, D.F. Targeting Metastatic Hormone Sensitive Prostate Cancer: Chemohormonal Therapy and New Combinatorial Approaches. J Urol 2019, 201, 876–885. [Google Scholar] [CrossRef]
- James, N.D.; de Bono, J.S.; Spears, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Ritchie, A.W.S.; Amos, C.L.; Gilson, C.; Jones, R.J.; et al. Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy. N Engl J Med 2017, 377, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Fizazi, K.; Foulon, S.; Carles, J.; Roubaud, G.; McDermott, R.; Fléchon, A.; Tombal, B.; Supiot, S.; Berthold, D.; Ronchin, P.; et al. Abiraterone plus prednisone added to androgen deprivation therapy and docetaxel in de novo metastatic castration-sensitive prostate cancer (PEACE-1): a multicentre, open-label, randomised, phase 3 study with a 2 × 2 factorial design. Lancet 2022, 399, 1695–1707. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Hussain, M.; Saad, F.; Fizazi, K.; Sternberg, C.N.; Crawford, E.D.; Kopyltsov, E.; Park, C.H.; Alekseev, B.; Montesa-Pino, Á.; et al. Darolutamide and Survival in Metastatic, Hormone-Sensitive Prostate Cancer. N Engl J Med 2022, 386, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- FDA approves darolutamide tablets for metastatic hormone-sensitive prostate cancer. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-darolutamide-tablets-metastatic-hormone-sensitive-prostate-cancer (accessed on 6 April 2024).
- Hoeh, B.; Garcia, C.C.; Wenzel, M.; Tian, Z.; Tilki, D.; Steuber, T.; Karakiewicz, P.I.; Chun, F.K.H.; Mandel, P. Triplet or Doublet Therapy in Metastatic Hormone-sensitive Prostate Cancer: Updated Network Meta-analysis Stratified by Disease Volume. Eur Urol Focus 2023, 9, 838–842. [Google Scholar] [CrossRef]
- Riaz, I.B.; Naqvi, S.A.A.; He, H.; Asghar, N.; Siddiqi, R.; Liu, H.; Singh, P.; Childs, D.S.; Ravi, P.; Hussain, S.A.; et al. First-line Systemic Treatment Options for Metastatic Castration-Sensitive Prostate Cancer: A Living Systematic Review and Network Meta-analysis. JAMA Oncol 2023, 9, 635–645. [Google Scholar] [CrossRef]
- Esposito, M.; Ganesan, S.; Kang, Y. Emerging strategies for treating metastasis. Nat Cancer 2021, 2, 258–270. [Google Scholar] [CrossRef]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: a hallmark of cancer revisited. Signal Transduction and Targeted Therapy 2020, 5, 28. [Google Scholar] [CrossRef]
- Parker, C.C.; James, N.D.; Brawley, C.D.; Clarke, N.W.; Ali, A.; Amos, C.L.; Attard, G.; Chowdhury, S.; Cook, A.; Cross, W.; et al. Radiotherapy to the prostate for men with metastatic prostate cancer in the UK and Switzerland: Long-term results from the STAMPEDE randomised controlled trial. PLoS Med 2022, 19, e1003998. [Google Scholar] [CrossRef]
- Boevé, L.M.S.; Hulshof, M.; Vis, A.N.; Zwinderman, A.H.; Twisk, J.W.R.; Witjes, W.P.J.; Delaere, K.P.J.; Moorselaar, R.; Verhagen, P.; van Andel, G. Effect on Survival of Androgen Deprivation Therapy Alone Compared to Androgen Deprivation Therapy Combined with Concurrent Radiation Therapy to the Prostate in Patients with Primary Bone Metastatic Prostate Cancer in a Prospective Randomised Clinical Trial: Data from the HORRAD Trial. Eur Urol 2019, 75, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, I.; Matsukawa, A.; Kardoust Parizi, M.; Klemm, J.; Mancon, S.; Chiujdea, S.; Fazekas, T.; Miszczyk, M.; Laukhtina, E.; Kawada, T.; et al. A Systematic Review and Meta-analysis of the Impact of Local Therapies on Local Event Suppression in Metastatic Hormone-sensitive Prostate Cancer. Eur Urol Oncol 2024. [CrossRef] [PubMed]
- Baboudjian, M.; Roubaud, G.; Fromont, G.; Gauthé, M.; Beauval, J.-B.; Barret, E.; Brureau, L.; Créhange, G.; Dariane, C.; Fiard, G. What is the ideal combination therapy in de novo, oligometastatic, castration-sensitive prostate cancer? World Journal of Urology 2023, 41, 2033–2041. [Google Scholar] [CrossRef] [PubMed]
- Achard, V.; Putora, P.M.; Omlin, A.; Zilli, T.; Fischer, S. Metastatic prostate cancer: treatment options. Oncology 2022, 100, 48–59. [Google Scholar] [CrossRef]
- EAU Guidelines - Uroweb: Prostate Cancer. Available online: https://uroweb.org/guidelines/prostate-cancer/chapter/treatment (accessed on 7 August 2024).
- Kafka, M.; Giannini, G.; Artamonova, N.; Neuwirt, H.; Ofner, H.; Kramer, G.; Bauernhofer, T.; Luger, F.; Höfner, T.; Loidl, W.; et al. Real-World Evidence of Triplet Therapy in Metastatic Hormone-Sensitive Prostate Cancer: An Austrian Multicenter Study. Clin Genitourin Cancer 2024, 22, 458–466.e451. [Google Scholar] [CrossRef]
- Mondal, D.; Roy, S.; Bhattacharjee, A.; Roy, R.; Meyur, S.; Biswas, B. Triplet therapy in metastatic hormone-sensitive prostate cancer: A real-world Indian multicentre study. Journal of Clinical Oncology 2024, 42, e17086–e17086. [Google Scholar] [CrossRef]
- Kolinsky, M.P.; O’Sullivan, D.E.; Boyne, D.J.; Brenner, D.R.; Shokar, S.; Cheung, W.Y. A real-world observational study characterizing patients (pts) with metastatic castration-sensitive prostate cancer (mCSPC) treated with or without androgen receptor pathway inhibitors (ARPIs). Journal of Clinical Oncology 2024, 42, 59–59. [Google Scholar] [CrossRef]
- George, D.J.; Agarwal, N.; Rider, J.R.; Li, B.; Shirali, R.; Sandin, R.; Hong, A.; Russell, D.; Ramaswamy, K.; Freedland, S.J. Real-world treatment patterns among patients diagnosed with metastatic castration-sensitive prostate cancer (mCSPC) in community oncology settings. Journal of Clinical Oncology 2021, 39, 5074–5074. [Google Scholar] [CrossRef]
- Chen, K.; O’Brien, J.; McVey, A.; Jenjitranant, P.; Kelly, B.D.; Kasivisvanathan, V.; Lawrentschuk, N.; Murphy, D.G.; Azad, A.A. Combination treatment in metastatic prostate cancer: is the bar too high or have we fallen short? Nat Rev Urol 2023, 20, 116–123. [Google Scholar] [CrossRef]
- Barata, P.C.; Leith, A.; Ribbands, A.; Montgomery, R.; Last, M.; Arondekar, B.; Ivanova, J.; Niyazov, A. Real-World Treatment Trends Among Patients with Metastatic Castration-Sensitive Prostate Cancer: Results from an International Study. Oncologist 2023, 28, 780–789. [Google Scholar] [CrossRef]
- Swami, U.; Sinnott, J.A.; Haaland, B.; Sayegh, N.; McFarland, T.R.; Tripathi, N.; Maughan, B.L.; Rathi, N.; Sirohi, D.; Nussenzveig, R.; et al. Treatment Pattern and Outcomes with Systemic Therapy in Men with Metastatic Prostate Cancer in the Real-World Patients in the United States. Cancers 2021, 13, 4951. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, E.; Medicine. Factors that affect health-care utilization. In Health-care utilization as a proxy in disability determination; National Academies Press (US): 2018.
- Medlinskiene, K.; Tomlinson, J.; Marques, I.; Richardson, S.; Stirling, K.; Petty, D. Barriers and facilitators to the uptake of new medicines into clinical practice: a systematic review. BMC Health Serv Res 2021, 21, 1198. [Google Scholar] [CrossRef] [PubMed]
- Lai-Kwon, J.; Heynemann, S.; Hart, N.H.; Chan, R.J.; Smith, T.J.; Nekhlyudov, L.; Jefford, M. Evolving Landscape of Metastatic Cancer Survivorship: Reconsidering Clinical Care, Policy, and Research Priorities for the Modern Era. Journal of Clinical Oncology 2023, 41, 3304–3310. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Wen, F.; Wu, Y.; Wheeler, J.; Li, Q. Cost-effectiveness analysis of additional docetaxel for metastatic hormone-sensitive prostate cancer treated with androgen-deprivation therapy from a Chinese perspective. European Journal of Cancer Care 2017, 26, e12505. [Google Scholar] [CrossRef]
- Yanagisawa, T.; Kawada, T.; Rajwa, P.; Mostafaei, H.; Motlagh, R.S.; Quhal, F.; Laukhtina, E.; König, F.; Pallauf, M.; Pradere, B. Sequencing impact and prognostic factors in metastatic castration-resistant prostate cancer patients treated with cabazitaxel: a systematic review and meta-analysis. In Proceedings of the Urologic Oncology: Seminars and Original Investigations; 2023; pp. 177–191. [Google Scholar]
- Sun, B.; Lovell, J.F.; Zhang, Y. Current development of cabazitaxel drug delivery systems. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology 2023, 15, e1854. [Google Scholar] [CrossRef]
- Sartor, O.; De Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G. Lutetium-177–PSMA-617 for metastatic castration-resistant prostate cancer. New England Journal of Medicine 2021, 385, 1091–1103. [Google Scholar] [CrossRef]
- Hofman, M.S.; Emmett, L.; Sandhu, S.; Iravani, A.; Joshua, A.M.; Goh, J.C.; Pattison, D.A.; Tan, T.H.; Kirkwood, I.D.; Ng, S. [177Lu] Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. The Lancet 2021, 397, 797–804. [Google Scholar] [CrossRef]
- Sorrentino, C.; Di Carlo, E. Molecular targeted therapies in metastatic prostate cancer: recent advances and future challenges. Cancers 2023, 15, 2885. [Google Scholar] [CrossRef]
- Bourlon, M.T.; Valdez, P.; Castro, E. Development of PARP inhibitors in advanced prostate cancer. Therapeutic Advances in Medical Oncology 2024, 16, 17588359231221337. [Google Scholar] [CrossRef]
- Ryan, C.J.; Cao, Q.; Souza, A.L.D.; VanderWeele, D.J.; Bryce, A.H.; Kelly, W.K.; Jha, G.G.; Barata, P.C.; Sartor, A.O.; Stadler, W.M.; et al. Early results from CASCARA: A phase 2 study of cabazitaxel/carboplatin plus abiraterone in high-volume metastatic castrate-sensitive prostate cancer (mCSPC). Journal of Clinical Oncology 2023, 41, 5071–5071. [Google Scholar] [CrossRef]
- Azad, A.; Dhiantravan, N.; Emmett, L.; Joshua, A.M.; Vela, I.; Pattison, D.A.; Francis, R.J.; Williams, S.; Sandhu, S.K.; Davis, I.D.; et al. UpFrontPSMA: A randomized phase II study of sequential 177Lu-PSMA617 and docetaxel versus docetaxel in metastatic hormone-naïve prostate cancer (mHNPC). Journal of Clinical Oncology 2021, 39, TPS180–TPS180. [Google Scholar] [CrossRef]
- Rathkopf, D.E.; Chi, K.N.; Olmos, D.; Cheng, H.H.; Agarwal, N.; Graff, J.N.; Sandhu, S.K.; Hayreh, V.; Lopez-Gitlitz, A.; Francis, P.S.J.; et al. AMPLITUDE: A study of niraparib in combination with abiraterone acetate plus prednisone (AAP) versus AAP for the treatment of patients with deleterious germline or somatic homologous recombination repair (HRR) gene-altered metastatic castration-sensitive prostate cancer (mCSPC). Journal of Clinical Oncology 2021, 39, TPS176–TPS176. [Google Scholar] [CrossRef]
- Zhu, Q.; Chen, J.; Liu, H.; Zhao, J.; Xu, C.; Sun, G.; Zeng, H. The efficacy and safety of PARP inhibitors in mCRPC with HRR mutation in second-line treatment: a systematic review and bayesian network meta-analysis. BMC Cancer 2024, 24, 706. [Google Scholar] [CrossRef] [PubMed]
- Kostos, L.; Tran, B.; Azad, A.A. Combination of PARP Inhibitors and Androgen Receptor Pathway Inhibitors in Metastatic Castration-Resistant Prostate Cancer. Drugs 2024. [Google Scholar] [CrossRef]
- Kwon, W.-A. PARP Inhibitors in the Treatment of Prostate Cancer: From Scientific Rationale to Clinical Development. World J Mens Health 2024, 42, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Fizazi, K.; Azad, A.A.; Matsubara, N.; Carles, J.; Fay, A.P.; De Giorgi, U.; Joung, J.Y.; Fong, P.C.C.; Voog, E.; Jones, R.J.; et al. First-line talazoparib with enzalutamide in HRR-deficient metastatic castration-resistant prostate cancer: the phase 3 TALAPRO-2 trial. Nature Medicine 2024, 30, 257–264. [Google Scholar] [CrossRef]
- Fu, X.; Li, P.; Zhou, Q.; He, R.; Wang, G.; Zhu, S.; Bagheri, A.; Kupfer, G.; Pei, H.; Li, J. Mechanism of PARP inhibitor resistance and potential overcoming strategies. Genes Dis 2024, 11, 306–320. [Google Scholar] [CrossRef]
- Atiq, M.; Chandran, E.; Karzai, F.; Madan, R.A.; Aragon-Ching, J.B. Emerging treatment options for prostate cancer. Expert review of anticancer therapy 2023, 23, 625–631. [Google Scholar] [CrossRef]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med 2021, 385, 1091–1103. [Google Scholar] [CrossRef]
- Hofman, M.S.; Emmett, L.; Sandhu, S.; Iravani, A.; Joshua, A.M.; Goh, J.C.; Pattison, D.A.; Tan, T.H.; Kirkwood, I.D.; Ng, S.; et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet 2021, 397, 797–804. [Google Scholar] [CrossRef]
- Jadvar, H.; Colletti, P.M. Clinical Trials of Prostate-Specific Membrane Antigen Radiopharmaceutical Therapy. Journal of Nuclear Medicine Technology 2023, 51, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.; Taheriazam, A.; Daneii, P.; Hassanpour, A.; Kakavand, A.; Rezaei, S.; Hejazi, E.S.; Aboutalebi, M.; Gholamrezaie, H.; Saebfar, H.; et al. Targeting PI3K/Akt signaling in prostate cancer therapy. J Cell Commun Signal 2023, 17, 423–443. [Google Scholar] [CrossRef]
- Crabb, S.J.; Griffiths, G.; Dunkley, D.; Downs, N.; Ellis, M.; Radford, M.; Light, M.; Northey, J.; Whitehead, A.; Wilding, S.; et al. Overall Survival Update for Patients with Metastatic Castration-resistant Prostate Cancer Treated with Capivasertib and Docetaxel in the Phase 2 ProCAID Clinical Trial. European Urology 2022, 82, 512–515. [Google Scholar] [CrossRef]
- Fizazi, K.; George, D.J.; Santis, M.D.; Clarke, N.; Fay, A.P.; Uemura, H.; Grinsted, L.; Rooney, C.; Verheijen, R.B.; Anjum, R.; et al. A phase III trial of capivasertib and abiraterone versus placebo and abiraterone in patients with de novo metastatic hormone-sensitive prostate cancer characterized by PTEN deficiency (CAPItello-281). Journal of Clinical Oncology 2021, 39, TPS178–TPS178. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Z. The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cellular & Molecular Immunology 2020, 17, 807–821. [Google Scholar] [CrossRef]
- Gratzke, C.; Kwiatkowski, M.; De Giorgi, U.; Martins da Trindade, K.; De Santis, M.; Armstrong, A.J.; Niu, C.; Liu, Y.; Poehlein, C.H. KEYNOTE-991: pembrolizumab plus enzalutamide and androgen deprivation for metastatic hormone-sensitive prostate cancer. Future Oncol 2023. [Google Scholar] [CrossRef] [PubMed]
- Isgandarov, A.; Darr, C.; Posdzich, P.; Hermann, K.; Hadaschik, B.; Grünwald, V. New treatment approaches for and ongoing trials in metastatic hormone-sensitive prostate cancer. Urologie (Heidelberg, Germany) 2023.
- Sagaram, S.; Rao, A. Rapidly evolving treatment paradigm and considerations for sequencing therapies in metastatic prostate cancer-a narrative review. Transl Androl Urol 2021, 10, 3188–3198. [Google Scholar] [CrossRef]
- Gebrael, G.; Fortuna, G.G.; Sayegh, N.; Swami, U.; Agarwal, N. Advances in the treatment of metastatic prostate cancer. Trends Cancer 2023, 9, 840–854. [Google Scholar] [CrossRef]
- Chen, K.; Kostos, L.; Azad, A.A. Future directions in systemic treatment of metastatic hormone-sensitive prostate cancer. World J Urol 2023, 41, 2021–2031. [Google Scholar] [CrossRef]
- Jian, T.; Zhan, Y.; Yu, Y.; Yu, K.; Hu, R.; Wang, J.; Lu, J. Combination therapy for high-volume versus low-volume metastatic hormone-sensitive prostate cancer: A systematic review and network meta-analysis. Front Pharmacol 2023, 14, 1148021. [Google Scholar] [CrossRef]
- Agarwal, N.; Azad, A.; Fizazi, K.; Mateo, J.; Matsubara, N.; Shore, N.D.; Chakrabarti, J.; Chen, H.-C.; Lanzalone, S.; Niyazov, A.; et al. Talapro-3: A phase 3, double-blind, randomized study of enzalutamide (ENZA) plus talazoparib (TALA) versus placebo plus enza in patients with DDR gene mutated metastatic castration-sensitive prostate cancer (mCSPC). Journal of Clinical Oncology 2022, 40, TPS221–TPS221. [Google Scholar] [CrossRef]
- Smith, M.R.; Hussain, M.; Saad, F.; Fizazi, K.; Sternberg, C.N.; Crawford, E.D.; Kopyltsov, E.; Park, C.H.; Alekseev, B.; Montesa-Pino, Á.; et al. Darolutamide and Survival in Metastatic, Hormone-Sensitive Prostate Cancer. New England Journal of Medicine 2022, 386, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- Sartor, A.O.; Tagawa, S.T.; Saad, F.; Bono, J.S.D.; Feng, F.Y.; Fizazi, K.; Sakharova, O.V.; Morris, M.J. PSMAddition: A phase 3 trial to compare treatment with 177Lu-PSMA-617 plus standard of care (SOC) versus SOC alone in patients with metastatic hormone-sensitive prostate cancer. Journal of Clinical Oncology 2022, 40, TPS210–TPS210. [Google Scholar] [CrossRef]
- Attard, G.; Murphy, L.; Clarke, N.W.; Cross, W.; Jones, R.J.; Parker, C.C.; Gillessen, S.; Cook, A.; Brawley, C.; Amos, C.L.; et al. Abiraterone acetate and prednisolone with or without enzalutamide for high-risk non-metastatic prostate cancer: a meta-analysis of primary results from two randomised controlled phase 3 trials of the STAMPEDE platform protocol. The Lancet 2022, 399, 447–460. [Google Scholar] [CrossRef]
- Agarwal, N.; Tangen, C.; Hussain, M.H.A.; Gupta, S.; Plets, M.; Lara, P.L.N.; Harzstark, A.; Twardowski, P.; Paller, C.J.; Zylla, D.M.; et al. SWOG S1216: A phase III randomized trial comparing androgen deprivation therapy (ADT) plus TAK-700 with ADT plus bicalutamide in patients (pts) with newly diagnosed metastatic hormone-sensitive prostate cancer (mHSPC) (NCT01809691). Journal of Clinical Oncology 2021, 39, 5001–5001. [Google Scholar] [CrossRef]
| Topic | Details | Related ongoing trials |
|---|---|---|
| Optimal agent following triplet therapy | Cabazitaxel: ability to overcome taxane resistance and manageable toxicity profile LuPSMA: targeted approach for PSMA-positive disease PARPis: potential benefits in HRRm patients Each agent offers distinct advantages depending on patient’s disease characteristics and prior treatment history |
CASCARA: Assessing quadruplet therapy (ADT + cabazitaxel/carboplatin + abiraterone) in high-volume mCSPC [106] UpFront PSMA: Evaluating LuPSMA in mCSPC [107] AMPLITUDE: Assessing efficacy of PARPis in mCSPC with HRRm [108] |
| Post-triplet therapy strategy for HRRm patients | - PARPi monotherapy: leveraging synthetic lethality in HRR-deficient cells with high efficacy and manageable safety profile - PARPis and ARPIs combination: potential synergistic effects but increased risk of compounded toxicities Early data from TALAPRO-3 suggest potential benefits |
TALAPRO-3: Assessing combination therapy of PARPi and ARPi in mCSPC [128] |
| Comparative efficacy of triplet therapy vs. PARPis in the HRRm population | Active research comparing triplet therapy and PARPis TALAPRO-3 and AMPLITUDE trials expected to provide critical insights Preliminary data suggest significant advantages of PARPis in HRRm patients |
AMPLITUDE and TALAPRO-3: expect to provide critical insights [108,128] |
| Need for head-to-head (combination of ADT with ARPIs vs. triplet combination of ADT, docetaxel, and ARPIs) |
Limited direct comparisons currently available Further research is needed to directly compare ADT + ARPIs vs. triplet therapy |
PEACE-1: Comparing SOC (ADT alone or with docetaxel) vs. SOC plus abiraterone in mCSPC [76] ARASENS: Comparing ADT with docetaxel + placebo vs. ADT with docetaxel + darolutamide in mCSPC [129] |
| Novel emerging therapeutic agents | LuPSMA: promising results in VISION and TheraP trials Capivasertib: targeting the PI3K-AKT pathway Pembrolizumab: evaluating the potential role in immunotherapy Emerging therapies likely to be approved for earlier-stage CSPC |
UpFront PSMA: evaluating LuPSMA in mCSPC [107] PSMAddition: comparing SOC (ADT with ARPI) vs. SOC plus 177Lu-PSMA-617 in mCSPC [130] CAPItello-281: assessing Capivasertib, abiraterone, and ADT in PTEN-deficient mCSPC [120] KEYNOTE-991: evaluating pembrolizumab, enzalutamide, and ADT in mCSPC [122] |
| Treatment intensification and de-escalation | Combining multiple agents raises questions about duration of therapy Intermittent regimens should be considered after initial treatment intensification in selected patients PSA kinetics may serve as biomarkers to guide treatment de-escalation; further trials needed |
STAMPEDE: Evaluating efficacy of ADT with various drug combinations [131] SWOG S1216: Evaluating treatment intensification and de-escalation strategies based on PSA response [132] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




