Submitted:
23 October 2024
Posted:
24 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Biosorption/Desorption Experiments and Data Analysis
2.3. Analytical Methods
3. Results and Discussion
3.1. Characterization of SB, SBW and BC-SBW Biosorbents
3.2. Establishing Optimal Biosorption Conditions
3.3. Evaluation of Biosorption Performances
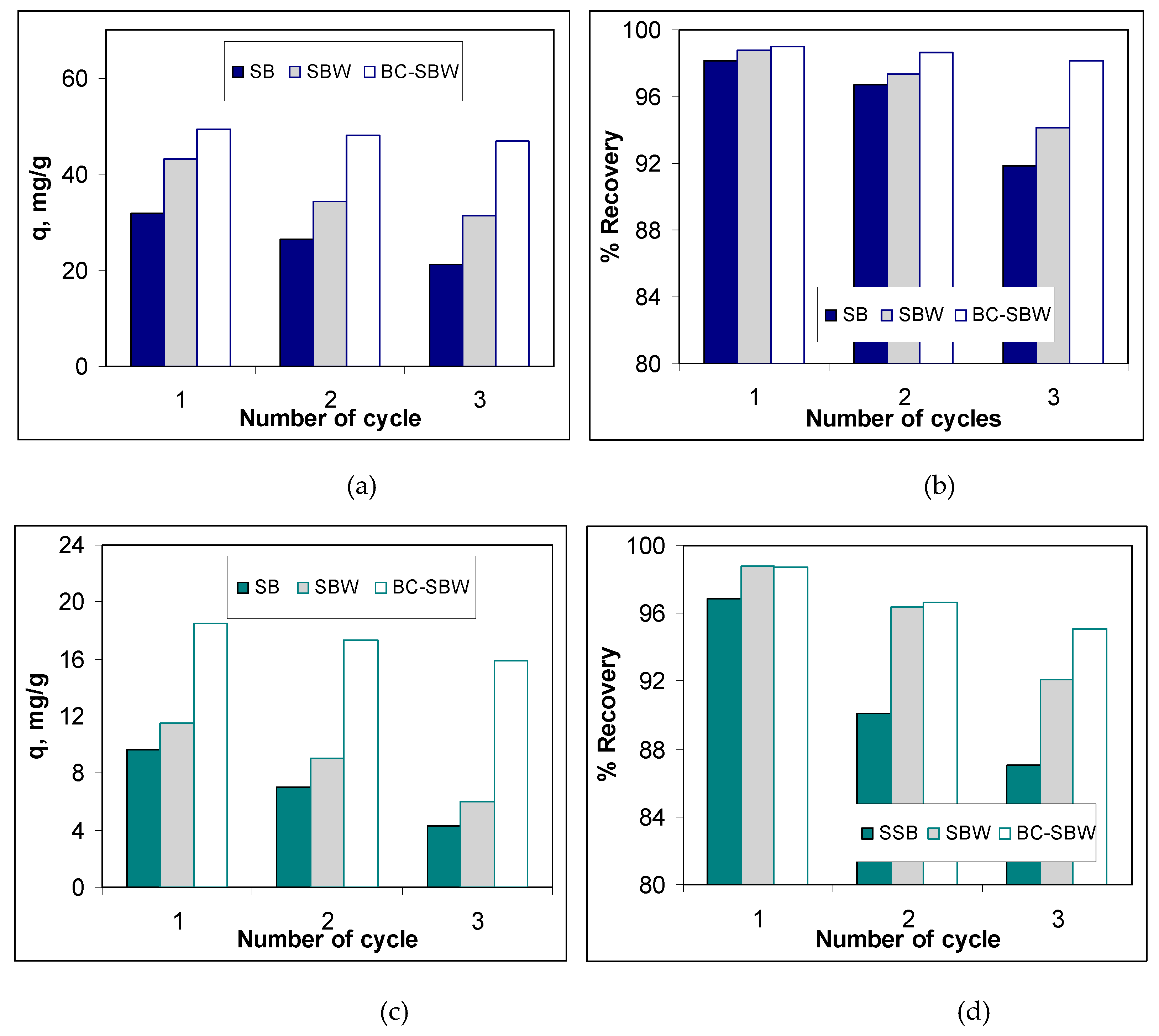
3.4. Evaluation of the Practical Applicability of Biosorbents
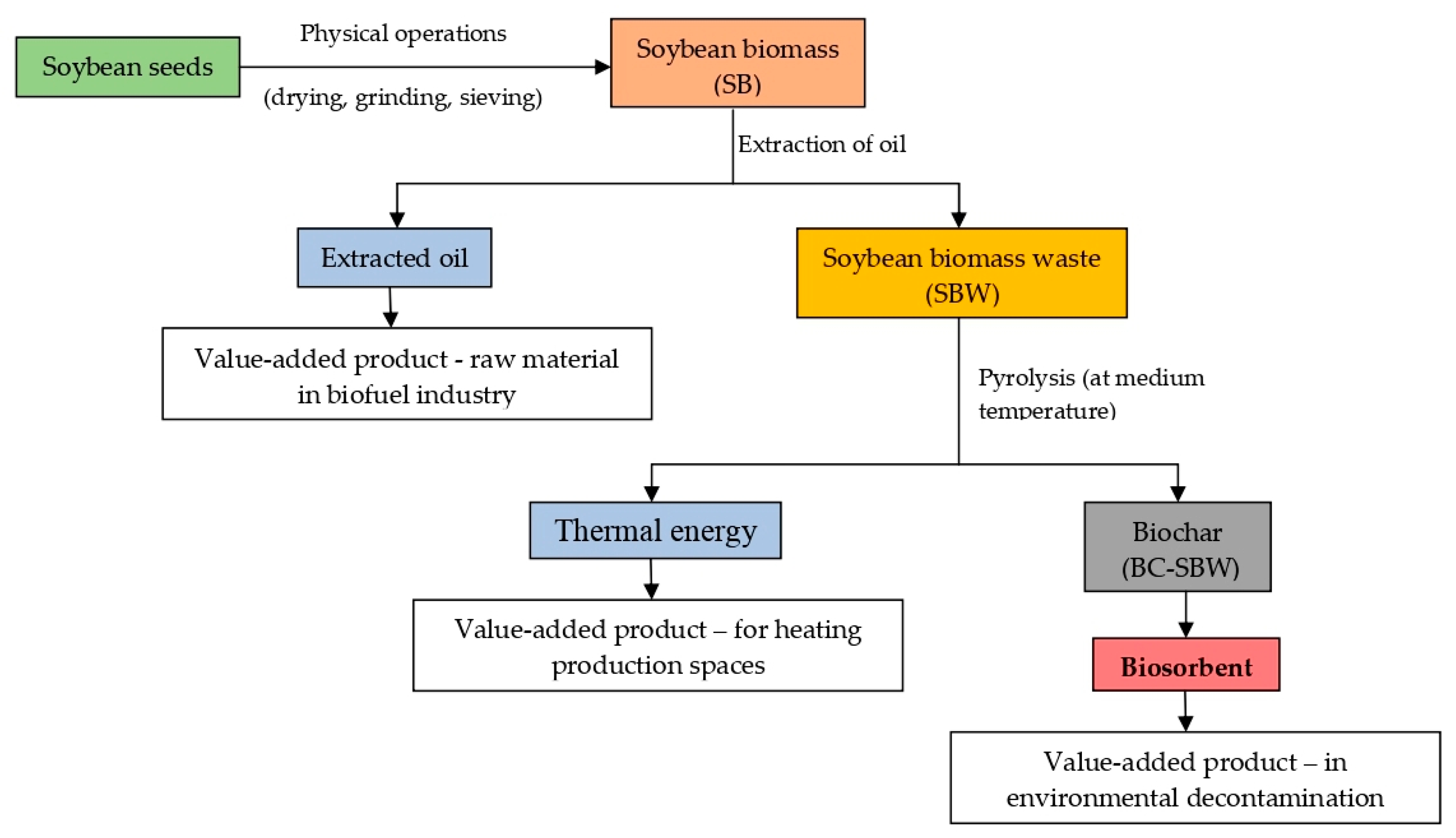
3.5. Circular Approach of Using Soybean Biomass in Environmental Decontamination
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Vareda, J.P.; Valente, A.J.M.; Duraes, L. Assessment of heavy metal pollution from anthropogenic activities and remediation strategies: A review. J. Environ. Manag. 2019, 246, 101–118. [Google Scholar] [CrossRef] [PubMed]
- Kakade, A.; Sharma, M.; El-Sayed, S.; Zhang, P.; Zhang, L.; Xing, X.; Yue, J.; Song, Z.; Nan, L.; Yujun, S.; Li, X. Heavy metals (HMs) pollution in the aquatic environment: Role of probiotics and gut microbiota in HMs remediation. Environ. Res. 2023, 115186. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett., 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Akpor, O.B.; Muchie, M. Environmental and public health implications of wastewater quality. Afr. J. Biotechnol. 2011, 10, 2379–2387. [Google Scholar]
- Briffa, J.; Sinagra, E.; Blundell, R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon, 2020, 6, e04691. [Google Scholar] [CrossRef]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, Q.M.R. Heavy metals and human health: mechanistic insight into toxicity and counter defense system of antioxidants. Int. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef]
- Martin, S.; Griswold, W. Human health effects of heavy metals. Environ. Sci. Technol. Briefs for Citizens, 2009, 15, 1–6. [Google Scholar]
- Saleh, T.A.; Mustaqeem, M.; Khaled, M. Water treatment technologies in removing heavy metal ions from wastewater: A review. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100617. [Google Scholar] [CrossRef]
- Razzak, S.A.; Faruque, M.O.; Alsheikh, Z.; Alsheikhmohamad, L.; Alkuroud, D.; Alfayez, A.; Zakir Hossain, S.M.; Hossain, M.M. A comprehensive review on conventional and biological-driven heavy metals removal from industrial wastewater. Environ. Adv. 2022, 7, 100168. [Google Scholar] [CrossRef]
- Bashir, A.; Malik, L.A.; Ahad, S.; Manzoor, T.; Bhat, M.A.; Dar, G.N.; Pandith, A.H. Removal of heavy metal ions from aqueous system by ion-exchange and biosorption methods. Environ. Chem. Lett. 2019, 17, 729–754. [Google Scholar] [CrossRef]
- Karic, N.; Maia, A.S.; Teodorovic, A.; Atanasova, N.; Langergraberf, G.; Crini, G.; Ribeiro, A.R.L.; Dolich, M. Bio-waste valorisation: Agricultural wastes as biosorbents for removal of (in)organic pollutants in wastewater treatment. Chem. Eng. J. Adv. 2022, 9, 100239. [Google Scholar] [CrossRef]
- Zhang, Y.; Duan, X. Chemical precipitation of heavy metals from wastewater by using the synthetical magnesium hydroxy carbonate. Water Sci. Technol. 2020, 81, 1130–1136. [Google Scholar] [CrossRef]
- Xiang, H.; Min, X.; Tang, C.J.; Sillanpaa, M.; Zhao, F. Recent advances in membrane filtration for heavy metal removal from wastewater: A mini review. J. Water Proc. Eng. 2022, 49, 103023. [Google Scholar] [CrossRef]
- Lumami Kapepula, V.; García Alvarez, M.; Sang Sefidi, V.; Buleng Njoyim Tamungang, E.; Ndikumana, T.; Musibono, D.-D.; Van Der Bruggen, B.; Luis, P. Evaluation of Commercial Reverse Osmosis and Nanofiltration Membranes for the Removal of Heavy Metals from Surface Water in the Democratic Republic of Congo. Clean Technol. 2022, 4, 1300–1316. [Google Scholar] [CrossRef]
- Razzak, S.A.; Faruque, M.O.; Alsheikh, Z.; Alsheikhmohamad, L.; Alkuroud, D.; Alfayez, A.; Zakir Hossain, S.M.; Hossain, M.M. A comprehensive review on conventional and biological-driven heavy metals removal from industrial wastewater. Environ. Adv. 2022, 7, 100168. [Google Scholar] [CrossRef]
- Syeda, H.I.; Sultan, I.; Razavi, K.S.; Yap, P.S. Biosorption of heavy metals from aqueous solution by various chemically modified agricultural wastes: A review. J. Water Proc. Eng. 2022, 46, 102446. [Google Scholar] [CrossRef]
- Zahar, M.N.; Parveen, A.; Nadeem, R. A pretreated green biosorbent based on Neem leaves biomass for the removal of lead from wastewater. Desalin. Water Treat. 2013, 51, 4459–4466. [Google Scholar]
- Vasic, V.; Kukic, D.; Sciban, M.; Durisic-Mladenovic, N.; Velic, N.; Pajin, B.; Crespo, J.; Farre, M.; Seres, Z. Lignocellulose-Based Biosorbents for the Removal of Contaminants of Emerging Concern (CECs) from Water: A Review. Water, 2023, 15, 1853. [Google Scholar] [CrossRef]
- Rakhym, A.B.; Seilkhanova, G.A.; Kurmanbayeva, T.S. Adsorption of lead (II) ions from water solutions with natural zeolite and chamotte clay. Mater. Today: Proc. 2020, 31, 482–485. [Google Scholar] [CrossRef]
- Kamali, M; Esmaeili, H. ; Tamjidi, S. Synthesis of Zeolite Clay/Fe-Al Hydrotalcite Composite as a Reusable Adsorbent for Adsorption/Desorption of Cationic Dyes. Arabian J. Sci. Eng. 2022, 47, 6651–6665. [Google Scholar] [CrossRef]
- Sánchez-Ponce, L.; Díaz-de-Alba, M.; Casanueva-Marenco, M.J.; Gestoso-Rojas, J.; Ortega-Iguña, M.; Galindo-Riaño, M.D.; Granado-Castro, M.D. Potential Use of Low-Cost Agri-Food Waste as Biosorbents for the Removal of Cd(II), Co(II), Ni(II) and Pb(II) from Aqueous Solutions. Separations 2022, 9, 309. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, Y.; Tang, J.; Yang, Z.; Zhang, L.; Huang, X. New insights into the interactions between Pb(II) and fruit waste biosorbent. Chemosphere 2022, 303. [Google Scholar] [CrossRef]
- Torres, E. Biosorption: A Review of the Latest Advances. Processes 2020, 8, 1584. [Google Scholar] [CrossRef]
- Cheng, M.H.; Dien, B.S.; Sing, V. Economics of plant oil recovery: A review. Biocatal. Agric. Biotechnol. 2019, 18, 2019–101056. [Google Scholar] [CrossRef]
- Liu, Z.; Gui, M.; Xu, T.; Zhang, L.; Kong, L.; Qin, L.; Zou, Z. Efficient aqueous enzymatic-ultrasonication extraction of oil from Sapindus mukorossi seed kernels. Ind. Crops. Prod. 2019, 134, 124–133. [Google Scholar] [CrossRef]
- Tran, K.Q.; Werle, S.; Trinh, T.T.; Magdziarz, A.; Sobek, S.; Pogrzeba, M. Fuel characterization and thermal degradation kinetics of biomass from phytoremediation plants. Biomass Bioenerg. 2020, 134, 105469. [Google Scholar] [CrossRef]
- Ugwu, E.I. , Agunwamba, J.C. A review on the applicability of activated carbon derived from plant biomass in adsorption of chromium, copper, and zinc from industrial wastewater. Environ. Monit. Assess. 2020, 192, 240. [Google Scholar] [CrossRef]
- Joshua, O. Ighalo, J.O.; Iwuchukwu, F.U.; Eyankware, O.E.; Iwuozor, K.O.; Olotu, K.; Bright, O.C.; Igwegbe, C.A. Flash pyrolysis of biomass: a review of recent advances. Clean Technol. Environ. Policy 2022, 24, 2349–2363. [Google Scholar]
- Huiyan Zhang, H.; Yang, K.; Tao, Y.; Yang, Q.; Xu, L.; Liu, C.; Ma, L.; Xiao, R. Biomass directional pyrolysis based on element economy to produce high-quality fuels, chemicals, carbon materials – A review. Biotechnol. Adv. 2023, 69, 108262. [Google Scholar]
- Tan, K.L.; Hameed, B.H. Insight into the adsorption kinetics models for the removal of contaminants from aqueous solutions. J.Taiwan Inst. Chem. Eng. 2017, 74, 25–48. [Google Scholar] [CrossRef]
- Demiral, I.; Samdan, C.; Demiral, H. Enrichment of the surface functional groups of activated carbon by modification method. Surf. Interfaces 2021, 22, 100873. [Google Scholar] [CrossRef]
- Sahmoune, M.N. Evaluation of thermodynamic parameters for adsorption of heavy metals by green adsorbents. Environ. Chem. Lett. 2019, 17, 697–704. [Google Scholar] [CrossRef]
- Rangabhashiyam, S.; Anu, N.; Nandagopal Giri, M.S.; Selvaraju, N. Relevance of isotherm models in biosorption of pollutants by agricultural by-products. J. Environ. Chem. Eng. 2014, 2, 398–414. [Google Scholar] [CrossRef]
- Fresenius, W.; Quentin, K.E.; Schneider, W. Water Analysis. A Practical Guide to Physico-Chemical, Chemical and Microbiological Water Examination and Quality Assurance; Springer: Berlin, Germany, 1988. [Google Scholar]
- NTPA 002, Governmental Decision no. 352 of April 21, 2005 on amending and supplementing Governmental Decision no. 188/2002 for the approval of some norms regarding the conditions for discharging wastewater into the aquatic environment, Romanian Official Monitor, no. 398 of May 11, 2005, http://www.rowater.ro/dacrisuri/20352_21.04.2005.pdf. Accessed on 15 June, 2024.
- Redha, A.A. Removal of heavy metals from aqueous media by biosorption. Arab J. Basi Appl. Sci. 2020, 1, 183–193. [Google Scholar]
- Gu, S.; Lan, C.Q. Biosorption of heavy metal ions by green alga Neochloris oleoabundans: Effects of metal ion properties and cell wall structure. J. Hazard. Mater. 2021, 418, 126336. [Google Scholar] [CrossRef]
- Sheikh, Z.; Amin, M.; Khan, N.; Khan, M.N.; Sami, S.K.; Khan, S.B.; Hafeez, I.; Khan, S.S.; Bakhsh, E.M.; Cheng, C.K. Potential application of Allium Cepa seeds as a novel biosorbent for efficient biosorption of heavy metals ions from aqueous solution. Chemosphere 2021, 279, 130545. [Google Scholar] [CrossRef]
- Bansal, M.; Garg, R.; Garg, V.K.; Garg, R.; Singh, D. Sequestration of heavy metal ions from multi-metal simulated wastewater systems using processed agricultural biomass. Chemosphere 2022, 296, 133966. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K.; Witek-Krowiak, A. State of the art for the biosorption process - A review. Appl. Biochem. Biotechnol. 2013, 170, 1389–1416. [Google Scholar] [CrossRef] [PubMed]
- Hoang, A.T.; Nizeti, S.; Cheng, C.K.; Luque, R.; Thomas, S.; Banh, T.L.; Pham, V.V.; Nguyen, X.P. Heavy metal removal by biomassderived carbon nanotubes as a greener environmental remediation: A comprehensive review. Chemosphere 2022, 287, 131959. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, T.; Zhang, H.; Liu, Y.; Xing, B. Adsorption of Pb(II) and Cd(II) by magnetic activated carbon and its mechanism. Sci. Total Environ. 2021, 757, 143910. [Google Scholar] [CrossRef]
- Madela, M. , Skuza, M.Towards a Circular Economy: Analysis of the Use of Biowaste as Biosorbent for the Removal of Heavy Metals. Energies 2021, 14, 5427. [Google Scholar] [CrossRef]
- Ali, R.M.; Hamad, H.A.; Hussein, M.M.; Malash, G.F. Potential of using green adsorbent of heavy metal removal from aqueous solutions: Adsorption kinetics, isotherm, thermodynamic, mechanism and economic analysis. Ecol. Eng. 2016, 91, 317–332. [Google Scholar] [CrossRef]
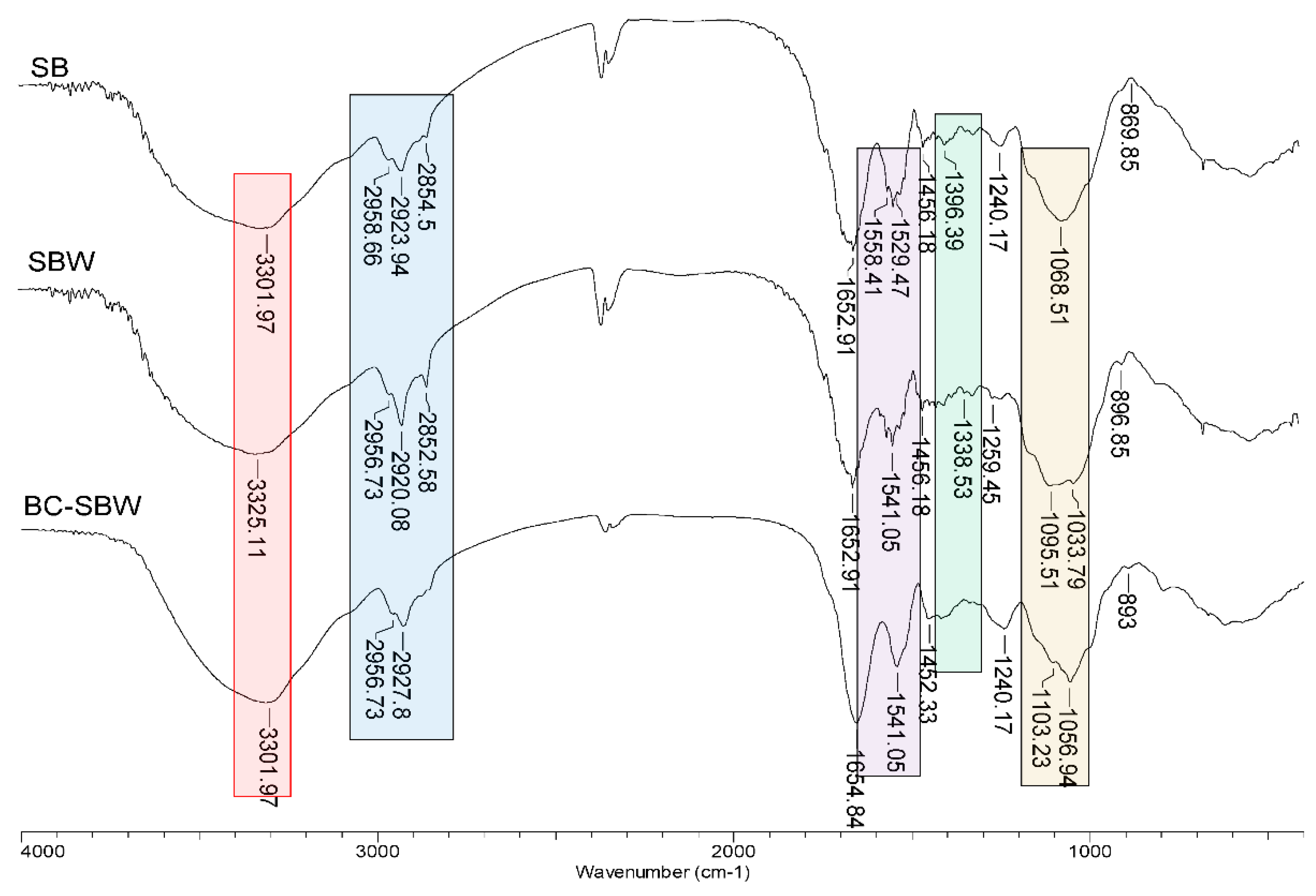

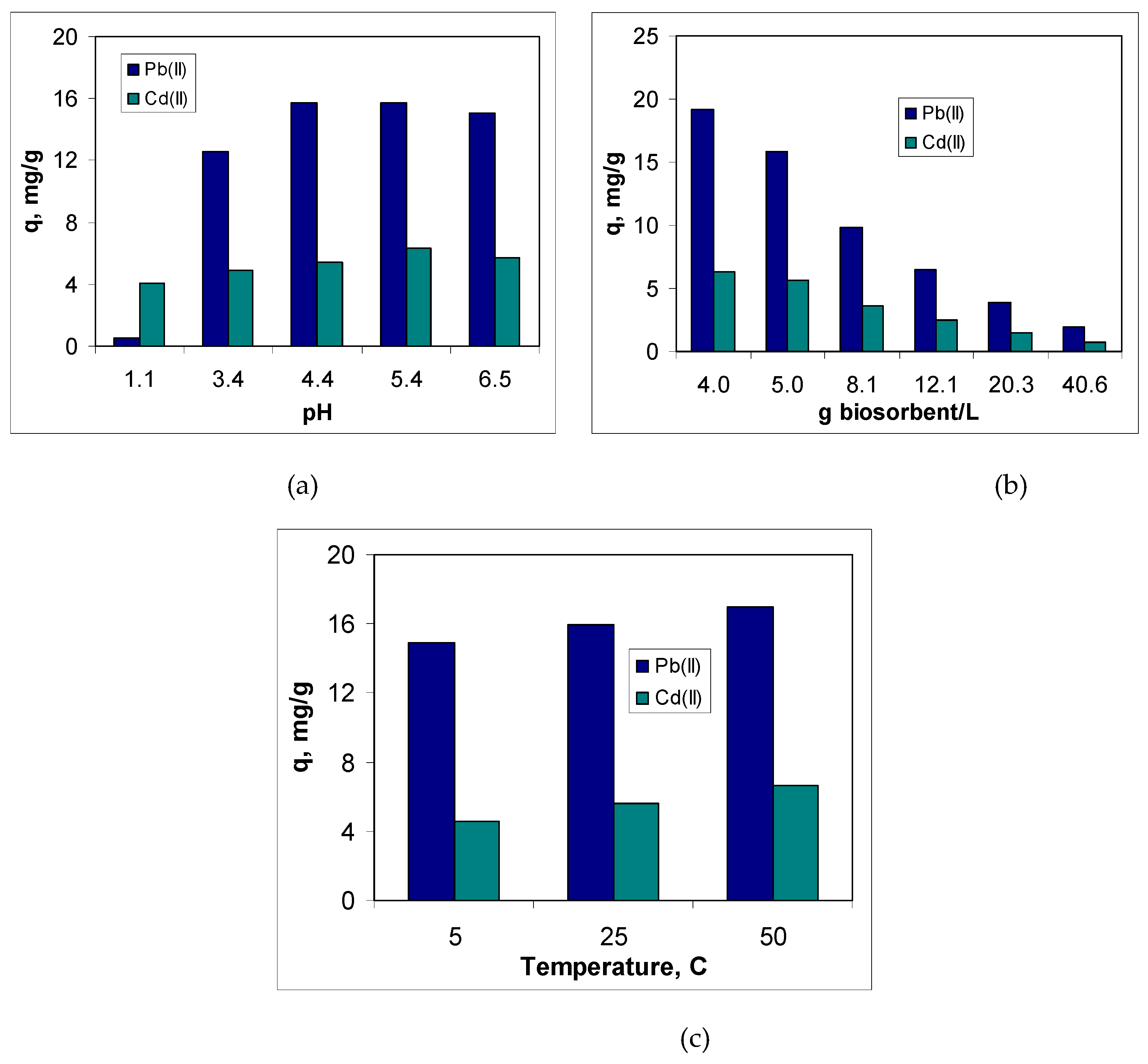
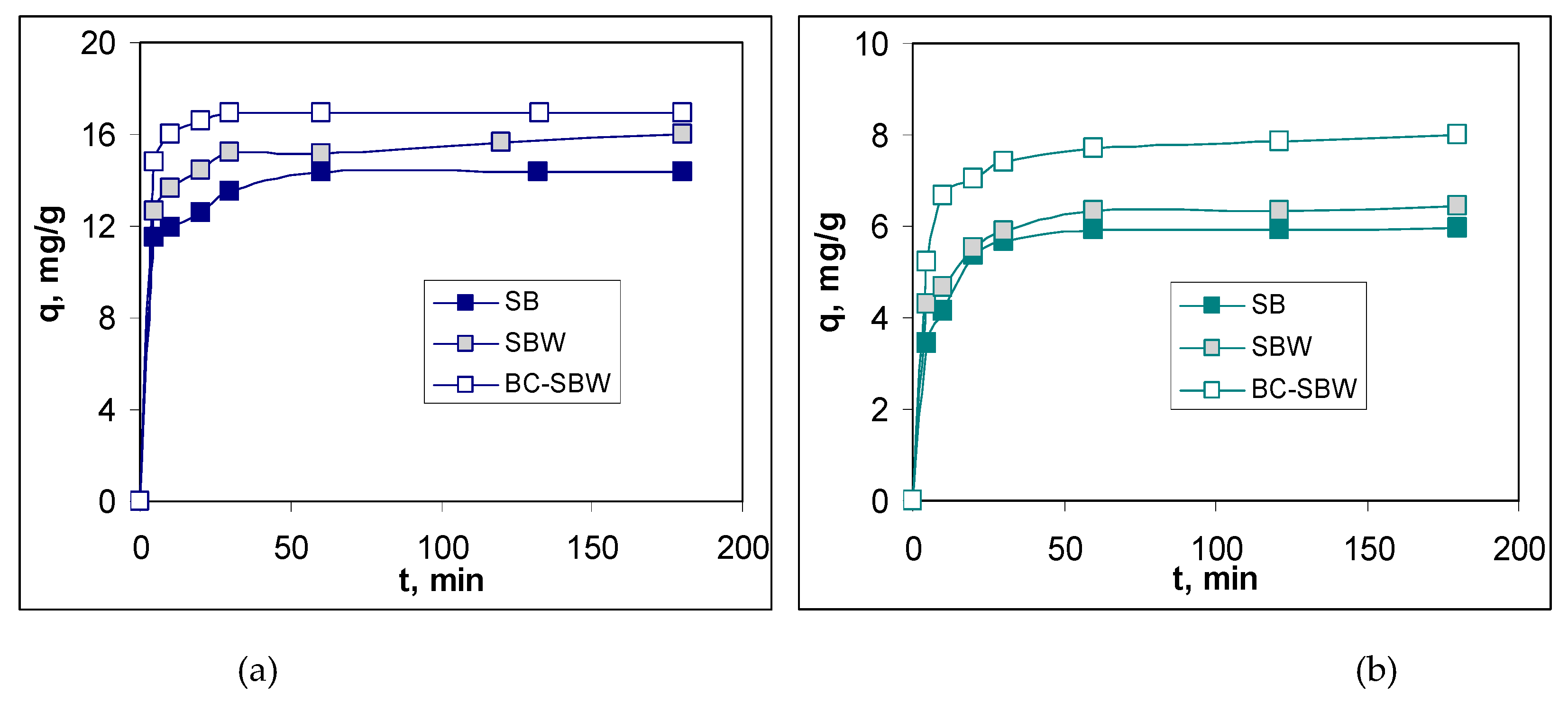
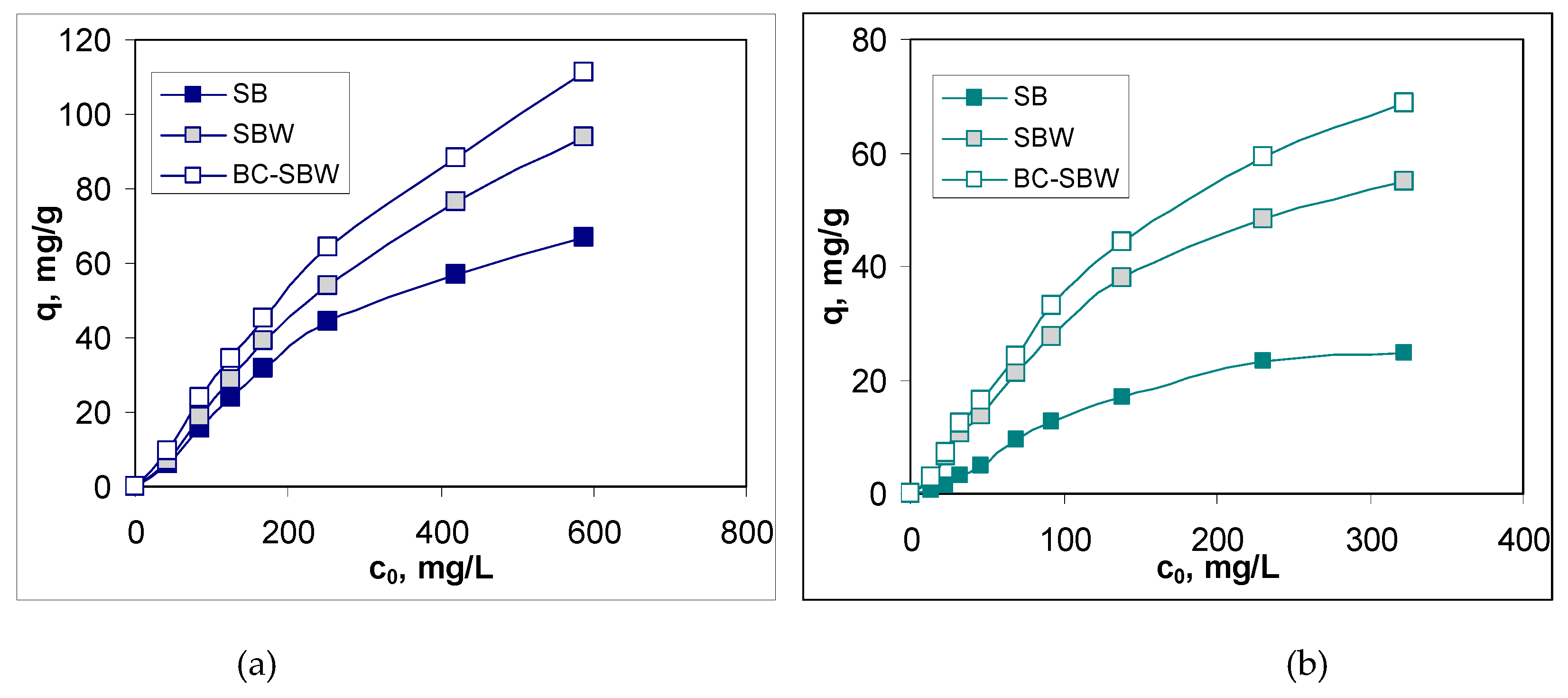
| Biosorbent | Pb(II) | Cd(II) | ||
| pHi | pHf | pHi | pHf | |
| SB | 5.40 | 5.21 | 5.40 | 5.18 |
| SBW | 5.40 | 5.52 | 5.40 | 5.61 |
| BC-SBW | 5.40 | 6.07 | 5.40 | 5.98 |
| Biosorbent | Pb(II) | Cd(II) | ||
| 60 min | 180 min | 60 min | 180 min | |
| SB | 95.32 % | 95.56 % | 64.48 % | 65.35 % |
| SBW | 96.43 % | 96.18 % | 66.16 % | 72.43 % |
| BC-SBW | 98.71 % | 99.47 % | 79.71 % | 84.86 % |
| Biosorbent | Pb(II) | Cd(II) | ||
| 41.93 mg/L | 587.03 mg/L | 13.83 mg/L | 322.81 mg/L | |
| SB | 79.84 % | 61.20 % | 50.47 % | 39.43 % |
| SBW | 90.78 % | 80.23 % | 76.88 % | 59.13 % |
| BC-SBW | 99.49 % | 89.87 % | 89.32 % | 63.41 % |
| Model parameter | Pb(II) | Cd(II) | ||||
| SB | SBW | BC-SBW | SB | SBW | BC-SBW | |
| Pseudo-first order model | ||||||
| R2 | 0.9554 | 0.9751 | 0.9642 | 0.9517 | 0.9684 | 0.9616 |
| qecalc, mg/g | 3.5237 | 4.2143 | 4.6751 | 0.4533 | 0.7621 | 1.0732 |
| k1, 1/min | 0.0175 | 0.0234 | 0.0276 | 0.0252 | 0.0276 | 0.0281 |
| Pseudo-second order model | ||||||
| R2 | 0.9998 | 0.9997 | 0.9995 | 0.9998 | 0.9997 | 0.9998 |
| qecalc, mg/g | 16.1032 | 16.9814 | 17.4611 | 6.0423 | 7.7061 | 9.0237 |
| k2, g/mg min | 0.0384 | 0.0288 | 0.0214 | 0.0103 | 0.0121 | 0.0163 |
| Langmuir model | ||||||
| R2 | 0.9876 | 0.9899 | 0.9795 | 0.9465 | 0.9691 | 0.9892 |
| qmax, mg/g | 69.4314 | 99.8163 | 116.8314 | 25.6348 | 36.1207 | 49.1031 |
| KL, L/g | 0.3718 | 0.4047 | 0.4303 | 0.2876 | 0.2993 | 0.3012 |
| Freundlich | ||||||
| R2 | 0.8986 | 0.8769 | 0.7996 | 0.7993 | 0.8703 | 0.9021 |
| 1/n | 0.023 | 0.018 | 0.011 | 0.117 | 0.146 | 0.102 |
| KF, L/g | 1.0234 | 2.2537 | 2.8324 | 1.3561 | 1.5247 | 1.8314 |
| Quality indicator |
Maximum permissible limit [35] | Initial | SB | SBW | BC-SBW |
| Pb(II) ions | |||||
| CPb(II), mg/L | 0.5 | 35.0 | 3.52 | 1.94 | 0.61 |
| pH | 6.5 – 8.5 | 5.4 | 5.6 | 5.9 | 6.9 |
| TSM*, mg/L | 350 | 21.8 | 43.4 | 33.4 | 46.8 |
| COD, mg O2/L | 500 | 278.4 | 326.8 | 288.3 | 259.2 |
| BOD5, mg O2/L | 300 | 65.03 | 71.2 | 58.4 | 50.4 |
| Cd(II) ions | |||||
| CCd(II), mg/L | 0.3 | 15.0 | 2.42 | 1.09 | 0.43 |
| pH | 6.5 – 8.5 | 5.4 | 5.7 | 6.1 | 7.1 |
| TSM*, mg/L | 350 | 21.8 | 46.1 | 44.12 | 48.3 |
| COD, mg O2/L | 500 | 278.4 | 318.4 | 291.3 | 257.9 |
| BOD5, mg O2/L | 300 | 65.03 | 69.8 | 57.9 | 49.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




