Submitted:
27 August 2024
Posted:
27 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Effects on Normal HDFs
2.2. Damage Resistance in HDFs Subjected to Photoaging Stimuli (UV Radiation)
2.3. Damage Resistance in HDFs Subjected to Glycation Stress
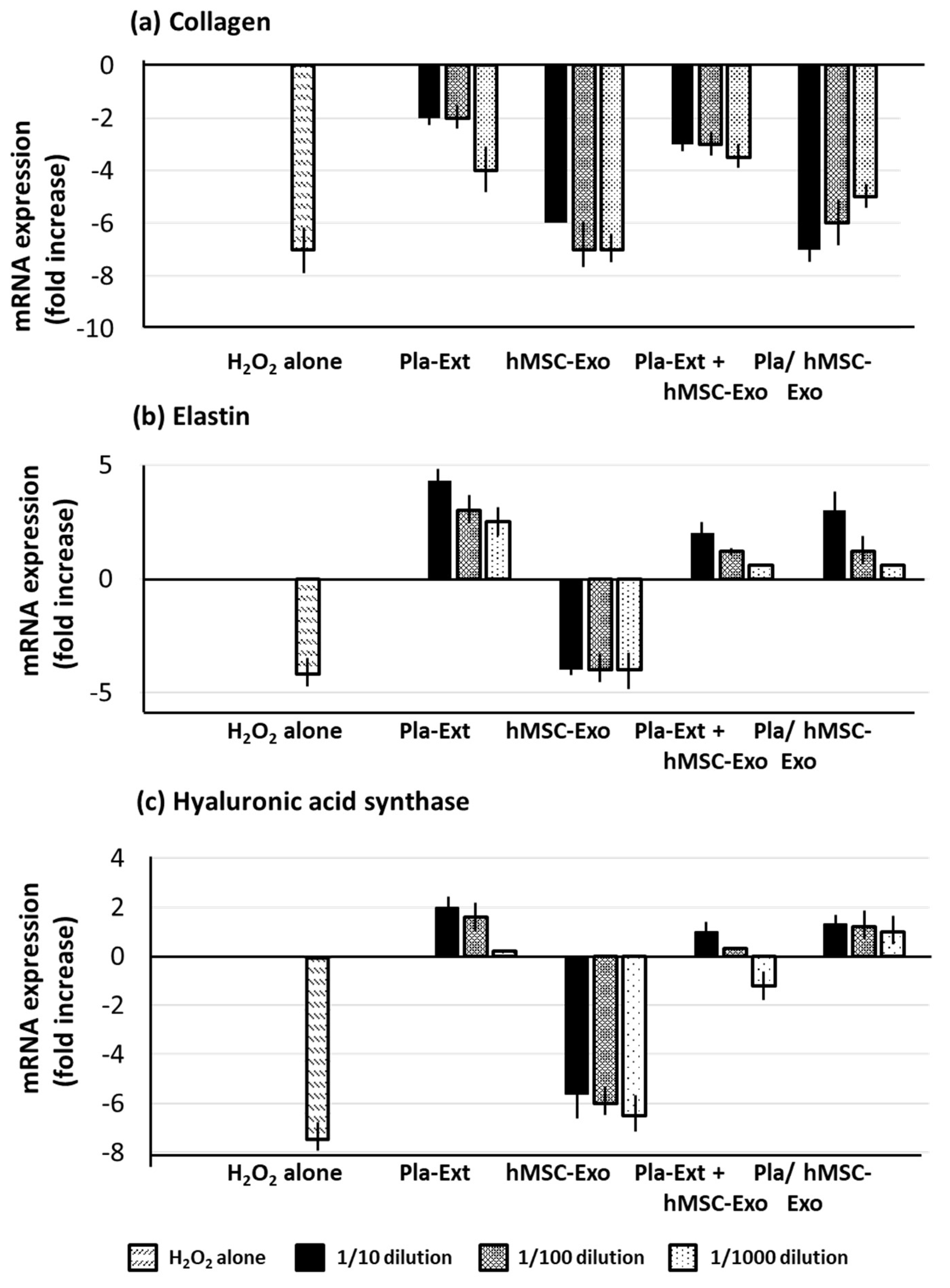
2.4. Damage Resistance in HDFs Subjected to Oxidative Stress
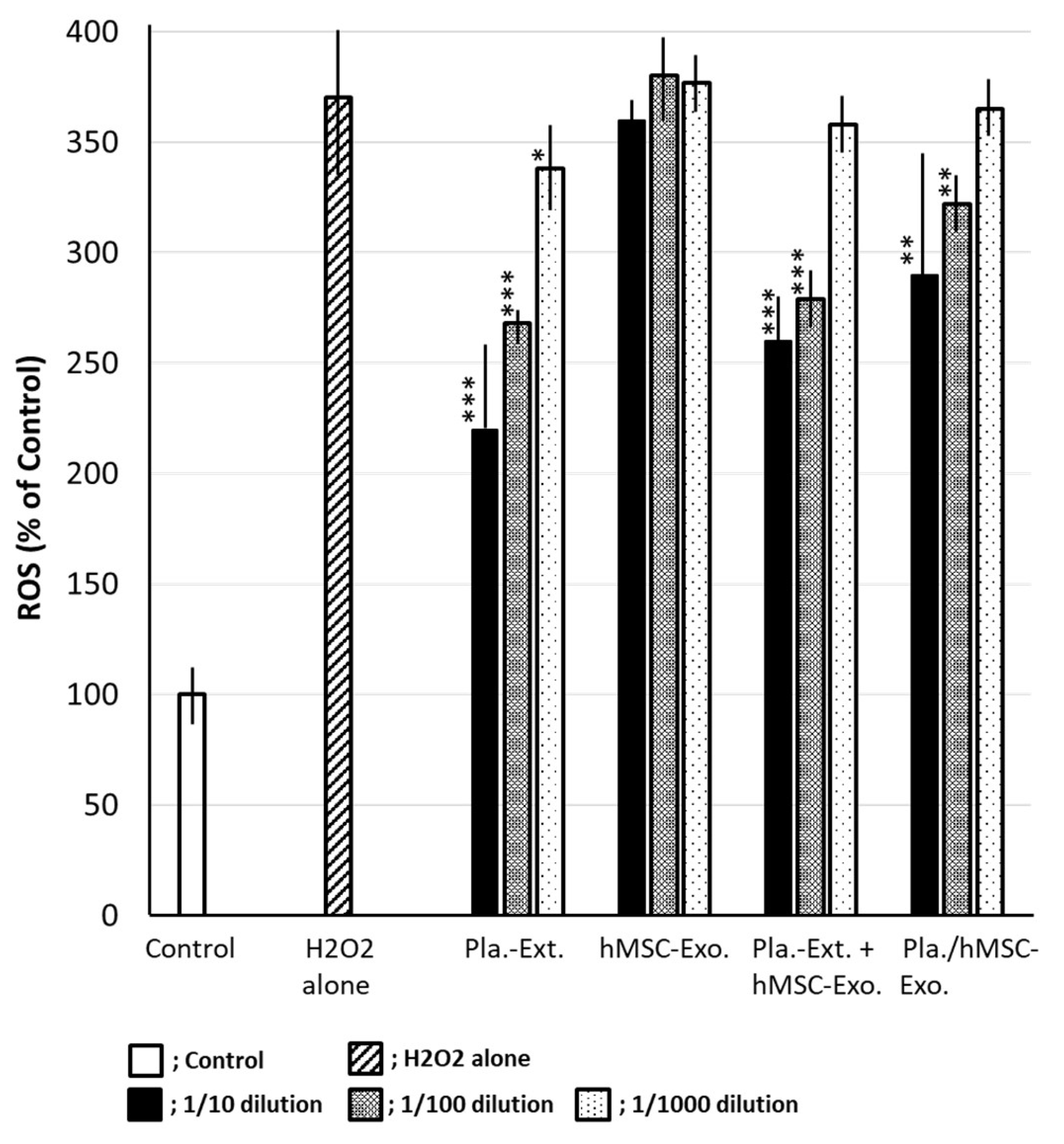
2.5. Inhibitory Efficacy against Intracellular ROS Accumulation in HDFs Subjected to Oxidative Stress
3. Discussion
4. Materials and Methods
4.1. Porcine Placental Extracts (Pla-Ext)
4.2. Cell
4.3. Isolation of Cultured Cell-Derived Exosomes
4.4. Photoaging (UV) Stimulation
4.5. AGE Generation
4.6. Oxidative Stress Reagent
4.7. Intracellular Total RNA Extraction
4.8. RT-qPCR
4.9. Determination of Intracellular Reactive Oxygen Species
4.10. Experimental Protocol
4.10.1. Effects on Normal HDFs
4.10.2. Resistance to Damage in HDFs Stimulated by Photoaging
4.10.3. AGE Receptor mRNA Expression in HDFs Subjected to Glycation Stress
4.10.4. Damage Resistance in HDFs Subjected to Oxidative Stress
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shin, S.H.; Lee, Y.H.; Rho, N.K.; Park, K.Y. Skin aging from mechanisms to interventions: focusing on dermal aging. Front. Physiol. 2023, 14, 1195272, Review. [Google Scholar] [CrossRef]
- Wang, Y.; Branicky, R.; Noë, A.; Hekimi, S. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J. Cell Biol. 2018, 217, 1915–1928. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Yang, S.; Lv, M.; Lv, J.; Sui, Y.; Guo, S. Protective roles of mesenchymal stem cells on skin photoaging: a narrative review. Tissue Cell 2022, 76, 101746. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Sun, Y.; Zhang, J.; Zhu, Q.; Yang, Y.; Niu, X.; Deng, Z.; Li, Q.; Wang, Y. Human embryonic stem cell-derived exosomes promote pressure ulcer healing in aged mice by rejuvenating senescent endothelial cells. Stem Cell Res. Ther. 2019, 10, 142. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.W.; Lee, W.J.; Hahn, S.B.; Kim, B.J.; Lew, D.H. The effect of human placenta extract in a wound healing model. Ann. Plast. Surg. 2010, 65, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Nath, S.; Bhattacharyya, D. Cell adhesion by aqueous extract of human placenta used as wound healer. Indian, J. Exp. Biol. 2007, 45, 732–738. [Google Scholar] [PubMed]
- Pan, S.Y.; Chan, M.; Wong, M.; Klokol, D.; Chernykh, V. Placental therapy: an insight to their biological and therapeutic properties. J. Med. Ther. 2017, 1, 12. [Google Scholar]
- Kong, M.H.; Lee, E.J.; Lee, S.Y.; Cho, S.J.; Hong, Y.S.; Park, S.B. Effect of human placental extract on menopausal symptoms, fatigue, and risk factors for cardiovascular disease in middle-aged Korean women. Menopause 2008, 15, 296–303. [Google Scholar] [CrossRef]
- Togashi, S.; Takahashi, N.; Iwama, M.; Watanabe, S.; Tamagawa, K.; Fukui, T. Antioxidative collagen-derived peptides in human-placenta extract. Placenta 2002, 23, 497–502. [Google Scholar] [CrossRef]
- Yoshikawa, C. Effect of porcine placental extract on collagen production in human skin fibroblasts in vitro. Gynecol. Obstet. 2013, 03, 0–4. [Google Scholar] [CrossRef]
- Tonello, G.; Daglio, M.; Zaccarelli, N.; Sottofattori, E.; Mazzei, M.; Balbi, A. Characterization and quantitation of the active polynucleotide fraction (PDRN) from human placenta, a tissue repair stimulating agent. J. Pharm. Biomed. Anal. 1996, 14, 1555–1560. [Google Scholar] [CrossRef]
- Takuma, K.; Mizoguchi, H.; Funatsu, Y.; Kitahara, Y.; Ibi, D.; Kamei, H.; Matsuda, T.; Koike, K.; Inoue, M.; Nagai, T.; Yamada, K. Placental extract improves hippocampal neuronal loss and fear memory impairment resulting from chronic restraint stress in ovariectomized mice. J. Pharmacol. Sci. 2012, 120, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Park, H.J.; Seo, H.G.; Kim, J.H.; Lim, G.S.; Lee, W.Y.; Kim, N.H.; Kim, J.H.; Lee, J.H.; Jung, H.S.; Sung, S.H.; Song, H. Immune modulation effect of porcine placenta extracts in weaned the pig. J. Anim. Sci. 2013, 91, 2405–2413. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.Y.; Kim, S.W.; Kim, B.; Lee, H.N.; Kim, S.J.; Song, M.; Kim, S.; Kim, J.; Kim, Y.B.; Kim, J.H.; Cho, S.G. Alpha-fetoprotein, identified as a novel marker for the antioxidant effect of placental extract, exhibits synergistic antioxidant activity in the presence of estradiol. PLOS ONE 2014, 9, e99421. [Google Scholar] [CrossRef]
- Matsuoka, T.; Takanashi, K.; Dan, K.; Yamamoto, K.; Tomobe, K.; Shinozuka, T. Effects of mesenchymal stem cell-derived exosomes on oxidative stress responses in skin cells. Mol. Biol. Rep. 2021, 48, 4527–4535. [Google Scholar] [CrossRef] [PubMed]
- Eckes, B.; Mauch, C.; Hüppe, G.; Krieg, T. Differential regulation of transcription and transcript stability of pro-alpha 1 (I) collagen and fibronectin in activated fibroblasts derived from patients with systemic scleroderma. Biochem. J. 1996, 315, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.A.; Yi, B.R.; Choi, K.C. Molecular mechanisms and in vivo mouse models of skin aging associated with dermal matrix alterations. Lab. Anim. Res. 2011, 27, 1–8. [Google Scholar] [CrossRef]
- Pal, P.; Roy, R.; Datta, P.K.; Dutta, A.K.; Biswas, B.; Bhadra, R. Hydroalcoholic human placental extract: skin pigmenting activity and gross chemical composition. Int. J. Dermatol. 1995, 34, 61–66. [Google Scholar] [CrossRef]
- Phan, J.; Kumar, P.; Hao, D.; Gao, K.; Farmer, D.; Wang, A. Engineering mesenchymal stem cells to improve their exosome efficacy and yield for cell-free therapy. J. Extracell. Vesicles 2018, 7, 1522236. [Google Scholar] [CrossRef]
- Cha, J.M.; Shin, E.K.; Sung, J.H.; Moon, G.J.; Kim, E.H.; Cho, Y.H.; Park, H.D.; Bae, H.; Kim, J.; Bang, O.Y. Efficient scalable production of therapeutic microvesicles derived from human mesenchymal stem cells. Sci. Rep. 2018, 8, 1171. [Google Scholar] [CrossRef]
- Ferreira, J.R.; Teixeira, G.Q.; Santos, S.G.; Barbosa, M.A.; Almeida-Porada, G.A.; Gonçalves, R.M. Mesenchymal stromal cell secretome: influencing therapeutic potential by cellular pre-conditioning. Front. Immunol. 2018, 9, 2837. [Google Scholar] [CrossRef] [PubMed]
- Pendse, S.; Kale, V.; Vaidya, A. Extracellular vesicles isolated from mesenchymal stromal cells primed with hypoxia: novel strategy in regenerative medicine. Curr. Stem Cell Res. 2021, 16, 243.e61. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Kim, H.W.; Gong, M.; Wang, J.; Millard, R.W.; Wang, Y.; Ashraf, M.; Xu, M. Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int. J. Cardiol. 2015, 182, 349–360. [Google Scholar] [CrossRef]
- Liang, L.; Zheng, D.; Lu, C.; Xi, Q.; Bao, H.; Li, W.; Gu, Y.; Mao, Y.; Xu, B.; Gu, X. Exosomes derived from miR-301a-3p-overexpressing adipose-derived mesenchymal stem cells reverse hypoxia-induced erectile dysfunction in rat models. Stem Cell Res. Ther. 2021, 12, 87. [Google Scholar] [CrossRef] [PubMed]
- Mazini, L.; Rochette, L.; Admou, B.; Amal, S.; Malka, G. Hopes and limits of adipose-derived stem cells (ADSCs) and mesenchymal stem cells (MSCs) in wound healing. Int. J. Mol. Sci. 2020, 21, 1306. [Google Scholar] [CrossRef]
- Gnecchi, M.; He, H.; Noiseux, N.; Liang, O.D.; Zhang, L.; Morello, F.; Mu, H.; Melo, L.G.; Pratt, R.E.; Ingwall, J.S.; Dzau, V.J. Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. F.A.S.E.B. J. 2006, 20, 661–669. [Google Scholar] [CrossRef]
- Maeda, S.; Matsui, T.; Ojima, A.; Takeuchi, M.; Yamagishi, S.I. Sulforaphane inhibits advanced glycation end product-induced pericyte damage by reducing expression of receptor for advanced glycation end products. Nutr. Res. 2014, 34, 807–813. [Google Scholar] [CrossRef]
- Kartal, B.; Akçay, A.; Palabiyik, B. Oxidative stress upregulates the transcription of genes involved in thiamine metabolism. Turk. J. Biol. 2018, 42, 447–452. [Google Scholar] [CrossRef]
- Liu, W.; Ma, C.; Li, H.Y.; Chen, L.; Yuan, S.S.; Li, K.J. MicroRNA-146a downregulates the production of hyaluronic acid and collagen I in Graves’ ophthalmopathy orbital fibroblasts. Exp. Ther. Med. 2020, 20, 38. [Google Scholar]
- Deslee, G.; Woods, J.C.; Moore, C.M.; Liu, L.; Conradi, S.H.; Milne, M.; Gierada, D.S.; Pierce, J.; Patterson, A.; Lewit, R.A.; Battaile, J.T.; Holtzman, M.J.; Hogg, J.C.; Pierce, R.A. Elastin expression in very severe human COPD. Eur. Respir. J. 2009, 34, 324–331. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
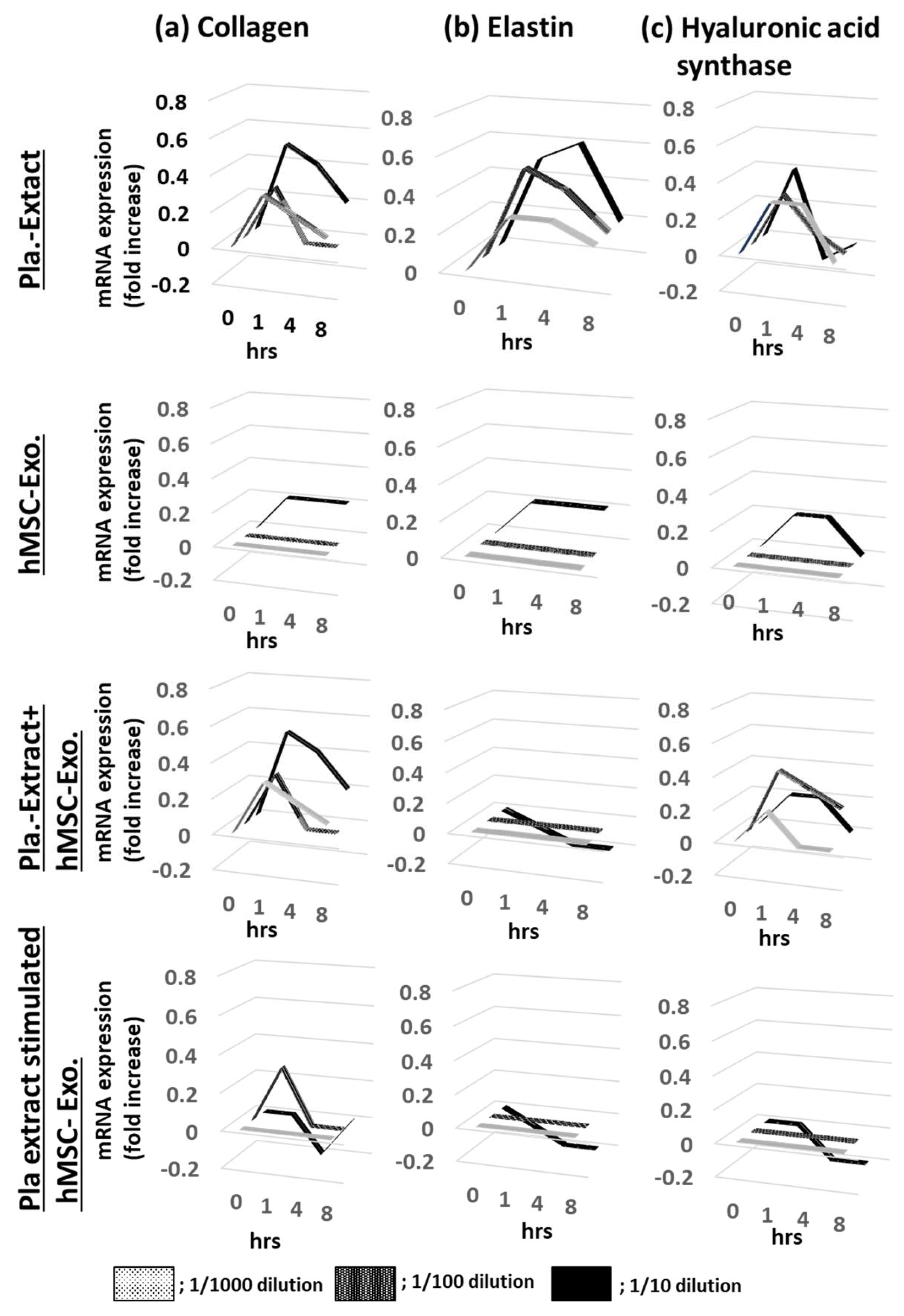
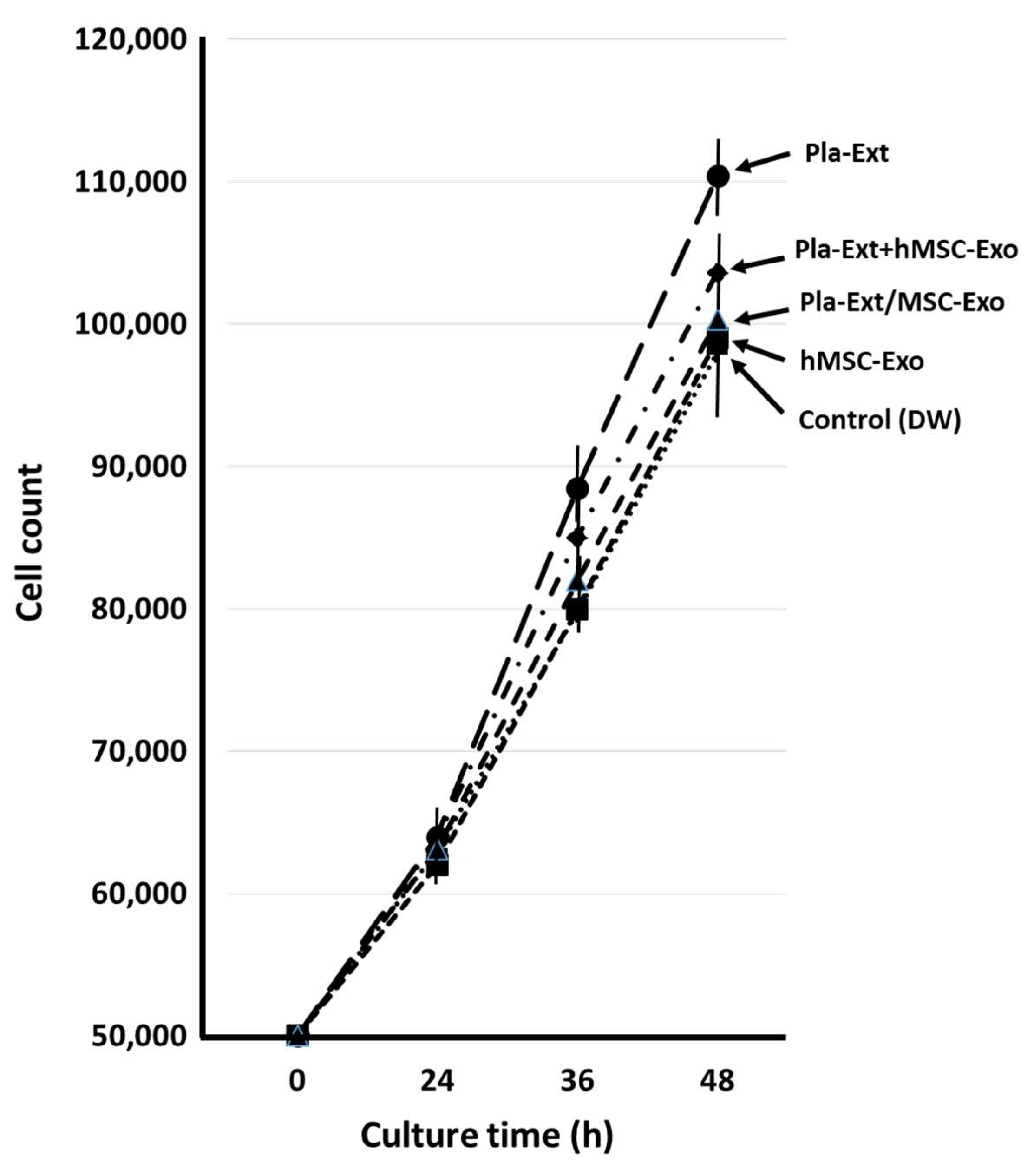
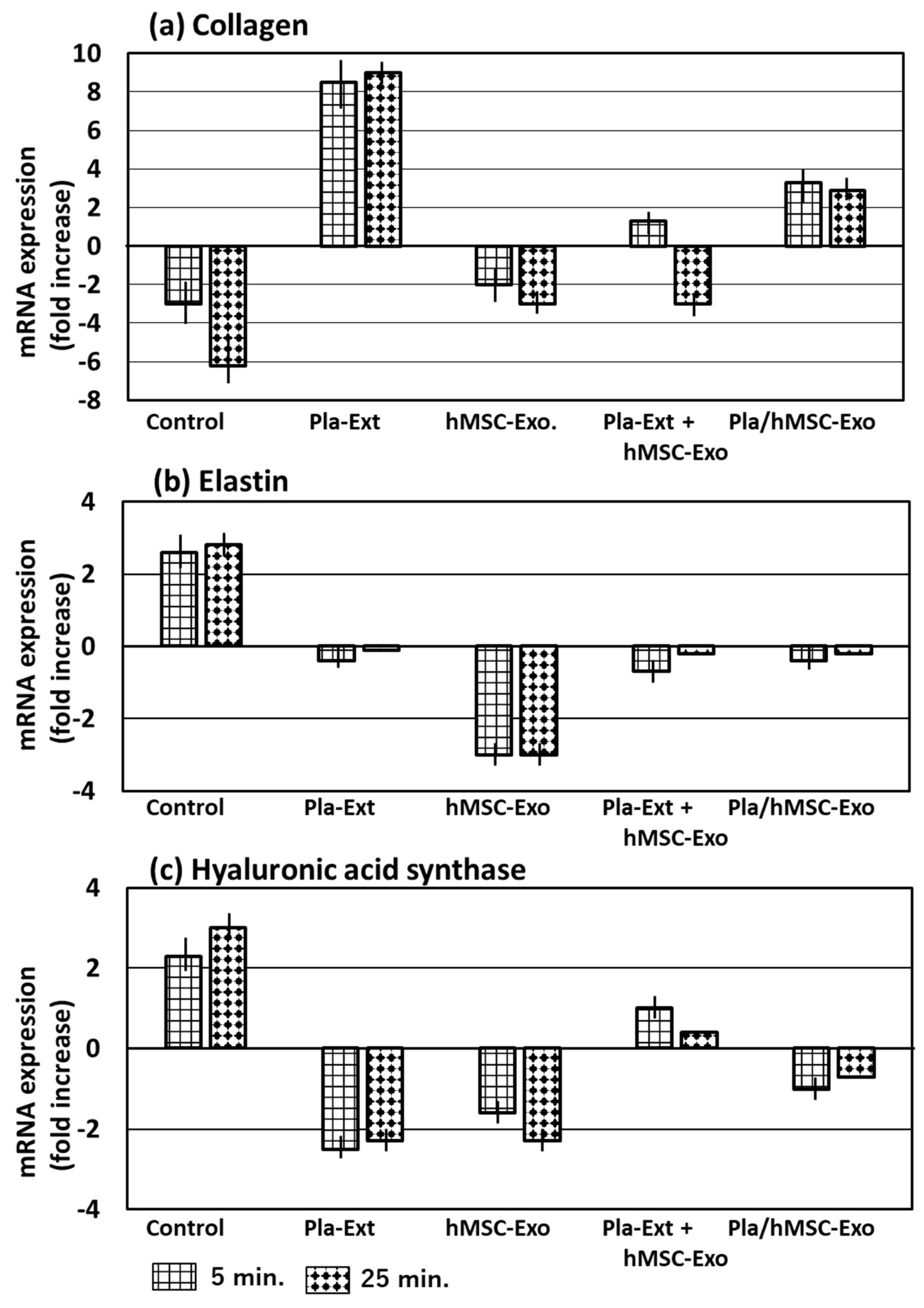
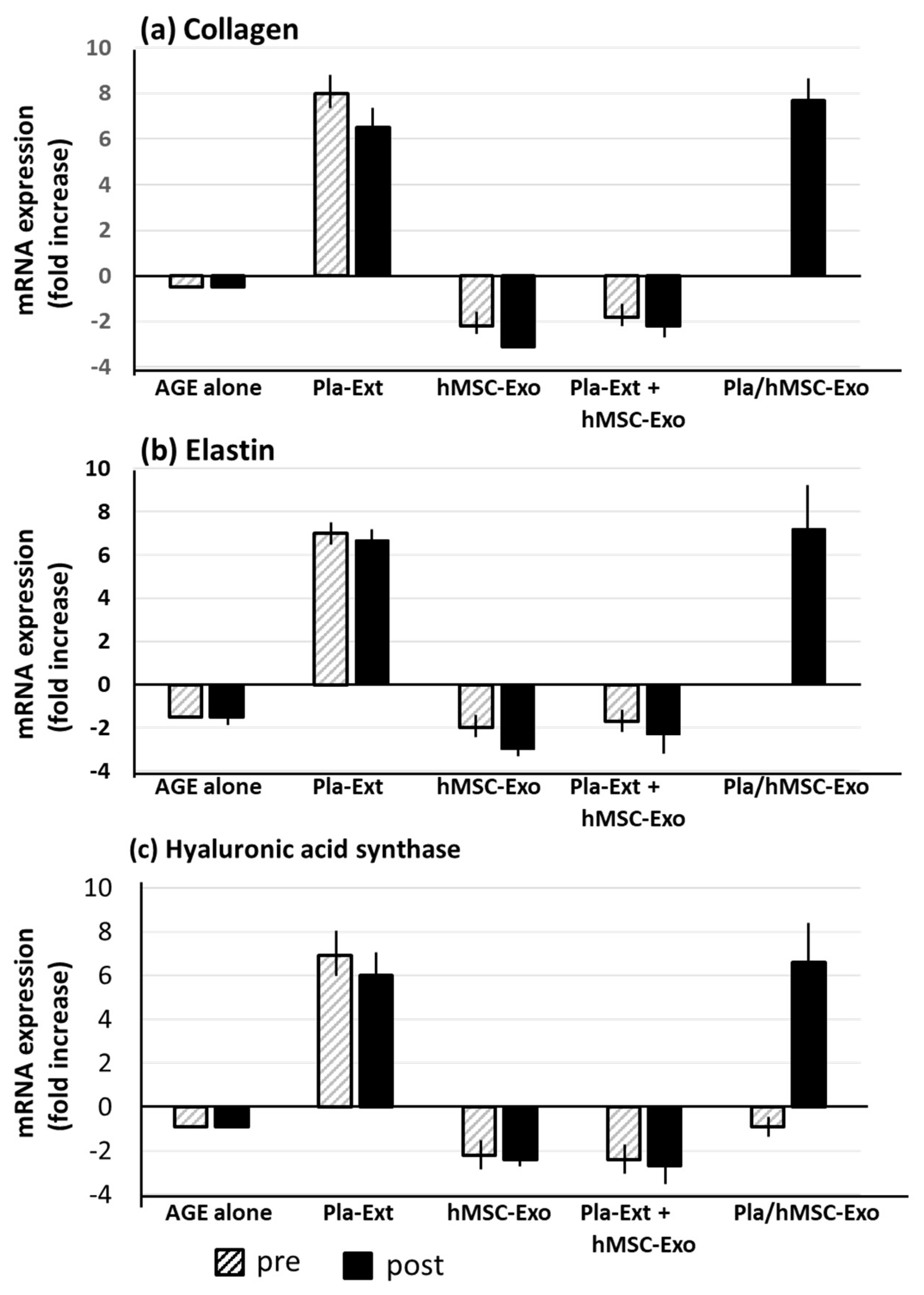
| Gene | Primer | Ref. |
|---|---|---|
| Collagen 1 A2 | Forward: CTGGACCTCCAGGTGTAAGC | [29] |
| Reverse: TGGCTGAGTCTCAAGTCACG | ||
| Elastin | Forward: GGCCATTCCTGGTGGAGTTCC | [30] |
| Reverse: AACTGGCTTAAGAGGTTTGCCTCCA | ||
| Hyaluronic acid synthase | Forward: CACGTAACGCAATTGGTCTTGTCC | [29] |
| Reverse: CCAGTGCTCTGAAGGCTGTGTAC | ||
| GAPDH | Forward: GACATGCCGCCTGGAGAAAC | [29] |
| Reverse: AGCCCAGGATGCCCTTTAGT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




