Submitted:
26 August 2024
Posted:
27 August 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Results
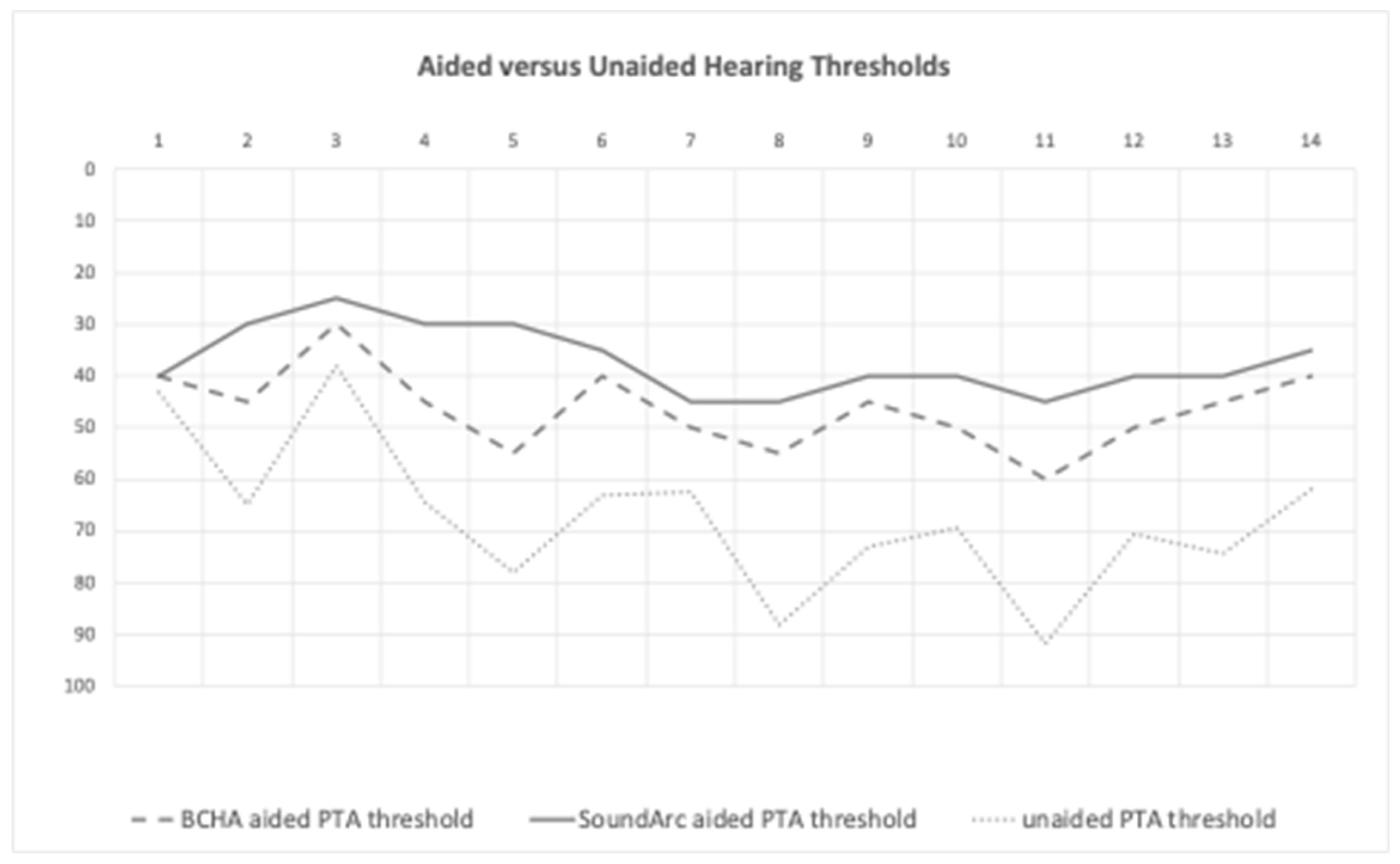
Aided versus Unaided Hearing Thresholds
Subjective Outcomes/Questionnaires
Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgements
Conflict of Interest
References
- Maier, H.; Lenarz, T.; Agha-Mir-Salim, P.; Agterberg, M.J.H.; Anagiotos, A.; Arndt, S.; Ball, G.; Bance, M.; Barbara, M.; Baumann, U.; et al. Consensus Statement on Bone Conduction Devices and Active Middle Ear Implants in Conductive and Mixed Hearing Loss. Otol. Neurotol. 2022, 43, 513–529. [Google Scholar] [CrossRef]
- Tjellström, A. Dimensions of Importance in Reconstructive Middle Ear Surgery. Acta Oto-Laryngologica 1977, 83, 488–490. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.P.; Symms, J.T.; Beasley, K.; Coffman, H.M. Bone conduction implants. Curr. Opin. Otolaryngol. Head Neck Surg. 2020, 28, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Wazen JJ, Wycherly B, Daugherty J. Complications of bone-anchored hearing devices. Adv Otorhinolaryngol. 2011;71:63-72. [CrossRef] [PubMed]
- Candreia, C.; Birrer, R.; Fistarol, S.; Kompis, M.; Caversaccio, M.; Arnold, A.; Stieger, C. Predisposing factors for adverse skin reactions with percutaneous bone anchored hearing devices implanted with skin reduction techniques. 2016, 273, 4185–4192. [CrossRef]
- Kurz, A.; Flynn, M.; Caversaccio, M.; Kompis, M. Speech Understanding with a New Implant Technology: A Comparative Study with a New Nonskin Penetrating Baha System. BioMed Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mclean, T.; Pai, I.; Philipatos, A.; Gordon, M. The Sophono Bone-Conduction System: Surgical, Audiologic, and Quality-of-Life Outcomes. Ear, Nose Throat J. 2017, 96, E28–E33. [Google Scholar] [CrossRef] [PubMed]
- Riss, D.; Arnoldner, C.; Baumgartner, W.-D.; Blineder, M.; Flak, S.; Bachner, A.; Gstoettner, W.; Hamzavi, J.-S. Indication criteria and outcomes with the Bonebridge transcutaneous bone-conduction implant. Laryngoscope 2014, 124, 2802–2806. [Google Scholar] [CrossRef]
- Sylvester, D.C.; Gardner, R.; Reilly, P.G.; Rankin, K.; Raine, C.H. Audiologic and Surgical Outcomes of a Novel, Nonpercutaneous, Bone Conducting Hearing Implant. Otol. Neurotol. 2013, 34, 922–926. [Google Scholar] [CrossRef]
- Powell, H.R.; Rolfe, A.M.; Birman, C.S. A Comparative Study of Audiologic Outcomes for Two Transcutaneous Bone-Anchored Hearing Devices. Otol. Neurotol. 2015, 36, 1525–1531. [Google Scholar] [CrossRef]
- Gerdes, T.; Salcher, R.B.; Schwab, B.; Lenarz, T.; Maier, H. Comparison of Audiological Results Between a Transcutaneous and a Percutaneous Bone Conduction Instrument in Conductive Hearing Loss. Otol. Neurotol. 2016, 37, 685–691. [Google Scholar] [CrossRef]
- Magele, A.; Schoerg, P.; Stanek, B.; Gradl, B.; Sprinzl, G.M. Active transcutaneous bone conduction hearing implants: Systematic review and meta-analysis. PLOS ONE 2019, 14, e0221484. [Google Scholar] [CrossRef]
- Rauch, A.-K.; Wesarg, T.; Aschendorff, A.; Speck, I.; Arndt, S. Long-term data of the new transcutaneous partially implantable bone conduction hearing system Osia®. Eur. Arch. Oto-Rhino-Laryngology 2022, 279, 4279–4288. [Google Scholar] [CrossRef]
- Almuhawas, F.; Alzhrani, F.; Saleh, S.; Alsanosi, A.; Yousef, M. Auditory Performance and Subjective Satisfaction with the ADHEAR System. Audiol. Neurotol. 2020, 26, 1–10. [Google Scholar] [CrossRef]
- Larsson, A.; Tjellström, A.; Stalfors, J. Implant Losses for the Bone-Anchored Hearing Devices Are More Frequent in Some Patients. Otol. Neurotol. 2015, 36, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Skarzynski, P.H.; Ratuszniak, A.; Osinska, K.; Koziel, M.; Krol, B.; Cywka, K.B.; Skarzynski, H. A Comparative Study of a Novel Adhesive Bone Conduction Device and Conventional Treatment Options for Conductive Hearing Loss. Otol. Neurotol. 2019, 40, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Verheij, E.; Bezdjian, A.; Grolman, W.; Thomeer, H.G.X.M. A Systematic Review on Complications of Tissue Preservation Surgical Techniques in Percutaneous Bone Conduction Hearing Devices. Otol. Neurotol. 2016, 37, 829–837. [Google Scholar] [CrossRef]
- Gawliczek, T.; Wimmer, W.; Munzinger, F.; Caversaccio, M.; Kompis, M. Speech Understanding and Sound Localization with a New Nonimplantable Wearing Option for Baha. BioMed Res. Int. 2018, 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- BOCCA, E. L'audiometria vocale [Vocal audiometry]. Otorinolaringol Ital. 1951;19(6):461-500. Undetermined Language. [PubMed]
- UNI EN ISO 8253-3 Acoustics — Audiometric test methods — Part 1: Pure-tone air and bone conduction audiometry. https://www.iso.org/standard/43601.html.
- UNI EN ISO 389-7 Acoustics — Reference zero for the calibration of audiometric equipment — Part 7: Reference threshold of hearing under free-field and diffuse-field listening conditions. 2019 (https://www.iso.org/standard/77365.html ).
- Newman, C.W.; Weinstein, B.E.; Jacobson, G.P.; Hug, G.A. The Hearing Handicap Inventory for Adults. Ear Hear. 1990, 11, 430–433. [Google Scholar] [CrossRef] [PubMed]
- Monzani, D.; Genovese, E.; Palma, S.; Rovatti, V.; Borgonzoni, M.; Martini, A. Measuring the psychosocial consequences of hearing loss in a working adult population: focus on validity and reliability of the Italian translation of the hearing handicap inventory. . 2007, 27, 186–91. [Google Scholar]
- Ventry, I.M.; Weinstein, B.E. The Hearing Handicap Inventory for the Elderly. Ear Hear. 1982, 3, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Di Berardino, F.; Nocini, R.; Ariotti, B.; Apa, E.; Gherpelli, C.; Monzani, D. Validity of the Italian adaptation of the Hearing Handicap Inventory for the Elderly (HHIE-It). Acta Otorhinolaryngol. Ital. 2023, 43, 74–81. [Google Scholar] [CrossRef]
- Cox, R.M.; Stephens, D.; Kramer, S.E. Translations of the International Outcome Inventory for Hearing Aids (IOI-HA): Traducciones del Inventario Internacional de Resultados para Auxiliares Auditivos (IOI-HA). Int. J. Audiol. 2002, 41, 3–26. [Google Scholar] [CrossRef]
- Gatehouse, S.; Noble, W. The Speech, Spatial and Qualities of Hearing Scale (SSQ). Int. J. Audiol. 2004, 43, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Fisk, J.D.; Ritvo, P.G.; Ross, L.; Haase, D.A.; Marrie, T.J.; Schlech, W.F. Measuring the functional impact of fatigue: Initial validation of the Fatigue Impact Scale. Clin. Infect. Dis. 1994, 18 (Suppl 1), S79–S83. [Google Scholar] [CrossRef]
- Cardano G: De subtilitate libri XXI. Parisiis, Fezandat & Roberri, 1550, liber XIII, p 234.
- Zarowski AJ, Verstraeten N, Somers T, Riff D, Offeciers EF. Headbands, testbands and softbands in preoperative testing and application of bone-anchored devices in adults and children. Adv Otorhinolaryngol. 2011;71:124-131. [CrossRef] [PubMed]
- Kompis, M.; Wimmer, W.; Caversaccio, M. Long term benefit of bone anchored hearing systems in single sided deafness. Acta Oto-Laryngologica 2016, 137, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Siau, R.T.K.; Dhillon, B.; Siau, D.; Green, K.M.J. Bone-anchored hearing aids in conductive and mixed hearing losses: why do patients reject them? . 2016, 273, 3117–3122. [Google Scholar] [CrossRef]
- Powell, R.; Wearden, A.; Pardesi, S.M.; Green, K. Understanding the low uptake of bone-anchored hearing aids: a review. J. Laryngol. Otol. 2017, 131, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Kurz, A.; Flynn, M.; Caversaccio, M.; Kompis, M. Speech Understanding with a New Implant Technology: A Comparative Study with a New Nonskin Penetrating Baha System. BioMed Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Mandavia, R.; Carter, A.; Haram, N.; Mossialos, E.; Schilder, A. An evaluation of the quality of evidence available to inform current bone conducting hearing device national policy. Clin. Otolaryngol. 2017, 42, 1000–1024. [Google Scholar] [CrossRef] [PubMed]
| ID | Sex | Age (years) |
Etiology of HL | Prior surgery | Years of eye-glass BCHA use |
PTA4 AC (dB HL) |
ABG (dB HL) |
| 1 | M | 75 | Chronic otitis media, radical surgery | yes | 11 | 43 | 28 |
| 2 | F | 80 | Unknown | no | 11 | 65 | 44 |
| 3 | F | 70 | Chronic otitis media | no | 16 | 38 | 25 |
| 4 | F | 74 | Otosclerosis | no | 3 | 64 | 48 |
| 5 | F | 79 | Chronic otitis media | no | 3 | 78 | 49 |
| 6 | F | 83 | Chronic otitis media | no | 10 | 63 | 34 |
| 7 | M | 68 | Chronic otitis media, myringoossiculoplasty | yes | 9 | 63 | 32 |
| 8 | M | 81 | Chronic otitis media | no | 8 | 88 | 45 |
| 9 | F | 78 | Otosclerosis, otitis media | no | 9 | 73 | 39 |
| 10 | M | 79 | Chronic otitis media, myringoplasty | yes | 14 | 69 | 39 |
| 11 | M | 88 | Chronic otitis media | no | 17 | 92 | 39 |
| 12 | F | 77 | Chronic otitis media, narrow external auditory canal | no | 6 | 71 | 35 |
| 13 | F | 76 | Chronic otitis media | no | 8 | 74 | 33 |
| 14 | M | 81 | Chronic otitis media | no | 12 | 62 | 32 |
| Overall mean ± SD | 8F; 6M | 77.8 ± 5.1 | - | - | 9.8 ± 4.2 | 67.4 ± 14.7 | 37.3 ± 7.3 |
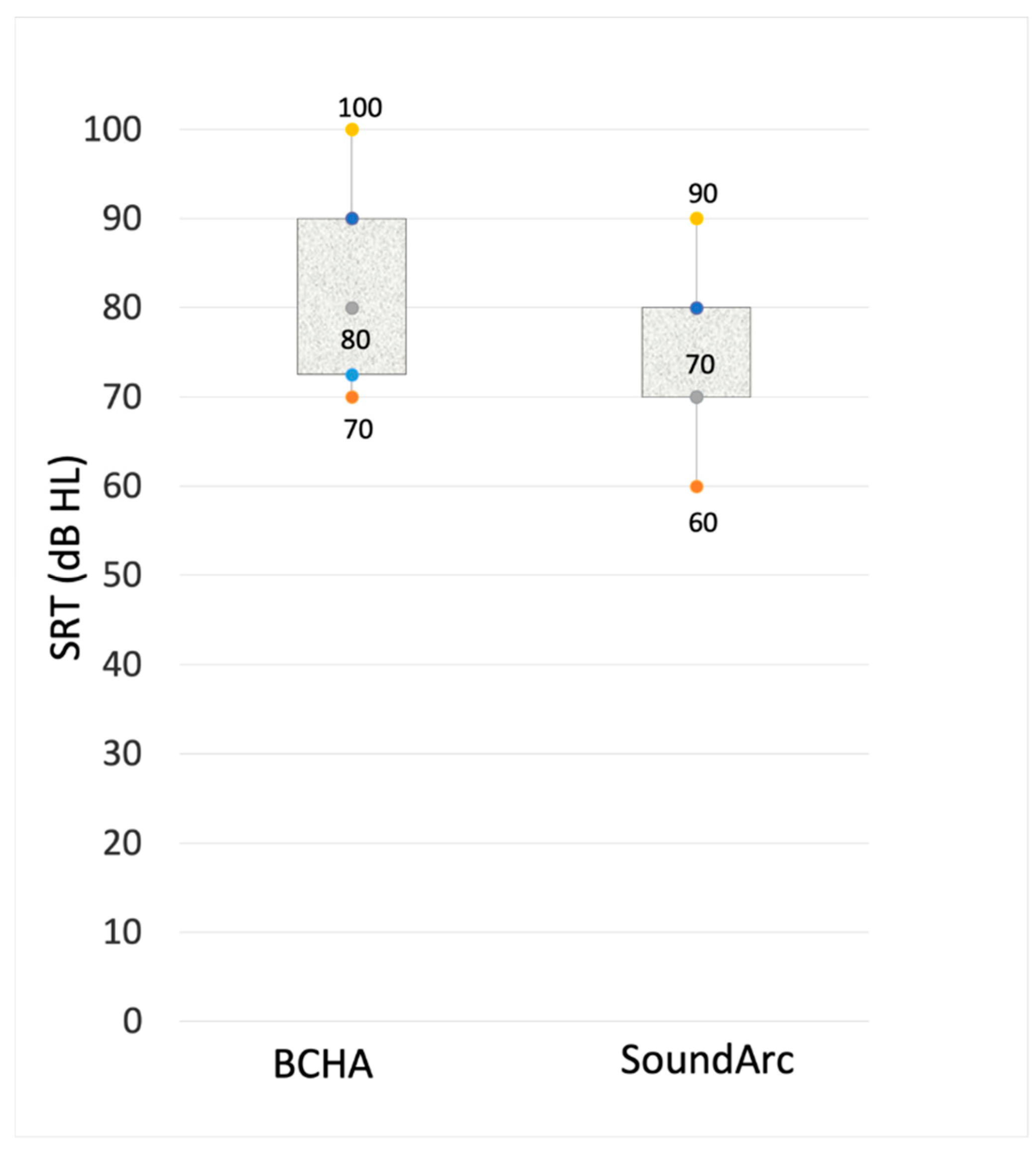
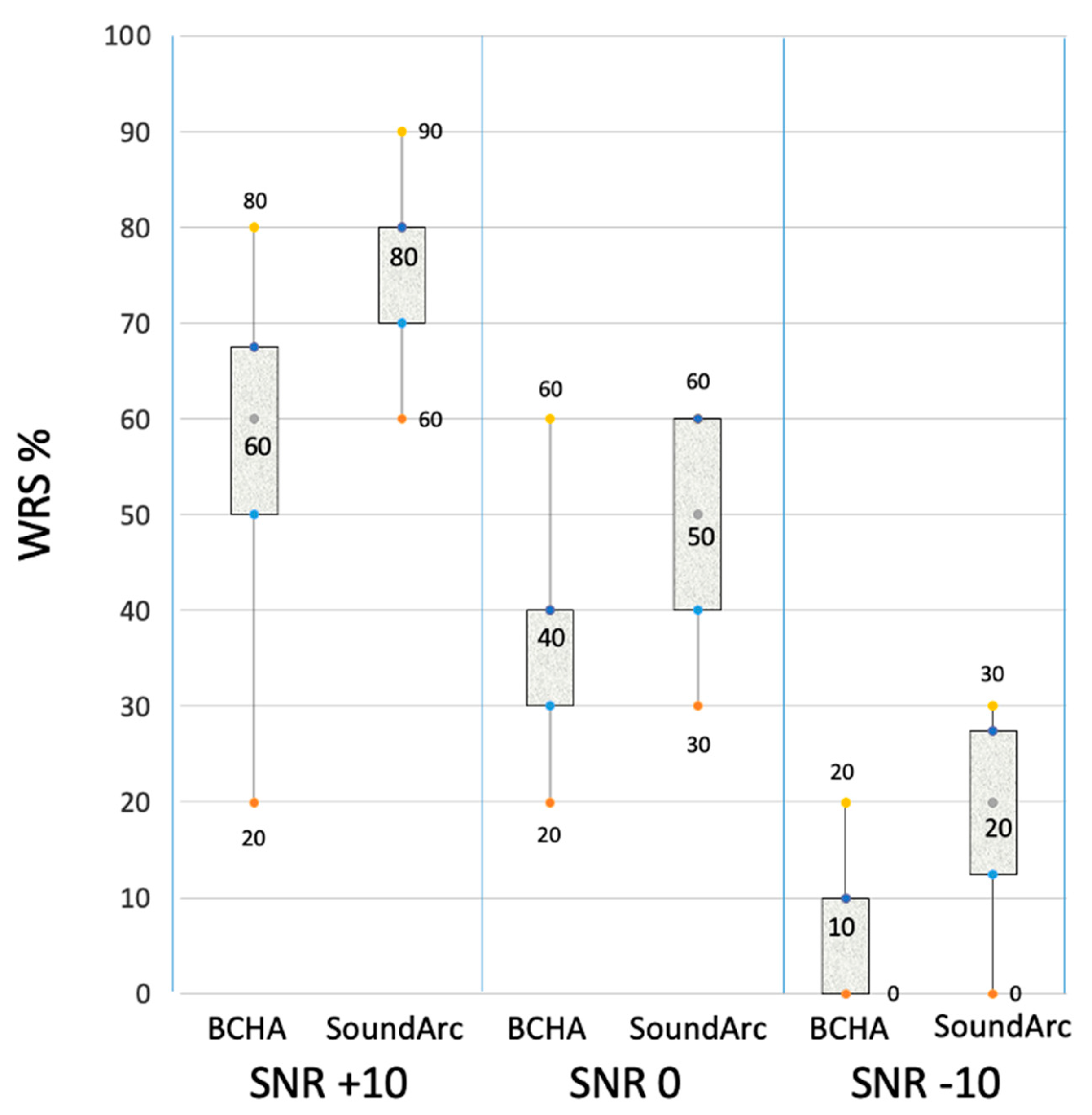
| Speech in quiet | Speech in noise (% of correct responses - WRS at 60 dB HL) | |||||||
|---|---|---|---|---|---|---|---|---|
| SRT dB HL | SNR +10 | SNR 0 | SNR-10 | |||||
| ID | BCHA | SoundArc | BCHA | SoundArc | BCHA | SoundArc | BCHA | SoundArc |
| 1 | 70 | 60 | 20 | 70 | 20 | 30 | 0 | 0 |
| 2 | 70 | 60 | 60 | 90 | 40 | 60 | 10 | 30 |
| 3 | 100 | 80 | 50 | 70 | 30 | 50 | 0 | 10 |
| 4 | 100 | 70 | 60 | 80 | 30 | 60 | 10 | 30 |
| 5 | 90 | 80 | 60 | 60 | 40 | 40 | 0 | 20 |
| 6 | 80 | 80 | 70 | 80 | 60 | 60 | 20 | 30 |
| 7 | 80 | 70 | 30 | 80 | 20 | 40 | 0 | 10 |
| 8 | 90 | 80 | 50 | 70 | 30 | 40 | 0 | 10 |
| 9 | 70 | 70 | 80 | 80 | 50 | 50 | 20 | 20 |
| 10 | 80 | 70 | 50 | 80 | 40 | 60 | 10 | 20 |
| 11 | 100 | 90 | 60 | 60 | 40 | 40 | 10 | 20 |
| 12 | 80 | 70 | 60 | 80 | 30 | 50 | 0 | 20 |
| 13 | 80 | 80 | 70 | 70 | 50 | 50 | 20 | 30 |
| 14 | 70 | 70 | 70 | 90 | 40 | 60 | 10 | 20 |
| Overall mean ± SD | 82.9 ± 11.4 | 73.6 ± 8.4 | 56.4 ± 16- | 75.7 ± 9.4 | 37.1 ± 11.4 | 49.3 ± 10.0 | 7.9 ± 8.0 | 19.3 ± 9.2 |
| Wilcokon paired test (p_value) | p= .005 | p= .005 | p= .006 | p= .002 | ||||
| SSQ | ||||||
|---|---|---|---|---|---|---|
| Speech | Spatial | Qualities | ||||
| ID | BCHA | SoundArc | BCHA | SoundArc | BCHA | SoundArc |
| 1 | 3 | 7.5 | 3 | 7 | 3 | 8 |
| 2 | 6 | 7 | 6 | 7.5 | 5.5 | 7 |
| 3 | 5 | 7.5 | 5 | 8 | 4.5 | 7.5 |
| 4 | 4.5 | 8 | 4.5 | 7.5 | 5 | 7.5 |
| 5 | 6 | 6 | 6 | 6.5 | 6 | 6 |
| 6 | 6 | 6.5 | 6.5 | 6.5 | 6 | 7 |
| 7 | 3 | 8 | 3 | 7.5 | 3.5 | 8 |
| 8 | 6 | 7.5 | 6 | 7 | 5.5 | 7 |
| 9 | 7 | 7 | 7 | 7 | 7.5 | 7.5 |
| 10 | 4.5 | 7 | 4.5 | 7 | 4 | 6.5 |
| 11 | 5 | 5 | 4 | 5 | 5 | 5 |
| 12 | 5.5 | 8 | 5 | 7.5 | 5 | 8 |
| 13 | 7 | 7 | 7 | 7 | 7 | 7.5 |
| 14 | 7 | 8.5 | 6.5 | 8 | 7 | 8.5 |
| Overall mean ± SD | 5.5 ± 1.4 | 7.0 ± 1.0 | 5.2 ± 1.5 | 7.0 ± 0.9 | 5.3 ± 1.3 | 7.0 ± 1.1 |
| Wilcokon paired test (p_value) | .005 | .003 | .003 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




