Submitted:
07 September 2024
Posted:
09 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Updated Mechanisms of Phage Action
2.1. Phage Adsorption and Receptor Recognition
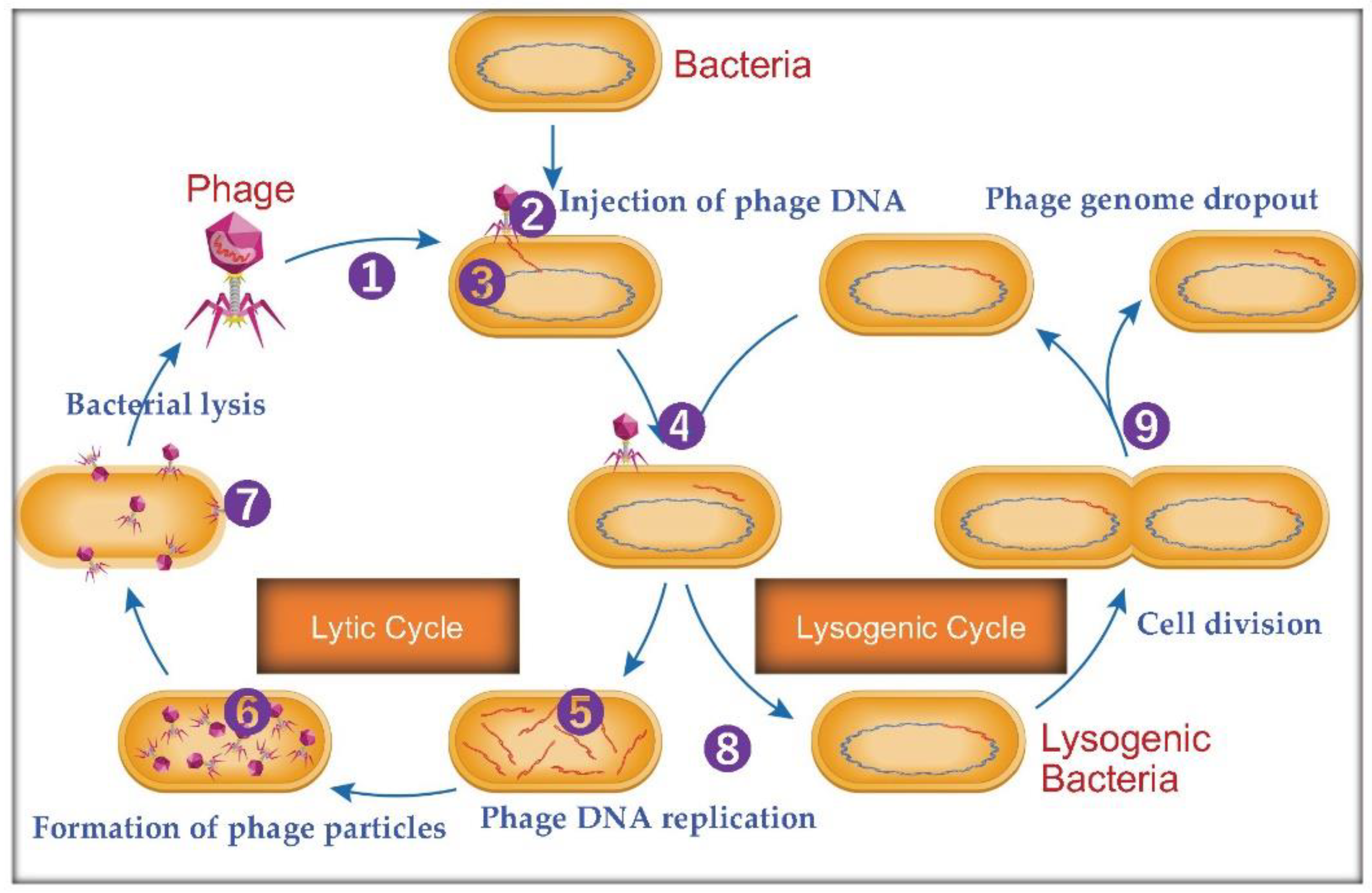
2.2. Genome Injection and Host Takeover
2.3. Replication Strategies
2.4. Phage-Encoded Toxins and Enzymes
2.5. Horizontal Gene Transfer and Phage Therapy
2.6. Phage-Host Co-Evolution
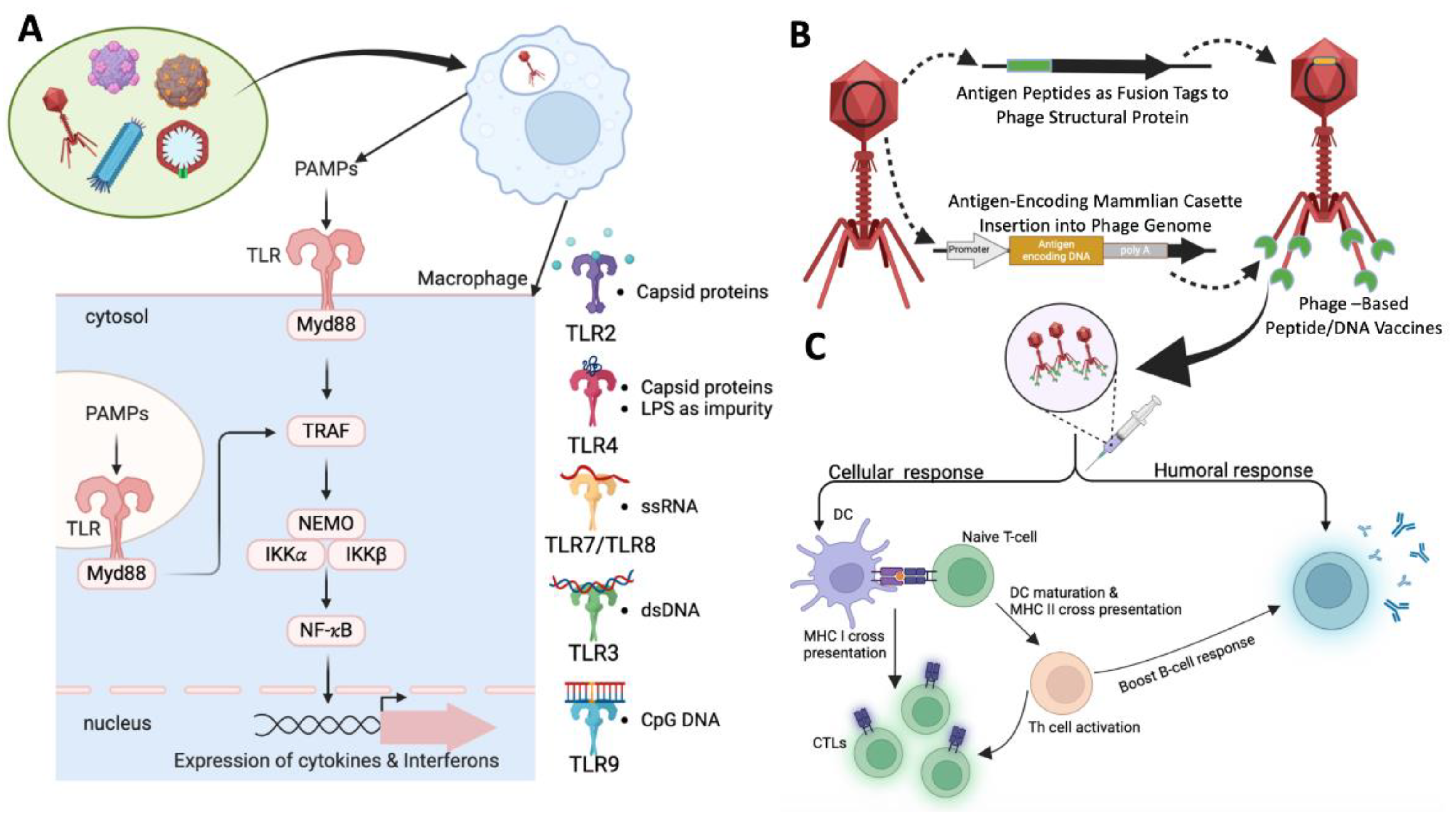
2.7. Immune System Interactions
2.8. Synthetic and Recombinant Phages
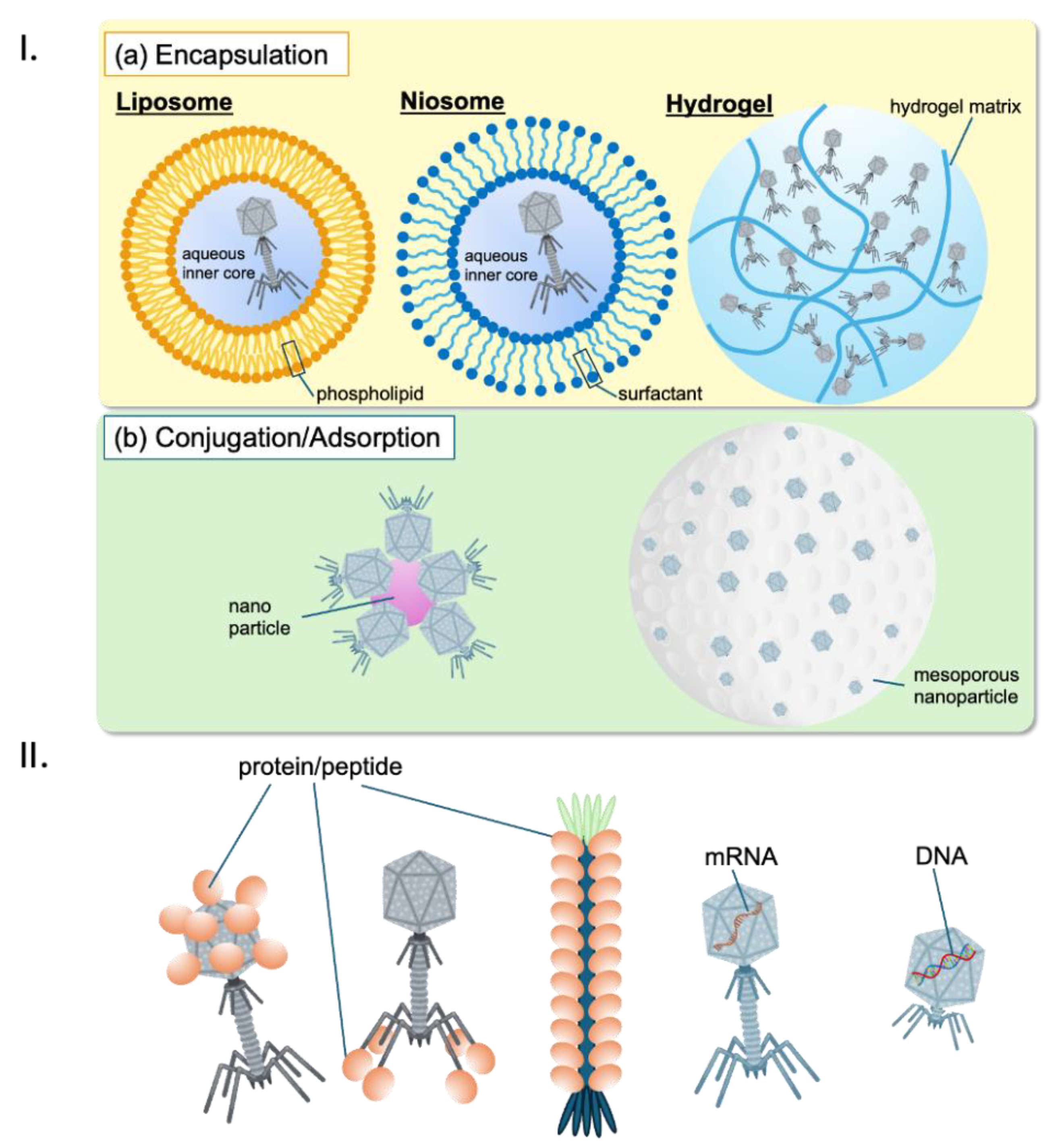
2.9. Phage Delivery Systems
2.10. Regulatory and Ethical Considerations
3. Phage Therapy for Drug-Resistant Bacterial Infections
3.1. The Growing Threat of Antibiotic Resistance
3.2. Mechanisms and Advantages of Phage Therapy
3.3. Targeting Specific Drug-Resistant Bacteria
3.4. Enhancing Therapeutic Potential by Engineered Phages
3.5. Phage Therapy in Combination with Antibiotics
3.6. Regulatory and Safety Considerations
4. Phage-Based Treatments for Biofilm-Generating Bacteria
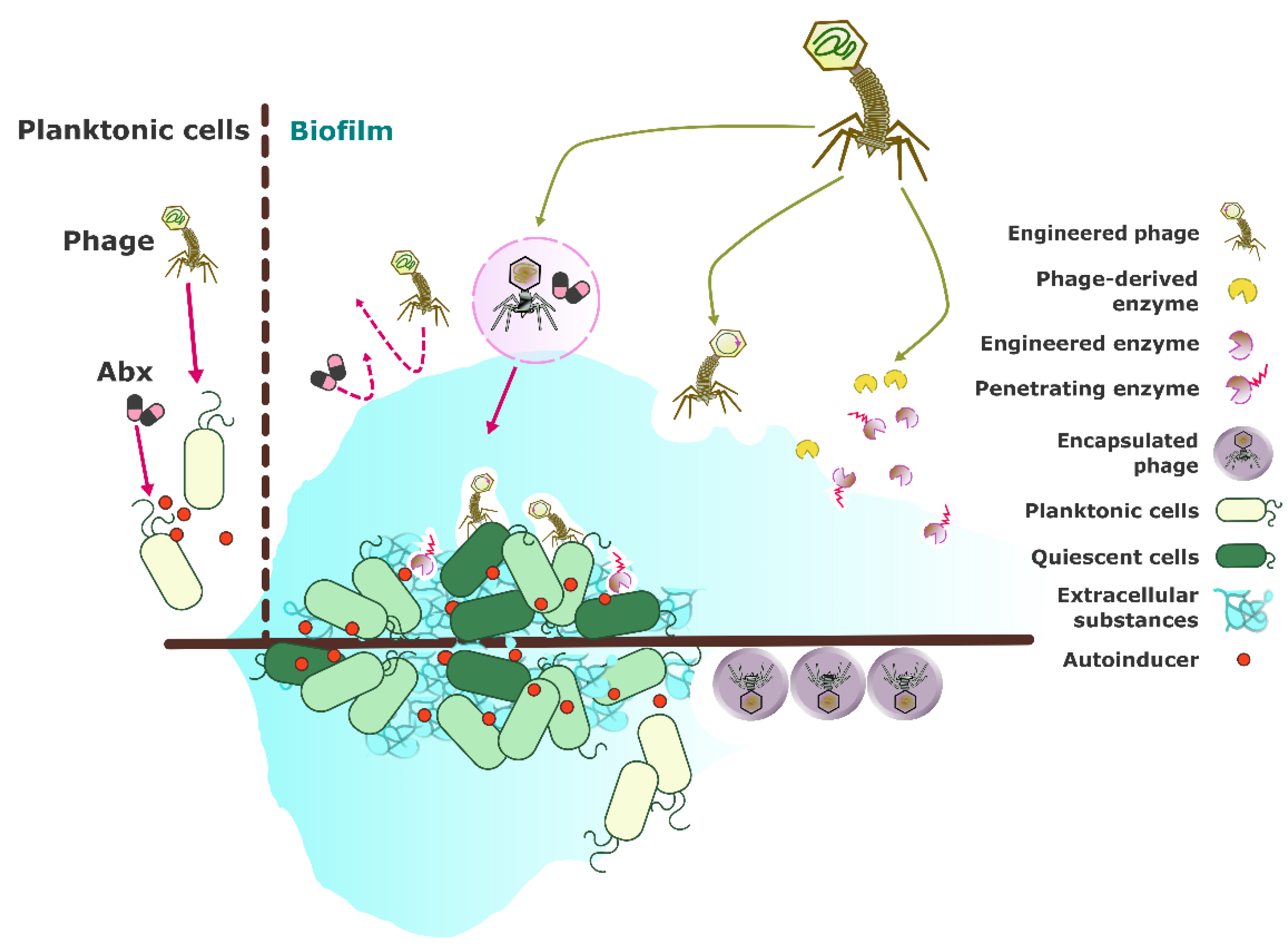
4.1. Mechanisms of Phage Action Against Biofilms
4.2. Advantages of Phage-Based Biofilm Treatments
4.3. Case Studies and Applications
4.4. Challenges and Future Directions
5. Phage Therapy for Intracellular Bacteria
5.1. Mechanisms of Phage Action Against Intracellular Bacteria
5.2. Advantages of Phage Therapy for Intracellular Bacterial Infections
5.3. Case Studies and Applications
5.4. Challenges and Future Directions
6. Phage-Based Vaccines
6.1. Principles of Phage-Based Vaccine Design
6.2. Mechanisms of Immune Stimulation
6.3. Applications of Phage-Based Vaccines
6.4. Challenges and Future Directions
7. Phage Therapy as Anti-Cancer Agents
7.1. Mechanisms of Phage-Mediated Anti-Cancer Activity
7.2. Development of Phage-Based Cancer Therapies
7.3. Applications of Phage-Based Cancer Therapies
7.4. Challenges and Future Directions
8. Phages as Drug Delivery Systems (DDS)
8.1. Principles of Phage-Based Drug Delivery
8.2. Engineering of Phage Vectors for Drug Delivery
8.3. Applications of Phage-Based Drug Delivery Systems
8.4. Challenges and Future Directions
9. Phage Display Technology in Drug Discovery
9.1. Applications in Drug Discovery
9.2. Recent Advances in Phage Display for Drug Discovery
9.3. Challenges and Future Directions
10. Safety and Regulatory Considerations
10.1. Safety Considerations
10.2. Regulatory Frameworks
11. Challenges and Future Directions
11.1. Standardization and Regulatory Considerations
11.2. Therapeutic and Technological Challenges
11.3. Future Directions
12. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jain, P.; Hartman, T.E.; Eisenberg, N.; O'Donnell, M.R.; Kriakov, J.; Govender, K.; Makume, M.; Thaler, D.S.; Hatfull, G.F.; Sturm, A.W.; et al. φ(2)GFP10, a high-intensity fluorophage, enables detection and rapid drug susceptibility testing of Mycobacterium tuberculosis directly from sputum samples. Journal of clinical microbiology 2012, 50, 1362–1369. [Google Scholar] [CrossRef] [PubMed]
- Colom, J.; Cano-Sarabia, M.; Otero, J.; Cortés, P.; Maspoch, D.; Llagostera, M. Liposome-Encapsulated Bacteriophages for Enhanced Oral Phage Therapy against Salmonella spp. Appl Environ Microbiol 2015, 81, 4841–4849. [Google Scholar] [CrossRef] [PubMed]
- Sartorius, R.; Trovato, M.; Manco, R.; D'Apice, L.; De Berardinis, P. Exploiting viral sensing mediated by Toll-like receptors to design innovative vaccines. NPJ Vaccines 2021, 6, 127–127. [Google Scholar] [CrossRef] [PubMed]
- Green, S.I.; Clark, J.R.; Santos, H.H.; Weesner, K.E.; Salazar, K.C.; Aslam, S.; Campbell, J.W.; Doernberg, S.B.; Blodget, E.; Morris, M.I.; et al. A Retrospective, Observational Study of 12 Cases of Expanded-Access Customized Phage Therapy: Production, Characteristics, and Clinical Outcomes. Clin Infect Dis 2023, 77, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Tripathi, P.; Gupta, R.; Pandey, S. Niosomes: A review on niosomal research in the last decade. Journal of Drug Delivery Science and Technology 2020, 56, 101581. [Google Scholar] [CrossRef]
- Chen, J.; Alcaine, S.D.; Jackson, A.A.; Rotello, V.M.; Nugen, S.R. Development of Engineered Bacteriophages for Escherichia coli Detection and High-Throughput Antibiotic Resistance Determination. ACS Sensors 2017, 2, 484–489. [Google Scholar] [CrossRef]
- Xu, H.; Li, L.; Li, R.; Guo, Z.; Lin, M.; Lu, Y.; Hou, J.; Govinden, R.; Deng, B.; Chenia, H.Y. Evaluation of dendritic cell-targeting T7 phages as a vehicle to deliver avian influenza virus H5 DNA vaccine in SPF chickens. Front Immunol 2022, 13, 1063129–1063129. [Google Scholar] [CrossRef]
- Jaroszewicz, W.; Morcinek-Orłowska, J.; Pierzynowska, K.; Gaffke, L.; Węgrzyn, G. Phage display and other peptide display technologies. FEMS Microbiology Reviews 2021, 46. [Google Scholar] [CrossRef]
- Wei, J.; Peng, N.; Liang, Y.; Li, K.; Li, Y. Phage Therapy: Consider the Past, Embrace the Future. Applied Sciences 2020, 10, 7654. [Google Scholar] [CrossRef]
- Matsui, H.; Uchiyama, J.; Ogata, M.; Nasukawa, T.; Takemura-Uchiyama, I.; Kato, S.-I.; Murakami, H.; Higashide, M.; Hanaki, H. Use of Recombinant Endolysin to Improve Accuracy of Group B Streptococcus Tests. Microbiol Spectr 2021, 9, e0007721–e0007721. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Bhardwaj, S.K.; Mehta, J.; Mohanta, G.C.; Deep, A. Bacteriophage immobilized graphene electrodes for impedimetric sensing of bacteria (Staphylococcus arlettae). Analytical Biochemistry 2016, 505, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Rotman, S.G.; Sumrall, E.; Ziadlou, R.; Grijpma, D.W.; Richards, R.G.; Eglin, D.; Moriarty, T.F. Local Bacteriophage Delivery for Treatment and Prevention of Bacterial Infections. Frontiers in microbiology 2020, 11, 538060–538060. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Gao, Y.; Dong, J.; Xue, Y.; Fan, M.; Shen, B.; Liu, C.; Shao, N. A novel peptide isolated from phage library to substitute a complex system for a vaccine against staphylococci infection. Vaccine 2006, 24, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Shabani, A.; Marquette, C.A.; Mandeville, R.; Lawrence, M.F. Carbon microarrays for the direct impedimetric detection of Bacillus anthracis using Gamma phages as probes. The Analyst 2013, 138, 1434. [Google Scholar] [CrossRef] [PubMed]
- Meile, S.; Kilcher, S.; Loessner, M.J.; Dunne, M. Reporter Phage-Based Detection of Bacterial Pathogens: Design Guidelines and Recent Developments. Viruses 2020, 12, 944. [Google Scholar] [CrossRef]
- Denyes, J.M.; Dunne, M.; Steiner, S.; Mittelviefhaus, M.; Weiss, A.; Schmidt, H.; Klumpp, J.; Loessner, M.J. Modified Bacteriophage S16 Long Tail Fiber Proteins for Rapid and Specific Immobilization and Detection of Salmonella Cells. Appl Environ Microbiol 2017, 83, e00277–00217. [Google Scholar] [CrossRef]
- Park, J.Y.; Moon, B.Y.; Park, J.W.; Thornton, J.A.; Park, Y.H.; Seo, K.S. Genetic engineering of a temperate phage-based delivery system for CRISPR/Cas9 antimicrobials against Staphylococcus aureus. Scientific reports 2017, 7, 44929–44929. [Google Scholar] [CrossRef]
- Kiga, K.; Tan, X.E.; Ibarra-Chavez, R.; Watanabe, S.; Aiba, Y.; Sato'o, Y.; Li, F.Y.; Sasahara, T.; Cui, B.; Kawauchi, M.; et al. Development of CRISPR-Cas13a-based antimicrobials capable of sequence-specific killing of target bacteria. Nat Commun 2020, 11, 2934. [Google Scholar] [CrossRef]
- Twort, F.W. An Investigation on the Nature of Ultra-Microscopic Viruses. The Lancet 1915, 186, 1241–1243. [Google Scholar] [CrossRef]
- Corner, E.s. On an invisible microbe antagonistic to dysentery bacilli. Note by M. F. d’Herelle, presented by M. Roux. Comptes Rendus Academie des Sciences 1917; 165:373–5. Bacteriophage 2014, 1, 3–5. [Google Scholar] [CrossRef]
- de Vries, C.R.; Chen, Q.; Demirdjian, S.; Kaber, G.; Khosravi, A.; Liu, D.; Van Belleghem, J.D.; Bollyky, P.L. Phages in vaccine design and immunity; mechanisms and mysteries. Current opinion in biotechnology 2021, 68, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Ilhan, H.; Tayyarcan, E.K.; Caglayan, M.G.; Boyaci, İ.H.; Saglam, N.; Tamer, U. Replacement of antibodies with bacteriophages in lateral flow assay of Salmonella Enteritidis. Biosensors and Bioelectronics 2021, 189, 113383. [Google Scholar] [CrossRef] [PubMed]
- Thiel, K. Old dogma, new tricks - 21st century phage therapy. Nature Biotechnology 2004, 22, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Frenzel, A.; Schirrmann, T.; Hust, M. Phage display-derived human antibodies in clinical development and therapy. MAbs 2016, 8, 1177–1194. [Google Scholar] [CrossRef]
- Stevens, R.H.; Zhang, H.; Kajsik, M.; Płoski, R.; Rydzanicz, M.; Sabaka, P.; Šutovský, S. Successful use of a phage endolysin for treatment of chronic pelvic pain syndrome/chronic bacterial prostatitis. Front Med (Lausanne) 2023, 10, 1238147–1238147. [Google Scholar] [CrossRef]
- Keller, C.M.; Kendra, C.G.; Bruna, R.E.; Craft, D.; Pontes, M.H. Genetic Modification of Sodalis Species by DNA Transduction. mSphere 2021, 6, e01331–01320. [Google Scholar] [CrossRef]
- Stachurska, X.; Cendrowski, K.; Pachnowska, K.; Piegat, A.; Mijowska, E.; Nawrotek, P. Nanoparticles Influence Lytic Phage T4-like Performance In Vitro. International journal of molecular sciences 2022, 23, 7179. [Google Scholar] [CrossRef]
- Benler, S.; Koonin, E.V. Phage lysis-lysogeny switches and programmed cell death: Danse macabre. BioEssays 2020, 42. [Google Scholar] [CrossRef]
- Abedon, S.T.; Kuhl, S.J.; Blasdel, B.G.; Kutter, E.M. Phage treatment of human infections. Bacteriophage 2011, 1, 66–85. [Google Scholar] [CrossRef]
- Srinivasan, S.; Alexander, J.F.; Driessen, W.H.; Leonard, F.; Ye, H.; Liu, X.; Arap, W.; Pasqualini, R.; Ferrari, M.; Godin, B. Bacteriophage Associated Silicon Particles: Design and Characterization of a Novel Theranostic Vector with Improved Payload Carrying Potential. J Mater Chem B 2013, 1, 10–1039. [Google Scholar] [CrossRef]
- Donlan, R.M. Preventing biofilms of clinically relevant organisms using bacteriophage. Trends Microbiol 2009, 17, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Dedrick, R.M.; Guerrero-Bustamante, C.A.; Garlena, R.A.; Russell, D.A.; Ford, K.; Harris, K.; Gilmour, K.C.; Soothill, J.; Jacobs-Sera, D.; Schooley, R.T.; et al. Engineered bacteriophages for treatment of a patient with a disseminated drug-resistant Mycobacterium abscessus. Nat Med 2019, 25, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Azam, A.H.; Tan, X.E.; Veeranarayanan, S.; Kiga, K.; Cui, L. Bacteriophage Technology and Modern Medicine. Antibiotics (Basel) 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Veeranarayanan, S.; Thitiananpakorn, K.; Wannigama, D.L. Bacteriophage Bioengineering: A Transformative Approach for Targeted Drug Discovery and Beyond. Pathogens 2023, 12. [Google Scholar] [CrossRef]
- González-Mora, A.; Hernández-Pérez, J.; Iqbal, H.M.N.; Rito-Palomares, M.; Benavides, J. Bacteriophage-Based Vaccines: A Potent Approach for Antigen Delivery. Vaccines (Basel) 2020, 8, 504. [Google Scholar] [CrossRef]
- Zalewska-Piątek, B.; Piątek, R. Bacteriophages as Potential Tools for Use in Antimicrobial Therapy and Vaccine Development. Pharmaceuticals (Basel) 2021, 14, 331. [Google Scholar] [CrossRef]
- Veeranarayanan, S.; Azam, A.H.; Kiga, K.; Watanabe, S.; Cui, L. Bacteriophages as Solid Tumor Theragnostic Agents. International journal of molecular sciences 2021, 23, 402. [Google Scholar] [CrossRef]
- Shen, Y.; Wang, J.; Li, Y.; Yang, C.-T.; Zhou, X. Modified Bacteriophage for Tumor Detection and Targeted Therapy. Nanomaterials (Basel) 2023, 13, 665. [Google Scholar] [CrossRef]
- Nagano, K.; Tsutsumi, Y. Development of novel drug delivery systems using phage display technology for clinical application of protein drugs. Proc Jpn Acad Ser B Phys Biol Sci 2016, 92, 156–166. [Google Scholar] [CrossRef]
- Wang, H.; Yang, Y.; Xu, Y.; Chen, Y.; Zhang, W.; Liu, T.; Chen, G.; Wang, K. Phage-based delivery systems: engineering, applications, and challenges in nanomedicines. J Nanobiotechnology 2024, 22, 365–365. [Google Scholar] [CrossRef]
- Citorik, R.J.; Mimee, M.; Lu, T.K. Sequence-specific antimicrobials using efficiently delivered RNA-guided nucleases. Nature biotechnology 2014, 32, 1141–1145. [Google Scholar] [CrossRef] [PubMed]
- Bikard, D.; Euler, C.W.; Jiang, W.; Nussenzweig, P.M.; Goldberg, G.W.; Duportet, X.; Fischetti, V.A.; Marraffini, L.A. Exploiting CRISPR-Cas nucleases to produce sequence-specific antimicrobials. Nature biotechnology 2014, 32, 1146–1150. [Google Scholar] [CrossRef] [PubMed]
- Ram, G.; Ross, H.F.; Novick, R.P.; Rodriguez-Pagan, I.; Jiang, D. Conversion of staphylococcal pathogenicity islands to CRISPR-carrying antibacterial agents that cure infections in mice. Nature biotechnology 2018, 36, 971–976. [Google Scholar] [CrossRef]
- Selle, K.; Fletcher, J.R.; Tuson, H.; Schmitt, D.S.; McMillan, L.; Vridhambal, G.S.; Rivera, A.J.; Montgomery, S.A.; Fortier, L.-C.; Barrangou, R.; et al. In Vivo Targeting of Clostridioides difficile Using Phage-Delivered CRISPR-Cas3 Antimicrobials. Mbio 2020, 11, e00019–00020. [Google Scholar] [CrossRef] [PubMed]
- Westwater, C.; Kasman, L.M.; Schofield, D.A.; Werner, P.A.; Dolan, J.W.; Schmidt, M.G.; Norris, J.S. Use of genetically engineered phage to deliver antimicrobial agents to bacteria: an alternative therapy for treatment of bacterial infections. Antimicrob Agents Chemother 2003, 47, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Krom, R.J.; Bhargava, P.; Lobritz, M.A.; Collins, J.J. Engineered Phagemids for Nonlytic, Targeted Antibacterial Therapies. Nano Letters 2015, 15, 4808–4813. [Google Scholar] [CrossRef]
- Ojima, S.; Azam, A.H.; Kondo, K.; Nie, W.; Wang, S.; Chihara, K.; Tamura, A.; Yamashita, W.; Nakamura, T.; Sugawara, Y.; et al. Systematic Discovery of Phage Genes that Inactivate Bacterial Immune Systems. 2024. [Google Scholar] [CrossRef]
- Yamashita, W.; Ojima, S.; Tamura, A.; Azam, A.H.; Kondo, K.; Yuancheng, Z.; Cui, L.; Shintani, M.; Suzuki, M.; Takahashi, Y.; et al. Harnessing a T1 Phage-Derived Spanin for Developing Phage-Based Antimicrobial Development. Biodes Res 2024, 6, 0028. [Google Scholar] [CrossRef]
- Yacoby, I.; Bar, H.; Benhar, I. Targeted drug-carrying bacteriophages as antibacterial nanomedicines. Antimicrob Agents Chemother 2007, 51, 2156–2163. [Google Scholar] [CrossRef]
- Cao, J. Helicobacter pylori-antigen-binding fragments expressed on the filamentous M13 phage prevent bacterial growth. Biochimica et Biophysica Acta (BBA) - General Subjects 2000, 1474, 107–113. [Google Scholar] [CrossRef]
- Dong, X.; Pan, P.; Zheng, D.-W.; Bao, P.; Zeng, X.; Zhang, X.-Z. Bioinorganic hybrid bacteriophage for modulation of intestinal microbiota to remodel tumor-immune microenvironment against colorectal cancer. Sci Adv 2020, 6, eaba1590–eaba1590. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Borg, R.E.; Dow, L.P.; Pruitt, B.L.; Chen, I.A. Controlled phage therapy by photothermal ablation of specific bacterial species using gold nanorods targeted by chimeric phages. Proceedings of the National Academy of Sciences of the United States of America 2020, 117, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
- Kilcher, S.; Studer, P.; Muessner, C.; Klumpp, J.; Loessner, M.J. Cross-genus rebooting of custom-made, synthetic bacteriophage genomes in L-form bacteria. Proceedings of the National Academy of Sciences of the United States of America 2018, 115, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.K.; Collins, J.J. Dispersing biofilms with engineered enzymatic bacteriophage. Proceedings of the National Academy of Sciences of the United States of America 2007, 104, 11197–11202. [Google Scholar] [CrossRef] [PubMed]
- Pei, R.; Lamas-Samanamud, G.R. Inhibition of biofilm formation by T7 bacteriophages producing quorum-quenching enzymes. Appl Environ Microbiol 2014, 80, 5340–5348. [Google Scholar] [CrossRef]
- Pires, D.P.; Cleto, S.; Sillankorva, S.; Azeredo, J.; Lu, T.K. Genetically Engineered Phages: a Review of Advances over the Last Decade. Microbiol Mol Biol Rev 2016, 80, 523–543. [Google Scholar] [CrossRef]
- Ando, H.; Lemire, S.; Pires, D.P.; Lu, T.K. Engineering Modular Viral Scaffolds for Targeted Bacterial Population Editing. Cell Syst 2015, 1, 187–196. [Google Scholar] [CrossRef]
- Yoichi, M.; Abe, M.; Miyanaga, K.; Unno, H.; Tanji, Y. Alteration of tail fiber protein gp38 enables T2 phage to infect Escherichia coli O157:H7. Journal of Biotechnology 2005, 115, 101–107. [Google Scholar] [CrossRef]
- Mahichi, F.; Synnott, A.J.; Yamamichi, K.; Osada, T.; Tanji, Y. Site-specific recombination of T2 phage using IP008 long tail fiber genes provides a targeted method for expanding host range while retaining lytic activity. FEMS Microbiology Letters 2009, 295, 211–217. [Google Scholar] [CrossRef]
- Pouillot, F.; Blois, H.; Iris, F. Genetically engineered virulent phage banks in the detection and control of emergent pathogenic bacteria. Biosecur Bioterror 2010, 8, 155–169. [Google Scholar] [CrossRef]
- Yehl, K.; Lemire, S.; Yang, A.C.; Ando, H.; Mimee, M.; Der Torossian Torres, M.; de la Fuente-Nunez, C.; Lu, T.K. Engineering Phage Host-Range and Suppressing Bacterial Resistance Through Phage Tail Fiber Mutagenesis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kašpárek, P.; Pantůček, R.; Kahánková, J.; Růžičková, V.; Doškař, J. Genome rearrangements in host-range mutants of the polyvalent staphylococcal bacteriophage 812. Folia Microbiologica 2007, 52, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Azam, A.H.; Chihara, K.; Kondo, K.; Nakamura, T.; Ojima, S.; Tamura, A.; Yamashita, W.; Cui, L.; Takahashi, Y.; Watashi, K.; et al. Viruses encode tRNA and anti-retron to evade bacterial immunity. 2023. [Google Scholar] [CrossRef]
- Merril, C.R.; Biswas, B.; Carlton, R.; Jensen, N.C.; Creed, G.J.; Zullo, S.; Adhya, S. Long-circulating bacteriophage as antibacterial agents. Proceedings of the National Academy of Sciences of the United States of America 1996, 93, 3188–3192. [Google Scholar] [CrossRef]
- Vitiello, C.L.; Merril, C.R.; Adhya, S. An amino acid substitution in a capsid protein enhances phage survival in mouse circulatory system more than a 1000-fold. Virus Research 2005, 114, 101–103. [Google Scholar] [CrossRef]
- Kim, K.-P.; Cha, J.-D.; Jang, E.-H.; Klumpp, J.; Hagens, S.; Hardt, W.-D.; Lee, K.-Y.; Loessner, M.J. PEGylation of bacteriophages increases blood circulation time and reduces T-helper type 1 immune response. Microb Biotechnol 2008, 1, 247–257. [Google Scholar] [CrossRef]
- Nobrega, F.L.; Costa, A.R.; Santos, J.F.; Siliakus, M.F.; van Lent, J.W.M.; Kengen, S.W.M.; Azeredo, J.; Kluskens, L.D. Genetically manipulated phages with improved pH resistance for oral administration in veterinary medicine. Scientific reports 2016, 6, 39235–39235. [Google Scholar] [CrossRef]
- Favor, A.H.; Llanos, C.D.; Youngblut, M.D.; Bardales, J.A. Optimizing bacteriophage engineering through an accelerated evolution platform. Scientific reports 2020, 10, 13981–13981. [Google Scholar] [CrossRef]
- Lu, T.K.; Collins, J.J. Engineered bacteriophage targeting gene networks as adjuvants for antibiotic therapy. Proceedings of the National Academy of Sciences of the United States of America 2009, 106, 4629–4634. [Google Scholar] [CrossRef]
- Edgar, R.; Friedman, N.; Molshanski-Mor, S.; Qimron, U. Reversing bacterial resistance to antibiotics by phage-mediated delivery of dominant sensitive genes. Appl Environ Microbiol 2012, 78, 744–751. [Google Scholar] [CrossRef]
- Yosef, I.; Manor, M.; Kiro, R.; Qimron, U. Temperate and lytic bacteriophages programmed to sensitize and kill antibiotic-resistant bacteria. Proceedings of the National Academy of Sciences of the United States of America 2015, 112, 7267–7272. [Google Scholar] [CrossRef] [PubMed]
- Libis, V.K.; Bernheim, A.G.; Basier, C.; Jaramillo-Riveri, S.; Deyell, M.; Aghoghogbe, I.; Atanaskovic, I.; Bencherif, A.C.; Benony, M.; Koutsoubelis, N.; et al. Silencing of Antibiotic Resistance in <i>E. coli</i> with Engineered Phage Bearing Small Regulatory RNAs. ACS Synthetic Biology 2014, 3, 1003–1006. [Google Scholar] [CrossRef] [PubMed]
- Hagens, S.; Blasi, U. Genetically modified filamentous phage as bactericidal agents: a pilot study. Letters in Applied Microbiology 2003, 37, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Paul, V.D.; Sundarrajan, S.; Rajagopalan, S.S.; Hariharan, S.; Kempashanaiah, N.; Padmanabhan, S.; Sriram, B.; Ramachandran, J. Lysis-deficient phages as novel therapeutic agents for controlling bacterial infection. BMC Microbiol 2011, 11, 195–195. [Google Scholar] [CrossRef] [PubMed]
- Tao, P.; Zhu, J.; Mahalingam, M.; Batra, H.; Rao, V.B. Bacteriophage T4 nanoparticles for vaccine delivery against infectious diseases. Advanced drug delivery reviews 2019, 145, 57–72. [Google Scholar] [CrossRef]
- Menéndez, T.; Santiago-Vispo, N.F.; Cruz-Leal, Y.; Coizeau, E.; Garay, H.; Reyes, O.; Batista, Y.; Cobas, K.; Carmenate, T.; Chinea, G. Identification and characterization of phage-displayed peptide mimetics of Neisseria meningitidis serogroup B capsular polysaccharide. International Journal of Medical Microbiology 2011, 301, 16–25. [Google Scholar] [CrossRef]
- Tao, P.; Mahalingam, M.; Kirtley, M.L.; van Lier, C.J.; Sha, J.; Yeager, L.A.; Chopra, A.K.; Rao, V.B. Mutated and bacteriophage T4 nanoparticle arrayed F1-V immunogens from Yersinia pestis as next generation plague vaccines. PLoS Pathog 2013, 9, e1003495–e1003495. [Google Scholar] [CrossRef]
- Sathaliyawala, T.; Rao, M.; Maclean, D.M.; Birx, D.L.; Alving, C.R.; Rao, V.B. Assembly of human immunodeficiency virus (HIV) antigens on bacteriophage T4: a novel in vitro approach to construct multicomponent HIV vaccines. J Virol 2006, 80, 7688–7698. [Google Scholar] [CrossRef]
- Hashemi, H.; Pouyanfard, S.; Bandehpour, M.; Noroozbabaei, Z.; Kazemi, B.; Saelens, X.; Mokhtari-Azad, T. Immunization with M2e-displaying T7 bacteriophage nanoparticles protects against influenza A virus challenge. PLoS One 2012, 7, e45765–e45765. [Google Scholar] [CrossRef]
- Shi, H.; Dong, S.; Zhang, X.; Chen, X.; Gao, X.; Wang, L. Phage vaccines displaying YGKDVKDLFDYAQE epitope induce protection against systemic candidiasis in mouse model. Vaccine 2018, 36, 5717–5724. [Google Scholar] [CrossRef]
- Ou, C.; Tian, D.; Ling, Y.; Pan, Q.; He, Q.; Eko, F.O.; He, C. Evaluation of an ompA-based phage-mediated DNA vaccine against Chlamydia abortus in piglets. International Immunopharmacology 2013, 16, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Suwan, K.; Yata, T.; Waramit, S.; Przystal, J.M.; Stoneham, C.A.; Bentayebi, K.; Asavarut, P.; Chongchai, A.; Pothachareon, P.; Lee, K.-Y.; et al. Next-generation of targeted AAVP vectors for systemic transgene delivery against cancer. Proceedings of the National Academy of Sciences of the United States of America 2019, 116, 18571–18577. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Tao, P.; Mahalingam, M.; Sha, J.; Kilgore, P.; Chopra, A.K.; Rao, V. A prokaryotic-eukaryotic hybrid viral vector for delivery of large cargos of genes and proteins into human cells. Sci Adv 2019, 5, eaax0064–eaax0064. [Google Scholar] [CrossRef] [PubMed]
- Lauster, D.; Klenk, S.; Ludwig, K.; Nojoumi, S.; Behren, S.; Adam, L.; Stadtmüller, M.; Saenger, S.; Zimmler, S.; Hönzke, K.; et al. Phage capsid nanoparticles with defined ligand arrangement block influenza virus entry. Nature Nanotechnology 2020, 15, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Tamura, A.; Azam, A.H.; Nakamura, T.; Lee, K.; Iyoda, S.; Kondo, K.; Ojima, S.; Chihara, K.; Yamashita, W.; Cui, L.; et al. Synthetic phage-based approach for sensitive and specific detection of Escherichia coli O157. Commun Biol 2024, 7, 535. [Google Scholar] [CrossRef]
- Wang, J.; Yang, M.; Zhu, Y.; Wang, L.; Tomsia, A.P.; Mao, C. Phage nanofibers induce vascularized osteogenesis in 3D printed bone scaffolds. Adv Mater 2014, 26, 4961–4966. [Google Scholar] [CrossRef]
- Cao, B.; Yang, M.; Mao, C. Phage as a Genetically Modifiable Supramacromolecule in Chemistry, Materials and Medicine. Acc Chem Res 2016, 49, 1111–1120. [Google Scholar] [CrossRef]
- Jin, H.-E.; Lee, S.-W. Engineering of M13 Bacteriophage for Development of Tissue Engineering Materials. Methods in Molecular Biology 2018, 487–502. [Google Scholar] [CrossRef]
- Zhou, N.; Li, Y.; Loveland, C.H.; Wilson, M.J.; Cao, B.; Qiu, P.; Yang, M.; Mao, C. Hierarchical Ordered Assembly of Genetically Modifiable Viruses into Nanoridge-in-Microridge Structures. Adv Mater 2019, 31, e1905577–e1905577. [Google Scholar] [CrossRef]
- Shrestha, K.R.; Lee, D.H.; Chung, W.; Lee, S.-W.; Lee, B.Y.; Yoo, S.Y. Biomimetic virus-based soft niche for ischemic diseases. Biomaterials 2022, 288, 121747. [Google Scholar] [CrossRef]
- Verbeken, G.; De Vos, D.; Vaneechoutte, M.; Merabishvili, M.; Zizi, M.; Pirnay, J.P. European regulatory conundrum of phage therapy. Future Microbiol 2007, 2, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Dowah, A.S.A.; Clokie, M.R.J. Review of the nature, diversity and structure of bacteriophage receptor binding proteins that target Gram-positive bacteria. Biophys Rev 2018, 10, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Labrie, S.J.; Samson, J.E.; Moineau, S. Bacteriophage resistance mechanisms. Nature Reviews Microbiology 2010, 8, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Margolin, W.; Molineux, I.J.; Liu, J. The bacteriophage t7 virion undergoes extensive structural remodeling during infection. Science (New York, N.Y.) 2013, 339, 576–579. [Google Scholar] [CrossRef] [PubMed]
- Scholl, D.; Adhya, S.; Merril, C. Escherichia coli K1's capsule is a barrier to bacteriophage T7. Appl Environ Microbiol 2005, 71, 4872–4874. [Google Scholar] [CrossRef]
- Salmond, G.P.C.; Fineran, P.C. A century of the phage: past, present and future. Nature Reviews Microbiology 2015, 13, 777–786. [Google Scholar] [CrossRef]
- Weigel, C.; Seitz, H. Bacteriophage replication modules. FEMS Microbiology Reviews 2006, 30, 321–381. [Google Scholar] [CrossRef]
- Penadés, J.R.; Christie, G.E. The Phage-Inducible Chromosomal Islands: A Family of Highly Evolved Molecular Parasites. Annual Review of Virology 2015, 2, 181–201. [Google Scholar] [CrossRef]
- Harper, D.; Parracho, H.; Walker, J.; Sharp, R.; Hughes, G.; Werthén, M.; Lehman, S.; Morales, S. Bacteriophages and Biofilms. Antibiotics 2014, 3, 270–284. [Google Scholar] [CrossRef]
- Lerminiaux, N.A.; Cameron, A.D.S. Horizontal transfer of antibiotic resistance genes in clinical environments. Can J Microbiol 2019, 65, 34–44. [Google Scholar] [CrossRef]
- Usman, S.S.; Uba, A.I.; Christina, E. Bacteriophage genome engineering for phage therapy to combat bacterial antimicrobial resistance as an alternative to antibiotics. Molecular Biology Reports 2023, 50, 7055–7067. [Google Scholar] [CrossRef] [PubMed]
- Hampton, H.G.; Watson, B.N.J.; Fineran, P.C. The arms race between bacteria and their phage foes. Nature 2020, 577, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.C.T.; Friman, V.-P.; Smith, M.C.M.; Brockhurst, M.A. Cross-resistance is modular in bacteria-phage interactions. PLoS Biol 2018, 16, e2006057–e2006057. [Google Scholar] [CrossRef] [PubMed]
- Hodyra-Stefaniak, K.; Miernikiewicz, P.; Drapała, J.; Drab, M.; Jończyk-Matysiak, E.; Lecion, D.; Kaźmierczak, Z.; Beta, W.; Majewska, J.; Harhala, M.; et al. Mammalian Host-Versus-Phage immune response determines phage fate in vivo. Scientific reports 2015, 5, 14802–14802. [Google Scholar] [CrossRef]
- Sweere, J.M.; Van Belleghem, J.D.; Ishak, H.; Bach, M.S.; Popescu, M.; Sunkari, V.; Kaber, G.; Manasherob, R.; Suh, G.A.; Cao, X.; et al. Bacteriophage trigger antiviral immunity and prevent clearance of bacterial infection. Science (New York, N.Y.) 2019, 363, eaat9691. [Google Scholar] [CrossRef]
- Durr, H.A.; Leipzig, N.D. Advancements in bacteriophage therapies and delivery for bacterial infection. Mater Adv 2023, 4, 1249–1257. [Google Scholar] [CrossRef]
- Malik, D.J.; Sokolov, I.J.; Vinner, G.K.; Mancuso, F.; Cinquerrui, S.; Vladisavljevic, G.T.; Clokie, M.R.J.; Garton, N.J.; Stapley, A.G.F.; Kirpichnikova, A. Formulation, stabilisation and encapsulation of bacteriophage for phage therapy. Advances in Colloid and Interface Science 2017, 249, 100–133. [Google Scholar] [CrossRef]
- Shein, A.M.S.; Wannigama, D.L.; Hurst, C.; Monk, P.N.; Amarasiri, M.; Badavath, V.N.; Phattharapornjaroen, P.; Ditcham, W.G.F.; Ounjai, P.; Saethang, T.; et al. Novel intranasal phage-CaEDTA-ceftazidime/avibactam triple combination therapy demonstrates remarkable efficacy in treating Pseudomonas aeruginosa lung infection. Biomedicine & Pharmacotherapy 2023, 168, 115793. [Google Scholar] [CrossRef]
- Pirnay, J.-P.; Verbeken, G.; Ceyssens, P.-J.; Huys, I.; De Vos, D.; Ameloot, C.; Fauconnier, A. The Magistral Phage. Viruses 2018, 10, 64. [Google Scholar] [CrossRef]
- Goel, N.; Hashmi, Z.; Khan, N.; Ahmad, R.; Khan, W.H. Recent Strategies to Combat Multidrug Resistance. In Non-traditional Approaches to Combat Antimicrobial Drug Resistance; 2023; pp. 1–27. [Google Scholar] [CrossRef]
- Abedon, S.T. Phage therapy of pulmonary infections. Bacteriophage 2015, 5, e1020260–e1020260. [Google Scholar] [CrossRef]
- Sarker, S.A.; Sultana, S.; Reuteler, G.; Moine, D.; Descombes, P.; Charton, F.; Bourdin, G.; McCallin, S.; Ngom-Bru, C.; Neville, T.; et al. Oral Phage Therapy of Acute Bacterial Diarrhea With Two Coliphage Preparations: A Randomized Trial in Children From Bangladesh. EBioMedicine 2016, 4, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Pirnay, J.-P.; Djebara, S.; Steurs, G.; Griselain, J.; Cochez, C.; De Soir, S.; Glonti, T.; Spiessens, A.; Vanden Berghe, E.; Green, S.; et al. Personalized bacteriophage therapy outcomes for 100 consecutive cases: a multicentre, multinational, retrospective observational study. Nat Microbiol 2024, 9, 1434–1453. [Google Scholar] [CrossRef] [PubMed]
- Kebriaei, R.; Lehman, S.M.; Shah, R.M.; Stamper, K.C.; Kunz Coyne, A.J.; Holger, D.; El Ghali, A.; Rybak, M.J. Optimization of Phage-Antibiotic Combinations against Staphylococcus aureus Biofilms. Microbiol Spectr 2023, 11, e0491822–e0491822. [Google Scholar] [CrossRef] [PubMed]
- Khalifa, L.; Shlezinger, M.; Beyth, S.; Houri-Haddad, Y.; Coppenhagen-Glazer, S.; Beyth, N.; Hazan, R. Phage therapy against Enterococcus faecalis in dental root canals. J Oral Microbiol 2016, 8, 32157–32157. [Google Scholar] [CrossRef]
- El-Telbany, M.; El-Didamony, G.; Askora, A.; Ariny, E.; Abdallah, D.; Connerton, I.F.; El-Shibiny, A. Bacteriophages to Control Multi-Drug Resistant Enterococcus faecalis Infection of Dental Root Canals. Microorganisms 2021, 9, 517. [Google Scholar] [CrossRef]
- El-Telbany, M.; Lin, C.-Y.; Abdelaziz, M.N.; Maung, A.T.; El-Shibiny, A.; Mohammadi, T.N.; Zayda, M.; Wang, C.; Zar Chi Lwin, S.; Zhao, J.; et al. Potential application of phage vB_EfKS5 to control Enterococcus faecalis and its biofilm in food. AMB Express 2023, 13, 130–130. [Google Scholar] [CrossRef]
- Bolocan, A.S.; Upadrasta, A.; Bettio, P.H.d.A.; Clooney, A.G.; Draper, L.A.; Ross, R.P.; Hill, C. Evaluation of Phage Therapy in the Context of Enterococcus faecalis and Its Associated Diseases. Viruses 2019, 11, 366. [Google Scholar] [CrossRef]
- Chung, K.M.; Liau, X.L.; Tang, S.S. Bacteriophages and Their Host Range in Multidrug-Resistant Bacterial Disease Treatment. Pharmaceuticals (Basel) 2023, 16, 1467. [Google Scholar] [CrossRef]
- Ong, S.P.; Azam, A.H.; Sasahara, T.; Miyanaga, K.; Tanji, Y. Characterization of Pseudomonas lytic phages and their application as a cocktail with antibiotics in controlling Pseudomonas aeruginosa. Journal of Bioscience and Bioengineering 2020, 129, 693–699. [Google Scholar] [CrossRef]
- Chan, B.K.; Abedon, S.T.; Loc-Carrillo, C. Phage Cocktails and the Future of Phage Therapy. Future Microbiology 2013, 8, 769–783. [Google Scholar] [CrossRef]
- Kaneko, T.; Osaka, T.; Tsuneda, S. Tailoring Effective Phage Cocktails for Long-Term Lysis of Escherichia coli Based on Physiological Properties of Constituent Phages. Phage (New Rochelle) 2023, 4, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Gordillo Altamirano, F.L.; Barr, J.J. Phage Therapy in the Postantibiotic Era. Clin Microbiol Rev 2019, 32, e00066–00018. [Google Scholar] [CrossRef] [PubMed]
- Dunne, M.; Rupf, B.; Tala, M.; Qabrati, X.; Ernst, P.; Shen, Y.; Sumrall, E.; Heeb, L.; Plückthun, A.; Loessner, M.J.; et al. Reprogramming Bacteriophage Host Range through Structure-Guided Design of Chimeric Receptor Binding Proteins. Cell Reports 2019, 29, 1336–1350. [Google Scholar] [CrossRef] [PubMed]
- Ronayne, E.A.; Wan, Y.C.S.; Boudreau, B.A.; Landick, R.; Cox, M.M. P1 Ref Endonuclease: A Molecular Mechanism for Phage-Enhanced Antibiotic Lethality. PLoS Genet 2016, 12, e1005797–e1005797. [Google Scholar] [CrossRef] [PubMed]
- Zalewska-Piątek, B. Phage Therapy-Challenges, Opportunities and Future Prospects. Pharmaceuticals (Basel) 2023, 16, 1638. [Google Scholar] [CrossRef]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J Gastrointest Pharmacol Ther 2017, 8, 162–173. [Google Scholar] [CrossRef]
- Międzybrodzki, R.; Kłak, M.; Jończyk-Matysiak, E.; Bubak, B.; Wójcik, A.; Kaszowska, M.; Weber-Dąbrowska, B.; Łobocka, M.; Górski, A. Means to Facilitate the Overcoming of Gastric Juice Barrier by a Therapeutic Staphylococcal Bacteriophage A5/80. Frontiers in microbiology 2017, 8, 467–467. [Google Scholar] [CrossRef]
- Moghtader, F.; Eğri, S.; Piskin, E. Phages in modified alginate beads. Artificial Cells, Nanomedicine, and Biotechnology 2016, 45, 357–363. [Google Scholar] [CrossRef]
- Moghtader, F.; Solakoglu, S.; Piskin, E. Alginate- and Chitosan-Modified Gelatin Hydrogel Microbeads for Delivery of E. coli Phages. Gels 2024, 10, 244. [Google Scholar] [CrossRef]
- Yan, J.; Mao, J.; Xie, J. Bacteriophage Polysaccharide Depolymerases and Biomedical Applications. BioDrugs 2013, 28, 265–274. [Google Scholar] [CrossRef]
- Pires, D.P.; Oliveira, H.; Melo, L.D.R.; Sillankorva, S.; Azeredo, J. Bacteriophage-encoded depolymerases: their diversity and biotechnological applications. Applied Microbiology and Biotechnology 2016, 100, 2141–2151. [Google Scholar] [CrossRef] [PubMed]
- Amankwah, S.; Abdella, K.; Kassa, T. Bacterial Biofilm Destruction: A Focused Review On The Recent Use of Phage-Based Strategies With Other Antibiofilm Agents. Nanotechnol Sci Appl 2021, 14, 161–177. [Google Scholar] [CrossRef] [PubMed]
- Petrovic Fabijan, A.; Iredell, J.; Danis-Wlodarczyk, K.; Kebriaei, R.; Abedon, S.T. Translating phage therapy into the clinic: Recent accomplishments but continuing challenges. PLoS Biol 2023, 21, e3002119–e3002119. [Google Scholar] [CrossRef] [PubMed]
- Fischetti, V.A. Bacteriophage endolysins: a novel anti-infective to control Gram-positive pathogens. Int J Med Microbiol 2010, 300, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Pizarro-Bauerle, J.; Ando, H. Engineered Bacteriophages for Practical Applications. Biological and Pharmaceutical Bulletin 2020, 43, 240–249. [Google Scholar] [CrossRef]
- Li, F.-Y.; Tan, X.-E.; Shimamori, Y.; Nishikawa, Y.; Watanabe, S.; Sasahara, T.; Miyanaga, K.; Aiba, Y.; Veeranarayanan, S.; Thitiananpakorn, K.; et al. Phagemid-Based Capsid System for CRISPR-Cas13a Antimicrobials Targeting Methicillin-Resistant Staphylococcus aureus. Commun Biol 2024, 7, s42003–s42024. [Google Scholar]
- Garenne, D.; Noireaux, V. Cell-free transcription–translation: engineering biology from the nanometer to the millimeter scale. Current Opinion in Biotechnology 2019, 58, 19–27. [Google Scholar] [CrossRef]
- Oślizło, A.; Miernikiewicz, P.; Piotrowicz, A.; Owczarek, B.; Kopciuch, A.; Figura, G.; Dąbrowska, K. Purification of phage display-modified bacteriophage T4 by affinity chromatography. BMC Biotechnol 2011, 11, 59–59. [Google Scholar] [CrossRef]
- Shimamori, Y.; Tan, X.-E.; Li, F.-Y.; Nishikawa, Y.; Watanabe, S.; Sasahara, T.; Miyanaga, K.; Aiba, Y.; Veeranarayanan, S.; Thitiananpakorn, K.; et al. Efficient synthesis of CRISPR-Cas13a-antimicrobial capsids against MRSA facilitated by silent mutation incorporation. Scientific reports 2024, 14, 16225–16225. [Google Scholar] [CrossRef]
- Mitsunaka, S.; Yamazaki, K.; Pramono, A.K.; Ikeuchi, M.; Kitao, T.; Ohara, N.; Kubori, T.; Nagai, H.; Ando, H. Synthetic engineering and biological containment of bacteriophages. Proceedings of the National Academy of Sciences of the United States of America 2022, 119, e2206739119–e2206739119. [Google Scholar] [CrossRef]
- Racenis, K.; Lacis, J.; Rezevska, D.; Mukane, L.; Vilde, A.; Putnins, I.; Djebara, S.; Merabishvili, M.; Pirnay, J.-P.; Kalnina, M.; et al. Successful Bacteriophage-Antibiotic Combination Therapy against Multidrug-Resistant Pseudomonas aeruginosa Left Ventricular Assist Device Driveline Infection. Viruses 2023, 15, 1210. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, J.; Nakamura, K.; Ishiguro, Y.; Iwano, H. Using phage to drive selections toward restoring antibiotic sensitivity in Pseudomonas aeruginosa via chromosomal deletions. Frontiers in microbiology 2024, 15, 1401234–1401234. [Google Scholar] [CrossRef] [PubMed]
- Gordillo Altamirano, F.L.; Kostoulias, X.; Subedi, D.; Korneev, D.; Peleg, A.Y.; Barr, J.J. Phage-antibiotic combination is a superior treatment against Acinetobacter baumannii in a preclinical study. EBioMedicine 2022, 80, 104045–104045. [Google Scholar] [CrossRef] [PubMed]
- Cano, E.J.; Caflisch, K.M.; Bollyky, P.L.; Van Belleghem, J.D.; Patel, R.; Fackler, J.; Brownstein, M.J.; Horne, B.A.; Biswas, B.; Henry, M.; et al. Phage Therapy for Limb-threatening Prosthetic Knee Klebsiella pneumoniae Infection: Case Report and In Vitro Characterization of Anti-biofilm Activity. Clin Infect Dis 2021, 73, e144–e151. [Google Scholar] [CrossRef] [PubMed]
- Petrovic Fabijan, A.; Lin, R.C.Y.; Ho, J.; Maddocks, S.; Ben Zakour, N.L.; Iredell, J.R.; Khalid, A.; Venturini, C.; Chard, R.; Morales, S.; et al. Safety of bacteriophage therapy in severe Staphylococcus aureus infection. Nat Microbiol 2020, 5, 465–472. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.-A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): a randomised, controlled, double-blind phase 1/2 trial. The Lancet Infectious Diseases 2019, 19, 35–45. [Google Scholar] [CrossRef]
- Osman, A.-H.; Kotey, F.C.N.; Odoom, A.; Darkwah, S.; Yeboah, R.K.; Dayie, N.T.K.D.; Donkor, E.S. The Potential of Bacteriophage-Antibiotic Combination Therapy in Treating Infections with Multidrug-Resistant Bacteria. Antibiotics (Basel) 2023, 12, 1329. [Google Scholar] [CrossRef]
- Plaut, R.D.; Stibitz, S. Regulatory Considerations for Bacteriophage Therapy Products: USA. Bacteriophages 2021, 1151–1163. [Google Scholar] [CrossRef]
- Fedorov, E.; Samokhin, A.; Kozlova, Y.; Kretien, S.; Sheraliev, T.; Morozova, V.; Tikunova, N.; Kiselev, A.; Pavlov, V. Short-Term Outcomes of Phage-Antibiotic Combination Treatment in Adult Patients with Periprosthetic Hip Joint Infection. Viruses 2023, 15, 499. [Google Scholar] [CrossRef]
- Ghanaim, A.M.; Foaad, M.A.; Gomaa, E.Z.; Dougdoug, K.A.E.; Mohamed, G.E.; Arisha, A.H.; Khamis, T. Bacteriophage therapy as an alternative technique for treatment of multidrug-resistant bacteria causing diabetic foot infection. Int Microbiol 2023, 26, 343–359. [Google Scholar] [CrossRef]
- Abdelhamid, A.G.; Yousef, A.E. Combating Bacterial Biofilms: Current and Emerging Antibiofilm Strategies for Treating Persistent Infections. Antibiotics (Basel) 2023, 12, 1005. [Google Scholar] [CrossRef] [PubMed]
- Akturk, E.; Melo, L.D.R.; Oliveira, H.; Crabbé, A.; Coenye, T.; Azeredo, J. Combining phages and antibiotic to enhance antibiofilm efficacy against an in vitro dual species wound biofilm. Biofilm 2023, 6, 100147–100147. [Google Scholar] [CrossRef] [PubMed]
- Melo, L.D.R.; Ferreira, R.; Costa, A.R.; Oliveira, H.; Azeredo, J. Efficacy and safety assessment of two enterococci phages in an in vitro biofilm wound model. Scientific reports 2019, 9, 6643–6643. [Google Scholar] [CrossRef] [PubMed]
- Olszak, T.; Danis-Wlodarczyk, K.; Arabski, M.; Gula, G.; Maciejewska, B.; Wasik, S.; Lood, C.; Higgins, G.; Harvey, B.J.; Lavigne, R.; et al. Pseudomonas aeruginosa PA5oct Jumbo Phage Impacts Planktonic and Biofilm Population and Reduces Its Host Virulence. Viruses 2019, 11, 1089. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.P.; Dötsch, A.; Anderson, E.M.; Hao, Y.; Khursigara, C.M.; Lam, J.S.; Sillankorva, S.; Azeredo, J. A Genotypic Analysis of Five P. aeruginosa Strains after Biofilm Infection by Phages Targeting Different Cell Surface Receptors. Frontiers in microbiology 2017, 8, 1229–1229. [Google Scholar] [CrossRef]
- Wang, F.; Liu, P.; Sun, L.; Li, C.; Petrenko, V.A.; Liu, A. Bio-mimetic nanostructure self-assembled from Au@Ag heterogeneous nanorods and phage fusion proteins for targeted tumor optical detection and photothermal therapy. Scientific reports 2014, 4, 6808–6808. [Google Scholar] [CrossRef]
- Ribeiro, J.M.; Pereira, G.N.; Durli Junior, I.; Teixeira, G.M.; Bertozzi, M.M.; Verri, W.A., Jr.; Kobayashi, R.K.T.; Nakazato, G. Comparative analysis of effectiveness for phage cocktail development against multiple Salmonella serovars and its biofilm control activity. Scientific reports 2023, 13, 13054–13054. [Google Scholar] [CrossRef]
- Gutiérrez, D.; Briers, Y.; Rodríguez-Rubio, L.; Martínez, B.; Rodríguez, A.; Lavigne, R.; García, P. Role of the Pre-neck Appendage Protein (Dpo7) from Phage vB_SepiS-phiIPLA7 as an Anti-biofilm Agent in Staphylococcal Species. Frontiers in microbiology 2015, 6, 1315–1315. [Google Scholar] [CrossRef]
- Zuo, P.; Metz, J.; Yu, P.; Alvarez, P.J.J. Biofilm-responsive encapsulated-phage coating for autonomous biofouling mitigation in water storage systems. Water Research 2022, 224, 119070. [Google Scholar] [CrossRef]
- Kim, H.Y.; Chang, R.Y.K.; Morales, S.; Chan, H.-K. Bacteriophage-Delivering Hydrogels: Current Progress in Combating Antibiotic Resistant Bacterial Infection. Antibiotics (Basel) 2021, 10, 130. [Google Scholar] [CrossRef]
- Pires, D.P.; Meneses, L.; Brandão, A.C.; Azeredo, J. An overview of the current state of phage therapy for the treatment of biofilm-related infections. Current Opinion in Virology 2022, 53, 101209. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, Y.; Bai, C.; Leung, S.S.Y. Translating bacteriophage-derived depolymerases into antibacterial therapeutics: Challenges and prospects. Acta Pharm Sin B 2024, 14, 155–169. [Google Scholar] [CrossRef] [PubMed]
- Sanmukh, S.G.; Admella, J.; Moya-Andérico, L.; Fehér, T.; Arévalo-Jaimes, B.V.; Blanco-Cabra, N.; Torrents, E. Accessing the In Vivo Efficiency of Clinically Isolated Phages against Uropathogenic and Invasive Biofilm-Forming Escherichia coli Strains for Phage Therapy. Cells 2023, 12, 344. [Google Scholar] [CrossRef] [PubMed]
- Verbanic, S.; Deacon, J.M.; Chen, I.A. The Chronic Wound Phageome: Phage Diversity and Associations with Wounds and Healing Outcomes. Microbiol Spectr 2022, 10, e0277721–e0277721. [Google Scholar] [CrossRef]
- Fiscarelli, E.V.; Rossitto, M.; Rosati, P.; Essa, N.; Crocetta, V.; Di Giulio, A.; Lupetti, V.; Di Bonaventura, G.; Pompilio, A. In Vitro Newly Isolated Environmental Phage Activity against Biofilms Preformed by Pseudomonas aeruginosa from Patients with Cystic Fibrosis. Microorganisms 2021, 9, 478. [Google Scholar] [CrossRef]
- Tan, X.; Chen, H.; Zhang, M.; Zhao, Y.; Jiang, Y.; Liu, X.; Huang, W.; Ma, Y. Clinical Experience of Personalized Phage Therapy Against Carbapenem-Resistant Acinetobacter baumannii Lung Infection in a Patient With Chronic Obstructive Pulmonary Disease. Front Cell Infect Microbiol 2021, 11, 631585–631585. [Google Scholar] [CrossRef]
- Mirzaei, A.; Wagemans, J.; Nasr Esfahani, B.; Lavigne, R.; Moghim, S. A Phage Cocktail To Control Surface Colonization by Proteus mirabilis in Catheter-Associated Urinary Tract Infections. Microbiol Spectr 2022, 10, e0209222–e0209222. [Google Scholar] [CrossRef]
- Kowalski, J.; Górska, R.; Cieślik, M.; Górski, A.; Jończyk-Matysiak, E. What Are the Potential Benefits of Using Bacteriophages in Periodontal Therapy? Antibiotics (Basel) 2022, 11, 446. [Google Scholar] [CrossRef]
- Chen, Z.; Guo, Z.; Lin, H.; Tian, Y.; Zhang, P.; Chen, H.; Wang, Y.; Shen, Y. The Feasibility of Phage Therapy for Periodontitis. Future Microbiology 2021, 16, 649–656. [Google Scholar] [CrossRef]
- Cooper, C.J.; Khan Mirzaei, M.; Nilsson, A.S. Adapting Drug Approval Pathways for Bacteriophage-Based Therapeutics. Frontiers in microbiology 2016, 7, 1209–1209. [Google Scholar] [CrossRef]
- Borin, J.M.; Avrani, S.; Barrick, J.E.; Petrie, K.L.; Meyer, J.R. Coevolutionary phage training leads to greater bacterial suppression and delays the evolution of phage resistance. Proceedings of the National Academy of Sciences of the United States of America 2021, 118, e2104592118. [Google Scholar] [CrossRef] [PubMed]
- Schmalstig, A.A.; Wiggins, A.; Badillo, D.; Wetzel, K.S.; Hatfull, G.F.; Braunstein, M. Bacteriophage infection and killing of intracellular Mycobacterium abscessus. Mbio 2024, 15, e0292423–e0292423. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Labani-Motlagh, A.; Bohorquez, J.A.; Moreira, J.D.; Ansari, D.; Patel, S.; Spagnolo, F.; Florence, J.; Vankayalapati, A.; Sakai, T.; et al. Bacteriophage therapy for the treatment of Mycobacterium tuberculosis infections in humanized mice. Commun Biol 2024, 7, 294–294. [Google Scholar] [CrossRef] [PubMed]
- Johansen, M.D.; Alcaraz, M.; Dedrick, R.M.; Roquet-Banères, F.; Hamela, C.; Hatfull, G.F.; Kremer, L. Mycobacteriophage-antibiotic therapy promotes enhanced clearance of drug-resistant Mycobacterium abscessus. Dis Model Mech 2021, 14, dmm049159. [Google Scholar] [CrossRef]
- Dedrick, R.M.; Jacobs-Sera, D.; Bustamante, C.A.G.; Garlena, R.A.; Mavrich, T.N.; Pope, W.H.; Reyes, J.C.C.; Russell, D.A.; Adair, T.; Alvey, R.; et al. Prophage-mediated defence against viral attack and viral counter-defence. Nat Microbiol 2017, 2, 16251–16251. [Google Scholar] [CrossRef]
- Shield, C.G.; Swift, B.M.C.; McHugh, T.D.; Dedrick, R.M.; Hatfull, G.F.; Satta, G. Application of Bacteriophages for Mycobacterial Infections, from Diagnosis to Treatment. Microorganisms 2021, 9, 2366. [Google Scholar] [CrossRef]
- Dedrick, R.M.; Freeman, K.G.; Nguyen, J.A.; Bahadirli-Talbott, A.; Cardin, M.E.; Cristinziano, M.; Smith, B.E.; Jeong, S.; Ignatius, E.H.; Lin, C.T.; et al. Nebulized Bacteriophage in a Patient With Refractory Mycobacterium abscessus Lung Disease. Open Forum Infect Dis 2022, 9, ofac194–ofac194. [Google Scholar] [CrossRef]
- Broxmeyer, L.; Sosnowska, D.; Miltner, E.; Chacón, O.; Wagner, D.; McGarvey, J.; Barletta, Raúl G.; Bermudez, Luiz E. Killing of Mycobacterium avium and Mycobacterium tuberculosis by a Mycobacteriophage Delivered by a Nonvirulent Mycobacterium: A Model for Phage Therapy of Intracellular Bacterial Pathogens. The Journal of Infectious Diseases 2002, 186, 1155–1160. [Google Scholar] [CrossRef]
- García, P.; Martínez, B.; Obeso, J.M.; Rodríguez, A. Bacteriophages and their application in food safety. Letters in Applied Microbiology 2008, 47, 479–485. [Google Scholar] [CrossRef]
- Kropinski, A.M.; Sulakvelidze, A.; Konczy, P.; Poppe, C. Salmonella Phages and Prophages—Genomics and Practical Aspects. Methods in Molecular Biology 2007, 133–175. [Google Scholar] [CrossRef]
- Khan Mirzaei, M.; Nilsson, A.S. Isolation of phages for phage therapy: a comparison of spot tests and efficiency of plating analyses for determination of host range and efficacy. PLoS One 2015, 10, e0118557–e0118557. [Google Scholar] [CrossRef] [PubMed]
- Mai, V.; Ukhanova, M.; Reinhard, M.K.; Li, M.; Sulakvelidze, A. Bacteriophage administration significantly reduces Shigella colonization and shedding by Shigella-challenged mice without deleterious side effects and distortions in the gut microbiota. Bacteriophage 2015, 5, e1088124–e1088124. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Liu, Q.; Lian, T.; Shao, L. The ΦCPG1 chlamydiaphage can infect Chlamydia trachomatis and significantly reduce its infectivity. Virus Research 2019, 267, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Śliwa-Dominiak, J.; Suszyńska, E.; Pawlikowska, M.; Deptuła, W. Chlamydia bacteriophages. Archives of Microbiology 2013, 195, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Beitzinger, B.; Gerbl, F.; Vomhof, T.; Schmid, R.; Noschka, R.; Rodriguez, A.; Wiese, S.; Weidinger, G.; Ständker, L.; Walther, P.; et al. Delivery by Dendritic Mesoporous Silica Nanoparticles Enhances the Antimicrobial Activity of a Napsin-Derived Peptide Against Intracellular Mycobacterium tuberculosis. Advanced Healthcare Materials 2021, 10. [Google Scholar] [CrossRef]
- Nick, J.A.; Dedrick, R.M.; Gray, A.L.; Vladar, E.K.; Smith, B.E.; Freeman, K.G.; Malcolm, K.C.; Epperson, L.E.; Hasan, N.A.; Hendrix, J.; et al. Host and pathogen response to bacteriophage engineered against Mycobacterium abscessus lung infection. Cell 2022, 185, 1860–1874. [Google Scholar] [CrossRef]
- Guerrero-Bustamante, C.A.; Dedrick, R.M.; Garlena, R.A.; Russell, D.A.; Hatfull, G.F. Toward a Phage Cocktail for Tuberculosis: Susceptibility and Tuberculocidal Action of Mycobacteriophages against Diverse Mycobacterium tuberculosis Strains. Mbio 2021, 12, e00973–00921. [Google Scholar] [CrossRef]
- Mohammad Hasani, S.; Ghafouri, E.; Kouhpayeh, S.; Amerizadeh, F.; Rahimmanesh, I.; Amirkhani, Z.; Khanahmad, H. Phage based vaccine: A novel strategy in prevention and treatment. Heliyon 2023, 9, e19925–e19925. [Google Scholar] [CrossRef]
- Bao, Q.; Li, X.; Han, G.; Zhu, Y.; Mao, C.; Yang, M. Phage-based vaccines. Advanced Drug Delivery Reviews 2019, 145, 40–56. [Google Scholar] [CrossRef]
- Zhu, J.; Jain, S.; Sha, J.; Batra, H.; Ananthaswamy, N.; Kilgore, P.B.; Hendrix, E.K.; Hosakote, Y.M.; Wu, X.; Olano, J.P.; et al. A Bacteriophage-Based, Highly Efficacious, Needle- and Adjuvant-Free, Mucosal COVID-19 Vaccine. Mbio 2022, 13, e0182222–e0182222. [Google Scholar] [CrossRef]
- Tao, P.; Mahalingam, M.; Zhu, J.; Moayeri, M.; Sha, J.; Lawrence, W.S.; Leppla, S.H.; Chopra, A.K.; Rao, V.B. A Bacteriophage T4 Nanoparticle-Based Dual Vaccine against Anthrax and Plague. Mbio 2018, 9, e01926–01918. [Google Scholar] [CrossRef] [PubMed]
- Jepson, C.D.; March, J.B. Bacteriophage lambda is a highly stable DNA vaccine delivery vehicle. Vaccine 2004, 22, 2413–2419. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Międzybrodzki, R.; Borysowski, J.; Dąbrowska, K.; Wierzbicki, P.; Ohams, M.; Korczak-Kowalska, G.; Olszowska-Zaremba, N.; Łusiak-Szelachowska, M.; Kłak, M.; et al. Phage as a Modulator of Immune Responses. Advances in Virus Research 2012, 41–71. [Google Scholar] [CrossRef]
- Krut, O.; Bekeredjian-Ding, I. Contribution of the Immune Response to Phage Therapy. The Journal of Immunology 2018, 200, 3037–3044. [Google Scholar] [CrossRef] [PubMed]
- Van Belleghem, J.D.; Dąbrowska, K.; Vaneechoutte, M.; Barr, J.J.; Bollyky, P.L. Interactions between Bacteriophage, Bacteria, and the Mammalian Immune System. Viruses 2018, 11, 10. [Google Scholar] [CrossRef]
- Popescu, M.; Van Belleghem, J.D.; Khosravi, A.; Bollyky, P.L. Bacteriophages and the Immune System. Annual Review of Virology 2021, 8, 415–435. [Google Scholar] [CrossRef]
- Carroll-Portillo, A.; Lin, H.C. Bacteriophage and the Innate Immune System: Access and Signaling. Microorganisms 2019, 7, 625. [Google Scholar] [CrossRef]
- Sartorius, R.; D'Apice, L.; Trovato, M.; Cuccaro, F.; Costa, V.; De Leo, M.G.; Marzullo, V.M.; Biondo, C.; D'Auria, S.; De Matteis, M.A.; et al. Antigen delivery by filamentous bacteriophage fd displaying an anti-DEC-205 single-chain variable fragment confers adjuvanticity by triggering a TLR9-mediated immune response. EMBO Mol Med 2015, 7, 973–988. [Google Scholar] [CrossRef]
- Chatterjee, A.; Duerkop, B.A. Beyond Bacteria: Bacteriophage-Eukaryotic Host Interactions Reveal Emerging Paradigms of Health and Disease. Frontiers in microbiology 2018, 9, 1394–1394. [Google Scholar] [CrossRef]
- Eriksson, F.; Tsagozis, P.; Lundberg, K.; Parsa, R.; Mangsbo, S.M.; Persson, M.A.A.; Harris, R.A.; Pisa, P. Tumor-Specific Bacteriophages Induce Tumor Destruction through Activation of Tumor-Associated Macrophages. The Journal of Immunology 2009, 182, 3105–3111. [Google Scholar] [CrossRef]
- Ragothaman, M.; Yoo, S.Y. Engineered Phage-Based Cancer Vaccines: Current Advances and Future Directions. Vaccines (Basel) 2023, 11, 919. [Google Scholar] [CrossRef]
- Gong, Y.-M.; Wei, X.-F.; Zheng, Y.-Y.; Li, Y.; Yu, Q.; Li, P.-F.; Zhu, B. Combining Phage Display Technology with In Silico-Designed Epitope Vaccine to Elicit Robust Antibody Responses against Emerging Pathogen Tilapia Lake Virus. J Virol 2023, 97, e0005023–e0005023. [Google Scholar] [CrossRef] [PubMed]
- Iwagami, Y.; Casulli, S.; Nagaoka, K.; Kim, M.; Carlson, R.I.; Ogawa, K.; Lebowitz, M.S.; Fuller, S.; Biswas, B.; Stewart, S.; et al. Lambda phage-based vaccine induces antitumor immunity in hepatocellular carcinoma. Heliyon 2017, 3, e00407–e00407. [Google Scholar] [CrossRef] [PubMed]
- Staquicini, D.I.; Tang, F.H.F.; Markosian, C.; Yao, V.J.; Staquicini, F.I.; Dodero-Rojas, E.; Contessoto, V.G.; Davis, D.; O'Brien, P.; Habib, N.; et al. Design and proof of concept for targeted phage-based COVID-19 vaccination strategies with a streamlined cold-free supply chain. Proceedings of the National Academy of Sciences of the United States of America 2021, 118, e2105739118. [Google Scholar] [CrossRef] [PubMed]
- Ul Haq, I.; Krukiewicz, K.; Yahya, G.; Haq, M.U.; Maryam, S.; Mosbah, R.A.; Saber, S.; Alrouji, M. The Breadth of Bacteriophages Contributing to the Development of the Phage-Based Vaccines for COVID-19: An Ideal Platform to Design the Multiplex Vaccine. International journal of molecular sciences 2023, 24, 1536. [Google Scholar] [CrossRef] [PubMed]
- Henein, A. What are the limitations on the wider therapeutic use of phage? Bacteriophage 2013, 3, e24872–e24872. [Google Scholar] [CrossRef] [PubMed]
- Furfaro, L.L.; Payne, M.S.; Chang, B.J. Bacteriophage Therapy: Clinical Trials and Regulatory Hurdles. Front Cell Infect Microbiol 2018, 8, 376–376. [Google Scholar] [CrossRef] [PubMed]
- Verbeken, G.; Pirnay, J.-P.; De Vos, D.; Jennes, S.; Zizi, M.; Lavigne, R.; Casteels, M.; Huys, I. Optimizing the European Regulatory Framework for Sustainable Bacteriophage Therapy in Human Medicine. Archivum Immunologiae et Therapiae Experimentalis 2012, 60, 161–172. [Google Scholar] [CrossRef]
- Faltus, T. The Medicinal Phage-Regulatory Roadmap for Phage Therapy under EU Pharmaceutical Legislation. Viruses 2024, 16, 443. [Google Scholar] [CrossRef]
- Strathdee, S.A.; Hatfull, G.F.; Mutalik, V.K.; Schooley, R.T. Phage therapy: From biological mechanisms to future directions. Cell 2023, 186, 17–31. [Google Scholar] [CrossRef]
- Manohar, P.; Tamhankar, A.J.; Leptihn, S.; Ramesh, N. Pharmacological and Immunological Aspects of Phage Therapy. Infectious Microbes and Diseases 2019, 1, 34–42. [Google Scholar] [CrossRef]
- Przystal, J.M.; Waramit, S.; Pranjol, M.Z.I.; Yan, W.; Chu, G.; Chongchai, A.; Samarth, G.; Olaciregui, N.G.; Tabatabai, G.; Carcaboso, A.M.; et al. Efficacy of systemic temozolomide-activated phage-targeted gene therapy in human glioblastoma. EMBO Mol Med 2019, 11, e8492. [Google Scholar] [CrossRef] [PubMed]
- Chongchai, A.; Bentayebi, K.; Chu, G.; Yan, W.; Waramit, S.; Phitak, T.; Kongtawelert, P.; Pothacharoen, P.; Suwan, K.; Hajitou, A. Targeted treatment of chondrosarcoma with a bacteriophage-based particle delivering a secreted tumor necrosis factor-related apoptosis-inducing ligand. Mol Ther Oncol 2024, 32, 200805–200805. [Google Scholar] [CrossRef] [PubMed]
- Sittiju, P.; Wudtiwai, B.; Chongchai, A.; Hajitou, A.; Kongtawelert, P.; Pothacharoen, P.; Suwan, K. Bacteriophage-based particles carrying the TNF-related apoptosis-inducing ligand (TRAIL) gene for targeted delivery in hepatocellular carcinoma. Nanoscale 2024, 16, 6603–6617. [Google Scholar] [CrossRef] [PubMed]
- Al-Bahrani, M.; Asavarut, P.; Waramit, S.; Suwan, K.; Hajitou, A. Transmorphic phage-guided systemic delivery of TNFα gene for the treatment of human pediatric medulloblastoma. FASEB J 2023, 37, e23038–e23038. [Google Scholar] [CrossRef]
- Chongchai, A.; Waramit, S.; Suwan, K.; Al-Bahrani, M.; Udomruk, S.; Phitak, T.; Kongtawelert, P.; Pothacharoen, P.; Hajitou, A. Bacteriophage-mediated therapy of chondrosarcoma by selective delivery of the tumor necrosis factor alpha (TNFα) gene. The FASEB Journal 2021, 35. [Google Scholar] [CrossRef]
- Dong, X.; Pan, P.; Zhang, Q.; Ye, J.-J.; Zhang, X.-Z. Engineered Living Bacteriophage-Enabled Self-Adjuvanting Hydrogel for Remodeling Tumor Microenvironment and Cancer Therapy. Nano Letters 2023, 23, 1219–1228. [Google Scholar] [CrossRef]
- Zheng, D.-W.; Dong, X.; Pan, P.; Chen, K.-W.; Fan, J.-X.; Cheng, S.-X.; Zhang, X.-Z. Phage-guided modulation of the gut microbiota of mouse models of colorectal cancer augments their responses to chemotherapy. Nature Biomedical Engineering 2019, 3, 717–728. [Google Scholar] [CrossRef]
- Ulfo, L.; Cantelli, A.; Petrosino, A.; Costantini, P.E.; Nigro, M.; Starinieri, F.; Turrini, E.; Zadran, S.K.; Zuccheri, G.; Saporetti, R.; et al. Orthogonal nanoarchitectonics of M13 phage for receptor targeted anticancer photodynamic therapy. Nanoscale 2022, 14, 632–641. [Google Scholar] [CrossRef]
- Hwang, Y.J.; Myung, H. Engineered Bacteriophage T7 as a Potent Anticancer Agent in vivo. Frontiers in microbiology 2020, 11, 491001–491001. [Google Scholar] [CrossRef]
- Wang, J.; Lamolinara, A.; Conti, L.; Giangrossi, M.; Cui, L.; Morelli, M.B.; Amantini, C.; Falconi, M.; Bartolacci, C.; Andreani, C.; et al. HER2-Displaying M13 Bacteriophages induce Therapeutic Immunity against Breast Cancer. Cancers (Basel) 2022, 14, 4054. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Yan, J.; Xin, K.; Li, L.; Sun, Q.; Wang, Y.; Chen, T.; Wu, S.; Shao, J.; Liu, B.; et al. Engineered Bacteriophage-Based <i>In Situ</i> Vaccine Remodels a Tumor Microenvironment and Elicits Potent Antitumor Immunity. ACS Nano 2024, 18, 12194–12209. [Google Scholar] [CrossRef]
- Shoae-Hassani, A.; Keyhanvar, P.; Seifalian, A.M.; Mortazavi-Tabatabaei, S.A.; Ghaderi, N.; Issazadeh, K.; Amirmozafari, N.; Verdi, J. λ Phage nanobioparticle expressing apoptin efficiently suppress human breast carcinoma tumor growth in vivo. PLoS One 2013, 8, e79907–e79907. [Google Scholar] [CrossRef] [PubMed]
- Razazan, A.; Nicastro, J.; Slavcev, R.; Barati, N.; Arab, A.; Mosaffa, F.; Jaafari, M.R.; Behravan, J. Lambda bacteriophage nanoparticles displaying GP2, a HER2/neu derived peptide, induce prophylactic and therapeutic activities against TUBO tumor model in mice. Scientific reports 2019, 9, 2221–2221. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Batra, H.; Ananthaswamy, N.; Mahalingam, M.; Tao, P.; Wu, X.; Guo, W.; Fokine, A.; Rao, V.B. Design of bacteriophage T4-based artificial viral vectors for human genome remodeling. Nat Commun 2023, 14, 2928–2928. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.-L.; Xie, X.-T.; Tan, L.-F.; Zhang, F.; Fan, J.-X.; Chen, W.; Hu, Y.-G.; Zhao, Y.-D.; Liu, B.; Xu, Q.-R. T4 Phage Display Technology for Enhanced Photodynamic Therapy of Breast Cancer. ACS Materials Letters 2023, 5, 2270–2281. [Google Scholar] [CrossRef]
- Shukla, G.S.; Sun, Y.-J.; Pero, S.C.; Sholler, G.S.; Krag, D.N. Immunization with tumor neoantigens displayed on T7 phage nanoparticles elicits plasma antibody and vaccine-draining lymph node B cell responses. Journal of Immunological Methods 2018, 460, 51–62. [Google Scholar] [CrossRef]
- Islam, M.S.; Fan, J.; Pan, F. The power of phages: revolutionizing cancer treatment. Front Oncol 2023, 13, 1290296–1290296. [Google Scholar] [CrossRef]
- Łobocka, M.; Dąbrowska, K.; Górski, A. Engineered Bacteriophage Therapeutics: Rationale, Challenges and Future. BioDrugs 2021, 35, 255–280. [Google Scholar] [CrossRef]
- Manivannan, A.C.; Dhandapani, R.; Velmurugan, P.; Thangavelu, S.; Paramasivam, R.; Ragunathan, L.; Saravanan, M. Phage in cancer treatment – Biology of therapeutic phage and screening of tumor targeting peptide. Expert Opinion on Drug Delivery 2022, 19, 873–882. [Google Scholar] [CrossRef]
- Karimi, M.; Mirshekari, H.; Moosavi Basri, S.M.; Bahrami, S.; Moghoofei, M.; Hamblin, M.R. Bacteriophages and phage-inspired nanocarriers for targeted delivery of therapeutic cargos. Advanced drug delivery reviews 2016, 106, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Parkhey, P.; Mishra, S.K.; Paul, P.K.; Singh, A.; Singh, V. Phage for drug delivery vehicles. Progress in Molecular Biology and Translational Science 2023, 191–201. [Google Scholar] [CrossRef]
- Hajitou, A. Targeted Systemic Gene Therapy and Molecular Imaging of Cancer. Tissue-Specific Vascular Endothelial Signals and Vector Targeting, Part B 2010, 65–82. [Google Scholar] [CrossRef]
- Qu, X.; Qiu, P.; Zhu, Y.; Yang, M.; Mao, C. Guiding nanomaterials to tumors for breast cancer precision medicine: from tumor-targeting small-molecule discovery to targeted nanodrug delivery. NPG Asia Mater 2017, 9, e452. [Google Scholar] [CrossRef] [PubMed]
- Turrini, E.; Ulfo, L.; Costantini, P.E.; Saporetti, R.; Di Giosia, M.; Nigro, M.; Petrosino, A.; Pappagallo, L.; Kaltenbrunner, A.; Cantelli, A.; et al. Molecular engineering of a spheroid-penetrating phage nanovector for photodynamic treatment of colon cancer cells. Cell Mol Life Sci 2024, 81, 144–144. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, K.; Hayashi, T.; Yamamoto, M.; Sato, N.; Shimohigoshi, M.; Miyaoka, D.; Yokota, C.; Watanabe, M.; Hisaki, Y.; Kamei, Y.; et al. An enterococcal phage-derived enzyme suppresses graft-versus-host disease. Nature 2024, 632, 174–181. [Google Scholar] [CrossRef]
- Petrosino, A.; Saporetti, R.; Starinieri, F.; Sarti, E.; Ulfo, L.; Boselli, L.; Cantelli, A.; Morini, A.; Zadran, S.K.; Zuccheri, G.; et al. A modular phage vector platform for targeted photodynamic therapy of Gram-negative bacterial pathogens. iScience 2023, 26, 108032–108032. [Google Scholar] [CrossRef]
- Shahrivarkevishahi, A.; Luzuriaga, M.A.; Herbert, F.C.; Tumac, A.C.; Brohlin, O.R.; Wijesundara, Y.H.; Adlooru, A.V.; Benjamin, C.; Lee, H.; Parsamian, P.; et al. PhotothermalPhage: A Virus-Based Photothermal Therapeutic Agent. Journal of the American Chemical Society 2021, 143, 16428–16438. [Google Scholar] [CrossRef]
- Cui, L.; Arbaah, M.; Thuy, N.M.; Aiba, Y.; Watanabe, S. Microbial Warfare in the Gut: Unveiling the Battle between Enterotoxigenic Bacteroides Fragilis and Bacteriophages. Sci J Biol & Life Sci 2023, 3. [Google Scholar] [CrossRef]
- Petrenko, V.A.; Gillespie, J.W. Paradigm shift in bacteriophage-mediated delivery of anticancer drugs: from targeted 'magic bullets' to self-navigated 'magic missiles'. Expert opinion on drug delivery 2017, 14, 373–384. [Google Scholar] [CrossRef]
- Bar, H.; Yacoby, I.; Benhar, I. Killing cancer cells by targeted drug-carrying phage nanomedicines. BMC Biotechnol 2008, 8, 37–37. [Google Scholar] [CrossRef] [PubMed]
- Li, X.T.; Peng, S.Y.; Feng, S.M.; Bao, T.Y.; Li, S.Z.; Li, S.Y. Recent Progress in Phage-Based Nanoplatforms for Tumor Therapy. Small 2023, 20. [Google Scholar] [CrossRef] [PubMed]
- Sunderland, K.S.; Yang, M.; Mao, C. Phage-Enabled Nanomedicine: From Probes to Therapeutics in Precision Medicine. Angew Chem Int Ed Engl 2017, 56, 1964–1992. [Google Scholar] [CrossRef] [PubMed]
- Cortés, P.; Cano-Sarabia, M.; Colom, J.; Otero, J.; Maspoch, D.; Llagostera, M. Nano/Micro Formulations for Bacteriophage Delivery. Methods in Molecular Biology 2017, 271–283. [Google Scholar] [CrossRef]
- Barros, J.A.R.; Melo, L.D.R.d.; Silva, R.A.R.d.; Ferraz, M.P.; Azeredo, J.C.V.d.R.; Pinheiro, V.M.d.C.; Colaço, B.J.A.; Fernandes, M.H.R.; Gomes, P.d.S.; Monteiro, F.J. Encapsulated bacteriophages in alginate-nanohydroxyapatite hydrogel as a novel delivery system to prevent orthopedic implant-associated infections. Nanomedicine: Nanotechnology, Biology and Medicine 2020, 24, 102145. [Google Scholar] [CrossRef]
- Vera-Robles, L.I.; González-Gracida, J.; Hernández-Gordillo, A.; Campero, A. Using the M13 Phage as a Biotemplate to Create Mesoporous Structures Decorated with Gold and Platinum Nanoparticles. Langmuir 2015, 31, 9188–9197. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Watanabe, S.; Sharmin, S.; Kawaguchi, T.; Tan, X.E.; Wannigama, D.L.; Cui, L. RNA and Single-Stranded DNA Phages: Unveiling the Promise from the Underexplored World of Viruses. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Zhao, X.; Zhong, X.; Yang, S.; Deng, J.; Deng, K.; Huang, Z.; Li, Y.; Yin, Z.; Liu, Y.; Viel, J.H.; et al. Guiding antibiotics towards their target using bacteriophage proteins. Nat Commun 2024, 15, 5287–5287. [Google Scholar] [CrossRef]
- Aljabali, A.A.A.; Aljbaly, M.B.M.; Obeid, M.A.; Shahcheraghi, S.H.; Tambuwala, M.M. The Next Generation of Drug Delivery: Harnessing the Power of Bacteriophages. Methods in Molecular Biology 2023, 279–315. [Google Scholar] [CrossRef]
- Singla, S.; Harjai, K.; Katare, O.P.; Chhibber, S. Encapsulation of Bacteriophage in Liposome Accentuates Its Entry in to Macrophage and Shields It from Neutralizing Antibodies. PLoS One 2016, 11, e0153777–e0153777. [Google Scholar] [CrossRef]
- Naskalska, A.; Heddle, J.G. Virus-like particles derived from bacteriophage MS2 as antigen scaffolds and RNA protective shells. Nanomedicine (Lond) 2024, 19, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Ju, Z.; Sun, W. Drug delivery vectors based on filamentous bacteriophages and phage-mimetic nanoparticles. Drug Deliv 2017, 24, 1898–1908. [Google Scholar] [CrossRef] [PubMed]
- Garg, P. Filamentous bacteriophage. Journal of Cancer Research and Therapeutics 2019, 15, S1–S10. [Google Scholar] [CrossRef] [PubMed]
- Nieth, A.; Verseux, C.; Barnert, S.; Süss, R.; Römer, W. A first step toward liposome-mediated intracellular bacteriophage therapy. Expert Opinion on Drug Delivery 2015, 12, 1411–1424. [Google Scholar] [CrossRef] [PubMed]
- Hathaway, H.; Milo, S.; Sutton, J.M.; Jenkins, T.A. Recent Advances in Therapeutic Delivery Systems of Bacteriophage and Bacteriophage-Encoded Endolysins. Therapeutic Delivery 2017, 8, 543–556. [Google Scholar] [CrossRef]
- Cortés, P.; Cano-Sarabia, M.; Colom, J.; Otero, J.; Maspoch, D.; Llagostera, M. Nano/microformulations for Bacteriophage Delivery. Methods in Molecular Biology 2023, 117–130. [Google Scholar] [CrossRef]
- Rastogi, V.; Yadav, P.; Verma, A.; Pandit, J.K. Ex vivo and in vivo evaluation of microemulsion based transdermal delivery of E. coli specific T4 bacteriophage: A rationale approach to treat bacterial infection. European Journal of Pharmaceutical Sciences 2017, 107, 168–182. [Google Scholar] [CrossRef]
- Franca, R.K.A.; Studart, I.C.; Bezerra, M.R.L.; Pontes, L.Q.; Barbosa, A.M.A.; Brigido, M.M.; Furtado, G.P.; Maranhao, A.Q. Progress on Phage Display Technology: Tailoring Antibodies for Cancer Immunotherapy. Viruses 2023, 15, 1903. [Google Scholar] [CrossRef]
- Miki, T.; Namii, K.; Seko, K.; Kakehi, S.; Moro, G.; Mihara, H. Pattern enrichment analysis for phage selection of stapled peptide ligands. Chem Sci 2022, 13, 12634–12642. [Google Scholar] [CrossRef]
- Wang, L.; Xu, J.; Kong, Y.; Liang, R.; Li, W.; Li, J.; Lu, J.; Dimitrov, D.S.; Yu, F.; Wu, Y.; et al. Engineering a Novel Antibody-Peptide Bispecific Fusion Protein Against MERS-CoV. Antibodies (Basel) 2019, 8, 53. [Google Scholar] [CrossRef]
- Guerlavais, V.; Sawyer, T.K.; Carvajal, L.; Chang, Y.S.; Graves, B.; Ren, J.-G.; Sutton, D.; Olson, K.A.; Packman, K.; Darlak, K.; et al. Discovery of Sulanemadlin (ALRN-6924), the First Cell-Permeating, Stabilized α-Helical Peptide in Clinical Development. Journal of Medicinal Chemistry 2023, 66, 9401–9417. [Google Scholar] [CrossRef] [PubMed]
- DeKosky, B.J.; Lungu, O.I.; Park, D.; Johnson, E.L.; Charab, W.; Chrysostomou, C.; Kuroda, D.; Ellington, A.D.; Ippolito, G.C.; Gray, J.J.; et al. Large-scale sequence and structural comparisons of human naive and antigen-experienced antibody repertoires. Proc Natl Acad Sci U S A 2016, 113, E2636–2645. [Google Scholar] [CrossRef] [PubMed]
- Vaisman-Mentesh, A.; Wine, Y. Monitoring Phage Biopanning by Next-Generation Sequencing. Methods in Molecular Biology 2017, 463–473. [Google Scholar] [CrossRef]
- André, A.S.; Moutinho, I.; Dias, J.N.R.; Aires-da-Silva, F. In vivo Phage Display: A promising selection strategy for the improvement of antibody targeting and drug delivery properties. Frontiers in microbiology 2022, 13, 962124–962124. [Google Scholar] [CrossRef]
- Spoendlin, F.C.; Abanades, B.; Raybould, M.I.J.; Wong, W.K.; Georges, G.; Deane, C.M. Improved computational epitope profiling using structural models identifies a broader diversity of antibodies that bind to the same epitope. Front Mol Biosci 2023, 10, 1237621. [Google Scholar] [CrossRef]
- Simonetti, L.; Ivarsson, Y. Genetically Encoded Cyclic Peptide Phage Display Libraries. ACS Cent Sci 2020, 6, 336–338. [Google Scholar] [CrossRef]
- Górski, A.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Fortuna, W.; Letkiewicz, S.; Rogóż, P.; Jończyk-Matysiak, E.; Dąbrowska, K.; Majewska, J.; Borysowski, J. Phage Therapy: Combating Infections with Potential for Evolving from Merely a Treatment for Complications to Targeting Diseases. Frontiers in microbiology 2016, 7, 1515–1515. [Google Scholar] [CrossRef]
- Manrique, P.; Bolduc, B.; Walk, S.T.; van der Oost, J.; de Vos, W.M.; Young, M.J. Healthy human gut phageome. Proceedings of the National Academy of Sciences of the United States of America 2016, 113, 10400–10405. [Google Scholar] [CrossRef]
- Seed, K.D. Battling Phages: How Bacteria Defend against Viral Attack. PLoS Pathog 2015, 11, e1004847–e1004847. [Google Scholar] [CrossRef]
- Yang, Q.; Le, S.; Zhu, T.; Wu, N. Regulations of phage therapy across the world. Frontiers in microbiology 2023, 14, 1250848–1250848. [Google Scholar] [CrossRef]
- FDA Issues Guidance for Collection of Race and Ethnicity Data in Clinical Trials for FDA Regulated Products. PsycEXTRA Dataset 2003. [CrossRef]
- Safety-Related Drug Labeling Changes Approved by FDA. PsycEXTRA Dataset 1997. [CrossRef]
- Van der Schueren, B.; Vrijlandt, P.; Thomson, A.; Janssen, H.; Dunder, K. New guideline of the European Medicines Agency (EMA) on the clinical investigation of medicinal products in the treatment and prevention of diabetes mellitus. Diabetologia 2024, 67, 1159–1162. [Google Scholar] [CrossRef] [PubMed]
- Na. The European Medicines Agency (EMA) CHMP has published a position paper on medication errors. Reactions Weekly 2012, &NA; 5. [CrossRef]
- Verbeken, G.; Pirnay, J.-P.; Lavigne, R.; Jennes, S.; De Vos, D.; Casteels, M.; Huys, I. Call for a dedicated European legal framework for bacteriophage therapy. Archivum immunologiae et therapiae experimentalis 2014, 62, 117–129. [Google Scholar] [CrossRef]
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use ( ICH ). Wiley StatsRef: Statistics Reference Online 2014. [CrossRef]
- Abedon, S.T. Ecology and Evolutionary Biology of Hindering Phage Therapy: The Phage Tolerance vs. Phage Resistance of Bacterial Biofilms. Antibiotics (Basel) 2023, 12, 245. [Google Scholar] [CrossRef]
- Knezevic, P.; Hoyle, N.S.; Matsuzaki, S.; Gorski, A. Editorial: Advances in Phage Therapy: Present Challenges and Future Perspectives. Frontiers in microbiology 2021, 12, 701898–701898. [Google Scholar] [CrossRef]
- Sulakvelidze, A.; Kutter, E. Bacteriophage Therapy in Humans. Bacteriophages 2004. [Google Scholar] [CrossRef]
- Abedon, S. Bringing phage therapy soon to a clinic near you. The Lancet Infectious Diseases 2020, 20, 551. [Google Scholar] [CrossRef]
- Abedon, S.T. Phage-Antibiotic Combination Treatments: Antagonistic Impacts of Antibiotics on the Pharmacodynamics of Phage Therapy? Antibiotics (Basel) 2019, 8, 182. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, J.; Nakamura, T.; Nakamura, K.; Nishida, K.; Amano, Y.; Watanabe, Y.; Gondaira, S.; Usui, M.; Shimizu, M.; Miyanaga, K.; et al. Biological properties of Staphylococcus virus PhiSA012 for phage therapy. Sci Rep 2022, 12, 21297. [Google Scholar] [CrossRef] [PubMed]
- Semler, D.D.; Goudie, A.D.; Finlay, W.H.; Dennis, J.J. Aerosol phage therapy efficacy in Burkholderia cepacia complex respiratory infections. Antimicrob Agents Chemother 2014, 58, 4005–4013. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K. Phage therapy in the battle towards antibiotic resistance. In Proceedings of the International Conference on Modern Medicine and Global Health (ICMMGH 2023), 2023/09/07; 2023. [Google Scholar]
| Phage-Enabled Techniques and Their Applications in Modern Medicine and Biotechnology | Relevant Strategies and Methodologies | References |
|---|---|---|
| 1. Gene-Targeted Bacterial Killing |
|
[18,41,42,43,44] |
| 2. Delivery of Antimicrobial Genes |
|
[45,46,47,48] |
| 3. Phage-Mediated Antimicrobial Agent Delivery |
|
[49,50,51,52] |
| 4. Strict Lytic Cycle Maintenance |
|
[53] |
| 5. Inhibition of Biofilm Formation |
|
[54,55,56] |
| 6. Modification of Phage Host Range |
|
[57,58,59,60,61,62] |
| 7. Strategies to Overcome Phage Resistance |
|
[47,63] |
| 8. In Vivo Phage Stabilization |
|
[64,65,66,67,68] |
| 9. Enhancing Antibiotic Sensitivity |
|
[69,70,71,72] |
| 10. Endotoxin Shock Suppression |
|
[73,74] |
| 11. Phage-Based Vaccines |
|
[75,76,77,78,79,80,81] |
| 12. Phage in Gene Therapy Applications |
|
[82,83] |
| 13. Phage-Mediated Virus Suppression |
|
[84] |
| 14. Phage for Diagnostic Applications |
|
[85] |
| 15. Phage-Assisted Bone Regeneration |
|
[86,87] |
| 16. Phage-Assisted Skin Regeneration |
|
[88] |
| 17. Phage-Assisted Nerve Regeneration |
|
[89,90] |
| Status | Completed or terminated | On going or not yet recruiting | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phase | Pre-clinical or case study | Phase | Pre-clinical or case study | Phase | ||||||
| I | I/II | II | II/III | III | I/II | II | III | |||
| Staphylococcus aureus | 1 | 2 | 1 | 1 | 3 | 3 | 3 | |||
| Coagulase-negative Staphylococci | 1 | 3 | ||||||||
| Pseudomonas aeruginosa | 2 | 1 | 4 | 1 | 1 | 1 | 3 | 1 | ||
| Escherichia coli | 2 | 3 | 1 | 1 | 1 | 2 | 2 | 1 | 1 | |
| Enterococcus spp. | 1 | 1 | 1 | 2 | 1 | |||||
| Klebsiella | 1 | 1 | 1 | 1 | 1 | 2 | ||||
| Acinetobacter baumannii | 1 | 1 | 1 | 1 | ||||||
| Streptococcus | 1 | 1 | ||||||||
| Proteus | 1 | 1 | 1 | |||||||
| Non-tuberculosis Mycobacteria | 1 | 1 | ||||||||
| Achromobacter xylosoxidans | 1 | 1 | ||||||||
| Stenotrophomonas maltophilia | 1 | 1 | ||||||||
| Bacteroides fragilis | 1 | 1 | ||||||||
| Shigella | 1 | |||||||||
| *Note: The numbers in the table represent the number of research projects (clinical trials). In some cases, phage cocktails targeting different bacteria are included, with each counted as one project. Most of the research projects are currently ongoing, so many have not yet been published. The table was compiled by the authors using the ClinicalTrials.gov database from the National Library of Medicine (https://clinicaltrials.gov/), and there are no comprehensive references for the entire table. It provides an overview of the current status of clinical studies using bacteriophages. | ||||||||||
| Challenges in phage therapy | Conventional Approach | Synthetic Approach | |
|---|---|---|---|
| 1 | Narrow host range | The use of phage cocktail [120,121,122] | Genetic manipulation of receptor-binding protein [53,57] |
| 2 | Emergence of phage-resistant bacteria | Phage cocktail; combined therapy of antibiotic and phage [123] | Genetic manipulation of receptor-binding protein [124]; incorporation of small RNAs or CRISPR-Cas system to silence antibiotic resistance determinant [18,72] or delivery of genes encoding proteins to sensitize bacteria against antibiotics [125] |
| 3 | Necessitate identification of phage with therapeutic effect against patients’ isolates (personalized medicine) | Establish phage biobanks (isolating large phage collections) [126] | Engineering of phage tail fibers to alter host range [58,59,61] |
| 4 | Rapid clearance by reticuloendothelial system (RES) | Multiple phage dosing [127] | Phage capsid protein mutagenesis [64]; PEGylation of phage particles [66] |
| 5 | Phage pharmacokinetics (bioavailability through oral administration) | Pharmacological neutralization of gastric acid [128] | Encapsulation of phage in nanoparticles [129,130] |
| 6 | Limited accessibility to biofilm-producing bacteria | Use only phages with intrinsic biofilm degrading properties [131,132], or combined therapy using phage and biofilm-degrading enzymes [133] | Engineered phages expressing biofilm-degrading enzymes [54] |
| 7 | Difficulties in defining pharmacokinetics (eg. MIC) | Standardize routes and dosages of administration (required specified combinations of phage-host for each infection) [134] | Generation of non-proliferative anti-bacterial phage capsids [18] |
| 8 | Safety concern: risk of horizontal gene transfer | The use of phage-derived endolysin [135] | Development of well-characterized, non-propagating phages [136], introduction of antibacterial cargo using phagemids [137] or phage-inducible chromosomal islands (PICIs) [43] |
| 9 | Presence of potential hazardous genes in phage genome (toxin, virulence, antibiotic resistance genes, etc.) | Obligate virulent phage is preferred in therapy [123]; whole-genome analysis should be done in the first place | Custom-made phage can be generated easily using current techniques [53,57,138]; the use of self-destructive engineered phage (conjugation to gold nanorods) [52] |
| 10 | Low purity and potential toxin contamination in phage preparation | Purification by CsCl density gradient and ion exchange column [123] or affinity chromatography [139] | The use of cell-free system (cell-free-transcription-translation, TXTL) for phage production [138] |
| Study | Target bacteria | Phage (Dosages) | Antibiotic (Dosages) | References | |
|---|---|---|---|---|---|
| 1 | Racenis et al.,2023 | multidrug-resistant P. aeruginosa | Phages PNM and PT07 (Titer of 107 PFU/mL ) | Ceftazidime/Avibactam (2.5 g) and Amikacin (750 mg) | [142] |
| 2 | Kebriaei et al., 2023 | Methicillin-resistant S aureus (MRSA) strains and their daptomycin-nonsusceptible vancomycin-intermediate (DNS-VISA) | Phages Intesti13, Sb-1, and Romulus(107 PFU/well) | Daptomycin, vancomycin, and ceftaroline at 0.5× MBIC or 1× MBIC | [114] |
| 3 | Altamirano et al., 2022 | Acinetobacter baumannii AB900 | Phages øFG02 (range: 102-108 PFU/ml) | Ceftazidime (range: 1-512 mg/ml) | [144] |
| 4 | Canoet al., 2021 | K. pneumoniae complex KpJH46 | Phage KpJH46Φ2(6.3 × 1010 phages in 50 mL for a total of 40 doses) | Minocycline, 100 mg | [145] |
| 5 | Morales et al., 2020 | Staphylococcus aureus | Three Myoviridae bacteriophages AB-SA01 (109 PFU/ml) | lucloxacillin, Cefazolin, Vancomycin, Ciprofloxacin , Rifampicin | [146] |
| 6 | Jault et al., 2019 | Pseudomonas aeruginosa | cocktail of 12 natural lytic anti-P aeruginosa bacteriophages (PP1131; 1 × 106 PFU]/mL) | 1% sulfadiazine silver emulsion cream | [147] |
| 7 | Schooley et al., 2017 | MDR A. baumannii | Multiple phage cocktails (C2P24, AC4, C2P21, and C1P12) | Minocycline | [148] |
| Phage | Functional Peptide Display/Cargo | Tumor Type | Mode of Therapy | Preclinical Model | Therapy Outcome | References |
|---|---|---|---|---|---|---|
| M13 | WDC-2 phage displaying melanoma cell targeting peptide TRTKLPRLHLQS | Melanoma | Immunomodulatory | Subcutaneous B16-F10 tumor model in mice | Delayed tumor growth and increased survival | [201] |
| λ Phage | Display of human ASPH derived proteins | Hepatocellular carcinoma | Immunotherapy – Delivery of antigen for vaccine effect | Prophylactic vaccination schedule in BNL HCC subcutaneous model | Prophylactic and therapeutic immunization significantly delayed HCC growth and progression | [204] |
| Hybrid M13/AAV | RGD4C peptide CDCRGDCFC that binds to αvβ3 integrin cell surface receptor on Glioblastoma | Glioblastoma | Gene therapy - Grp78 expression | Intracranial implantation of U87 glioblastoma cells | Suppressed the growth of orthotopic glioblastoma | [213] |
| M13 phage | Fusobacterium nucleatum binding M13 phages | Colorectal cancer | Immunomodulatory | Orthotopic CT26 murine model | Precise scavenging of pro-tumor bacteria of Fusobacterium nucleatum, thereby blocking immunosuppressive myeloid-derived suppressor cells augmentation in the tumor microenvironment. | [51] |
| 2nd generation M13 vector | CDCRGDCFC (RGD4C) ligand that binds to αvβ3 integrin | Chondrosarcoma | Gene therapy –tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) expression | Subcutaneous implantation of SW1353-GFP-Luc cells | Decreased tumor size with nil side effects | [214] |
| TPA (transmorphic phage/AAV) | Tumour targeting ligand, CDCRGDCFC (RGD4C) | Hepatocellular carcinoma | Therapeutic gene cassette that expresses TRAIL | N/A | Selective and efficient delivery of the tmTRAIL gene to HCC cells that induced apoptotic death of HCC cells | [215] |
| Transmorphic phage/AAV, TPA | Double-cyclic tumour targeting ligand, RGD4C ligand | Medulloblastoma | Delivering transgene expressing the tumor necrosis factor-alpha (TNFα) | Subcutaneous Daoy medulloblastoma xenograft mice model | Selective tumor homing, targeted tumor expression of TNFα, apoptosis, and destruction of the tumor vasculature | [216] |
| M13/AAV | RGD4C ligand on the pIII minor coat protein for targeted therapyHistidine-rich endosomal escape peptide, H5WYG | Chondrosarcoma | Delivery of TNFα transgene | Subcutaneously established SW1353 xenograft in athymic mice | Complete elimination of tumor growth and eradication of the tumor size and tumor viability | [217] |
| M13 bacteriophage | Chemical cross-linking and biomineralization of palladium nanoparticles | Breast cancer | Delivery of palladium nanoparticle for photothermal therapy and NLG919, a nontoxic IDO1-selective inhibitor | Subcutaneous breast cancer model using 4T1 cells | Induced immunogenic death of tumor cells with down-regulated IDO1 expression | [218] |
| M13 | Fn-binding phages | Colon carcinoma | Immunomodulatory and reversing chemoresistance | Caecum implantation of CT26 cells in BALB/c mice | Modulated gut microbiota to augment chemotherapeutic effect | [219] |
| M13 | Peptide (SYPIPDT) that is able to bind the epidermal growth factor receptor (EGFR)Chemical conjugation of Rose Bengal (RB) photosensitizing molecules on the capsid surface | Epidermoid carcinoma | Photodynamic therapy | N/A | M13EGFR–RB derivatives generated intracellular reactive oxygen species activated by an ultralow intensity white light irradiation, thereby killing the cancer cells | [220] |
| T7 | Cancer homing peptide pep42 (CTVALPGGYVRVC) targeting the grp78 on cancer cells | Melanoma | Mammalian expression cassette of the cytokine granulocyte macrophage-colony stimulating factor (GM-CSF) | Subcutaneous B16F10 xenografts | Inhibited tumor growth by 72% compared to the untreated control. | [221] |
| M13 | Engineered to display the EC and TM domains of human HER2 (ECTM phages) or its splice variant Δ16HER2 | Breast carcinoma | Immunotherapy -Delivery of antigen for vaccine effect | Δ16HER2-expressing epithelial tumor cell lines mice | Anti-HER2 vaccination induced a significant anti-HER2 antibody response and controls the tumor growth. | [222] |
| M13 | Display of anti-CD40 DARPin into the gene of the pIII coat protein for CD40 targeting | Colon adenocarcinoma | Immunotherapy – In situ vaccines | Subcutaneous MC38 xenografts | Significant accumulation of the phages and activation of DCs at the tumor site, reversing the immunosuppressive tumor microenvironment | [223] |
| λ Phage | Tumor selectivity of the cargo, apoptin | Breast Carcinoma | Gene therapy | BT-474 cells subcutaneous xenograft | Implanted BT-474 human breast tumor was successfully responded to the systemic and local injection of untargeted recombinant λ NBPs | [224] |
| λ Phage | Display of displaying a HER2/neu derived peptide GP2 | Breast carcinoma | Immunotherapy – Delivery of antigen for vaccine effect | Subcutaneous TUBO cell implant | Robust CTL response against HER2/neu-positive tumor challenge in both prophylactic and therapeutic settings | [225] |
| T4-AAV | RGD peptide (CDCRGDCFC), a cell surface targeting ligand, when fused to the tip of Hoc fiber | HEK293T | Gene delivery, Protein Delivery & Genome editing | N/A | Delivered full-length dystrophin gene and performed genome editing, gene recombination, gene replacement, gene expression, and gene silencing. | [226] |
| T4 | Display of Catalase protein on phage heads Chemically coupled chlorin e6 (Ce6), a photosensitizer | Breast cancer | Photodynamic therapy | Subcutaneous 4T1 cancer cell model | Relieved tumor hypoxia and enabled Ce6 to produce ROS for effective tumor inhibition | [227] |
| T7 phage | Display of neoepitopes derived from mutated proteins of melanoma tumor cells | Melanoma | Immunotherapy - Delivery of neoepitopes for vaccine effect | Subcutaneous B16F10 xenografts | Rapid production of vaccines that can deliver mutated peptides and stimulate an appropriate B cell response | [228] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




