Submitted:
23 August 2024
Posted:
26 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
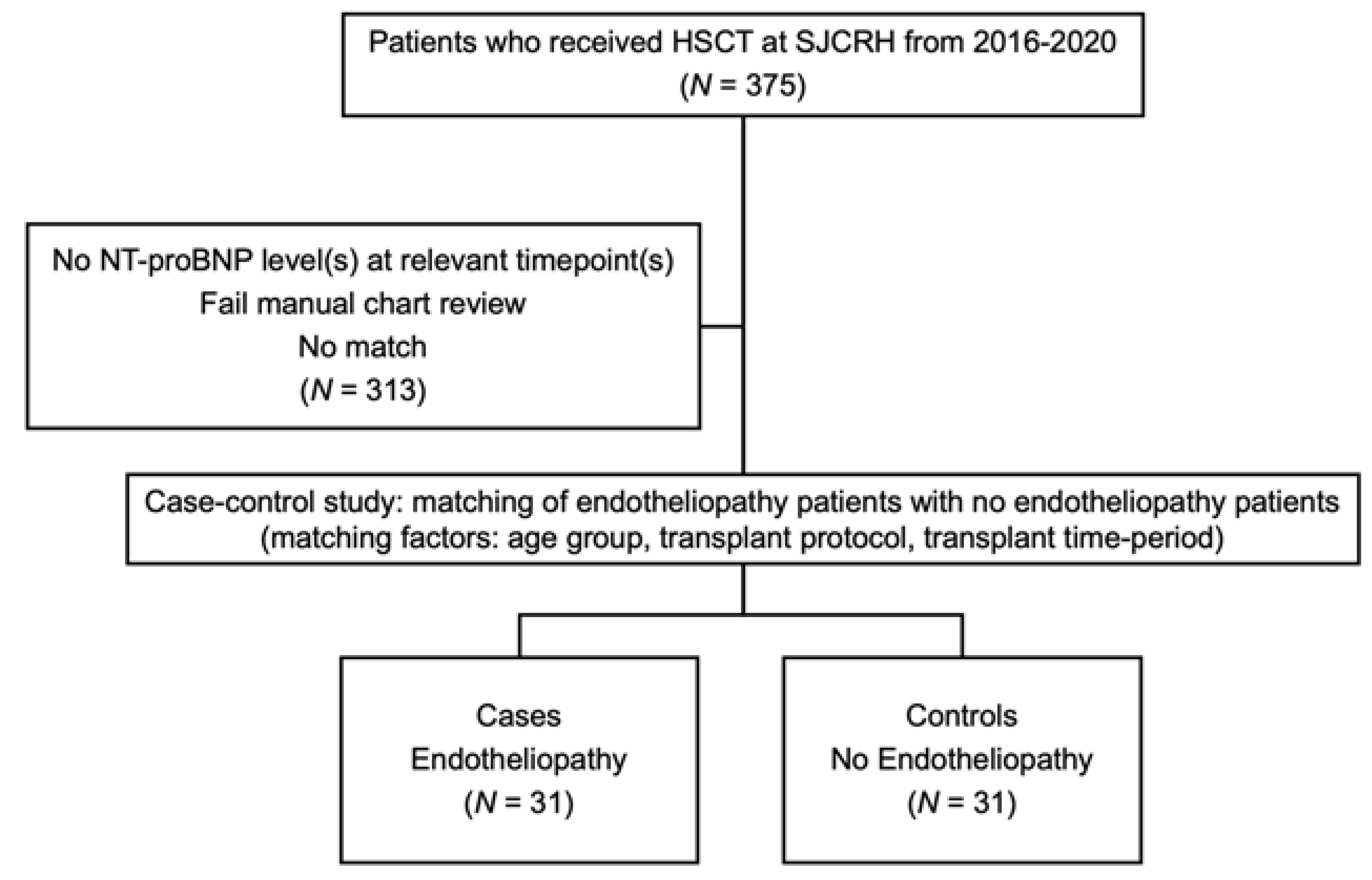
2. Materials and Methods
3. Results
3.1. Demographic and Baseline Clinical Characteristics
3.2. Endotheliopathy Data
3.3. NT-proBNP Levels
3.4. Conditional Logistic Regression
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, A.H.; Mahadeo, K.M. Perspective: A Framework to Screen Pediatric and Adolescent Hematopoietic Cellular Therapy Patients for Organ Dysfunction: Time for a Multi-Disciplinary and Longitudinal Approach. Front. Oncol. 2021, 11, 622630. [Google Scholar] [CrossRef] [PubMed]
- Carreras, E.; Diaz-Ricart, M. The Role of the Endothelium in the Short-Term Complications of Hematopoietic SCT. Bone Marrow Transplant. 2011, 46, 1495–1502. [Google Scholar] [CrossRef]
- Pagliuca, S.; Michonneau, D.; Sicre de Fontbrune, F.; Sutra del Galy, A.; Xhaard, A.; Robin, M.; Peffault de Latour, R.; Socie, G. Allogeneic Reactivity–Mediated Endothelial Cell Complications after HSCT: A Plea for Consensual Definitions. Blood Adv. 2019, 3, 2424–2435. [Google Scholar] [CrossRef]
- Cooke, K.R.; Jannin, A.; Ho, V. The Contribution of Endothelial Activation and Injury to End-Organ Toxicity Following Allogeneic Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2008, 14, 23–32. [Google Scholar] [CrossRef]
- Hildebrandt, G.C.; Chao, N. Endothelial Cell Function and Endothelial-related Disorders Following Haematopoietic Cell Transplantation. Br. J. Haematol. 2020, 190, 508–519. [Google Scholar] [CrossRef]
- Lia, G.; Giaccone, L.; Leone, S.; Bruno, B. Biomarkers for Early Complications of Endothelial Origin After Allogeneic Hematopoietic Stem Cell Transplantation: Do They Have a Potential Clinical Role? Front. Immunol. 2021, 12, 641427. [Google Scholar] [CrossRef] [PubMed]
- Zinter, M.S.; Dvorak, C.C.; Spicer, A.; Cowan, M.J.; Sapru, A. New Insights Into Multicenter PICU Mortality Among Pediatric Hematopoietic Stem Cell Transplant Patients*: Crit. Care Med. 2015, 43, 1986–1994. [Google Scholar] [CrossRef]
- Zaidman, I.; Mohamad, H.; Shalom, L.; Ben Arush, M.; Even-Or, E.; Averbuch, D.; Zilkha, A.; Braun, J.; Mandel, A.; Kleid, D.; et al. Survival of Pediatric Patients Requiring Admission in the Intensive Care Unit Post Hematopoietic Stem Cell Transplantation: Prognostic Factors Associated with Mortality. Pediatr. Blood Cancer 2022, 69, e29549. [Google Scholar] [CrossRef] [PubMed]
- Rubattu, S.; Sciarretta, S.; Valenti, V.; Stanzione, R.; Volpe, M. Natriuretic Peptides: An Update on Bioactivity, Potential Therapeutic Use, and Implication in Cardiovascular Diseases. Am. J. Hypertens. 2008, 21, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.; Lim, W.-C.; Sy, R.; Cunningham, I.; Trotman, J.; Kritharides, L. Subacute Cardiac Toxicity Following Autologous Haematopoietic Stem Cell Transplantation in Patients with Normal Cardiac Function. Heart 2008, 94, 911–918. [Google Scholar] [CrossRef]
- Masuko, M.; Ito, M.; Kurasaki, T.; Yano, T.; Takizawa, J.; Toba, K.; Aoki, S.; Fuse, I.; Kodama, M.; Furukawa, T.; et al. Plasma Brain Natriuretic Peptide during Myeloablative Stem Cell Transplantation. Intern. Med. 2007, 46, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Roziakova, L.; Bojtarova, E.; Mistrik, M.; Dubrava, J.; Gergel, J.; Lenkova, N.; Mladosievicova, B. Serial Measurements of Cardiac Biomarkers in Patients after Allogeneic Hematopoietic Stem Cell Transplantation. J. Exp. Clin. Cancer Res. 2012, 31, 13. [Google Scholar] [CrossRef]
- Horacek, J.M.; Tichy, M.; Pudil, R.; Jebavy, L.; Zak, P.; Ulrychova, M.; Slovacek, L.; Maly, J. Multimarker Approach to Evaluation of Cardiac Toxicity during Preparative Regimen and Hematopoietic Cell Transplantation.
- Snowden, J.; Hill, G.; Hunt, P.; Carnoutsos, S.; Spearing, R.; Espiner, E.; Hart, D. Assessment of Cardiotoxicity during Haemopoietic Stem Cell Transplantation with Plasma Brain Natriuretic Peptide. Bone Marrow Transplant. 2000, 26, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Roziakova, L.; Bojtarova, E.; Mistrik, M.; Krajcovicova, I.; Mladosievicova, B. Abnormal Cardiomarkers in Leukemia Patients Treated with Allogeneic Hematopoietic Stem Cell Transplantation. Bratisl. Med. J. 2012, 113, 159–162. [Google Scholar] [CrossRef]
- Se, Z.; Zhou, H.; Li, H.; Sun, J.; Zhan, Q.; Zeng, Q.; Liu, Q.; Xu, D. Clinical Characteristics of Patients with Different N-Terminal Probrain Natriuretic Peptide Levels after Hematopoietic Stem Cell Transplantation. Dis. Markers 2020, 2020, 1–7. [Google Scholar] [CrossRef]
- Roziakova, L.; Mistrik, M.; Batorova, A.; Kruzliak, P.; Bojtarova, E.; Dubrava, J.; Gergel, J.; Mladosievicova, B. Can We Predict Clinical Cardiotoxicity with Cardiac Biomarkers in Patients After Haematopoietic Stem Cell Transplantation? Cardiovasc. Toxicol. 2015, 15, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Niwa, N.; Watanabe, E.; Hamaguchi, M.; Kodera, Y.; Miyazaki, H.; Kodama, I.; Ohono, M. Early and Late Elevation of Plasma Atrial and Brain Natriuretic Peptides in Patients after Bone Marrow Transplantation. Ann. Hematol. 2001, 80, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Rotz, S.J.; Ryan, T.D.; Hlavaty, J.; George, S.A.; El-Bietar, J.; Dandoy, C.E. Cardiotoxicity and Cardiomyopathy in Children and Young Adult Survivors of Hematopoietic Stem Cell Transplant. Pediatr. Blood Cancer 2017, 64, e26600. [Google Scholar] [CrossRef]
- Rotz, S.J.; Dandoy, C.E.; Taylor, M.D.; Jodele, S.; Jefferies, J.L.; Lane, A.; El-Bietar, J.A.; Powell, A.W.; Davies, S.M.; Ryan, T.D. Long-Term Systolic Function in Children and Young Adults after Hematopoietic Stem Cell Transplant. Bone Marrow Transplant. 2017, 52, 1443–1447. [Google Scholar] [CrossRef]
- Horacek, J.M.; Pudil, R.; Tichy, M.; Jebavy, L.; Zak, P.; Slovacek, L.; Maly, J. Biochemical Markers and Assessment of Cardiotoxicity During Preparative Regimen and Hematopoietic Cell Transplantation in Acute Leukemia. Exp. Oncol. 2007. [Google Scholar]
- Zaucha-Prażmo, A.; Sadurska, E.; Drabko, K.; Kowalczyk, J.R. Can We Find a Good Biochemical Marker of Early Cardiotoxicity in Children Treated with Haematopoietic Stem Cell Transplantation? Współczesna Onkol. 2016, 3, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Huo, W.; Zhao, H.; Lv, J.; Lv, S.; An, Y. Risk Factors and Predictive Model for Mortality in Patients Undergoing Allogeneic Hematopoietic Stem Cell Transplantation Admitted to the Intensive Care Unit. Exp. Ther. Med. 2024, 27, 168. [Google Scholar] [CrossRef] [PubMed]
- McArthur, J.; Fitzgerald, J.; Bajwa, R.; Margossian, S.; Yates, A.; Duncan, C.; Talano, J.; Spear, D.; Luther, K.; Tamburro, R. 763: Brain Natriuretic Peptide (Bnp) Levels As A Predictor Of Need For Critcal Care Resources In Pediatric Hematopoietic Stem Cell Transplant (HSCT) Patients. Crit. Care Med. 2012, 40, 1–328. [Google Scholar] [CrossRef]
- Kim, D.H.; Ha, E.J.; Park, S.J.; Koh, K.-N.; Kim, H.; Im, H.J.; Jhang, W.K. Prognostic Factors of Pediatric Hematopoietic Stem Cell Transplantation Recipients Admitted to the Pediatric Intensive Care Unit. Acute Crit. Care 2021, 36, 380–387. [Google Scholar] [CrossRef]
- Fu, S.; Ping, P.; Wang, F.; Luo, L. Synthesis, Secretion, Function, Metabolism and Application of Natriuretic Peptides in Heart Failure. J. Biol. Eng. 2018, 12, 2. [Google Scholar] [CrossRef]
- Ma, K.K.; Ogawa, T.; De Bold, A.J. Selective Upregulation of Cardiac Brain Natriuretic Peptide at the Transcriptional and Translational Levels by Pro-Inflammatory Cytokines and by Conditioned Medium Derived from Mixed Lymphocyte Reactions via P38 MAP Kinase. J. Mol. Cell. Cardiol. 2004, 36, 505–513. [Google Scholar] [CrossRef]
- Kuhn, M. Endothelial Actions of Atrial and B-type Natriuretic Peptides. Br. J. Pharmacol. 2012, 166, 522–531. [Google Scholar] [CrossRef]
- Hall, C. NT-ProBNP: The Mechanism Behind the Marker. J. Card. Fail. 2005, 11, S81–S83. [Google Scholar] [CrossRef]
- Kataoka, K.; Nannya, Y.; Iwata, H.; Seo, S.; Kumano, K.; Takahashi, T.; Nagai, R.; Kurokawa, M. Plasma Brain Natriuretic Peptide Is Associated with Hepatic Veno-Occlusive Disease and Early Mortality after Allogeneic Hematopoietic Stem Cell Transplantation. Bone Marrow Transplant. 2010, 45, 1631–1637. [Google Scholar] [CrossRef]
- Corbacioglu, S.; Carreras, E.; Ansari, M.; Balduzzi, A.; Cesaro, S.; Dalle, J.-H.; Dignan, F.; Gibson, B.; Guengoer, T.; Gruhn, B.; et al. Diagnosis and Severity Criteria for Sinusoidal Obstruction Syndrome/Veno-Occlusive Disease in Pediatric Patients: A New Classification from the European Society for Blood and Marrow Transplantation. Bone Marrow Transplant. 2018, 53, 138–145. [Google Scholar] [CrossRef]
- Jodele, S.; Davies, S.M.; Lane, A.; Khoury, J.; Dandoy, C.; Goebel, J.; Myers, K.; Grimley, M.; Bleesing, J.; El-Bietar, J.; et al. Diagnostic and Risk Criteria for HSCT-Associated Thrombotic Microangiopathy: A Study in Children and Young Adults. Blood 2014, 124, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Navanandan, N.; Searns, J.; Ambroggio, L. Method/Ology of Phases of Biomarker Discovery. Hosp. Pediatr. 2023, 13, e181–e185. [Google Scholar] [CrossRef]
| Characteristic | Cases of Endotheliopathy N (%) |
Controls N (%) |
Total N (%) |
p-value (Pearson Exact Chi-Square) |
|---|---|---|---|---|
| Total patients in cohort | 31 (100) | 31 (100) | 62 (100) | N/A |
| Age Group <2 years 2-10 years >10 years |
3 (10) 22 (71) 6 (19) |
3 (10) 22 (71) 6 (19) |
6 (10) 44 (71) 12 (19) |
1.00 |
| Sex Female Male |
15 (48) 16 (52) |
11 (35) 20 (65) |
26 (42) 36 (58) |
0.44 |
| Transplant Type Autologous Haploidentical Matched sibling donor Matched unrelated donor |
10 (32) 12 (39) 3 (10) 6 (19) |
10 (32) 12 (39) 3 (10) 6 (19) |
20 (32) 24 (39) 6 (10) 12 (19) |
1.00 |
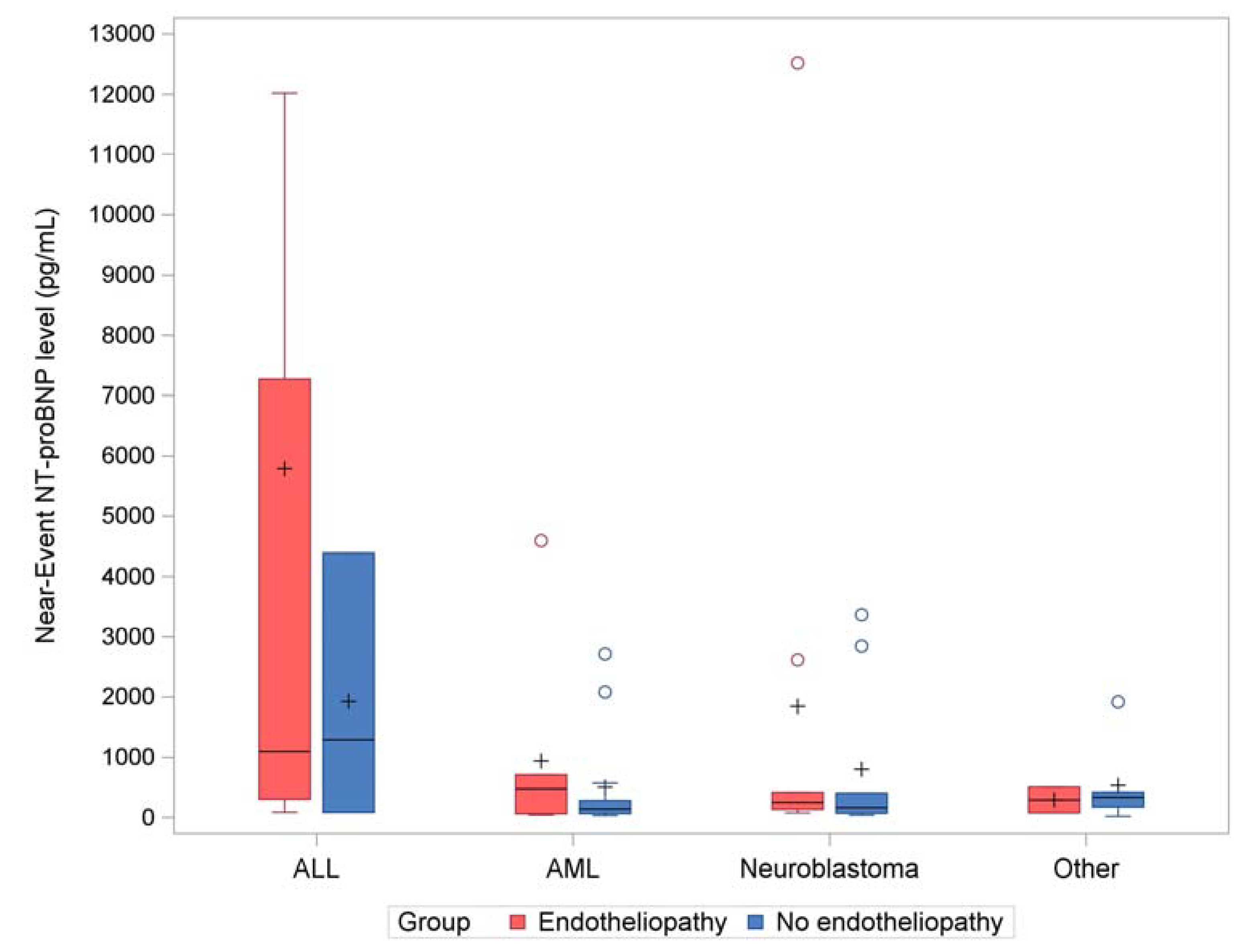
| Transplant Indication ALL AML Neuroblastoma Other Congenital neutropenia/ MDS derived leukemia Familial HLH Germ cell tumor MDS Non-Hodgkin Lymphoma Other acute leukemia Sickle cell disease |
13 (42) 7 (23) 9 (29) 2 (6) 0 (0) 0 (0) 1 (3) 0 (0) 0 (0) 0 (0) 1 (3) |
3 (10) 13 (42) 9 (29) 6 (19) 1 (3) 1 (3) 1 (3) 1 (3) 1 (3) 1 (3) 0 (0) |
16 (26) 20 (32) 18 (29) 8 (13) 1 (2) 1 (2) 2 (3) 1 (2) 1 (2) 1 (2) 1 (2) |
0.02* |
| Prior Transplant(s) 0 ≥1 |
19 (61) 12 (39) |
24 (77) 7 (23) |
43 (69) 19 (31) |
0.27 |
| Preparative Regimen Myeloablative Non-Myeloablative Reduced Intensity |
18 (58) 0 (0) 13 (42) |
18 (58) 1 (3) 12 (39) |
36 (58) 1 (2) 25 (40) |
1.00 |
| Baseline eGFR (mL/min/1.73m2) >60 30-60 <30 |
30 (97) 1 (3) 0 (0) |
30 (97) 0 (0) 1 (3) |
60 (97) 1 (2) 1 (2) |
1.00 |
| Baseline presence of pHTN by echocardiogram No |
31 (100) |
31 (100) |
62 (100) |
N/A |
| Baseline EF by Echocardiogram ≥50% |
31 (100) |
31 (100) |
62 (100) |
N/A |
| Characteristic | Cases of Endotheliopathy | Controls | Total | p-value* |
|---|---|---|---|---|
| Endotheliopathy Type, N (%) | ||||
| DAH | 1 (3) | 0 (0) | 1 (2) | <0.0001 |
| SOS | 22 (71) | 0 (0) | 22 (35) | |
| TMA | 8 (26) | 0 (0) | 8 (13) | |
| Days following HSCT of endotheliopathy diagnosis, median (range) | 15 (6, 85) | N/A | N/A | N/A |
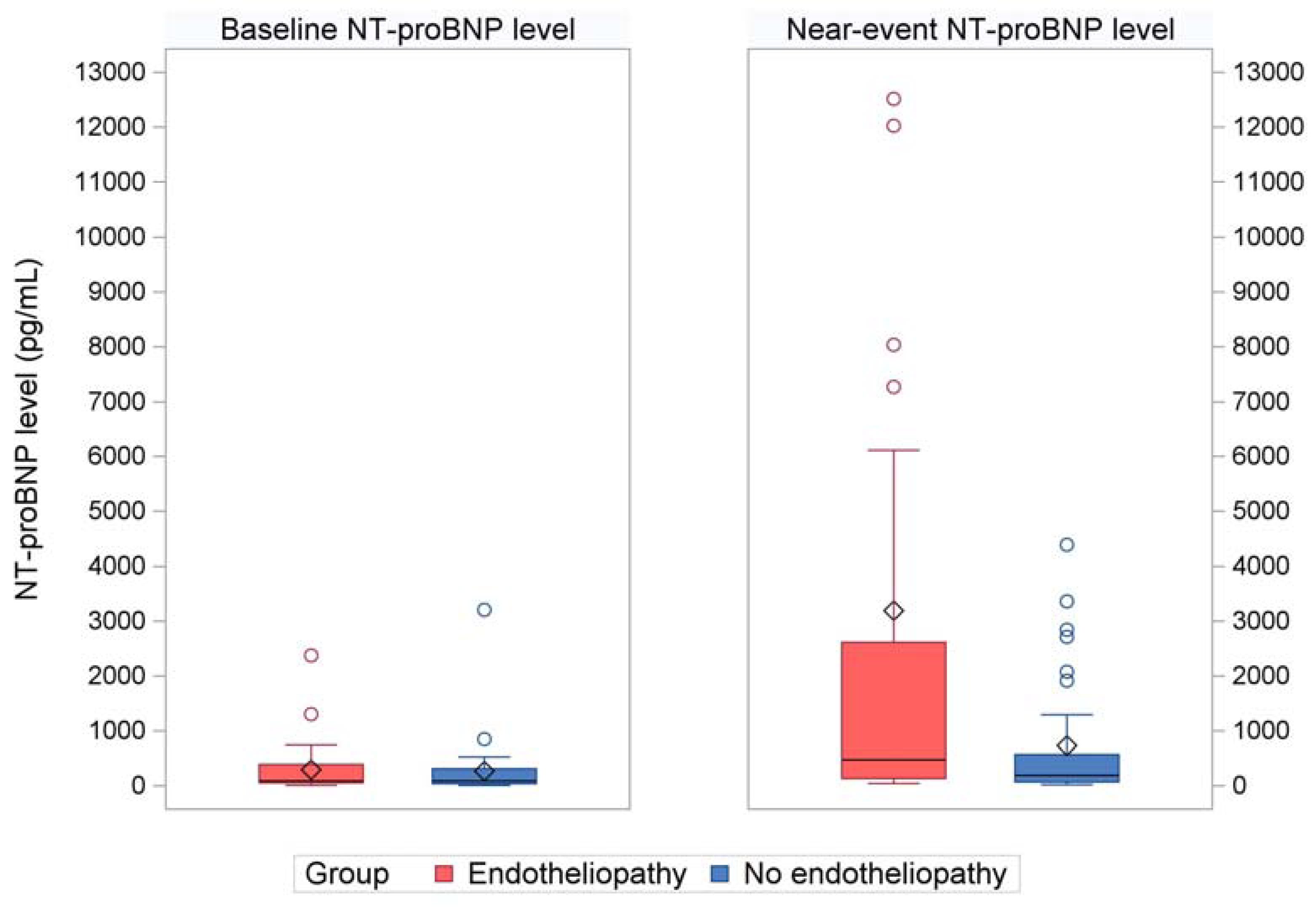
| Near-event NT-proBNP level in pg/mL Mean (standard deviation) Median (range) |
3190.2 (7151.8) 473 (40, 36875) |
729.7 (1158.4) 187 (18, 4385) |
1959.9 (5230) 262.5 (18, 36875) |
0.03 |
| Baseline NT-proBNP level in pg/mL Mean (standard deviation) Median (range) |
292.1 (477.3) 86 (6, 2373) |
267.1 (577.8) 86 (5, 3205) |
279.6 (525.7) 86 (5, 3205) |
0.51 |
| Days following HSCT of near-event NT-proBNP level collected Median (range) | 13 (5, 84) | 12 (4, 76) | 12.5 (4,84) | 0.25 |
| Near-event EF in percent by echocardiogram Median (range) |
70 (45, 80) | 70 (50, 75) | 70 (45, 80) | 0.57 |
| Near-event presence of pHTN by echocardiogram, N (%) No Yes |
29 (94) 2 (6) |
30 (97) 1 (3) |
59 (95) 3 (5) |
1.00 |
| Variable | Unit | Odds Ratio* (95% CI) | p-value | |
|---|---|---|---|---|
| Primary Model | Baseline NT-proBNP level | 1000 pg/mL | 1.08 (0.26-4.51) | 0.91 |
| AIC = 58.25 | Baseline EF | 10% | 0.31 (0.07-1.39) | 0.12 |
| Near-event NT-proBNP level | 1000 pg/mL | 1.32 (0.94-1.85) | 0.11 | |
| Near-event EF | 10% | 2.51 (0.90-7.06) | 0.08 | |
| Prior transplant(s) | 1 Transplant | 2.83 (0.54-14.77) | 0.22 | |
| Secondary Model | Baseline NT-proBNP level | 1000 pg/mL | 0.44 (0.07-2.76) | 0.38 |
| AIC = 52.57 | Baseline EF | 10% | 0.34 (0.06-1.86) | 0.21 |
| Near-event NT-proBNP level | 1000 pg/mL | 1.09 (0.83-1.43) | 0.54 | |
| Near-event EF | 10% | 2.32 (0.65-8.32) | 0.20 | |
| Prior transplant(s) | 1 Transplant | 6.36 (0.95-42.48) | 0.06 | |
| Age difference | 1 year | 0.75 (0.52-1.07) | 0.11 | |
| Transplant indication of ALL vs. all other indications | N/A | 20.16 (1.90-213.56) | 0.01 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




