Submitted:
20 August 2024
Posted:
22 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
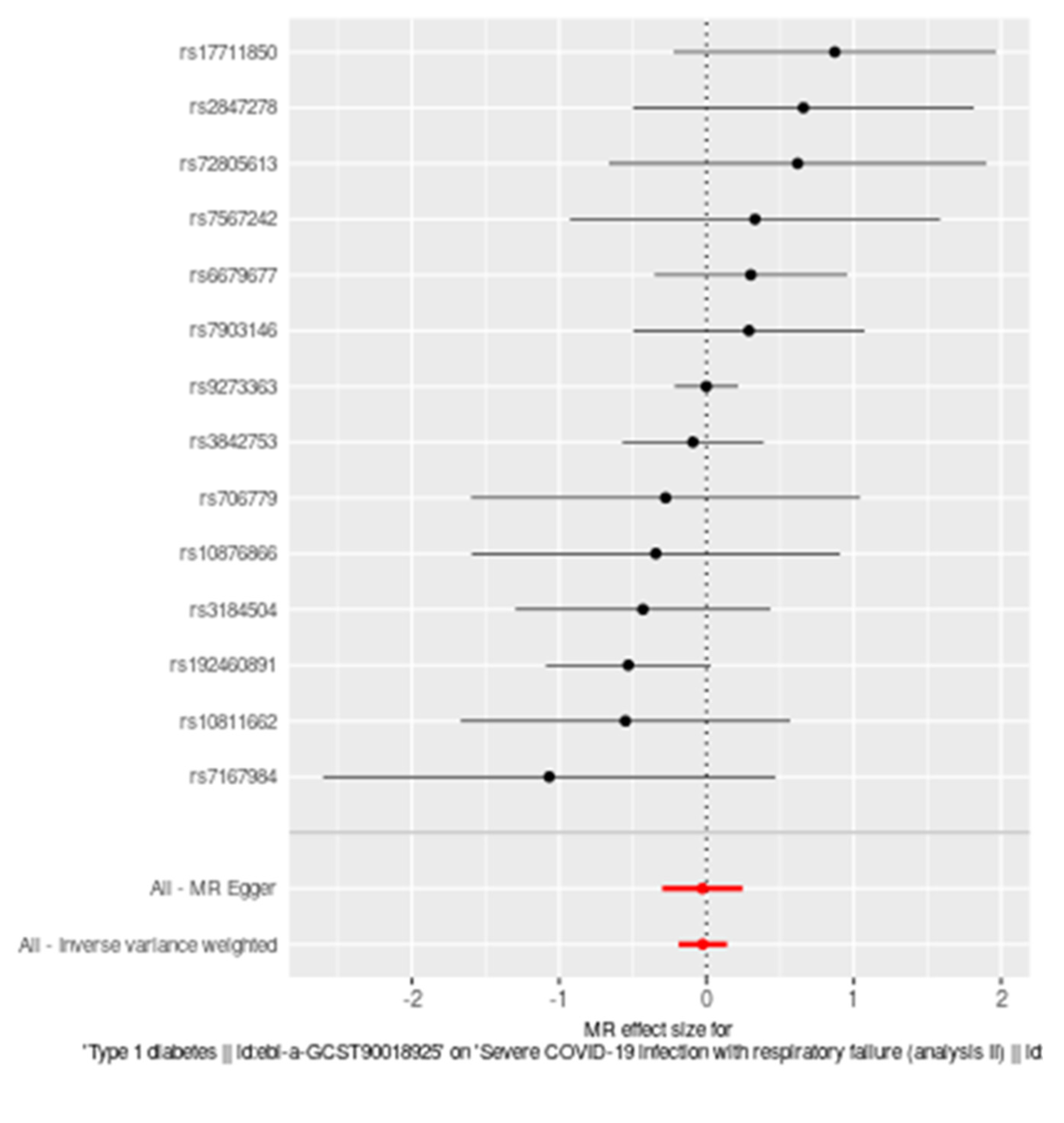
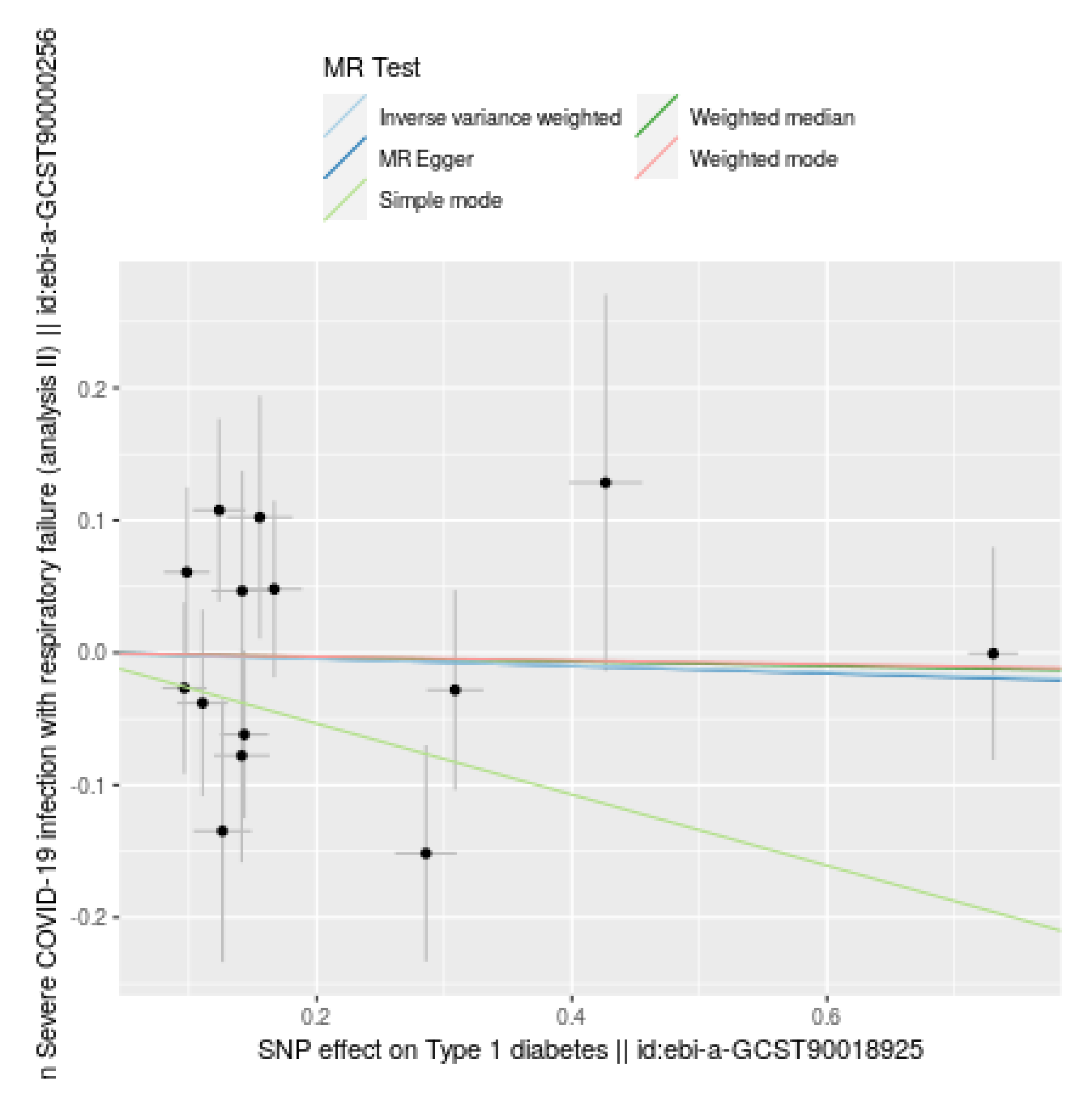
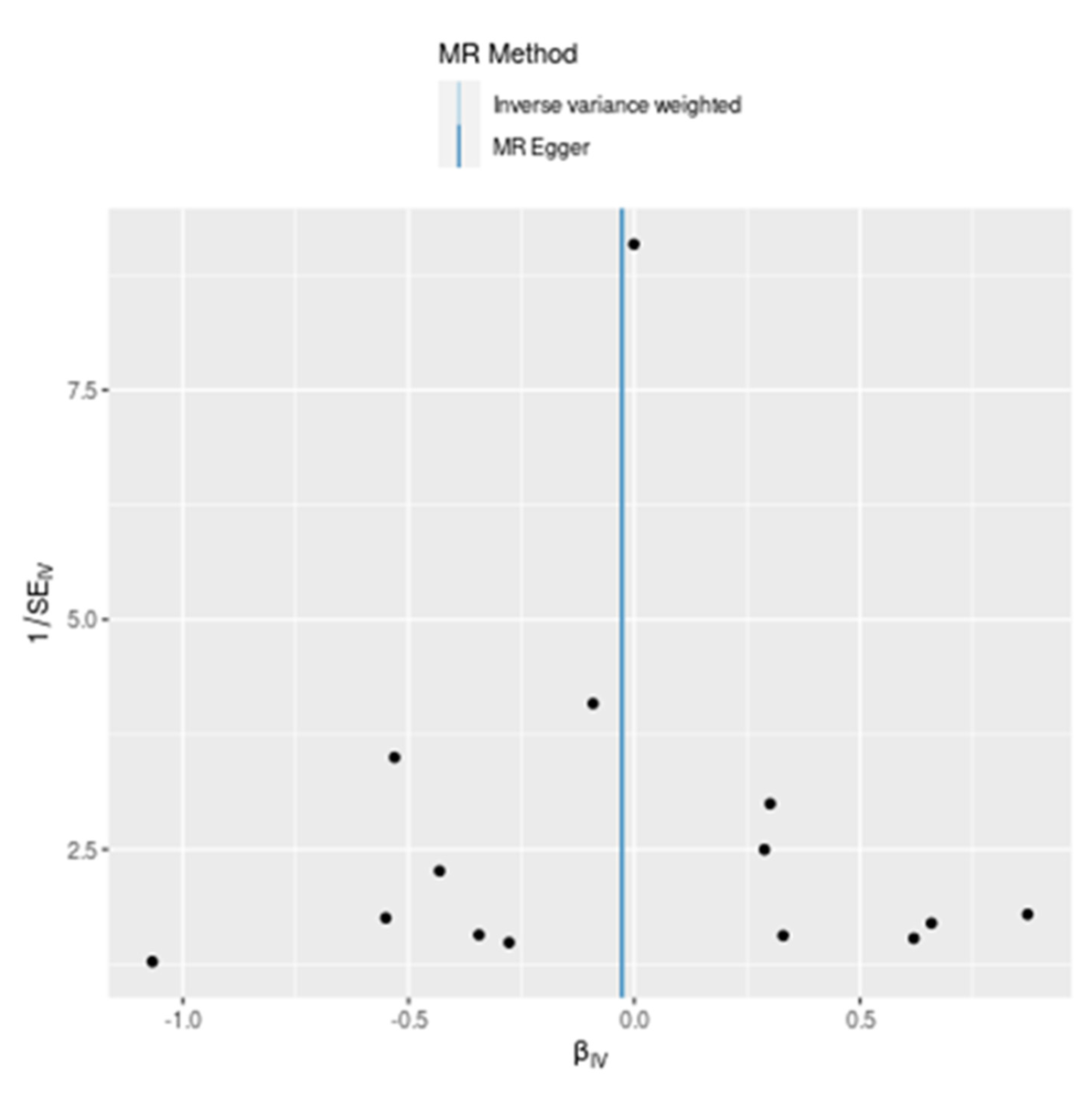
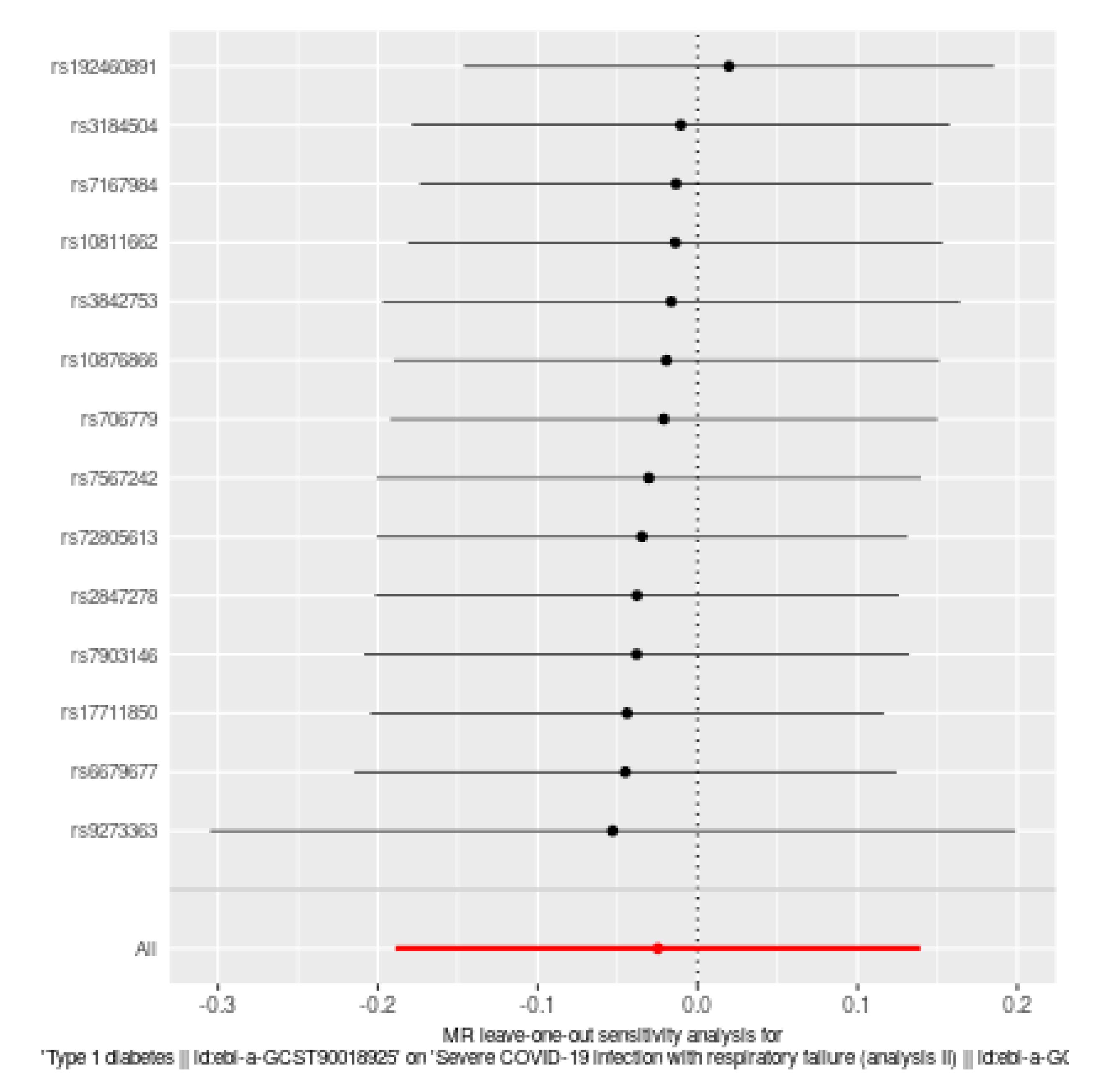
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Das S, Anu KR, Birangal SR, Nikam AN, Pandey A, Mutalik S, Joseph A. Role of comorbidities like diabetes on severe acute respiratory syndrome coronavirus-2: A review. Life Sci. 2020, 258, 118202. [Google Scholar] [CrossRef] [PubMed]
- Pal R, Yadav U, Grover S, Saboo B, Verma A, Bhadada SK. Knowledge, attitudes and practices towards COVID-19 among young adults with Type 1 Diabetes Mellitus amid the nationwide lockdown in India: A cross-sectional survey. Diabetes research and clinical practice. 2020, 166, 108344. [Google Scholar] [CrossRef]
- Nassar M, Nso N, Baraka B, Alfishawy M, Mohamed M, Nyabera A, Sachmechi I. The association between COVID-19 and type 1 diabetes mellitus: A systematic review. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2021, 15, 447–454. [Google Scholar]
- Mota M, Stefan AG. Covid-19 and Diabetes–A Bidirectional Relationship? Romanian Journal of Diabetes Nutrition and Metabolic Diseases. 2020, 27, 77–79. [Google Scholar]
- Alkundi A, Mahmoud I, Musa A, Naveed S, Alshawwaf M. Clinical characteristics and outcomes of COVID-19 hospitalized patients with diabetes in the United Kingdom: A retrospective single centre study. Diabetes research and clinical practice. 2020, 165, 108263. [Google Scholar] [CrossRef]
- Al Hayek AA, Robert AA, Alotaibi ZK, Al Dawish M. Clinical characteristics of hospitalized and home isolated COVID-19 patients with type 1 diabetes. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2020, 14, 1841–1845. [Google Scholar]
- Rysz S, Jonsson Fagerlund M, Rimes-Stigare C, Larsson E, Campoccia Jalde F, Mårtensson J. Chronic dysglycemia and risk of SARS-CoV-2 associated respiratory failure in hospitalized patients. Acta Anaesthesiologica Scandinavica. 2022, 66, 48–55. [Google Scholar] [CrossRef]
- Wargny M, Gourdy P, Ludwig L, Seret-Bégué D, Bourron O, Darmon P, Amadou C, Pichelin M, Potier L, Thivolet C, Gautier JF. Type 1 diabetes in people hospitalized for COVID-19: new insights from the CORONADO study. Diabetes Care. 2020, 43, e174. [Google Scholar] [CrossRef]
- Barron E, Bakhai C, Kar P, Weaver A, Bradley D, Ismail H, Knighton P, Holman N, Khunti K, Sattar N, Wareham NJ. Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: a whole-population study. The lancet Diabetes & endocrinology. 2020, 8, 813–822. [Google Scholar]
- Mazucanti CH, Egan JM. SARS-CoV-2 disease severity and diabetes: why the connection and what is to be done? Immunity & Ageing. 2020, 17, 21. [Google Scholar]
- Gardner G, Fraker CA. Natural killer cells as key mediators in type I diabetes immunopathology. Frontiers in immunology. 2021, 12, 722979. [Google Scholar] [CrossRef]
- Vargas-Rodriguez JR, Garza-Veloz I, Flores-Morales V, Badillo-Almaraz JI, Rocha-Pizana MR, Valdes-Aguayo JJ, Martinez-Fierro ML. Hyperglycemia and angiotensin-converting enzyme 2 in pulmonary function in the context of SARS-CoV-2 infection. Frontiers in Medicine. 2022, 8, 758414. [Google Scholar] [CrossRef]
- Kountouri A, Korakas E, Ikonomidis I, Raptis A, Tentolouris N, Dimitriadis G, Lambadiari V. Type 1 diabetes mellitus in the SARS-CoV-2 pandemic: oxidative stress as a major pathophysiological mechanism linked to adverse clinical outcomes. Antioxidants. 2021, 10, 752. [Google Scholar] [CrossRef] [PubMed]
- Kashfi K, Anbardar N, Asadipooya A, Asadipooya K. Type 1 Diabetes and COVID-19: A Literature Review and Possible Management. International Journal of Endocrinology and Metabolism.
- Nouri-Keshtkar M, Taghizadeh S, Farhadi A, Ezaddoustdar A, Vesali S, Hosseini R, Totonchi M, Kouhkan A, Chen C, Zhang JS, Bellusci S. Potential impact of diabetes and obesity on alveolar type 2 (AT2)-lipofibroblast (LIF) interactions after COVID-19 infection. Frontiers in Cell and Developmental Biology. 2021, 9, 676150. [Google Scholar]
- Zhang JY, Whalley JP, Knight JC, Wicker LS, Todd JA, Ferreira RC. SARS-CoV-2 infection induces a long-lived pro-inflammatory transcriptional profile. Genome Medicine. 2023, 15, 1–2. [Google Scholar]
- Chahal S, Raj RG, Kumar R. Risk of Type 1 Diabetes Mellitus in SARS CoV-2 Patients. Current Diabetes Reviews.
- Taylor JP, Hubert MT. The role of NADPH oxidases in infectious and inflammatory diseases. Redox biology. 2021, 48, 102159. [Google Scholar]
- Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? The lancet respiratory medicine. 2020, 8, e21. [Google Scholar] [CrossRef]
- Tonon F, Candido R, Toffoli B, Tommasi E, Cortello T, Fabris B, Bernardi S. Type 1 diabetes is associated with significant changes of ACE and ACE2 expression in peripheral blood mononuclear cells. Nutrition, Metabolism and Cardiovascular Diseases. 2022, 32, 1275–1282. [Google Scholar] [CrossRef]
- Hartmann-Boyce J, Rees K, Perring JC, Kerneis SA, Morris EM, Goyder C, Otunla AA, James OE, Syam NR, Seidu S, Khunti K. Risks of and from SARS-CoV-2 infection and COVID-19 in people with diabetes: a systematic review of reviews. Diabetes Care. 2021, 44, 2790–2811. [Google Scholar] [CrossRef]
- Gregory JM, Slaughter JC, Duffus SH, Smith TJ, LeStourgeon LM, Jaser SS, McCoy AB, Luther JM, Giovannetti ER, Boeder S, Pettus JH. COVID-19 severity is tripled in the diabetes community: a prospective analysis of the pandemic’s impact in type 1 and type 2 diabetes. Diabetes care. 2021, 44, 526–532. [Google Scholar] [CrossRef]
- Katikireddi SV, Green MJ, Taylor AE, Davey Smith G, Munafò MR. Assessing causal relationships using genetic proxies for exposures: an introduction to Mendelian randomization. Addiction. 2018, 113, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Davey Smith G, Hemani G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Human molecular genetics. 2014, 23, R89–R98. [Google Scholar] [CrossRef] [PubMed]
- Sekula P, Fabiola Del Greco M, Pattaro C, Köttgen A. Mendelian randomization as an approach to assess causality using observational data. Journal of the American Society of Nephrology. 2016, 27, 3253–3265. [Google Scholar] [CrossRef]
- Nitsch D, Molokhia M, Smeeth L, DeStavola BL, Whittaker JC, Leon DA. Limits to causal inference based on Mendelian randomization: a comparison with randomized controlled trials. American journal of epidemiology. 2006, 163, 397–403. [Google Scholar] [CrossRef]
- Ference BA, Holmes MV, Smith GD. Using Mendelian randomization to improve the design of randomized trials. Cold Spring Harbor perspectives in medicine. 2021, 11, a040980. [Google Scholar] [CrossRef] [PubMed]
- Li S, Yuan S, Schooling CM, Larsson SC. A Mendelian randomization study of genetic predisposition to autoimmune diseases and COVID-19. Scientific Reports. 2022, 12, 17703. [Google Scholar] [CrossRef]
- Boddu SK, Aurangabadkar G, Kuchay MS. New onset diabetes, type 1 diabetes and COVID-19. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2020, 14, 2211–2217. [Google Scholar]
- Badraoui R, Alrashedi MM, El-May MV, Bardakci F. Acute respiratory distress syndrome: a life-threatening associated complication of SARS-CoV-2 infection inducing COVID-19. Journal of Biomolecular Structure and Dynamics. 2021, 39, 6842–6851. [Google Scholar] [CrossRef]
- Yonekawa A, Shimono N. Clinical significance of COVID-19 and diabetes: in the pandemic situation of SARS-CoV-2 variants including Omicron (B. 1.1. 529). Biology. 2022, 11, 400. [Google Scholar]
- Trieu C, Sunil B, Ashraf AP, Cooper J, Yarbrough A, Pinninti S, Boppana S. SARS-CoV-2 infection in hospitalized children with type 1 and type 2 diabetes. Journal of clinical & translational endocrinology. 2021, 26, 100271. [Google Scholar]
- Roy S, Demmer RT. Impaired glucose regulation, SARS-CoV-2 infections and adverse COVID-19 outcomes. Translational Research. 2022, 241, 52–69. [Google Scholar] [CrossRef] [PubMed]
- Guo W, Li M, Dong Y, Zhou H, Zhang Z, Tian C, Qin R, Wang H, Shen Y, Du K, Zhao L. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes/metabolism research and reviews. 2020, 36, e3319. [Google Scholar] [CrossRef] [PubMed]
- Wu R, Mumtaz M, Maxwell AJ, Isaacs SR, Laiho JE, Rawlinson WD, Hyöty H, Craig ME, Kim KW. Respiratory infections and type 1 diabetes: potential roles in pathogenesis. Reviews in Medical Virology. 2023, 33, e2429. [Google Scholar] [CrossRef]
- Lampasona V, Secchi M, Scavini M, Bazzigaluppi E, Brigatti C, Marzinotto I, Davalli A, Caretto A, Laurenzi A, Martinenghi S, Molinari C. Antibody response to multiple antigens of SARS-CoV-2 in patients with diabetes: an observational cohort study. Diabetologia. 2020, 63, 2548–2558. [Google Scholar] [CrossRef] [PubMed]
- Ma XL, Shi QY, Zhao QG, Xu Q, Yan SS, Han BX, Fang C, Zhang L, Pei YF. Causal associations between type 1 diabetes and COVID-19 infection and prognosis: a two-sample Mendelian randomization study. BMJ Open Diabetes Research and Care. 2023, 11, e003167. [Google Scholar] [CrossRef]
- Iacobellis, G. COVID-19 and diabetes: Can DPP4 inhibition play a role? Diabetes research and clinical practice. 2020, 162. [Google Scholar] [CrossRef]
- Zhu L, She ZG, Cheng X, Qin JJ, Zhang XJ, Cai J, Lei F, Wang H, Xie J, Wang W, Li H. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell metabolism. 2020, 31, 1068–1077. [Google Scholar] [CrossRef]
- Pal R, Bhadada SK. Should anti-diabetic medications be reconsidered amid COVID-19 pandemic? Diabetes research and clinical practice. 2020, 163. [Google Scholar]
- Carboni E, Carta AR, Carboni E. Can pioglitazone be potentially useful therapeutically in treating patients with COVID-19? Medical Hypotheses. 2020, 140, 109776. [Google Scholar] [CrossRef]
- Lubel J, Garg M. Renin-angiotensin-aldosterone system inhibitors in Covid-19. N Engl J Med. 2020, 382, e92. [Google Scholar]
- Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. cell. 2020, 181, 271–280. [Google Scholar] [CrossRef]
- Koh H, Moh AM, Yeoh E, Lin Y, Low SK, Ooi ST, Tan SK, Lin JH, Hoong CW. Diabetes predicts severity of COVID-19 infection in a retrospective cohort: A mediatory role of the inflammatory biomarker C-reactive protein. Journal of medical virology. 2021, 93, 3023–3032. [Google Scholar] [CrossRef]
- Zhu L, She ZG, Cheng X, Qin JJ, Zhang XJ, Cai J, Lei F, Wang H, Xie J, Wang W, Li H. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell metabolism. 2020, 31, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- Concato J, Shah N, Horwitz RI. Randomized, controlled trials, observational studies, and the hierarchy of research designs. New England journal of medicine. 2000, 342, 1887–1892. [Google Scholar] [CrossRef] [PubMed]
- Emanuel EJ, Wendler D, Killen J, Grady C. What makes clinical research in developing countries ethical? The benchmarks of ethical research. Journal of infectious diseases. 2004, 189, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Boyko, EJ. Observational research—opportunities and limitations. Journal of Diabetes and its Complications. 2013, 27, 642–648. [Google Scholar] [CrossRef]
- Carlson MD, Morrison RS. Study design, precision, and validity in observational studies. Journal of palliative medicine. 2009, 12, 77–82. [Google Scholar] [CrossRef]
- Colditz, GA. Overview of the epidemiology methods and applications: strengths and limitations of observational study designs. Critical reviews in food science and nutrition. 2010, 50, 10–12. [Google Scholar] [CrossRef]
- Maki KC, Slavin JL, Rains TM, Kris-Etherton PM. Limitations of observational evidence: implications for evidence-based dietary recommendations. Advances in nutrition. 2014, 5, 7–15. [Google Scholar] [CrossRef]
- Gill D, Walker VM, Martin RM, Davies NM, Tzoulaki I. Comparison with randomized controlled trials as a strategy for evaluating instruments in Mendelian randomization. International journal of epidemiology. 2020, 49, 1404–1406. [Google Scholar] [CrossRef] [PubMed]
- Katikireddi SV, Green MJ, Taylor AE, Davey Smith G, Munafò MR. Assessing causal relationships using genetic proxies for exposures: an introduction to Mendelian randomization. Addiction. 2018, 113, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Davey Smith G, Hemani G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Human molecular genetics. 2014, 23, R89–R98. [Google Scholar] [CrossRef] [PubMed]
- Lee, YH. Overview of Mendelian randomization analysis. Journal of Rheumatic Diseases. 2020, 27, 241–246. [Google Scholar] [CrossRef]
- Nitsch D, Molokhia M, Smeeth L, DeStavola BL, Whittaker JC, Leon DA. Limits to causal inference based on Mendelian randomization: a comparison with randomized controlled trials. American journal of epidemiology. 2006, 163, 397–403. [Google Scholar] [CrossRef]
- Lee, YH. Overview of Mendelian randomization analysis. Journal of Rheumatic Diseases. 2020, 27, 241–246. [Google Scholar] [CrossRef]
| Method | NSNP | Odds Ratio (OR) | SE | 95%CI | P value |
|---|---|---|---|---|---|
| MR Egger | 14 | 0.9726 | 0.138930 | 0.7354-1.2859 | 0.844671 |
| Weighted Median | 14 | 0.9836 | 0.104530 | 0.8055-1.2012 | 0.873989 |
| Inverse Variance Weighted | 14 | 0.9756 | 0.083776 | 0.8290-1.1482 | 0.768234 |
| Simple Mode | 14 | 0.7648 | 0.219149 | 0.4913-1.1907 | 0.242919 |
| Weighted Mode | 14 | 0.9853 | 0.104027 | 0.8072-1.2031 | 0.889348 |
| Test | Metric | Value | Degrees of Freedom | P-value |
|---|---|---|---|---|
| Heterogeneity | ||||
| Q Statistic (MR Egger) | 13.869 | 12 | 0.3091776 | |
| Q Statistic (IVW) | 13.869 | 13 | 0.3830933 | |
| Directional Pleiotropy (MR Egger) | ||||
| Egger Intercept | 0.001012 | x | x | |
| Standard Error (SE) | 0.035030 | x | x | |
| P value | x | 0.977409 | ||
| Steiger Directionality | ||||
| SNP R2; (Exposure) | 0.004047 | x | ||
| SNP R2; (Outcome) | 0.0036851 | x | ||
| Correct Causal Direction | TRUE | x | ||
| Steiger P-value | 0.8578485 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




