Submitted:
13 August 2024
Posted:
14 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
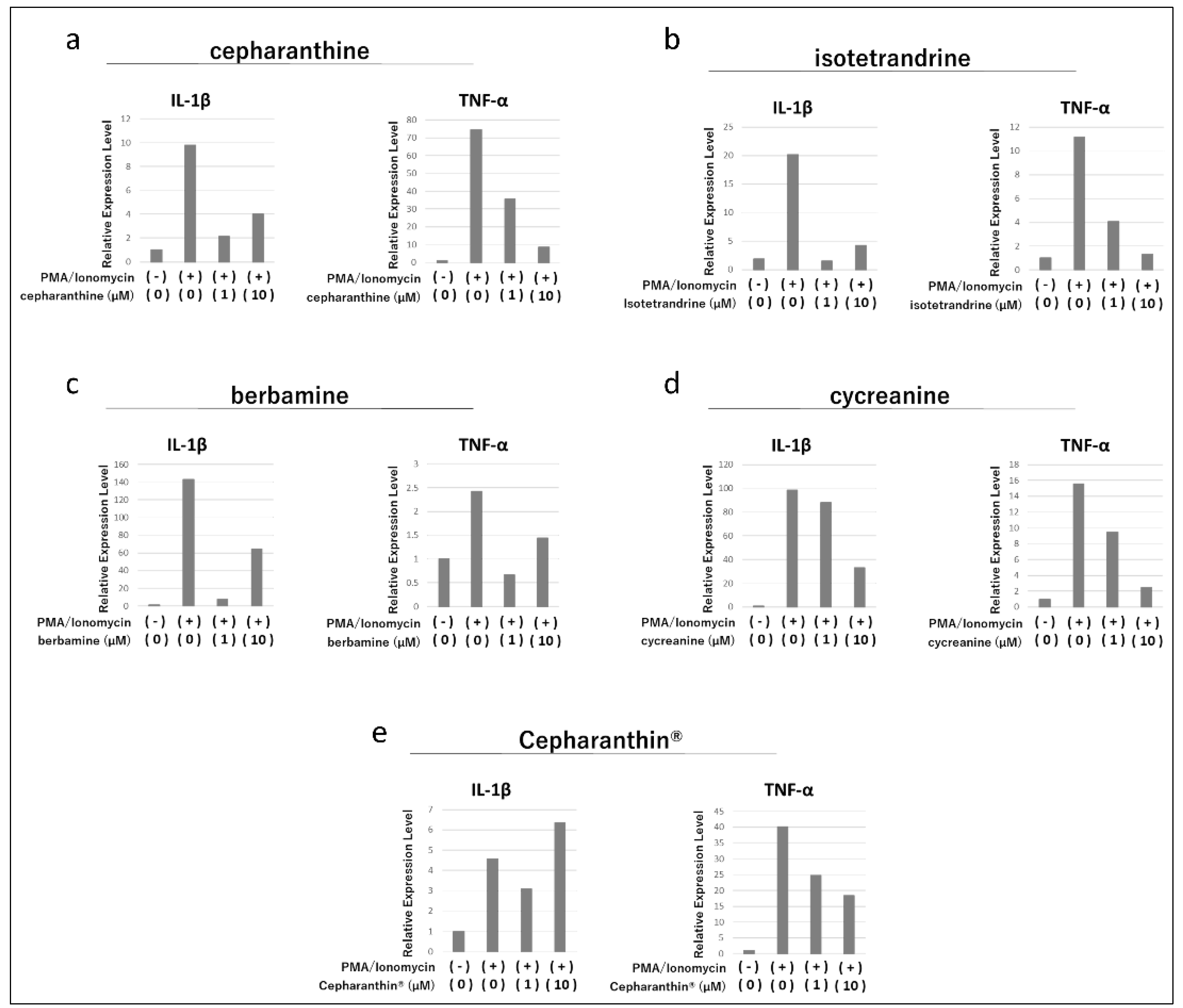
2.1. Effects of Cepharanthine, Isotetrandrine, Berbamine, Cycreanine, and Cepharanthin® on the Activation of Human Acute T-Cell Leukemia Cell Line (Jurkat E6.1)
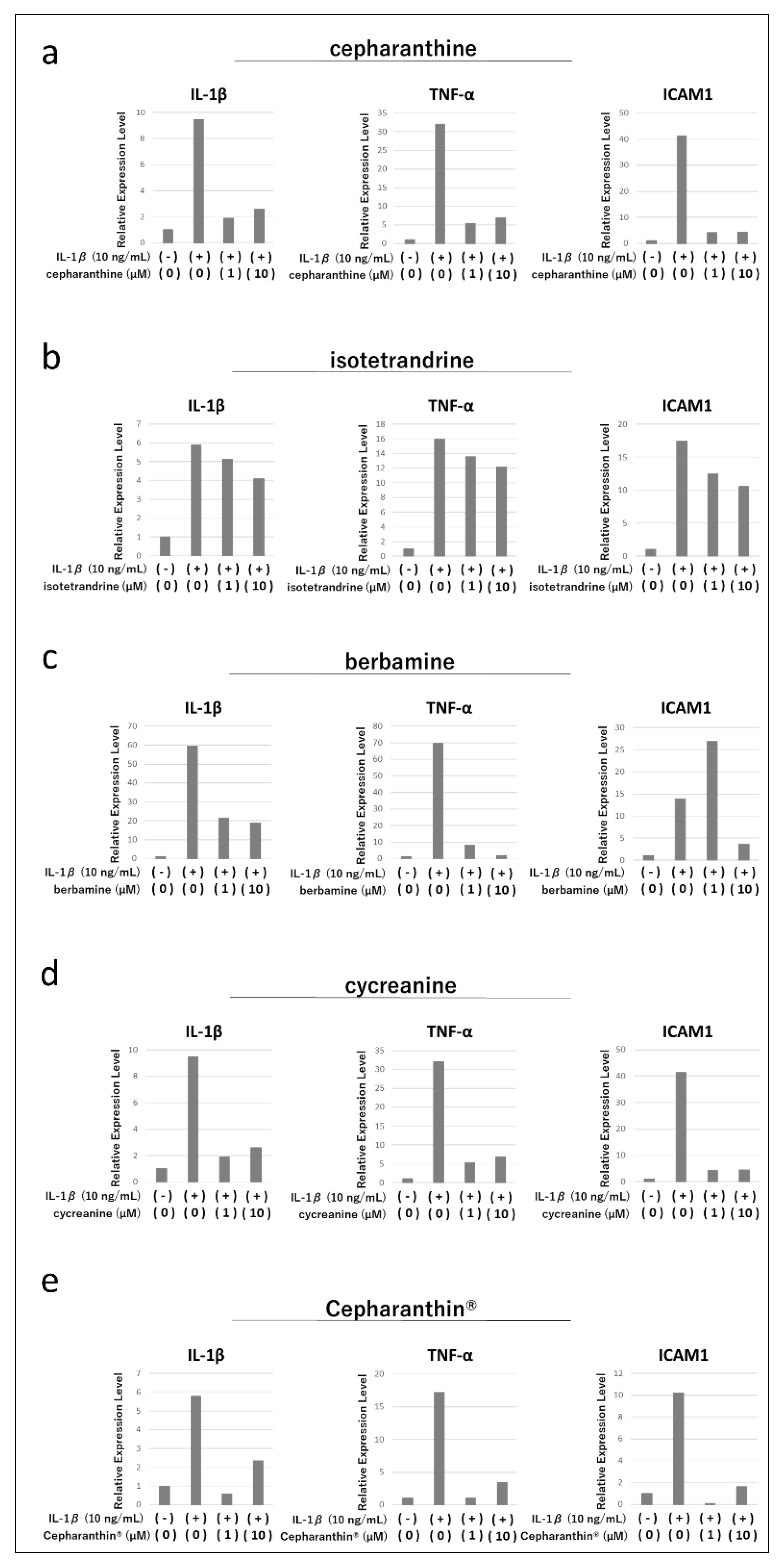
2.2. Effects of Cepharanthine, Isotetrandrine, Berbamine, Cycreanine, and Cepharanthin® on the Expression of Various Cytokine Genes Induced by Inflammatory Cytokine Stimulation in an Immortalized Human Oral Mucosa-Derived Epithelial Cell Line (hTERT-TIGKs)
2.3. Effect of Cepharanthin® on Cytokine Gene Expression in hTERT-TIGKs in an In Vitro OLP Model
2.4. Effect of Cepharanthin® and/or Activated T Lymphocytes on the Proliferation of hTERT-TIGKs
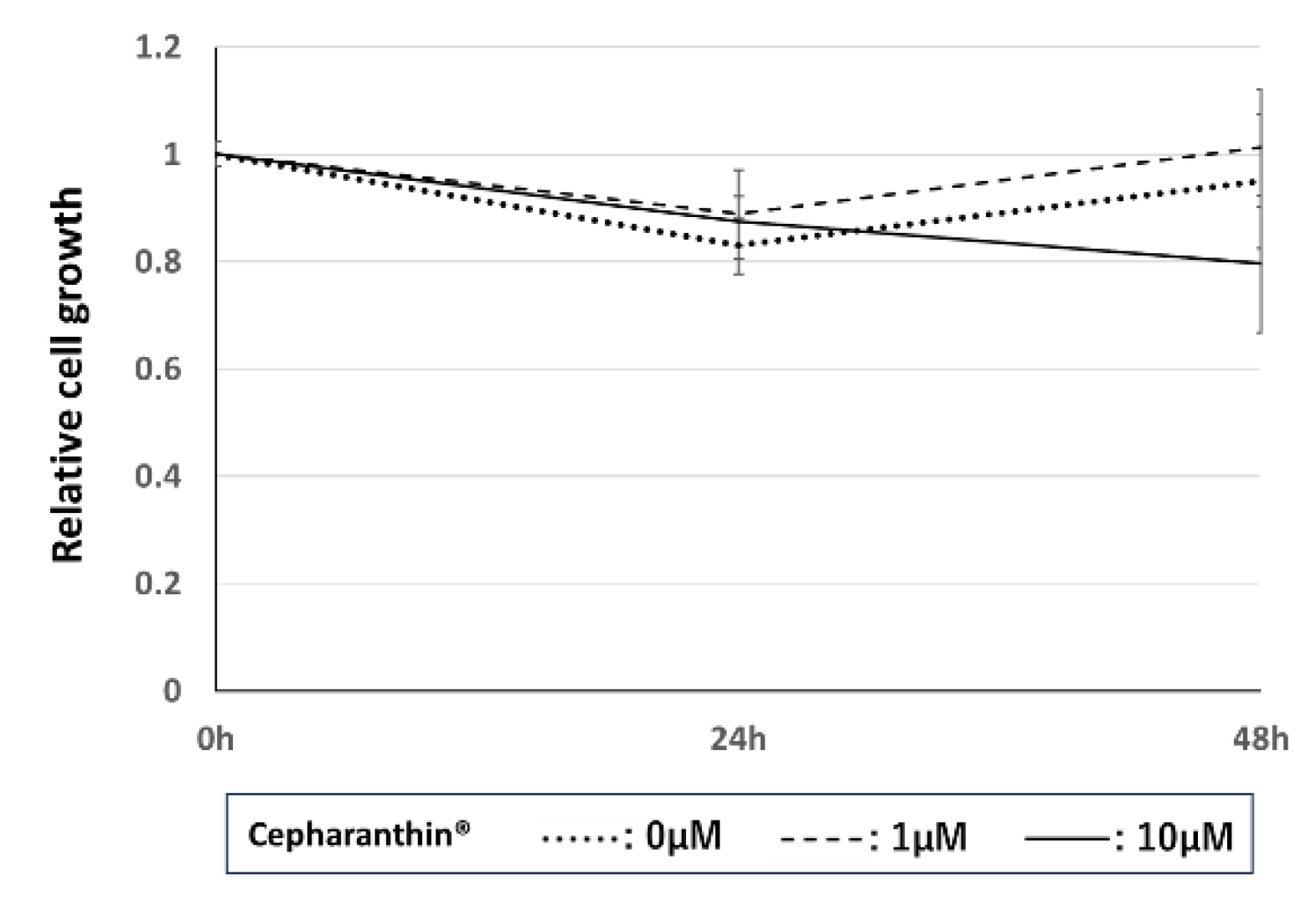
2.4.1. Effect of Cepharanthin® on the Proliferation of hTERT-TIGKs
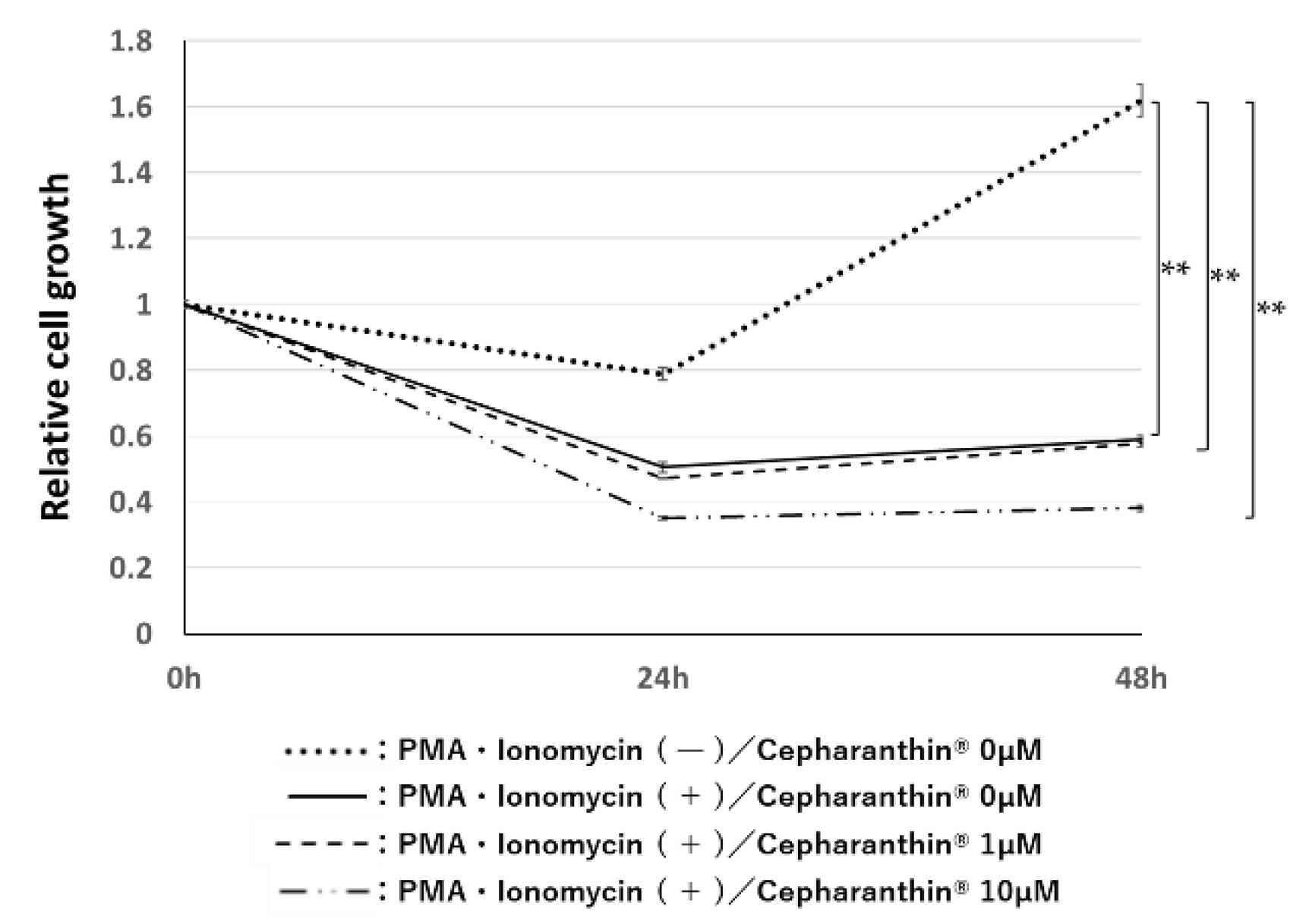
2.4.2. Effect of Cephalanthin® on the Influence of Activated T Cells for the Proliferation of hTERT-TIGKs in an In Vitro OLP Model
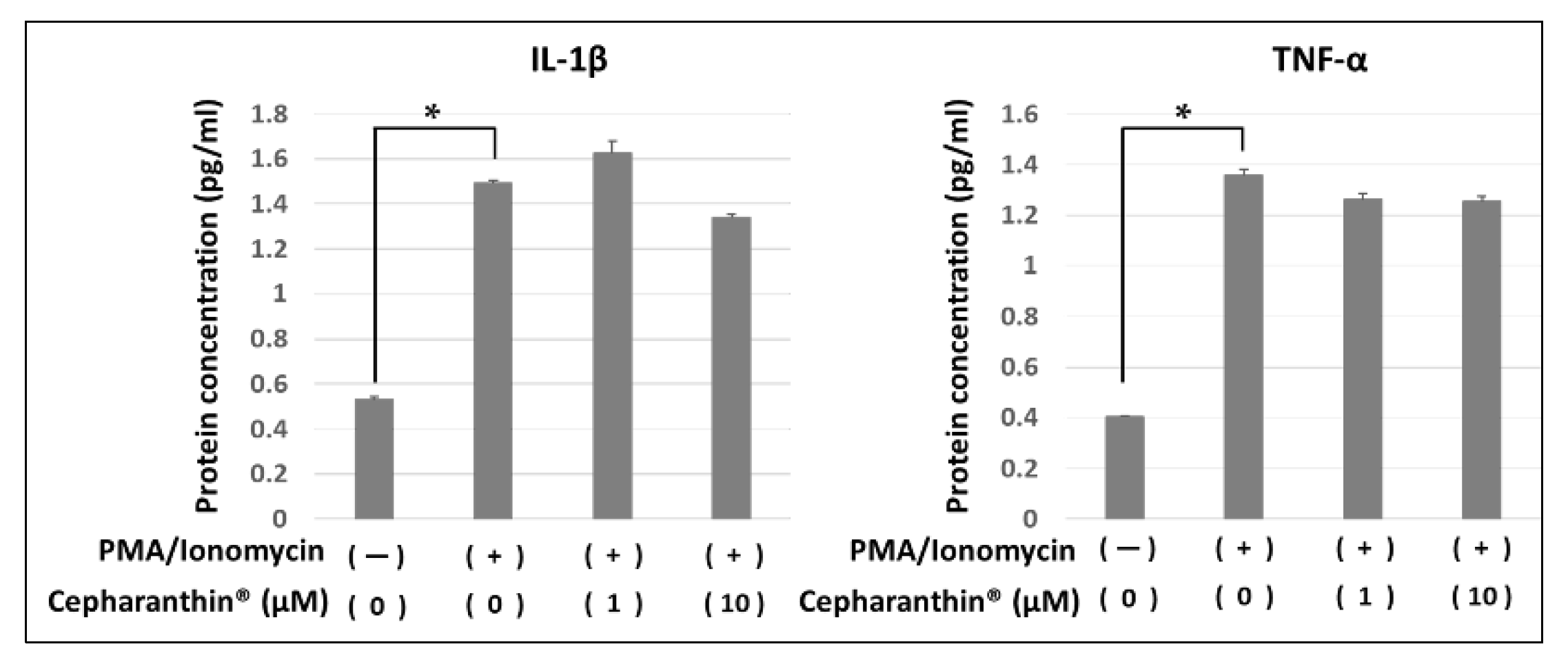
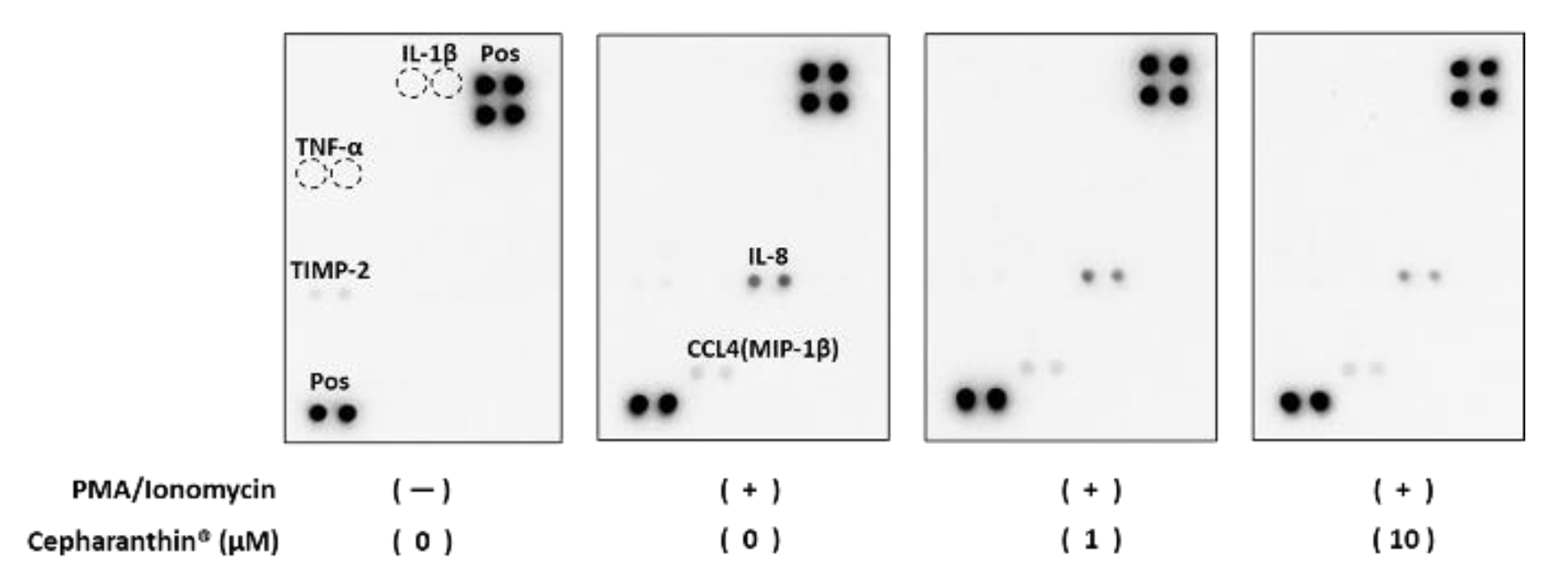
2.5. Effect of Cepharanthin® on the Production and the Accumulation of Inflammatory Factor Proteins from Activated T Cells and hTERT-TIGKs in an In Vitro OLP Model
3. Discussion
4. Materials and Methods
4.1. Cell Lines and Cell Culture
4.2. Effects of Cepharanthine, Isotetrandrine, Berbamine, Cycreanine, and Cepharanthin® on the Activation of Jurkat E6.1
4.3. Effects of Cepharanthine, Isotetrandrine, Berbamine, Cycreanine, and Cepharanthin® on the Expression of Various Cytokine Genes Induced by Inflammatory Cytokine Stimulation in hTERT-TIGKs
4.4. Effect of Cepharanthin® on Cytokine Gene Expression in hTERT-TIGKs in an In Vitro OLP Model
4.5. RT-qPCR
4.6. Effect of Cepharanthin® and/or Activated T Lymphocytes on the Proliferation of hTERT-TIGKs
4.6.1. Effect of Cepharanthin® on the Proliferation of hTERT-TIGKs
4.6.2. Effect of Cepharanthin® on the Influence of Activated T Cells on the Proliferation of hTERT-TIGKs in an In Vitro OLP Model
4.7. Effect of Cepharanthin® on the Production and Accumulation of Inflammatory Factor Proteins from Activated T Cells and hTERT-TIGKs in an In Vitro OLP Model
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sugerman P.B., Savage N.W., Walsh L.J., Zhao Z.Z., Zhou X.J., Khan A., Seymour G.J., Bigby M. The pathogenesis of oral lichen planus. Crit. Rev. Oral Biol. Med. 2002;13:350-365. doi: 10.1177/154411130201300405. [CrossRef]
- Lodi G., Scully C., Carrozzo M., Griths M., Sugerman P.B., Thongprasom K. Current controversies in oral lichen planus: Report of an international consensus meeting. Part 1. Viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:40-51. doi: 10.1016/j.tripleo.2004.06.077. [CrossRef]
- Deyhimi P,Arzhang E. Study of extrinsic apoptotic pathway in oral Lichen Planus using TNFR 1 and FasL immunohistochemical markers and TUNEL technique. Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology. 2018;30:380-385.
- Deng S., Wang S., Shi X., Zhou H. Microenvironment in Oral Potentially Malignant Disorders: Multi-Dimensional Characteristics and Mechanisms of Carcinogenesis. Int. J. Mol. Sci. 2022;23:8940. doi: 10.3390/ijms23168940. [CrossRef]
- Nosratzehi T. Oral lichen planus: an overview of potential risk factors, biomarkers and treatments. Asian Pac J Cancer Prev APJCP. 2018;19:1161-1167. doi: 10.22034/APJCP.2018.19.5.1161. [CrossRef]
- Thongprasom K., Chaimusig M., Korkij W., Sererat T., Luangjarmekorn L., Rojwattanasirivej S. A randomized-controlled trial to compare topical cyclosporin with triamcinolone acetonide for the treatment of oral lichen planus. J Oral Pathol Med. 2007;36(3):142-146. doi: 10.1111/j.1600-0714.2007.00510.x. [CrossRef]
- Fitzpatrick S.G., Hirsch S.A., Gordon S.C. The malignant transformation of oral lichen planus and oral lichenoid lesions: a systematic review. J Am Dent Assoc. 2014;145:45-56. doi: 10.14219/jada.2013.10. [CrossRef]
- Georgakopoulou E.A., Troupis T.G., Troupis G., Gorgoulis V.G. Update of the cancer-associated molecular mechanisms in oral lichen planus, a disease with possible premalignant nature. J BUON. 2011;16:613-6.
- Giuliani M., Troiano G., Cordaro M., Corsalini M., Gioco G., Lo Muzio L., Pignatelli P., Lajolo C. Rate of malignant transformation of oral lichen planus: a systematic review. Oral Dis. 2019;25(3):693-709. doi: 10.1111/odi.12885. [CrossRef]
- Landini G, Mylonas P, Shah Z.I, Hamburger J. The reported rates of transformation of oral lichen planus. Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology. 2014;26:213-220.
- Shirasuna K. Oral lichen planus: malignant potential and diagnosis. Oral Sci. Int. 2014;11:1-7. doi: 10.1016/S1348-8643(13)00030-X. [CrossRef]
- M. R. Roopashree., Rajesh V Gondhalekar., M. C. Shashikanth., Jiji George., S. H. Thippeswamy., Abhilasha Shukla. Pathogenesis of oral lichen planus - a review.J Oral Pathol Med. 2010;Nov;39(10):729-34. doi: 10.1111/j.1600-0714.2010.00946.x. [CrossRef]
- Scully C., Beyli M., Ferreiro M.C., Ficarra G., Gill Y., Griffiths M. Update on oral lichen planus: etiopathogenesis and management. Crit Rev Oral Biol Med. 1998;9:86-122. doi: 10.1177/10454411980090010501. [CrossRef]
- Ismail S.B., Kumar S.K., Zain R.B. Oral lichen planus and lichenoid reactions: Etiopathogenesis, diagnosis, management and malignant transformation. J. Oral Sci. 2007;49:89-106. doi: 10.2334/josnusd.49.89. [CrossRef]
- Lodi G., Scully C., Carrozzo M., Griffiths M., Sugerman P.B., Thongprasom K. Current controversies in oral lichen planus: report of an international consensus meeting. Part 2. Clinical management and malignant transformation. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:164-178. doi: 10.1016/j.tripleo.2004.06.076. doi: 10.1016/j.tripleo.2004.06.076. [CrossRef]
- El-Howati A, Thornhill MH, Colley HE, Murdoch C. Immune mechanisms in oral lichen planus. Oral Dis. 2023;29(4):1400-1415. doi: 10.1111/odi.14142. Epub 2022 Feb 8. [CrossRef]
- Kawamata H., Ito D., Tsushima F., Nakamura S., Kawano K., Sugawara Y., MoriyamaM., Iwabuchi H., Abiko Y., Maeda H., Sugita Y., Hasegawa H., Komiyama Y., Shiraishi R., Wakui T. Narrative Review and Task Force Consensus of Current Treatment Methods and Clinical Evaluation of the Outcome for Oral Lichen Planus. Journal of Japanese Society of Oral Medicine. 2023;29(2):21-35.
- Cepharanthin® ,KAKEN SYOYAKU CO, LTD. Available online: https://www.kakenshoyaku.com/product/cepharanthin.html (accessed on 5 August 2024).
- Seubwai W., Vaeteewoottacharn K., Hiyoshi M., Suzu S., Puapairoj A., Wongkham C., Okada S., Wongkham S. Cepharanthine exerts antitumor activity on cholangiocarcinoma by inhibiting NF-kappaB. Cancer Sci. 2010;101:1590-1595. doi: 10.1111/j.1349-7006.2010.01572.x. [CrossRef]
- Bailly C. Cepharanthine: an update of its mode of action, pharmacological properties and medical applications. Phytomedicine. 2019;62:152956. doi: 10.1016/j.phymed.2019.152956. [CrossRef]
- Chen Z., Huang C., Yang Y.L., Ding Y., Ou-Yang H.Q., Zhang Y.Y., Xu M. Inhibition of the STAT3 signaling pathway is involved in the antitumor activity of cepharanthine in SaOS2 cells. Acta Pharmacol Sin. 2012;33:101-108. doi: 10.1038/aps.2011.164. [CrossRef]
- Xu W., Kusano J., Chen S., Yamamoto R., Matsuda H., Hara Y., Fujii Y., Hayashi S., Tanaka S., Sugiyama K., Yamada H., Hirano T. Absolute configuration of tetrandrine and isotetrandrine influences their anti-proliferation effects in human T cells via different regulation of NF-κB. Z. Fur Naturforschung. C J. Biosci. 2021;76:21-25. doi: 10.1515/znc-2020-0064. [CrossRef]
- Han C., Wang Z., Chen S., Li L., Xu Y., Kang W., Wei C., Ma H., Wang M., Jin X. Berbamine suppresses the progression of bladder cancer by Modulating the ROS/NF-kappaB axis. Oxid Med Cell Longev. 2021;8851763. doi: 10.1155/2021/8851763. [CrossRef]
- Kondo Y., Takano F., Hojo H. Suppression of lipopolysaccharide-induced fulminant hepatitis and tumor necrosis factor production by bisbenzylisoquinoline alkaloids in bacillus Calmette-Guerin-treated mice. Biochem. Pharmacol. 1993;46:1861-1863. doi: 10.1016/0006-2952(93)90594-m. [CrossRef]
- Sugawara Y, Jinbu Y, Sasano T, Kusama M, Mori Y, Kitagawa Y, Takahashi T, Sugiyama Y, Fukuda M, Kawamata H, Shibata T, Ueno T, Kishimoto H, Iida S, Nakamura N, Sugiura T. Retrospective study on clinical efficacy of Cepharanthine for oral Lichen Planus as determined by the multiple institutes collaborative project. J Jpn Oral Medicine 2016;22:59-67.
- Liu K., Hong B., Wang S., Lou F., You Y., Hu R., Shafqat A., Fan H., Tong Y. Pharmacological activity of cepharanthine. Molecules. 2023;28(13):5019. doi: 10.3390/molecules28135019. [CrossRef]
- Manna S.K., Ramesh G.T. Interleukin-8 induces nuclear transcription factor-kappaB through a TRAF6-dependent pathway. J. Boil. Chem. 2005;280(8):7010-7021. doi: 10.1074/jbc.M410994200. [CrossRef]
- Kawamata H., Kawai K., Kameyama S., Johnson M.D., Stetler-Stevenson W.G., Oyasu R. Over-expression of tissue inhibitor of matrix metalloproteinases (TIMP1 and TIMP2) suppresses extravasation of pulmonary metastasis of a rat bladder carcinoma. Int. J. Cancer. 1995;63:680-687. doi: 10.1002/ijc.2910630513. [CrossRef]
- Kawamata H., Nakashiro K.i., Uchida D., Harada K., Yoshida H., Sato M. Possible contribution of active MMP2 to lymph-node metastasis and secreted cathepsin L to bone invasion of newly established human oral-squamous-cancer cell lines. Int. J. Cancer. 1997;70:120-127. doi: 10.1002/(sici)1097-0215(19970106)70:1<120::aid-ijc18>3.0.co;2-p.
- Sakai T., Furihata T., Kawamata H., Omotehara F., Shinagawa Y., Imura J., Kubota K., Terano A., Fujimori T. Molecular and genetic characterization of a non-metastatic human esophageal cancer cell line, T. Tn expressing non-functional mutated p53. Int. J. Oncol. 2002;21:547-552.
- Moffatt-Jauregui C.E., Robinson B., de Moya A.V., Brockman R.D., Roman A.V., Cash M.N., Culp D.J., Lamont R.J. Establishment and characterization of a telomerase immortalized human gingival epithelial cell line. Journal of Periodontal Research, 2013;48(6): 713-721. doi:10.1111/jre.12059. [CrossRef]
- Rasband W.S., ImageJ, U. S. National Institutes of Health, Bethesda, Maryland, USA, http://rsb.info.nih.gov/ij/, 1997-2012.


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



