Submitted:
11 August 2024
Posted:
13 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Recombinant Listeria Monocytogenes Culture and Infection
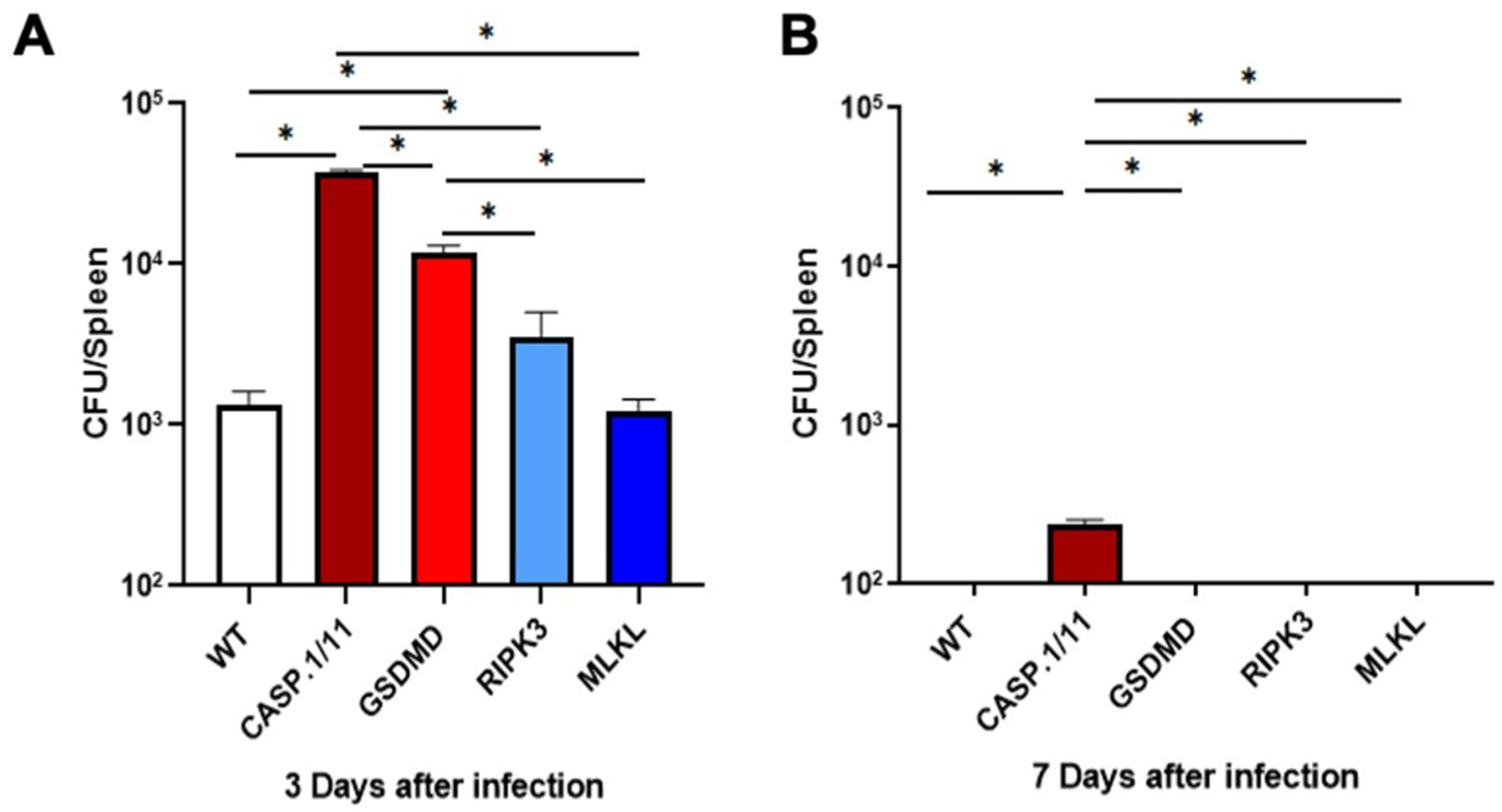
2.3. Bacterial Burden Per Spleen
2.4. In Vivo Evaluation of CD8 T Cell Effector Activity
2.5. In Vivo Tumor Growth
2.6. Statistical Analysis
3. Results
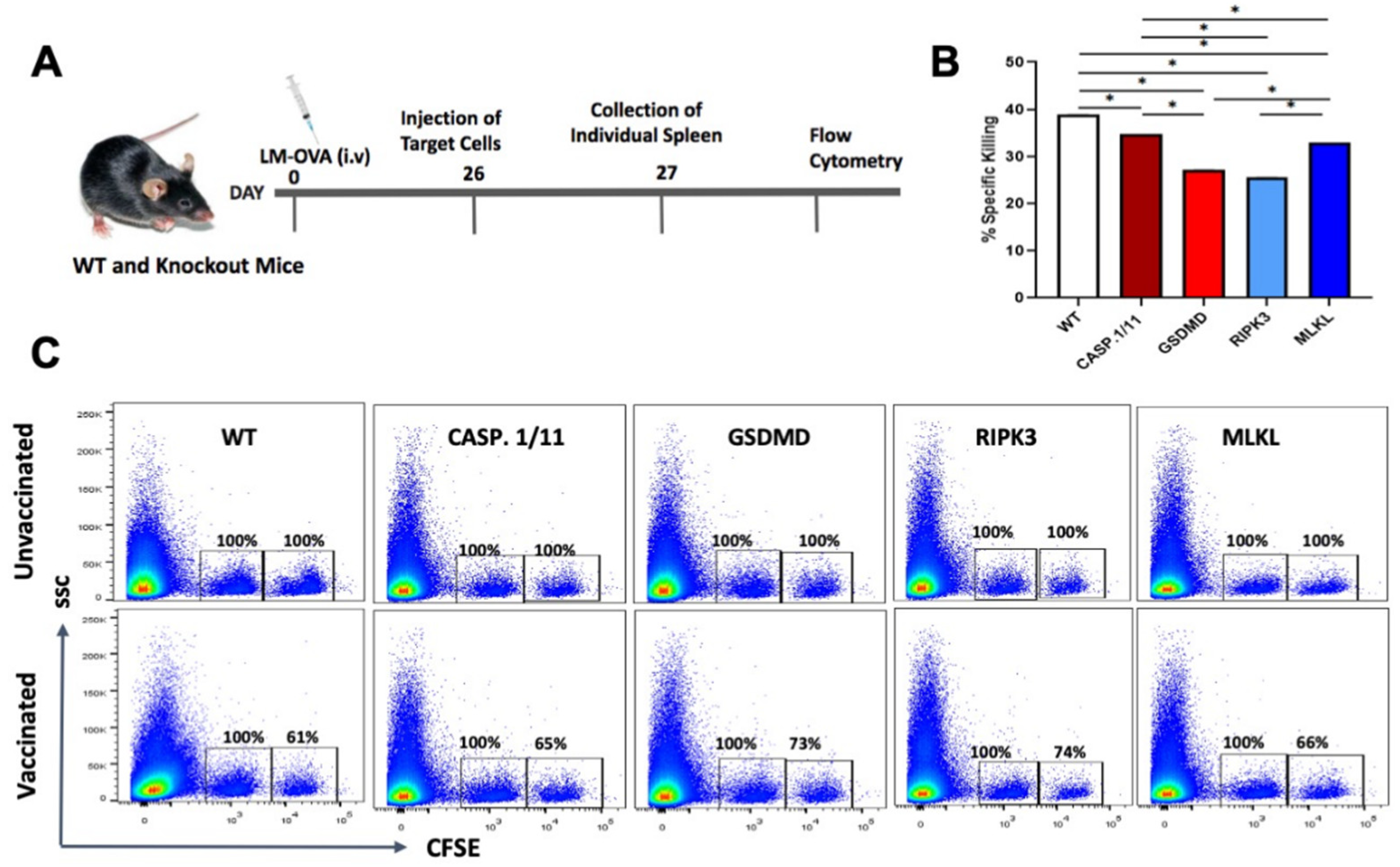
3.1. Pyroptosis- but Not Necroptosis-Related Proteins Affect LM-OVA Bacterial Burden
3.2. Deficiency of Pyroptosis- or Necroptosis-Related Proteins Did Not Impair the Early Antigen-Specific CTL Response Triggered by LM-OVA Vaccination
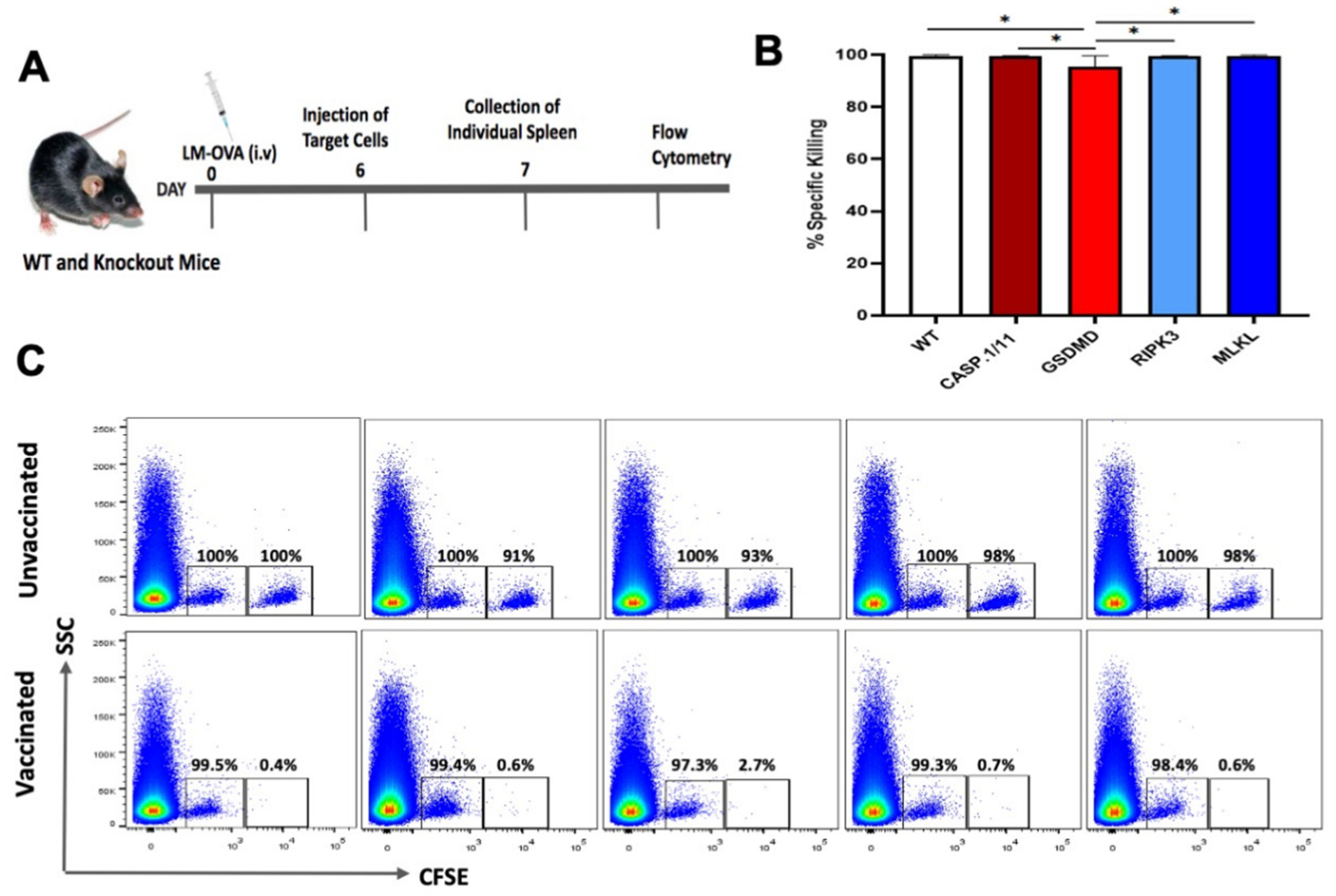
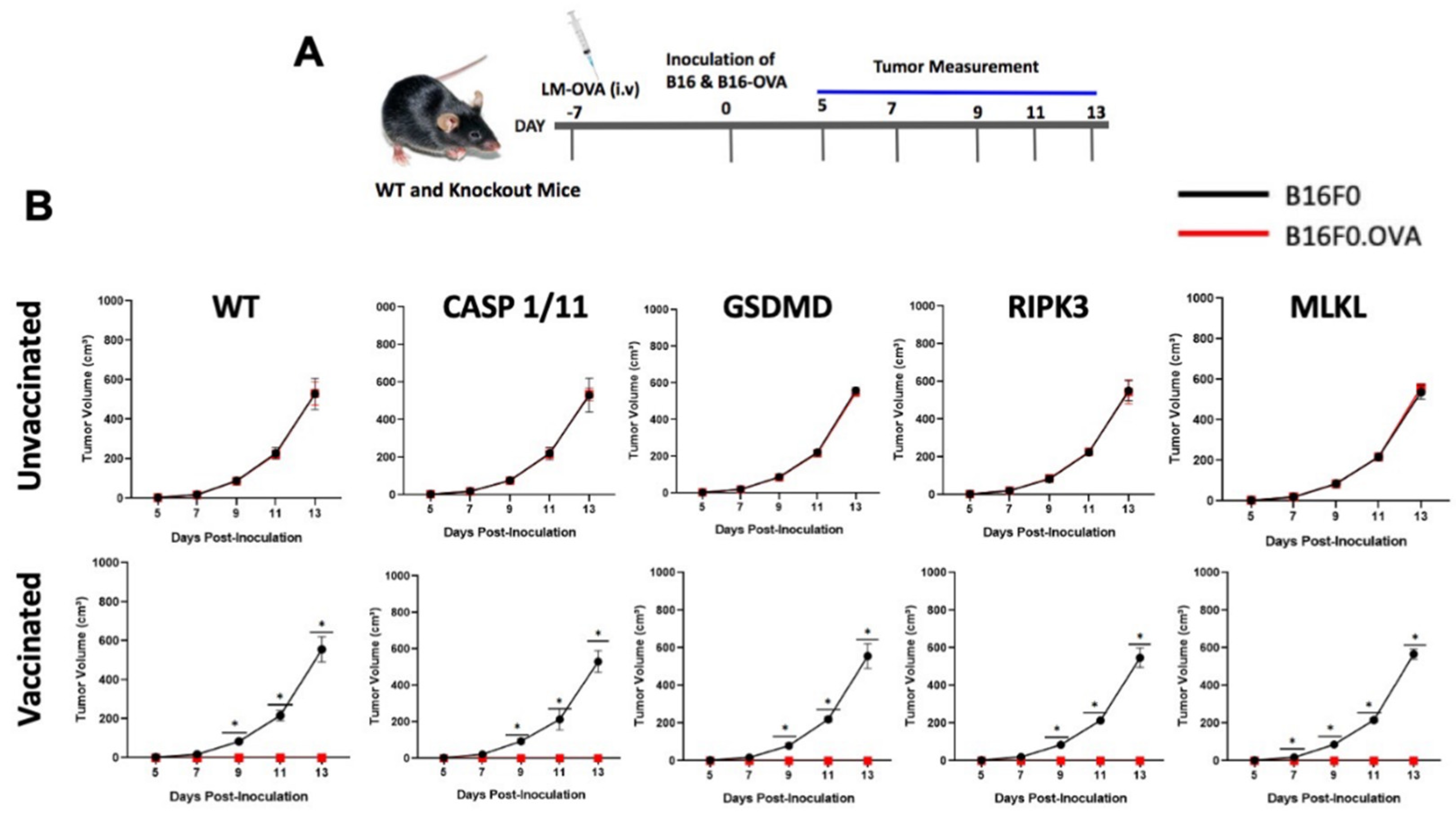
3.3. Late Antigen-Specific CTL Response Triggered by LM-OVA Vaccination Is Weakened in Pyroptosis- and Necroptosis-Deficient Mice
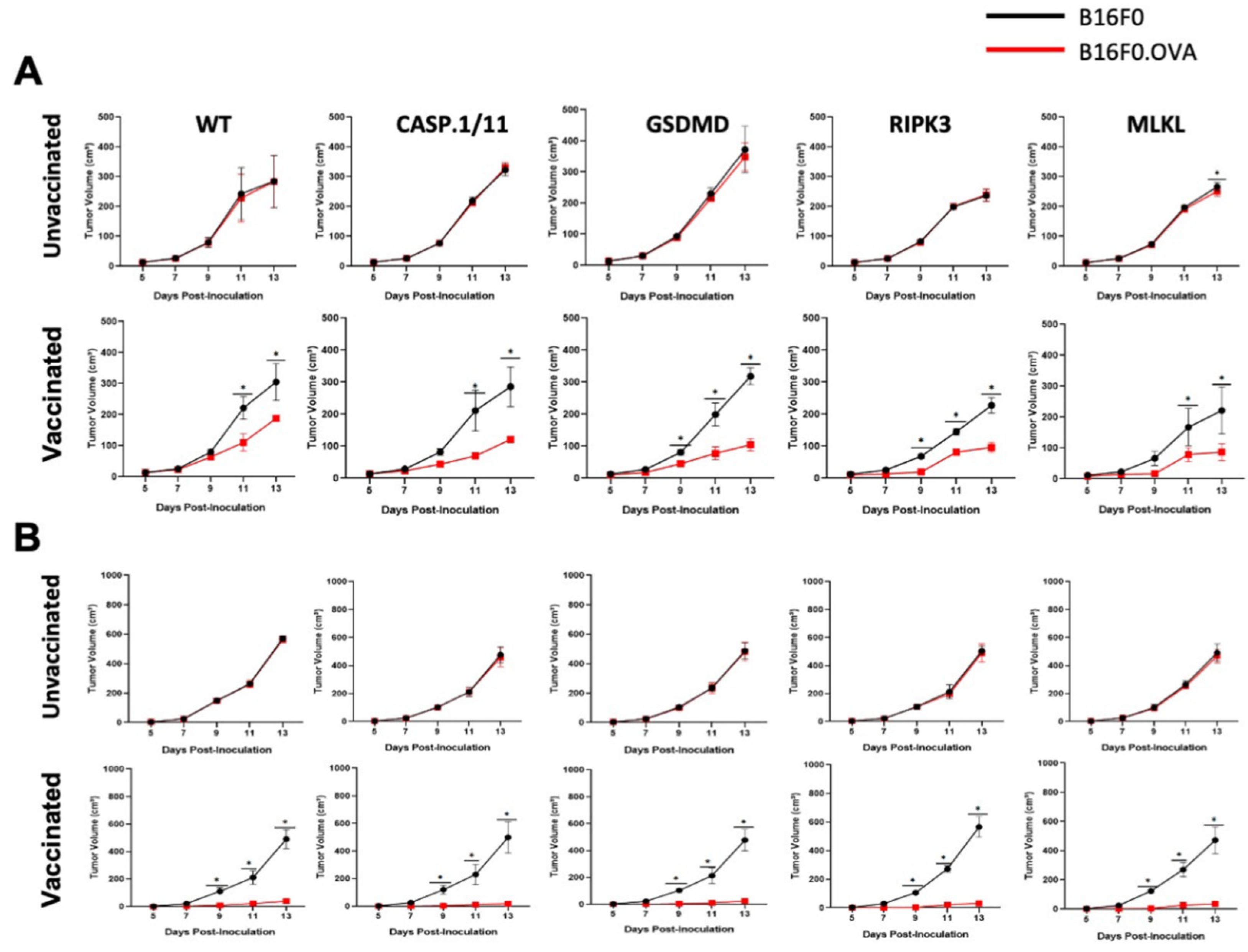
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gupta, S.L.; Basu, S.; Soni, V.; Jaiswal, R.K. Immunotherapy: An Alternative Promising Therapeutic Approach against Cancers. Mol. Biol. Rep. 2022, 49, 9903–9913. [Google Scholar] [CrossRef] [PubMed]
- Abbott, M.; Ustoyev, Y. Cancer and the Immune System: The History and Background of Immunotherapy. Semin. Oncol. Nurs. 2019, 35, 150923. [Google Scholar] [CrossRef] [PubMed]
- Milling, L.; Zhang, Y.; Irvine, D.J. Delivering Safer Immunotherapies for Cancer. Adv. Drug Deliv. Rev. 2017, 114, 79–101. [Google Scholar] [CrossRef]
- Zhao, Z.; Zheng, L.; Chen, W.; Weng, W.; Song, J.; Ji, J. Delivery Strategies of Cancer Immunotherapy: Recent Advances and Future Perspectives. J. Hematol. Oncol. 2019, 12, 126. [Google Scholar] [CrossRef] [PubMed]
- Hurez, V.; Padrón, Á.; Svatek, R.S.; Curiel, T.J. Considerations for Successful Cancer Immunotherapy in Aged Hosts. Exp. Gerontol. 2018, 107, 27–36. [Google Scholar] [CrossRef]
- Barnestein, R.; Galland, L.; Kalfeist, L.; Ghiringhelli, F.; Ladoire, S.; Limagne, E. Immunosuppressive Tumor Microenvironment Modulation by Chemotherapies and Targeted Therapies to Enhance Immunotherapy Effectiveness. OncoImmunology 2022, 11, 2120676. [Google Scholar] [CrossRef]
- Pitt, J.M.; Marabelle, A.; Eggermont, A.; Soria, J.-C.; Kroemer, G.; Zitvogel, L. Targeting the Tumor Microenvironment: Removing Obstruction to Anticancer Immune Responses and Immunotherapy. Ann. Oncol. 2016, 27, 1482–1492. [Google Scholar] [CrossRef]
- Ding, J.; Wang, K.; Liu, W.; She, Y.; Sun, Q.; Shi, J.; Sun, H.; Wang, D.-C.; Shao, F. Pore-Forming Activity and Structural Autoinhibition of the Gasdermin Family. Nature 2016, 535, 111–116. [Google Scholar] [CrossRef] [PubMed]
- https://clinicaltrials.gov/search?cond=Cancer&intr=bacteria&aggFilters=status:com.
- Witte, C.E.; Archer, K.A.; Rae, C.S.; Sauer, J.-D.; Woodward, J.J.; Portnoy, D.A. Innate Immune Pathways Triggered by Listeria Monocytogenes and Their Role in the Induction of Cell-Mediated Immunity. In Advances in Immunology; Elsevier, 2012; Volume 113, pp. 135–156. [Google Scholar] [CrossRef]
- D’Orazio, S.E.F. Innate and Adaptive Immune Responses during Listeria Monocytogenes Infection. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef]
- Morrow, Z.T.; Powers, Z.M.; Sauer, J.-D. Listeria Monocytogenes Cancer Vaccines: Bridging Innate and Adaptive Immunity. Curr. Clin. Microbiol. Rep. 2019, 6, 213–224. [Google Scholar] [CrossRef]
- Olagunju, A.S.; Rana, A.; Amarante-Mendes, G.P. Listeria Monocytogenes-Based Cancer Vaccines: Importance of Pathogen Interplay with Host’s Cell Death Machinery. Am J Biomed Sci & Res 2024, 21. [Google Scholar] [CrossRef]
- Ding, C.; Ma, J.; Dong, Q.; Liu, Q. Live Bacterial Vaccine Vector and Delivery Strategies of Heterologous Antigen: A Review. Immunol. Lett. 2018, 197, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Regan, T.; MacSharry, J.; Brint, E. Tracing Innate Immune Defences along the Path of Listeria Monocytogenes Infection. Immunol. Cell Biol. 2014, 92, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Amarante-Mendes, G.P.; Adjemian, S.; Branco, L.M.; Zanetti, L.C.; Weinlich, R.; Bortoluci, K.R. Pattern Recognition Receptors and the Host Cell Death Molecular Machinery. Front. Immunol. 2018, 9, 2379. [Google Scholar] [CrossRef]
- Schroder, K.; Tschopp, J. The Inflammasomes. Cell 2010, 140, 821–832. [Google Scholar] [CrossRef]
- Bortoluci, K.R.; Medzhitov, R. Control of Infection by Pyroptosis and Autophagy: Role of TLR and NLR. Cell. Mol. Life Sci. 2010, 67, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Deets, K.A.; Vance, R.E. Inflammasomes and Adaptive Immune Responses. Nat. Immunol. 2021, 22, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.R.; Dustin, M.L.; Sauer, J.-D. Inflammasome-Mediated Inhibition of Listeria Monocytogenes-Stimulated Immunity Is Independent of Myelomonocytic Function. PLoS ONE 2013, 8, e83191. [Google Scholar] [CrossRef] [PubMed]
- Theisen, E.; Sauer, J.-D. Listeria Monocytogenes-Induced Cell Death Inhibits the Generation of Cell-Mediated Immunity. Infect. Immun. 2017, 85, e00733-16. [Google Scholar] [CrossRef]
- Weber, K.; Roelandt, R.; Bruggeman, I.; Estornes, Y.; Vandenabeele, P. Nuclear RIPK3 and MLKL Contribute to Cytosolic Necrosome Formation and Necroptosis. Commun. Biol. 2018, 1, 6. [Google Scholar] [CrossRef]
- Sai, K.; Parsons, C.; House, J.S.; Kathariou, S.; Ninomiya-Tsuji, J. Necroptosis Mediators RIPK3 and MLKL Suppress Intracellular Listeria Replication Independently of Host Cell Killing. J. Cell Biol. 2019, 218, 1994–2005. [Google Scholar] [CrossRef] [PubMed]
- Dudani, R.; Chapdelaine, Y.; Faassen, H.V.; Smith, D.K.; Shen, H.; Krishnan, L.; Sad, S. Multiple Mechanisms Compensate to Enhance Tumor-Protective CD8(+) T Cell Response in the Long-Term despite Poor CD8(+) T Cell Priming Initially: Comparison between an Acute versus a Chronic Intracellular Bacterium Expressing a Model Antigen. J. Immunol. Baltim. Md 1950 2002, 168, 5737–5745. [Google Scholar] [CrossRef] [PubMed]
- Clemente, T.; Dominguez, M.R.; Vieira, N.J.; Rodrigues, M.M.; Amarante-Mendes, G.P. In Vivo Assessment of Specific Cytotoxic T Lymphocyte Killing. Methods San Diego Calif 2013, 61, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fu, M.; Wang, M.; Wan, D.; Wei, Y.; Wei, X. Cancer Vaccines as Promising Immuno-Therapeutics: Platforms and Current Progress. J. Hematol. Oncol.J Hematol Oncol 2022, 15, 28–28. [Google Scholar] [CrossRef] [PubMed]
- Maciag, P.C.; Radulovic, S.; Rothman, J. The First Clinical Use of a Live-Attenuated Listeria Monocytogenes Vaccine: A Phase I Safety Study of Lm-LLO-E7 in Patients with Advanced Carcinoma of the Cervix. Vaccine 2009, 27, 3975–3983. [Google Scholar] [CrossRef] [PubMed]
- Stein, M.N.; Fong, L.; Tutrone, R.; Mega, A.; Lam, E.T.; Parsi, M.; Vangala, S.; Gutierrez, A.A.; Haas, N.B. ADXS31142 Immunotherapy ± Pembrolizumab Treatment for Metastatic Castration-Resistant Prostate Cancer: Open-Label Phase I/II KEYNOTE-046 Study. The Oncologist 2022, 27, 453–461. [Google Scholar] [CrossRef]
- Le, D.T.; Brockstedt, D.G.; Nir-Paz, R.; Hampl, J.; Mathur, S.; Nemunaitis, J.; Sterman, D.H.; Hassan, R.; Lutz, E.; Moyer, B.; Giedlin, M.; Louis, J.-L.; Sugar, E.A.; Pons, A.; Cox, A.L.; Levine, J.; Murphy, A.L.; Illei, P.; Dubensky, T.W.; Eiden, J.E.; Jaffee, E.M.; Laheru, D.A. A Live-Attenuated Listeria Vaccine (ANZ-100) and a Live-Attenuated Listeria Vaccine Expressing Mesothelin (CRS-207) for Advanced Cancers: Phase I Studies of Safety and Immune Induction. Clin. Cancer Res. 2012, 18, 858–868. [Google Scholar] [CrossRef]
- Anderson, T.S.; McCormick, A.L.; Daugherity, E.A.; Oladejo, M.; Okpalanwaka, I.F.; Smith, S.L.; Appiah, D.; Wood, L.M.; Lowe, D.B. Listeria-Based Vaccination against the Pericyte Antigen RGS5 Elicits Anti-Vascular Effects and Colon Cancer Protection. Oncoimmunology 2023, 12, 2260620. [Google Scholar] [CrossRef]
- Tsuji, N.M.; Tsutsui, H.; Seki, E.; Kuida, K.; Okamura, H.; Nakanishi, K.; Flavell, R.A. Roles of Caspase-1 in Listeria Infection in Mice. Int. Immunol. 2004, 16, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Rana, A.; de Almeida, F.C.; Paico Montero, H.A.; Gonzales Carazas, M.M.; Bortoluci, K.R.; Sad, S.; Amarante-Mendes, G.P. RIPK3 and Caspase-1/11 Are Necessary for Optimal Antigen-Specific CD8 T Cell Response Elicited by Genetically Modified Listeria Monocytogenes. Front. Immunol. 2020, 11, 536–536. [Google Scholar] [CrossRef]
- Berche, P.; Gaillard, J.L.; Sansonetti, P.J. Intracellular Growth of Listeria Monocytogenes as a Prerequisite for in Vivo Induction of T Cell-Mediated Immunity. J. Immunol. Baltim. Md 1950 1987, 138, 2266–2271. [Google Scholar] [CrossRef]
- Chávez-Arroyo, A.; Portnoy, D.A. Why Is Listeria Monocytogenes Such a Potent Inducer of CD8+ T-Cells? Cell. Microbiol. 2020, 22, e13175–e13175. [Google Scholar] [CrossRef]
- Goossens, P.L.; Milon, G.; Cossart, P.; Saron, M.-F. Attenuated Listeria Monocytogenes as a Live Vector for Induction of CD8 + T Cells in vivo: A Study with the Nucleoprotein of the Lymphocytic Choriomeningitis Virus. Int. Immunol. 1995, 7, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Ikonomidis, G.; Paterson, Y.; Kos, F.J.; Portnoy, D.A. Delivery of a Viral Antigen to the Class I Processing and Presentation Pathway by Listeria Monocytogenes. J. Exp. Med. 1994, 180, 2209–2218. [Google Scholar] [CrossRef]
- Shen, H.; Slifka, M.K.; Matloubian, M.; Jensen, E.R.; Ahmed, R.; Miller, J.F. Recombinant Listeria Monocytogenes as a Live Vaccine Vehicle for the Induction of Protective Anti-Viral Cell-Mediated Immunity. Proc. Natl. Acad. Sci. 1995, 92, 3987–3991. [Google Scholar] [CrossRef]
- Qiu, Z.; Khairallah, C.; Sheridan, B.S. Listeria Monocytogenes: A Model Pathogen Continues to Refine Our Knowledge of the CD8 T Cell Response. Pathog. Basel Switz. 2018, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Farhood, B.; Najafi, M.; Mortezaee, K. CD8 + Cytotoxic T Lymphocytes in Cancer Immunotherapy: A Review. J. Cell. Physiol. 2019, 234, 8509–8521. [Google Scholar] [CrossRef]
- Nguyen, H.-M.; Oladejo, M.; Paulishak, W.; Wood, L.M. A Listeria-Based Vaccine Targeting ISG15 Exerts Anti-Tumor Efficacy in Renal Cell Carcinoma. Cancer Immunol. Immunother. CII 2023, 72, 2889–2903. [Google Scholar] [CrossRef]
- Oladejo, M.; Nguyen, H.-M.; Silwal, A.; Reese, B.; Paulishak, W.; Markiewski, M.M.; Wood, L.M. Listeria-Based Immunotherapy Directed against CD105 Exerts Anti-Angiogenic and Anti-Tumor Efficacy in Renal Cell Carcinoma. Front. Immunol. 2022, 13, 1038807. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.H.; Badovinac, V.P. Listeria Monocytogenes: A Model Pathogen to Study Antigen-Specific Memory CD8 T Cell Responses. Semin. Immunopathol. 2015, 37, 301–310. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



