Submitted:
11 August 2024
Posted:
13 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Pharmacokinetics of Arsenic Trioxide
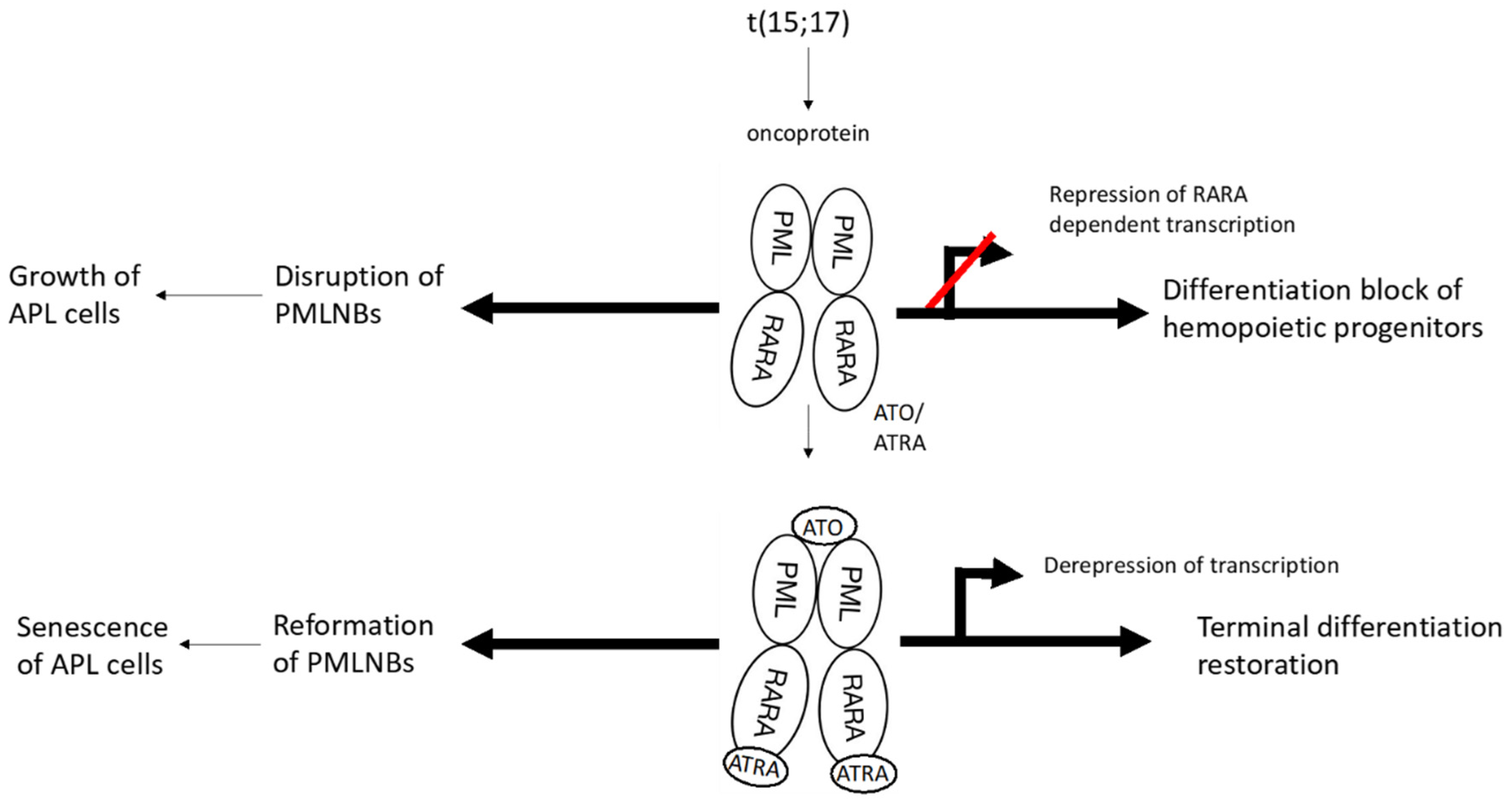
3. Mechanisms of Action of Arsenic Trioxide in Leukemia
4. Anti-Inflammatory Effects of Arsenic Trioxide
4.1. Systemic Lupus Erythematosus
4.2. Systemic Sclerosis
4.3. Inflammatory Arthritis
5. Prospect of Arsenic Trioxide Treatment for SLE
5.1. Unmet Needs in the Treatment of SLE
5.2. Disease Modifying Effects of SLE Therapies
5.3. Novel Small Molecules for SLE
5.4. Preliminary Clinical Data of ATO in SLE
5.5. Clinical Experience of ATO in Other Rheumatic Diseases
6. Conclusions
Conflicts of Interest
Abbreviations
References
- Miller WH, Schipper HM, Lee JS, Singer J, Waxman S. Mechanisms of action of arsenic trioxide. Cancer Res. 2002 Jul 15;62(14):3893–903.
- Kumana CR, Mak R, Kwong YL, Gill H. Resurrection of Oral Arsenic Trioxide for Treating Acute Promyelocytic Leukaemia: A Historical Account From Bedside to Bench to Bedside. Front Oncol. 2020 Aug 4;10.
- Chen X, Hong Y, Zheng P, You X, Feng J, Huang Z, et al. The economic research of arsenic trioxide for the treatment of newly diagnosed acute promyelocytic leukemia in China. Cancer. 2020 Jan 15;126(2):311–21.
- Shen ZX, Shi ZZ, Fang J, Gu BW, Li JM, Zhu YM, et al. All-trans retinoic acid/As2O3 combination yields a high quality remission and survival in newly diagnosed acute promyelocytic leukemia. Proc Natl Acad Sci U S A. 2004 Apr 13;101(15):5328–35.
- Drugs@FDA: FDA-Approved Drugs [Internet]. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=021248 (accessed on 9 July 2024).
- Zhu HH, Wu DP, Jin J, Li JY, Ma J, Wang JX, et al. Oral Tetra-Arsenic Tetra-Sulfide Formula Versus Intravenous Arsenic Trioxide As First-Line Treatment of Acute Promyelocytic Leukemia: A Multicenter Randomized Controlled Trial. J Clin Oncol. 2013 Nov 20;31(33):4215–21.
- Sanz MA, Fenaux P, Tallman MS, Estey EH, Löwenberg B, Naoe T, et al. Management of acute promyelocytic leukemia: updated recommendations from an expert panel of the European LeukemiaNet. Blood. 2019 Apr 11;133(15):1630–43.
- Kantarjian HM, Jain N, Garcia-Manero G, Welch MA, Ravandi F, Wierda WG, et al. The cure of leukemia through the optimist’s prism. Cancer. 2021 Oct 6.
- Vineetha VP, Raghu KG. An Overview on Arsenic Trioxide-Induced Cardiotoxicity. Cardiovasc Toxicol. 2019 Apr 15;19(2):105–19.
- Ravandi F, Koumenis I, Johri A, Tallman M, Roboz GJ, Strickland S, et al. Oral arsenic trioxide ORH-2014 pharmacokinetic and safety profile in patients with advanced hematologic disorders. Haematologica. 2020 Jun 1;105(6):1567–74.
- Kumana CR, Au WY, Lee NSL, Kou M, Mak RWM, Lam CW, et al. Systemic availability of arsenic from oral arsenic-trioxide used to treat patients with hematological malignancies. Eur J Clin Pharmacol. 2002;58(8):521–6.
- Au WY, Kwong YL. Arsenic trioxide: safety issues and their management. Acta Pharmacol Sin. 2008 Mar;29(3):296–304.
- Gill H, Yim R, Lee HKK, Mak V, Lin SY, Kho B, et al. Long-term outcome of relapsed acute promyelocytic leukemia treated with oral arsenic trioxide-based reinduction and maintenance regimens: A 15-year prospective study. Cancer. 2018 Jun 1;124(11):2316–26.
- Siu CW, Au WY, Yung C, Kumana CR, Lau CP, Kwong YL, et al. Effects of oral arsenic trioxide therapy on QT intervals in patients with acute promyelocytic leukemia: implications for long-term cardiac safety. Blood. 2006 Jul 1;108(1):103–6.
- Jansen RJ, Argos M, Tong L, Li J, Rakibuz-Zaman M, Islam MdT, et al. Determinants and Consequences of Arsenic Metabolism Efficiency among 4,794 Individuals: Demographics, Lifestyle, Genetics, and Toxicity. Cancer Epidemiol Biomarkers Prev. 2016 Feb 4;25(2):381–90.
- Fujisawa S, Ohno R, Shigeno K, Sahara N, Nakamura S, Naito K, et al. Pharmacokinetics of arsenic species in Japanese patients with relapsed or refractory acute promyelocytic leukemia treated with arsenic trioxide. Cancer Chemother Pharmacol. 2007 Mar;59(4):485–93.
- Li J, Packianathan C, Rossman TG, Rosen BP. Nonsynonymous Polymorphisms in the Human AS3MT Arsenic Methylation Gene: Implications for Arsenic Toxicity. Chem Res Toxicol. 2017 Jul 17;30(7):1481–91.
- Lu J, Hu S, Wang W, Li J, Dong Z, Zhou J, et al. AS3MT Polymorphisms, Arsenic Metabolism, and the Hematological and Biochemical Values in APL Patients Treated with Arsenic Trioxide. Toxicol Sci. 2018 Nov 1;166(1):219–27.
- Ghiuzeli CM, Stýblo M, Saunders J, Calabro A, Budman D, Allen S, et al. The pharmacokinetics of therapeutic arsenic trioxide in acute promyelocytic leukemia patients. Leuk Lymphoma. 2022 Mar;63(3):653–63.
- Lou Y, Ma Y, Jin J, Zhu H. Oral Realgar-Indigo Naturalis Formula Plus Retinoic Acid for Acute Promyelocytic Leukemia. Front Oncol. 2021 Feb 5;10:597601.
- Zhu HH, Hu J, Lo-Coco F, Jin J. The simpler, the better: oral arsenic for acute promyelocytic leukemia. Blood. 2019 Aug 15;134(7):597–605.
- Zhu HH, Wu DP, Du X, Zhang X, Liu L, Ma J, et al. Oral arsenic plus retinoic acid versus intravenous arsenic plus retinoic acid for non-high-risk acute promyelocytic leukaemia: a non-inferiority, randomised phase 3 trial. Lancet Oncol. 2018 Jul;19(7):871–9.
- de Thé H, Pandolfi PP, Chen Z. Acute Promyelocytic Leukemia: A Paradigm for Oncoprotein-Targeted Cure. Cancer Cell. 2017 Nov 13;32(5):552–60.
- Diaz Z, Colombo M, Mann KK, Su H, Smith KN, Bohle DS, et al. Trolox selectively enhances arsenic-mediated oxidative stress and apoptosis in APL and other malignant cell lines. Blood. 2005 Feb 1;105(3):1237–45.
- Zhang XW, Yan XJ, Zhou ZR, Yang FF, Wu ZY, Sun H Bin, et al. Arsenic trioxide controls the fate of the PML-RARalpha oncoprotein by directly binding PML. Science. 2010 Apr 9;328(5975):240–3.
- Zhang XW, Yan XJ, Zhou ZR, Yang FF, Wu ZY, Sun HB, et al. Arsenic Trioxide Controls the Fate of the PML-RARα Oncoprotein by Directly Binding PML. Science. 2010 Apr 9;328(5975):240–3.
- Yan M, Wang H, Wei R, Li W. Arsenic trioxide: applications, mechanisms of action, toxicity and rescue strategies to date. Arch Pharm Res. 2024 Mar 1;47(3):249–71.
- Chen GQ, Shi XG, Tang W, Xiong SM, Zhu J, Cai X, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): I. As2O3 exerts dose-dependent dual effects on APL cells. Blood. 1997 May 1;89(9):3345–53.
- Ji H, Li Y, Jiang F, Wang X, Zhang J, Shen J, et al. Inhibition of transforming growth factor beta/SMAD signal by MiR-155 is involved in arsenic trioxide-induced anti-angiogenesis in prostate cancer. Cancer Sci. 2014 Dec 1;105(12):1541–9.
- Gao JK, Wang LX, Long B, Ye XT, Su JN, Yin XY, et al. Arsenic Trioxide Inhibits Cell Growth and Invasion via Down- Regulation of Skp2 in Pancreatic Cancer Cells. Asian Pac J Cancer Prev APJCP. 2015;16(9):3805–10.
- Jiang F, Wang X, Liu Q, Shen J, Li Z, Li Y, et al. Inhibition of TGF-β/SMAD3/NF-κB signaling by microRNA-491 is involved in arsenic trioxide-induced anti-angiogenesis in hepatocellular carcinoma cells. Toxicol Lett. 2014 Nov 8;231(1):55–61.
- Zhang J, Zhang Y, Wang W, Zhang Z. Potential molecular mechanisms underlying the effect of arsenic on angiogenesis. Arch Pharm Res. 2019 Nov 1;42(11):962–76.
- Bobé P, Bonardelle D, Benihoud K, Opolon P, Chelbi-Alix MK. Arsenic trioxide: A promising novel therapeutic agent for lymphoproliferative and autoimmune syndromes in MRL/lpr mice. Blood. 2006 Dec 15;108(13):3967–75.
- Zhao Y, Wen G, Qiao Z, Xu H, Sun Q, Huang H, et al. Effects of tetra-arsenic tetra-sulfide on BXSB lupus-prone mice: a pilot study. Lupus. 2013 Apr;22(5):469–76.
- Hu H, Chen E, Li Y, Zhu X, Zhang T, Zhu X. Effects of Arsenic Trioxide on INF-gamma Gene Expression in MRL/lpr Mice and Human Lupus. Biol Trace Elem Res. 2018 Aug;184(2):391–7.
- Kavian N, Marut W, Servettaz A, Nicco C, Chéreau C, Lemaréchal H, et al. Reactive oxygen species-mediated killing of activated fibroblasts by arsenic trioxide ameliorates fibrosis in a murine model of systemic sclerosis. Arthritis Rheum. 2012 Oct;64(10):3430–40.
- Kavian N, Marut W, Servettaz A, Laude H, Nicco C, Chéreau C, et al. Arsenic Trioxide Prevents Murine Sclerodermatous Graft-versus-Host Disease. J Immunol. 2012 May 15;188(10):5142–9.
- Ye Y, Ricard L, Siblany L, Stocker N, De Vassoigne F, Brissot E, et al. Arsenic trioxide induces regulatory functions of plasmacytoid dendritic cells through interferon-α inhibition. Acta Pharm Sin B. 2020 Jun;10(6):1061–72.
- Cauvet A, Decellas A, Guignabert C, Rongvaux-Gaïda D, Thuillet R, Ottaviani M, et al. Arsenic trioxide demonstrates efficacy in a mouse model of preclinical systemic sclerosis. Arthritis Res Ther. 2023;25:167.
- Chêne C, Rongvaux-Gaïda D, Thomas M, Rieger F, Nicco C, Batteux F. Optimal combination of arsenic trioxide and copper ions to prevent autoimmunity in a murine HOCl-induced model of systemic sclerosis. Front Immunol. 2023 Mar 30;14:1149869.
- Mei Y, Zheng Y, Wang H, Gao J, Liu D, Zhao Y, et al. Arsenic Trioxide Induces Apoptosis of Fibroblast-like Synoviocytes and Represents Antiarthritis Effect in Experimental Model of Rheumatoid Arthritis. J Rheumatol. 2011 Jan 1;38(1):36–43.
- Li C, Zhang J, Wang W, Wang H, Zhang Y, Zhang Z. Data on arsenic trioxide modulates Treg/Th17/Th1/Th2 cells in treatment-naïve rheumatoid arthritis patients and collagen-induced arthritis model mice. Data Brief. 2019 Oct 15;27:104615.
- Li C, Chu T, Zhang Z, Zhang Y. Single Cell RNA-Seq Analysis Identifies Differentially Expressed Genes of Treg Cell in Early Treatment-Naive Rheumatoid Arthritis By Arsenic Trioxide. Front Pharmacol. 2021 May 24;12:656124.
- Niu S, Zhu X, Zhang J, Ma Y, Lang X, Luo L, et al. Arsenic trioxide modulates the composition and metabolic function of the gut microbiota in a mouse model of rheumatoid arthritis. Int Immunopharmacol. 2022 Oct 1;111:109159.
- Li C, Zhang J, Wang W, Wang H, Zhang Y, Zhang Z. Arsenic trioxide improves Treg and Th17 balance by modulating STAT3 in treatment-naïve rheumatoid arthritis patients. Int Immunopharmacol. 2019 Aug 1;73:539–51.
- Mok CC, Ho LY, Chan KL, Tse SM, To CH. Trend of Survival of a Cohort of Chinese Patients With Systemic Lupus Erythematosus Over 25 Years. Front Med. 2020 Sep 11;7.
- Tektonidou MG, Lewandowski LB, Hu J, Dasgupta A, Ward MM. Survival in adults and children with systemic lupus erythematosus: a systematic review and Bayesian meta-analysis of studies from 1950 to 2016. Ann Rheum Dis. 2017 Dec;76(12):2009–16.
- Tektonidou MG, Dasgupta A, Ward MM. Risk of End-Stage Renal Disease in Patients With Lupus Nephritis, 1971-2015: A Systematic Review and Bayesian Meta-Analysis. Arthritis Rheumatol Hoboken NJ. 2016 Jun;68(6):1432–41.
- Mok CC, Ho LY, Tse SM, Chan KL. Prevalence of remission and its effect on damage and quality of life in Chinese patients with systemic lupus erythematosus. Ann Rheum Dis. 2017 Aug 1;76(8):1420–5.
- Furie R, Rovin BH, Houssiau F, Malvar A, Teng YKO, Contreras G, et al. Two-Year, Randomized, Controlled Trial of Belimumab in Lupus Nephritis. N Engl J Med. 2020 Sep 17;383(12):1117–28.
- Navarra SV, Guzmán RM, Gallacher AE, Hall S, Levy RA, Jimenez RE, et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet Lond Engl. 2011 Feb 26;377(9767):721–31.
- Furie RA, Morand EF, Bruce IN, Manzi S, Kalunian KC, Vital EM, et al. Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1): a randomised, controlled, phase 3 trial. Lancet Rheumatol. 2019 Dec;1(4):e208–19.
- Morand EF, Furie R, Tanaka Y, Bruce IN, Askanase AD, Richez C, et al. Trial of Anifrolumab in Active Systemic Lupus Erythematosus. N Engl J Med. 2020 Jan 16;382(3):211–21.
- van Vollenhoven RF, Navarra SV, Levy RA, Thomas M, Heath A, Lustine T, et al. Long-term safety and limited organ damage in patients with systemic lupus erythematosus treated with belimumab: a Phase III study extension. Rheumatol Oxf Engl. 2020 Feb 1;59(2):281–91.
- Kalunian KC, Furie R, Morand EF, Bruce IN, Manzi S, Tanaka Y, et al. A Randomized, Placebo-Controlled Phase III Extension Trial of the Long-Term Safety and Tolerability of Anifrolumab in Active Systemic Lupus Erythematosus. Arthritis Rheumatol Hoboken NJ. 2023 Feb;75(2):253–65.
- Mok, C.C. Combination strategies for lupus nephritis: facts and controversies. Expert Rev Clin Immunol. 2023 May;19(5):527–36.
- van Vollenhoven R, Askanase AD, Bomback AS, Bruce IN, Carroll A, Dall’Era M, et al. Conceptual framework for defining disease modification in systemic lupus erythematosus: a call for formal criteria. Lupus Sci Med. 2022 Mar;9(1):e000634.
- Askanase AD, Furie RA, Dall’Era M, Bomback AS, Schwarting A, Zhao MH, et al. Disease-modifying therapies in systemic lupus erythematosus for extrarenal manifestations. Lupus Sci Med. 2024 May 1;11(1):e001124.
- Mok, C.C. Targeted Small Molecules for Systemic Lupus Erythematosus: Drugs in the Pipeline. Drugs. 2023 Apr;83(6):479–96.
- Mok, C.C. The Jakinibs in systemic lupus erythematosus: progress and prospects. Expert Opin Investig Drugs. 2019 Jan;28(1):85–92.
- Morand EF, Vital EM, Petri M, van Vollenhoven R, Wallace DJ, Mosca M, et al. Baricitinib for systemic lupus erythematosus: a double-blind, randomised, placebo-controlled, phase 3 trial (SLE-BRAVE-I). Lancet Lond Engl. 2023 Mar 25;401(10381):1001–10.
- Petri M, Bruce IN, Dörner T, Tanaka Y, Morand EF, Kalunian KC, et al. Baricitinib for systemic lupus erythematosus: a double-blind, randomised, placebo-controlled, phase 3 trial (SLE-BRAVE-II). The Lancet. 2023 Mar 25;401(10381):1011–9.
- Mok, C.C. Outlook of the jakinibs in systemic lupus erythematous after baricitinib failed. Int J Rheum Dis. 2024 Feb;27(2):e15082.
- Hamidou M, Néel A, Poupon J, Amoura Z, Ebbo M, Sibilia J, et al. Safety and efficacy of low-dose intravenous arsenic trioxide in systemic lupus erythematosus: an open-label phase IIa trial (Lupsenic). Arthritis Res Ther. 2021 Dec 1;23(1).
- Ge F, Zhang Y, Cao F, Li J, Hou J, Wang P, et al. Arsenic trioxide-based therapy is suitable for patients with psoriasis-associated acute promyelocytic leukemia - A retrospective clinical study. Hematol Amst Neth. 2016 Jun;21(5):287–94.
| Authors, years | Model | Clinical effects | Immunological effects |
|---|---|---|---|
| Bobe et al., 2006 [33] | Lupus mice | ↓ lymphoproliferation, skin, lung and kidney inflammation; significantly prolonged survival | Induced apoptosis and depletion of auto-reactive T cells, ↓ production of autoantibodies and cytokines |
| Zhao et al., 2013 [34] | Lupus mice | ↓splenomegaly, amelioration of skin, liver and renal lesions | ↓ immune complex deposition, mesangial proliferation, and inflammatory cell infiltration in kidney and liver tissues, ↓monocytosis in spleen and serum interleukin-6 level |
| Hu et al., 2018 [35] | Lupus mice and blood cells from SLE patients | In vitro experiments | ↓mRNA and protein expression of IFN-γ in mice splenocytes and human SLE PBMCs |
| Kavian et al., 2012 [36] | Hypochlorite induced SSc mice | ↓dermal thickness and collagen deposition in skin and lung tissues | ↓vascular cell adhesion molecule 1 level, autoantibody, IL-4 and IL-13 production by activated T cells; selectively killed activated fibroblasts through ROS generation |
| Kavian et al., 2012 [37] | Induced chronic GVHD in mice | ↓GVHD symptoms, fibrosis of skin and internal organs | ↓activated CD4+ T cells and plasmacytoid dendritic cells (pDCs) through depletion of glutathione |
| Ye et al., 2020 [38] | Blood cells from SSc patients | In vitro experiments | Induced apoptosis of pDCs, preferentially inhibited IFN-α secretion and phosphorylation of the interferon regulatory factor 7, ↓capacity of pDCs to induce CD4+ T cell proliferation, Th1/Th22 polarization and B cell differentiation to plasmablasts |
| Cauvet et al., 2023 [39] | Pre-clinical SSc mice | Improvement in lung histology, trend of reduction in fibrosis markers and strong reduction in vascular remodeling | ↓memory T cells, ↑% of naive T cells in the lungs; downregulated biological pathways associated with activity of the immune pathways, such as T-cell activation, regulation of leucocyte activation, leucocyte cell-cell adhesion, and regulation of lymphocyte activation. |
| Chene et al., 2023 [40] | Hypochlorite induced SSc mice | ↓skin thickening and fibrosis | ↓number of B cells and activation of CD4+ T cells, ↑ROS production and apoptosis of murine fibroblasts |
| Mei et al., 2011 [41] | Collagen induced arthritis in mice | ↓synovial hyperplasia and inflammation in the joints | Enhanced apoptosis of fibroblast-like synoviocytes |
| Li et al., 2019 [42] | Blood and synovial cells from RA patients | In vitro experiments | ↓Th17 differentiation through a reduction of STAT3 mRNA expression, ↑Treg cell generation through an augmentation of Foxp3 expression, downregulated the Th1/Th2 ratio |
| Li et al., 2021 [43] | Treg cells from early RA patients | In vitro experiments | Modulated expression of several genes associated with inflammation, Treg-cell activation and differentiation |
| Niu et al., 2022 [44] | Collagen-induced arthritis in mice | ↓arthritis | Modulated gut microbiota and improved fecal metabolite abnormalities |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




