Submitted:
07 August 2024
Posted:
08 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Canine Sera
2.2. PAX Design and Processing
2.2.1. Allergen Selection
2.2.2. Cartridge Processing
2.2.3. Cross-reactive Carbohydrate Determinant Blocking Strategy
2.2.4. Positivity Threshold Determination
2.3. Validation of the PAX
2.3.1. Verification of the Specificity of the Anti-IgE Monoclonal Antibody
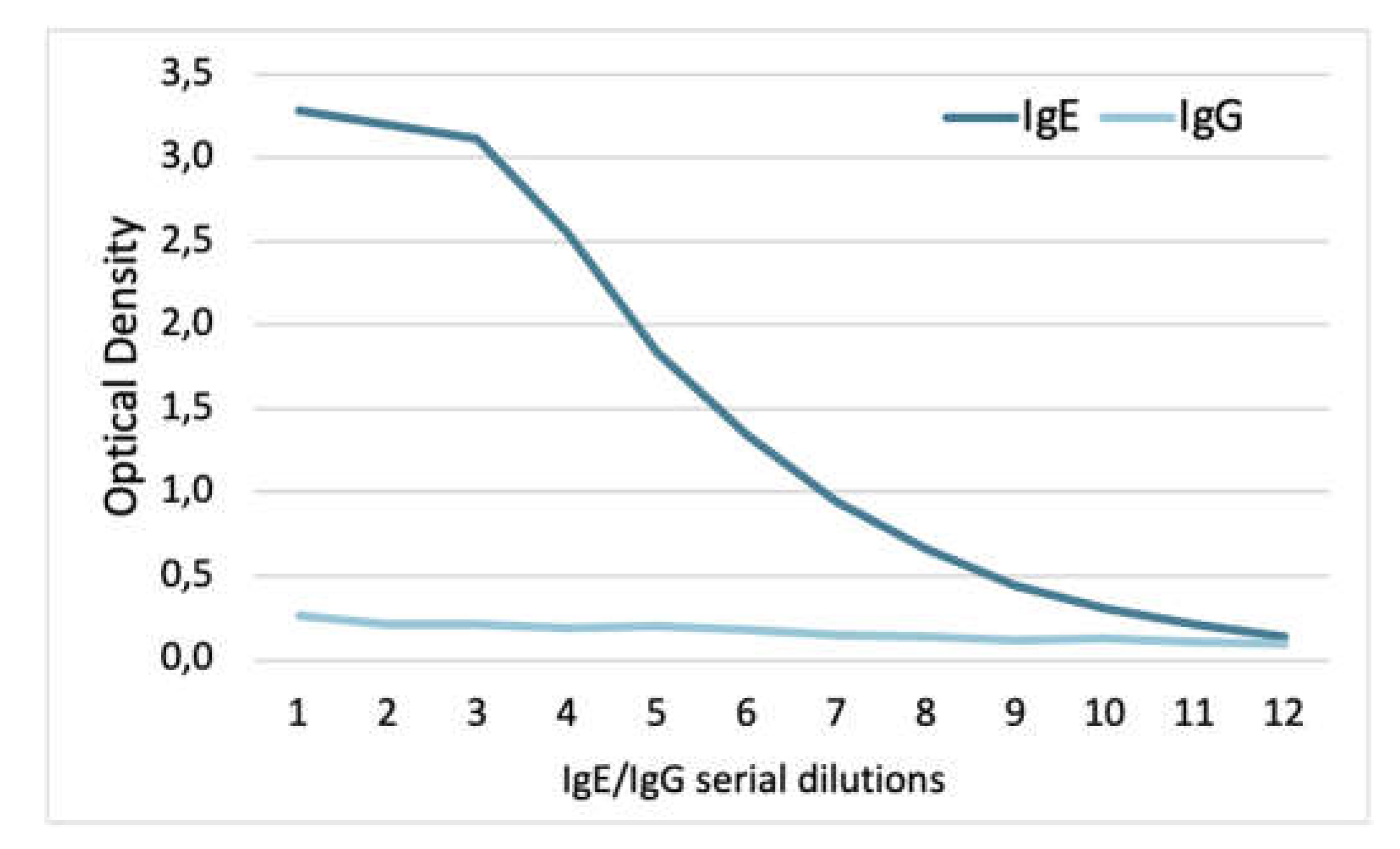
2.3.2. Verification of the Specificity of IgE Binding to Target Allergens
2.3.3. Assay Precision
2.3.4. Internal Consistency
2.3.5. Test Performance Characteristics (Sensitivity, Specificity, Accuracy, Predictive Values)
2.4. Influence of Possible Confounding Factors
2.4.1. Effect of Sample Type
2.4.2. Effect of Hemoglobin, Triglycerides, and Bilirubin
2.4.3. Effect of Sample Storage at Elevated Temperatures
2.4.4. Effect of the CCD-IgE Blocking Strategy
2.5. Sensitization of European Dogs to Environmental Allergens
2.6. Statistics
3. Results
3.1. Positivity Threshold Determination and Positivity Classes
3.1.1. Positivity Threshold Determination
3.1.2. Positivity Classes
- Class 1: 28.00–99.99 ng/mL
- Class 2: 100.00–399.99 ng/mL
- Class 3: 400.00–799.99 ng/mL
- Class 4: ≥ 800.00 ng/mL
3.2. Validation of the PAX
3.2.1. Verification of the Specificity of the Anti-IgE Monoclonal Antibody
3.2.2. Verification of the Specificity of IgE Binding to Target Allergens
3.2.3. Assay Precision
3.2.4. Internal Consistency
3.2.5. Test Performance Characteristics
- Sensitivity = 27/(27+4) = 87.1%
- Specificity = 81/(81+2) = 97.6%
- Accuracy = (27+81)/(27+2+4+81) = 94.7%
- Positive predictive value = 27/(27+2) = 93.1%
- Negative predictive value = 81/(81+4) = 95.3%
3.3. Influence of Possible Confounding Factors
3.3.1. Effect of Sample Type
3.3.2. Effect of Sample Storage at Elevated Temperatures
3.3.3. Effect of Hemoglobin, Triglycerides, and Bilirubin
3.3.4. Effect of CCD-IgE Blocking Strategy
3.4. Sensitization of European Dogs to Environmental Allergens3.4. General Metrics
- Class 1: 73,947 (80.8%)
- Class 2: 12,532 (13.7%)
- Class 3: 2,427 (2.7%)
- Class 4: 2,583 (2.8%)
4. Discussion
4.1. PAX Design
4.1.1. Allergen Selection
4.1.2. IgE Capture Reagent
4.1.3. Positivity Threshold Determination
4.2. PAX Validation
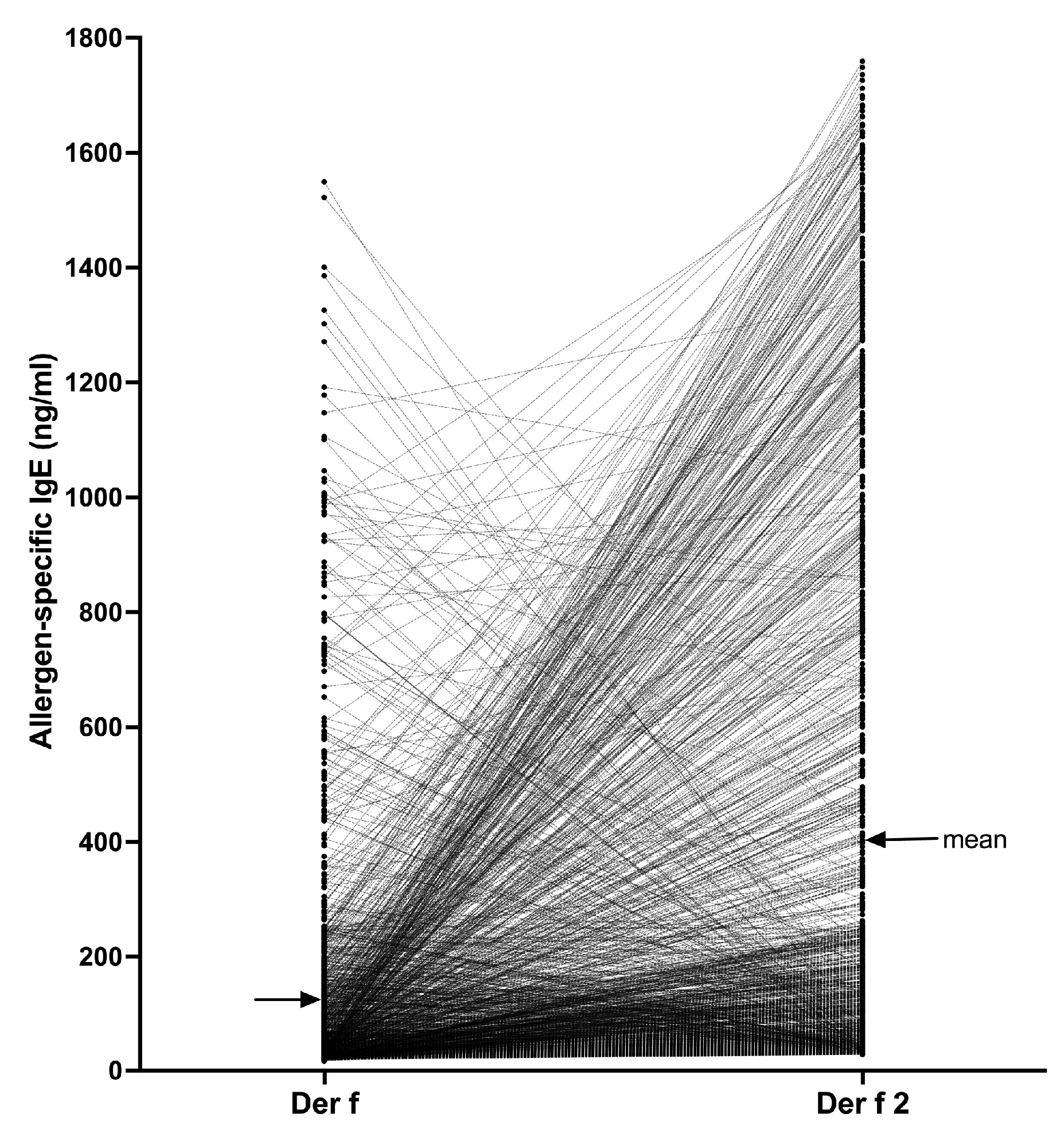
4.2.1. Verification of the Specificity of IgE Binding to Target Allergens
4.2.2. Assay Precision
4.2.3. Internal Consistency
4.2.4. Test Performance Characteristics
4.3. Influence of Possible Confounding Factors
4.4. Sensitization of European Dogs to Environmental Allergens.
4.4.1. Environmental and Venom Allergens
4.4.2. House Dust Mite Allergens
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mueller, R.S.; Jensen-Jarolim, E.; Roth-Walter, F.; Marti, E.; Janda, J.; Seida, A.A.; DeBoer, D. Allergen immunotherapy in people, dogs, cats and horses—Differences, similarities and research needs. Allergy 2018, 73, 1989–1999. [Google Scholar] [CrossRef]
- Di Bona, D.; Plaia, A.; Leto-Barone, M.S.; La Piana, S.; Macchia, L.; Di Lorenzo, G. Efficacy of allergen immunotherapy in reducing the likelihood of developing new allergen sensitizations: A systematic review. Allergy 2017, 72, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Loewenstein, C.; Mueller, R.S. A review of allergen-specific immunotherapy in human and veterinary medicine. Vet Dermatol 2009, 20, 84–98. [Google Scholar] [CrossRef]
- Olivry, T.; DeBoer, D.J.; Favrot, C.; Jackson, H.A.; Mueller, R.S.; Nuttall, T.; Prelaud, P. ; International Committee on Allergic Diseases of Animals Treatment of canine atopic dermatitis: 2015 updated guidelines from the International Committee on Allergic Diseases of Animals (ICADA). BMC Vet Res 2015, 11, 210. [Google Scholar] [CrossRef]
- van Ree, R. Indoor allergens: Relevance of major allergen measurements and standardization. J Allergy Clin Immunol 2007, 119, 270–279. [Google Scholar] [CrossRef]
- Brunetto, B.; Tinghino, R.; Braschi, M.C.; Antonicelli, L.; Pini, C.; Iacovacci, P. Characterization and comparison of commercially available mite extracts for in vivo diagnosis. Allergy 2010, 65, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Perez, R.; Poza-Guedes, P.; Barrios Del Pino, Y.; Matheu, V.; Sanchez-Machin, I. Evaluation of major mite allergens from European standardized commercial extracts for in vivo diagnosis: Addressing the need for precision medicine. Clin Transl Allergy 2019, 9, 14. [Google Scholar] [CrossRef]
- Abrams, S.B.; Brock, G.N.; Palettas, M.; Bolner, M.L.; Moore-Sowers, T.; Plunkett, G.A.; Cole, L.K.; Diaz, S.F.; Lorch, G. An evaluation of veterinary allergen extract content and resultant canine intradermal threshold concentrations. Vet Dermatol 2018, 29, e496–e167. [Google Scholar] [CrossRef] [PubMed]
- Antosegui, I.J.; Melioli, G.; Canonica, G.W.; Gómez, R.M.; Jensen-Jarolim, E.; Ebisawa, M.; Luengo, O. A WAO—ARIA—GA(2)LEN consensus document on molecular-based allergy diagnosis (PAMD@): Update 2020. World Allergy Organ J 2020, 13, 100091. [Google Scholar]
- Chen, J.; Cai, Z.; Fan, D.; Hu, J.; Hou, Y.; He, Y.; Zhang, Z.; Zhao, Z.; Gao, P.; Hu, W.; Sun, J.; Li, J.; Ji, K. Chromosome-level assembly of Dermatophagoides farinae genome and transcriptome reveals two novel allergens Der f 37 and Der f 39. World Allergy Organ J 2021, 14, 100590. [Google Scholar] [CrossRef]
- Moya, R.; Carnes, J.; Sinovas, N.; Ramio, L.; Brazis, P.; Puigdemont, A. Immunoproteomic characterization of a Dermatophagoides farinae extract used in the treatment of canine atopic dermatitis. Vet Immunol Immunopathol 2016, 180, 1–8. [Google Scholar] [CrossRef]
- Dramburg, S.; Hilger, C.; Santos, A.F.; de Las Vecillas, L.; Aalberse, R.C.; Acevedo, N.; Aglas, L.; Altmann, F.; Arruda, K.L.; Asero, R.; Ballmer-Weber, B.; Barber, D.; Beyer, K.; Biedermann, T.; Bilo, M.B.; Blank, S.; Bosshard, P.P.; Breiteneder, H.; Brough, H.A.; Bublin, M.; Campbell, D.; Caraballo, L.; Caubet, J.C.; Celi, G.; Chapman, M.D.; Chruszcz, M.; Custovic, A.; Czolk, R.; Davies, J.; Douladiris, N.; Eberlein, B.; Ebisawa, M.; Ehlers, A.; Eigenmann, P.; Gadermaier, G.; Giovannini, M.; Gomez, F.; Grohman, R.; Guillet, C.; Hafner, C.; Hamilton, R.G.; Hauser, M.; Hawranek, T.; Hoffmann, H.J.; Holzhauser, T.; Iizuka, T.; Jacquet, A.; Jakob, T.; Janssen-Weets, B.; Jappe, U.; Jutel, M.; Kalic, T.; Kamath, S.; Kespohl, S.; Kleine-Tebbe, J.; Knol, E.; Knulst, A.; Konradsen, J.R.; Korosec, P.; Kuehn, A.; Lack, G.; Le, T.; Lopata, A.; Luengo, O.; Makela, M.; Marra, A.M.; Mills, C.; Morisset, M.; Muraro, A.; Nowak-Wegrzyn, A.; Nugraha, R.; Ollert, M.; Palosuo, K.; Pastorello, E.A.; Patil, S.U.; Platts-Mills, T.; Pomes, A.; Poncet, P.; Potapova, E.; Poulsen, L.K.; Radauer, C.; Radulovic, S.; Raulf, M.; Rouge, P.; Sastre, J.; Sato, S.; Scala, E.; Schmid, J.M.; Schmid-Grendelmeier, P.; Schrama, D.; Senechal, H.; Traidl-Hoffmann, C.; Valverde-Monge, M.; van Hage, M.; van Ree, R.; Verhoeckx, K.; Vieths, S.; Wickman, M.; Zakzuk, J.; Matricardi, P.M.; Hoffmann-Sommergruber, K. EAACI Molecular Allergology User’s Guide 2.0. Pediatr Allergy Immunol 2023, 34, e13854. [Google Scholar] [CrossRef]
- Heffler, E.; Puggioni, F.; Peveri, S.; Montagni, M.; Canonica, G.W.; Melioli, G. Extended IgE profile based on an allergen macroarray: A novel tool for precision medicine in allergy diagnosis. World Allergy Organ J 2018, 11, 7. [Google Scholar] [CrossRef]
- Scala, E.; Caprini, E.; Abeni, D.; Meneguzzi, G.; Buzzulini, F.; Cecchi, L.; Villalta, D.; Asero, R. A qualitative and quantitative comparison of IgE antibody profiles with two multiplex platforms for component-resolved diagnostics in allergic patients. Clin Exp Allergy 2021, 51, 1603–1612. [Google Scholar] [CrossRef]
- Buzzulini, F.; Da Re, M.; Scala, E.; Martelli, P.; Conte, M.; Brusca, I.; Villalta, D. Evaluation of a new multiplex assay for allergy diagnosis. Clin Chim Acta 2019, 493, 73–78. [Google Scholar] [CrossRef]
- Scala, E.; Caprini, E.; Abeni, D.; Meneguzzi, G.; Buzzulini, F.; Cecchi, L.; Villalta, D.; Asero, R. A qualitative and quantitative comparison of IgE antibody profiles with two multiplex platforms for component-resolved diagnostics in allergic patients. Clin Exp Allergy 2021, 51, 1603–1612. [Google Scholar] [CrossRef]
- Bojcukova, J.; Vlas, T.; Forstenlechner, P.; Panzner, P. Comparison of two multiplex arrays in the diagnostics of allergy. Clinical and Translational Allergy 2019, 9, 31. [Google Scholar] [CrossRef]
- Koch, L.; Laipold, N.; Arzt-Gradwohl, L.; Cerpes, U.; Sturm, E.M.; Aberer, W.; Sturm, G.J. IgE multiplex testing in house dust mite allergy is utile, and sensitivity is comparable to extract-based singleplex testing. Allergy 2020, 75, 2091–2094. [Google Scholar] [CrossRef]
- Platteel, A.C.M.; van der Pol, P.; Murk, J.; Verbrugge-Bakker, I.; Hack-Steemers, M.; Roovers, T.H.W.M.; Heron, M. A comprehensive comparison between ISAC and ALEX(2) multiplex test systems. Clin Chem Lab Med 2022, 60, 1046–1052. [Google Scholar] [CrossRef]
- Sonneveld, L.J.H.; Emons, J.A.M.; Arends, N.J.T.; Landzaat, L.J.; Veenbergen, S.; Schreurs, M.W.J. ALEX versus ISAC multiplex array in analyzing food allergy in atopic children. Clin Mol Allergy 2022, 20, 10. [Google Scholar] [CrossRef]
- Thouvenot, B.; Roitel, O.; Tomasina, J.; Hilselberger, B.; Richard, C.; Jacquenet, S.; Codreanu-Morel, F.; Morisset, M.; Kanny, G.; Beaudouin, E.; Delebarre-Sauvage, C.; Olivry, T.; Favrot, C.; Bihain, B. Transcriptional frameshifts contribute to protein allergenicity. J Clin Invest 2020, 21, 126275. [Google Scholar] [CrossRef]
- Martin, A.; Sierra, M.P.; Gonzalez, J.L.; Arevalo, M.A. Identification of allergens responsible for canine cutaneous adverse food reactions to lamb, beef and cow’s milk. Vet Dermatol 2004, 15, 349–356. [Google Scholar] [CrossRef]
- Bexley, J.; Kingswell, N.; Olivry, T. Serum IgE cross-reactivity between fish and chicken meats in dogs. Vet Dermatol 2019, 30, e25–e28. [Google Scholar] [CrossRef]
- Olivry, T.; Pucheu-Haston, C.M.; Mayer, U.; Bergvall, K.; Bexley, J. Identification of major and minor chicken allergens in dogs. Vet Dermatol 2022, 33, e46–e16. [Google Scholar] [CrossRef]
- Chruszcz, M.; Kapingidza, A.B.; Dolamore, C.; Kowal, K. A robust method for the estimation and visualization of IgE cross-reactivity likelihood between allergens belonging to the same protein family. PLoS ONE 2018, 13, e0208276. [Google Scholar] [CrossRef]
- Olivry, T.; O’Malley, A.; Chruszcz, M. Evaluation of the theoretical risk of cross-reactivity among recently identified food allergens for dogs. Vet Dermatol 2022, 33, 523–526. [Google Scholar] [CrossRef]
- Hammerberg, B.; Bevier, D.; DeBoer, D.J.; Olivry, T.; Orton, S.M.; Gebhard, D.; Vaden, S.L. Auto IgG anti-IgE and IgG x IgE immune complex presence and effects on ELISA-based quantitation of IgE in canine atopic dermatitis, demodectic acariasis and helminthiasis. Vet Immunol Immunopathol 1997, 60, 33–46. [Google Scholar] [CrossRef]
- Altmann, F. Coping with cross-reactive carbohydrate determinants in allergy diagnosis. Allergo J Int 2016, 25, 98–105. [Google Scholar] [CrossRef]
- Leistungsbewertung von In-vitro-Diagnostika; Deutsche Fassung EN 13612:2002. Available online: https://www.austrian-standards.at/de/shop/din-en-13612-2002-08~p3038461 (accessed on 6 August 2024).
- Pierson-Perry, J.F. CLSI EP17: Evaluation of detection capability for clinical laboratory measurement procedures, 2nd ed.; CLSI: 2012.
- Hamilton, R.G.; Matsson, P.N.J. CLSI I/LA20: Analytical performance characteristics, quality assurance, and clinical utility of immunological assays for human immunoglobulin E antibodies of defined allergen specificities, 3rd. ed.; CLSI: 2016.
- Gebhard, D.; Orton, S.; Edmiston, D.; Nakagaki, K.; DeBoer, D.; Hammerberg, B. Canine IgE monoclonal antibody specific for a filarial antigen: Production by a canine x murine heterohybridoma using B cells from a clinically affected lymph node. Immunology 1995, 85, 429–434. [Google Scholar]
- Orton, S.M.; Arasu, P.; Hammerberg, B. A novel gene from Brugia sp. that encodes a cytotoxic fatty acid binding protein allergen recognized by canine monoclonal IgE and serum IgE from infected dogs. J Parasitol 2007, 93, 1378–1387. [Google Scholar] [CrossRef]
- Botoni, L.S.; Torres, S.M.F.; Koch, S.N.; Heinemann, M.B.; Costa-Val, A.P. Comparison of demographic data, disease severity and response to treatment, between dogs with atopic dermatitis and atopic-like dermatitis: A retrospective study. Vet Dermatol 2019, 30, 10–e4. [Google Scholar] [CrossRef]
- Thorpe, M.; Moverare, R.; Fischer, C.; Lidholm, J.; Rudengren, M.; Borres, M.P. History and utility of specific IgE cutoff levels: What is the relevance for allergy diagnosis? J Allergy Clin Immunol Pract 2023, 11, 3021–3029. [Google Scholar] [CrossRef]
- Zwickl, L.L.M.N.; Joekel, D.E.; Fischer, N.M.; Rostaher, A.; Thamsborg, K.; Deplazes, P.; Favrot, C. Total and Toxocara canis larval excretory/secretory antigen- and allergen-specific IgE in atopic and non-atopic dogs. Vet Dermatol 2018, 29, 222–e80. [Google Scholar] [CrossRef]
- Okayama, T.; Matsuno, Y.; Yasuda, N.; Tsukui, T.; Suzuta, Y.; Koyanagi, M.; Sakaguchi, M.; Ishii, Y.; Olivry, T.; Masuda, K. Establishment of a quantitative ELISA for the measurement of allergen-specific IgE in dogs using anti-IgE antibody cross-reactive to mouse and dog IgE. Vet Immunol Immunopathol 2011, 139, 99–106. [Google Scholar] [CrossRef]
- Lee, K.W.; Blankenship, K.D.; McCurry, Z.M.; Esch, R.E.; DeBoer, D.J.; Marsella, R. Performance characteristics of a monoclonal antibody cocktail-based ELISA for detection of allergen-specific IgE in dogs and comparison with a high affinity IgE receptor-based ELISA. Vet Dermatol 2009, 20, 157–164. [Google Scholar] [CrossRef]
- Rostaher, A.; Fischer, N.M.; Vigani, A.; Steblaj, B.; Martini, F.; Brem, S.; Favrot, C.; Kosnik, M. Hymenoptera venom immunotherapy in dogs: Safety and clinical efficacy. Animals (Basel) 2023, 13, 3002. [Google Scholar] [CrossRef]
- Christian, M.; Wagner, R.; Kahnt, E. Pilot study on the storage stability of allergen- specific immunoglobulin (Ig)e against house dust and storage mites in serum of dogs (abstract). Vet Dermatol 2024, 35, 105. [Google Scholar]
- Christian, M.; Wagner, R.; Müller, E. Storage stability of allergen-specific immunoglobulin E against house dust and storage mites in sera of dogs with a focus on warm temperature and exposure to sunlight (abstract). Vet Dermatol 2024, 35, 50. [Google Scholar]
- Platts-Mills, T.A.; Hilger, C.; Jappe, U.; van Hage, M.; Gadermaier, G.; Spillner, E.; Lidholm, J.; Keshavarz, B.; Aalberse, R.C.; van Ree, R.; Goodman, R.E.; Pomes, A. Carbohydrate epitopes currently recognized as targets for IgE antibodies. Allergy 2021, 76, 2383–2394. [Google Scholar] [CrossRef]
- Favrot, C.; Jacquenet, S.; Fischer, N.; Rostaher, A.; Ogier, V.; Bihain, B. Toxocara canis infestations influence seroconversions to Dermatophagoides farinae IgE (abstract). Vet Dermatol 2017, 28, 539. [Google Scholar]
- Olivry, T.; Jacquenet, S.; Mas-Fontao, A.; Morel, B.; Faye, L.; Gomord, V.; Favrot, C. Investigations on the cross-reactivity between the Dermatophagoides farinae house dust mite and the canine roundworm Toxocara canis (abstract). Vet Dermatol 2023, 34, 95. [Google Scholar]
- Burzynska, M.; Piasecka-Kwiatkowska, D. A Review of honeybee venom allergens and allergenicity. Int J Mol Sci 2021, 22, 8371. [Google Scholar] [CrossRef]
- Jakob, T.; Muller, U.; Helbling, A.; Spillner, E. Component resolved diagnostics for Hymenoptera venom allergy. Curr Opin Allergy Clin Immunol 2017, 17, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Bolte, A.; Czajkowski, T.; Kompa, T. The north-eastern distribution range of European beech—A review. Forestry 2007, 80, 413–429. [Google Scholar] [CrossRef]
- Huang, H.; Sarzsinszky, E.; Vrtala, S. House dust mite allergy: The importance of house dust mite allergens for diagnosis and immunotherapy. Mol Immunol 2023, 158, 54–67. [Google Scholar] [CrossRef] [PubMed]
- McCall, C.; Hunter, S.; Stedman, K.; Weber, E.; Hillier, A.; Bozic, C.; Rivoire, B.; Olivry, T. Characterization and cloning of a major high molecular weight house dust mite allergen (Der f 15) for dogs. Vet Immunol Immunopathol 2001, 78, 231–247. [Google Scholar] [CrossRef]
- Weber, E.; Hunter, S.; Stedman, K.; Dreitz, S.; Olivry, T.; Hillier, A.; McCall, C. Identification, characterization, and cloning of a complementary DNA encoding a 60-kd house dust mite allergen (Der f 18) for human beings and dogs. J Allergy Clin Immunol 2003, 112, 79–86. [Google Scholar] [CrossRef]
- Olivry, T.; Mas Fontao, A.; Jacquenet, S.; Aumayr, M.; Tsukui, T.; Gomord, V.; Faye, L.; Favrot, C. Identification of cross-reactive allergens between the Dermatophagoides farinae house dust mite and the Toxocara canis nematode in dogs with suspected allergies. Vet Dermatol 2024, in press. [Google Scholar]


| Class 1 | Class 2 | Class 3 | Class 4 | |
|---|---|---|---|---|
| Recovery percentage | 99.5% [98.1-101.0] |
99.0% [95.5-102.6] |
99.6% [84.1-115.0] |
98.9% [96.5-101.3] |
| Coefficient of variation | 4.2% [3.5-4.8] |
4.4% [2.3-6.4] |
9.7% [0.0-19.9] |
3.0% [1.8-4.2%] |
| Allergic Dogs | Healthy Dogs | |
|---|---|---|
| Der f sIgE ≥ 28.00 ng/mL | 27 | 2 |
| Der f sIgE < 28.00 ng/mL | 4 | 81 |
| Citrate | Heparin | EDTA | |
|---|---|---|---|
| Recovery percentage | 91.3% [88.3-94.2] | 100.5% [93.3-107.7] | 106.6% [89.4-123.9] |
| Coefficient of variation | 6.8% [4.6-9.1] | 7.7% [4.5-10.8] | 13.3% [7.2-19.4] |
| Day 3 | Day 7 | Day 14 | Day 21 | Day 28 | |
|---|---|---|---|---|---|
| Recovery percentage | 99.7% [95.1-104.2] |
104.5% [97.8-111.2] |
101.9% [94.5-109.3] |
103.1% [95.5-110.7] |
98.4% [91.6-105.2] |
| Coefficient of variation | 7.0% [4.8-9.3] |
10.0% [6.5-13.5] |
11.5% [7.9-15.0] |
10.9% [6.7-15.1%] |
10.4% [6.4-14.3%] |
| Day 3 | Day 7 | Day 14 | Day 21 | Day 28 | |
|---|---|---|---|---|---|
| Recovery percentage | 99.0% [92.4-105.7] |
98.5% [91.2-105.8] |
103.9% [93.7-114.0] |
96.6% [86.6-106.5] |
96.8% [85.5-108.1] |
| Coefficient of variation | 9.7% [6.0-13.4] |
4.5% [6.0-15.0] |
15.7% [10.9-20.5] |
17.0% [11.8-22.2%] |
18.9% [12.4-25.4%] |
| Hemolysis | Lipemia | Bilirubinemia | |
|---|---|---|---|
| Recovery percentage | 93.2% [86.3-100.2] | 99.1% [96.1-102.0] | 96.9% [93.0-100.8] |
| Coefficient of variation | 10.7% [6.4-14.9] | 4.4% [3.3-5.6] | 5.9% [4.1-7.7] |
| After the first CCD block |
After the second CCD block |
|
|---|---|---|
| Reduction in positive numbers | 54.9% [41.5–58.4] | 72.1% [56.0–88.1] |
| Reduction in positive IgE level | 33.5% [16.7–50.4] | 50.5% [28.4–72.5] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




