Submitted:
07 August 2024
Posted:
08 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
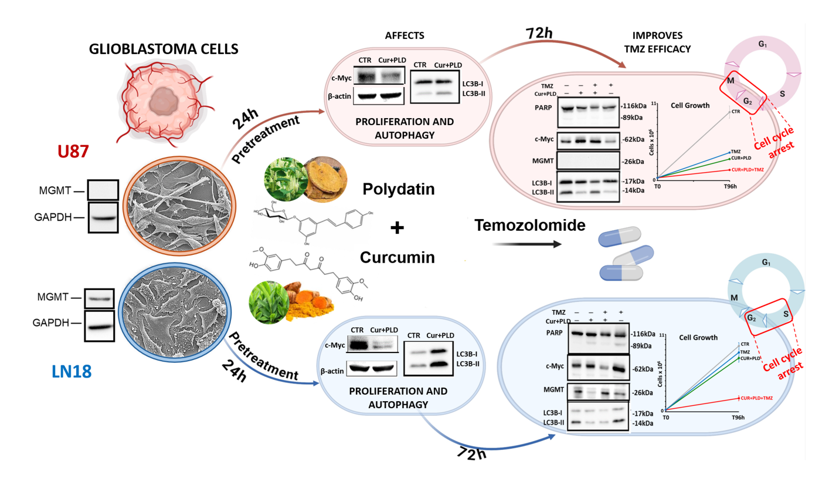
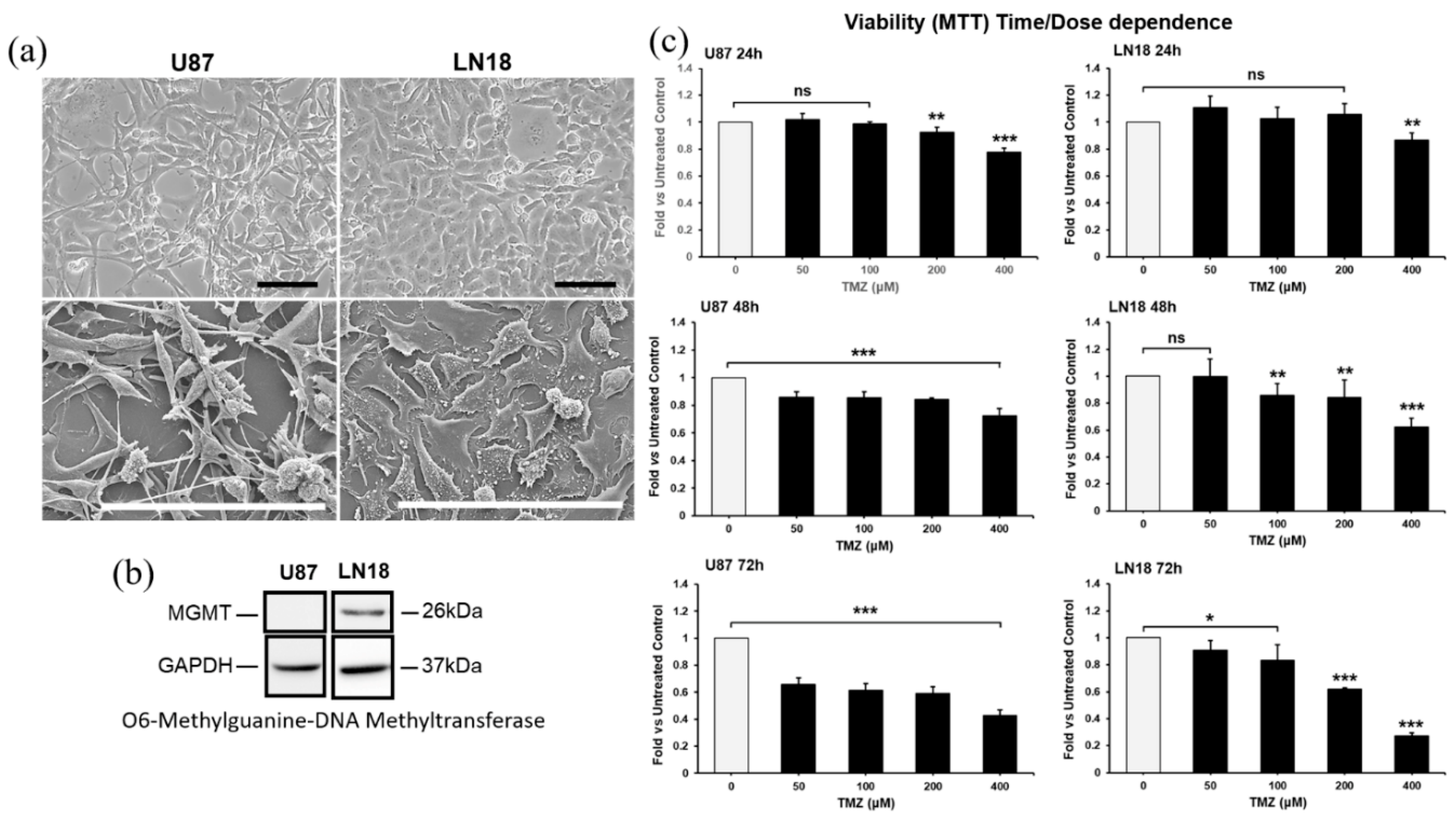
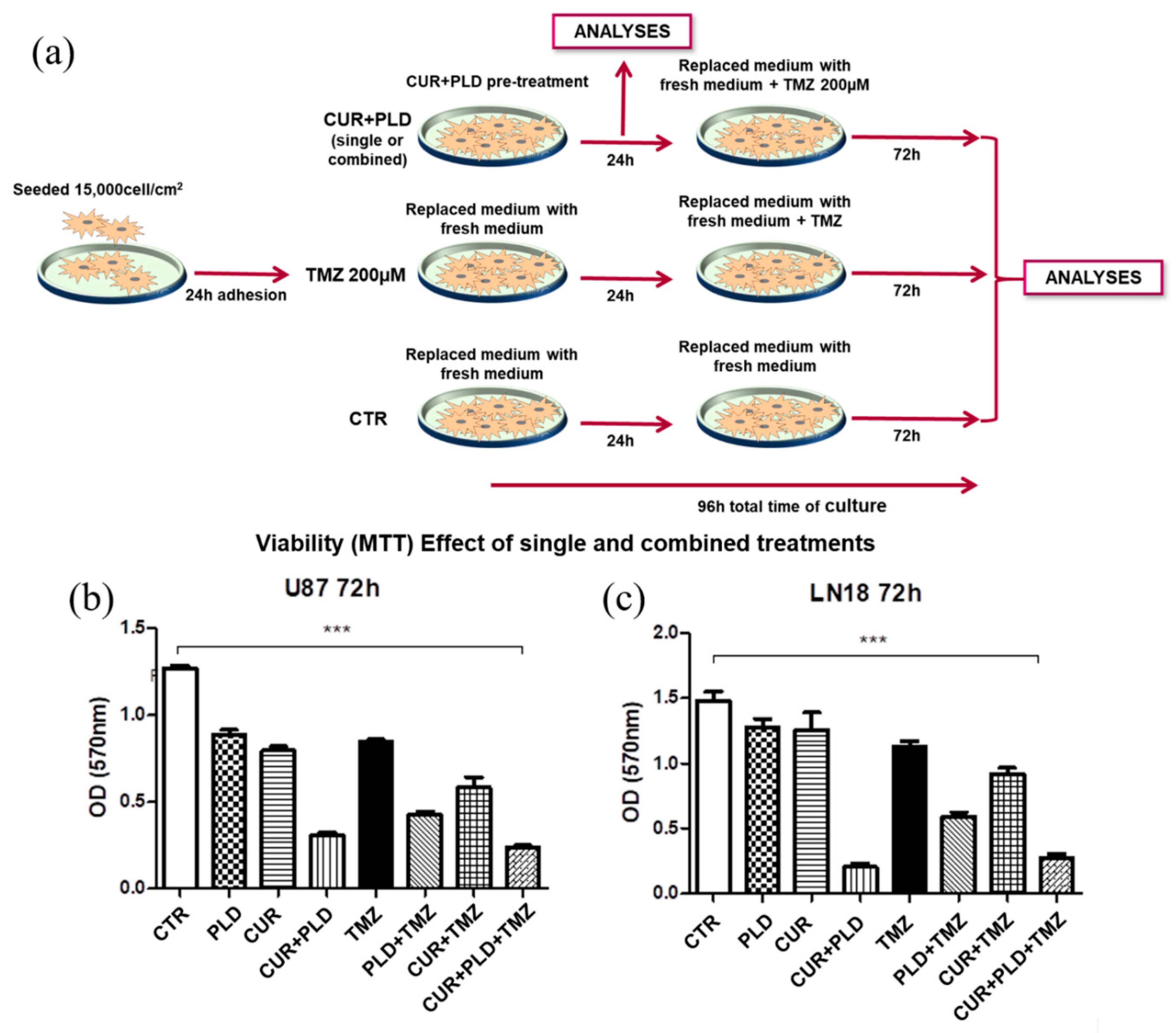
2.1. Establishment of the Dose- and Time-Dependent Response to TMZ, and of the Synergic Concentrations of CUR and PLD in U87 and LN18 Glioblastoma Cells
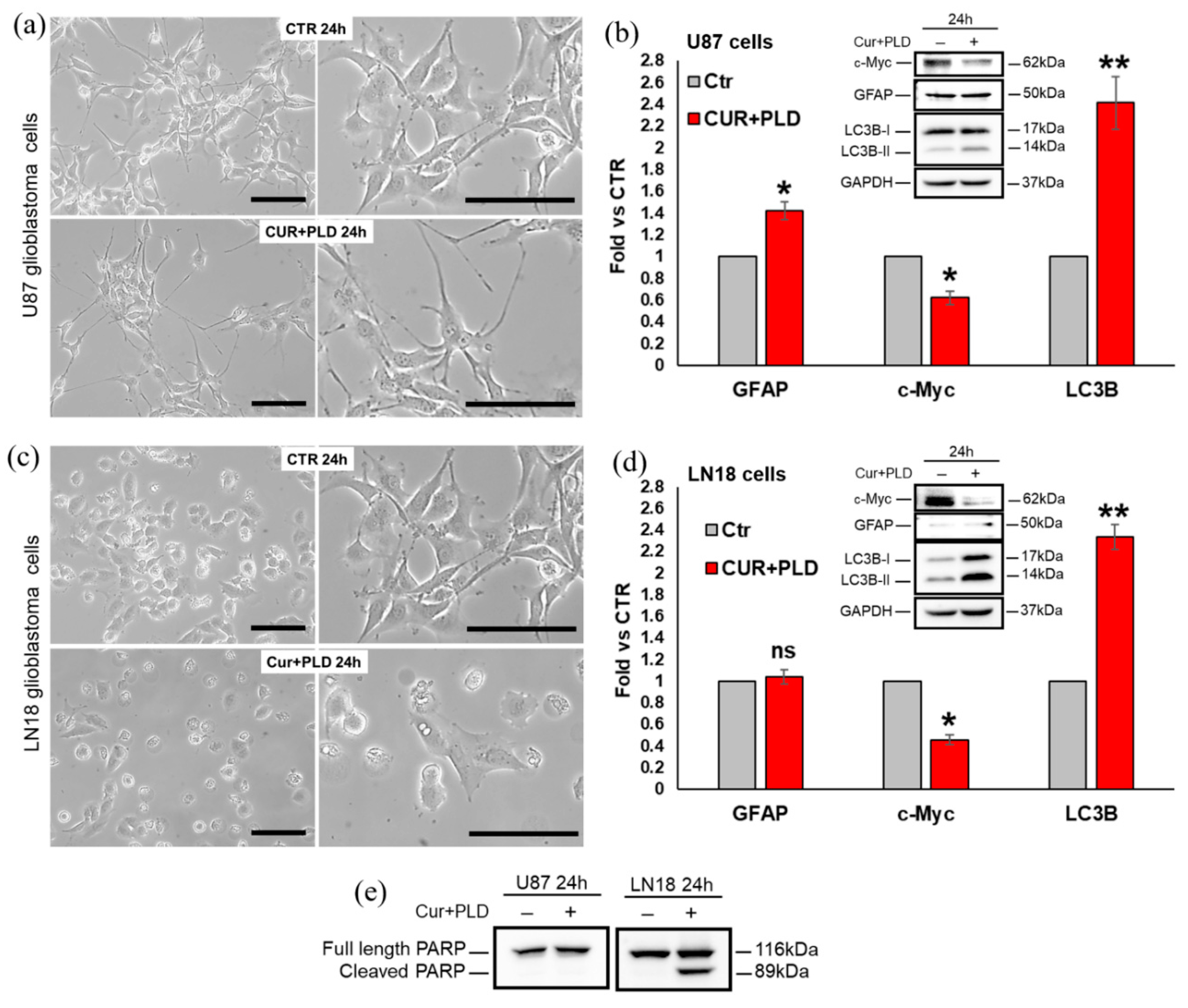
2.2. Effects of the Synergic Combination of CUR+PLD on U87 and LN18 Glioblastoma Cells before TMZ Treatment
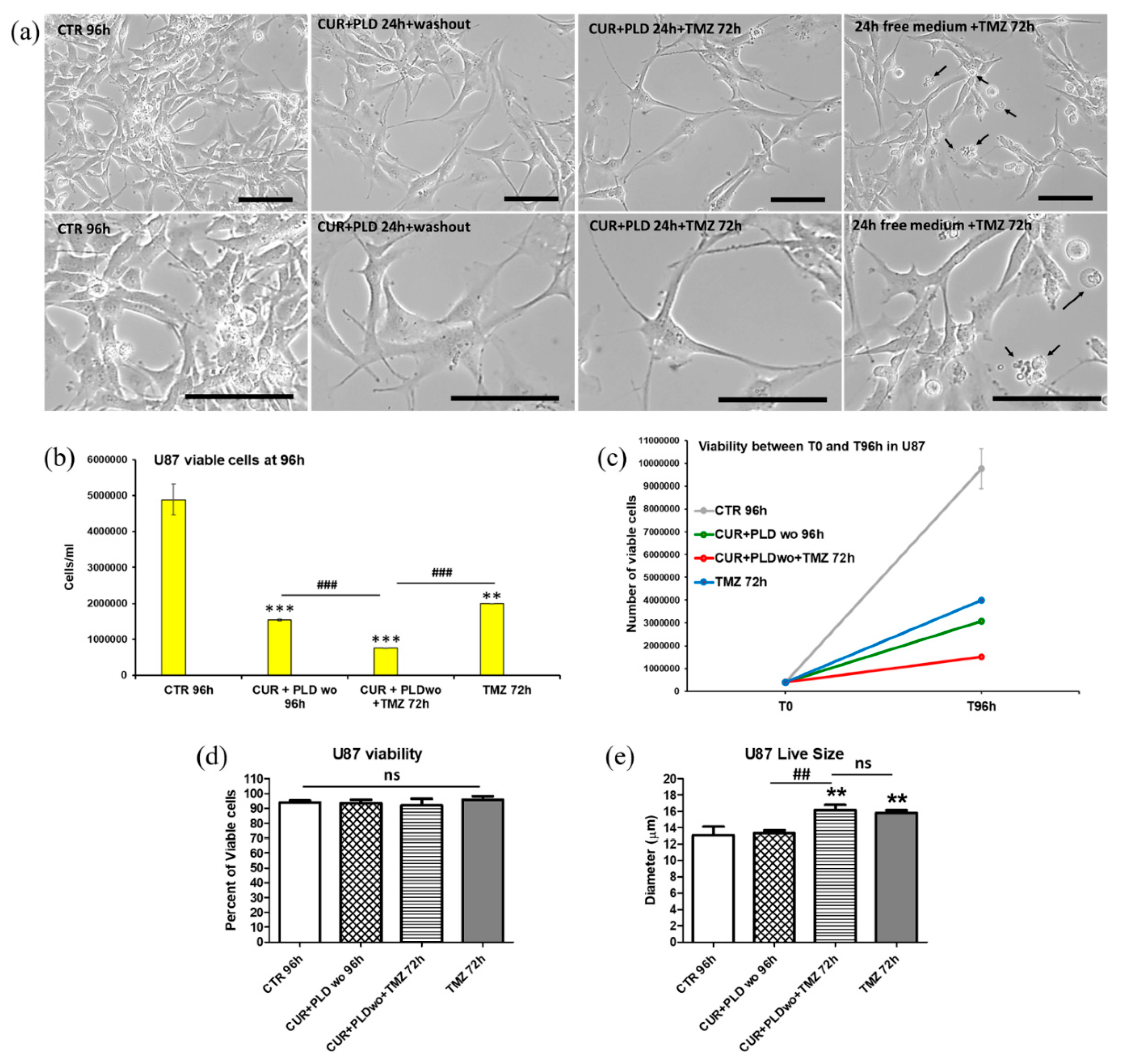
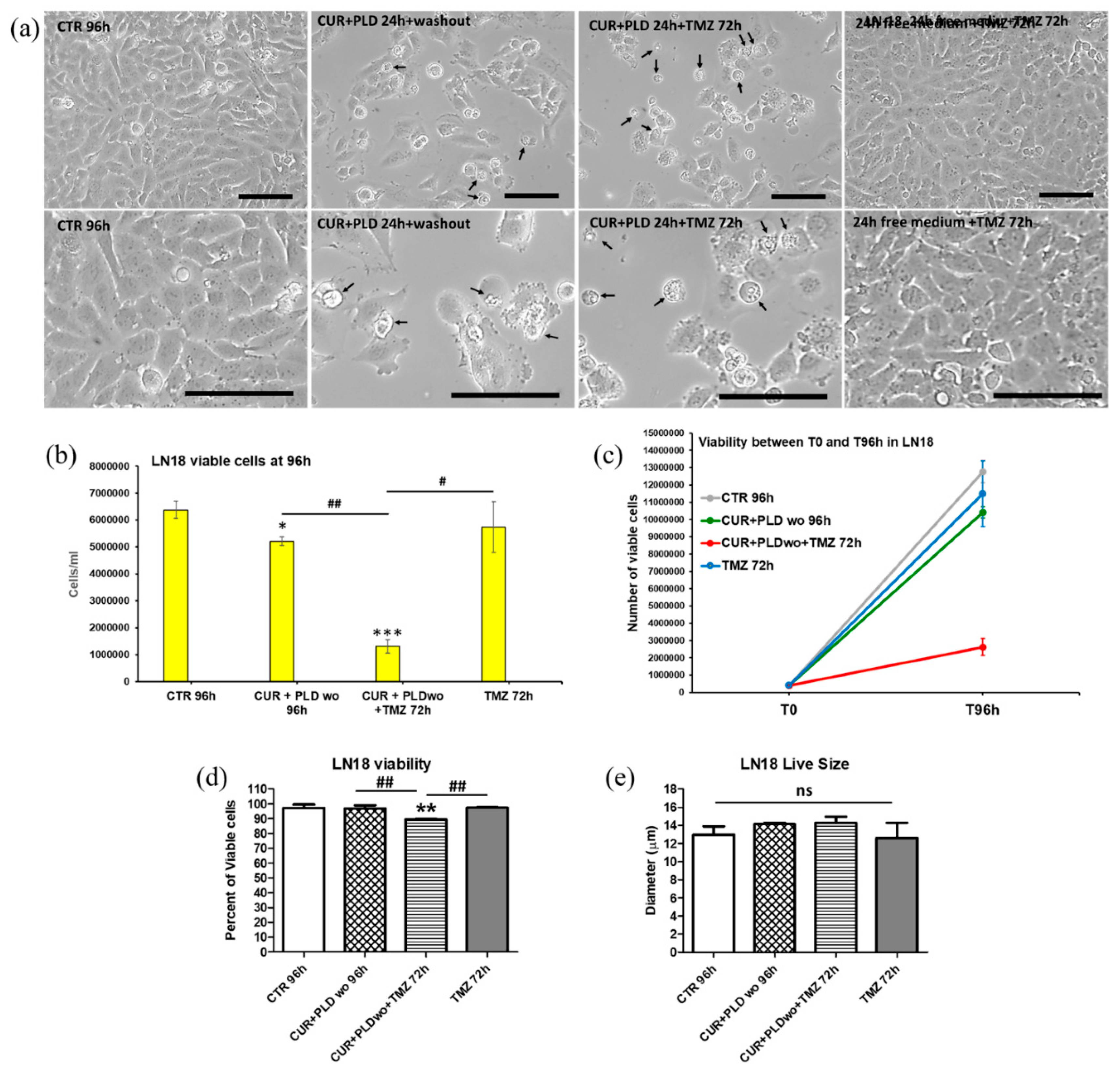
2.3. Pretreatment with CUR+PLD Significantly Improved the Effect of TMZ Treatment on the Number of Live U87 and LN18 Glioblastoma Cells
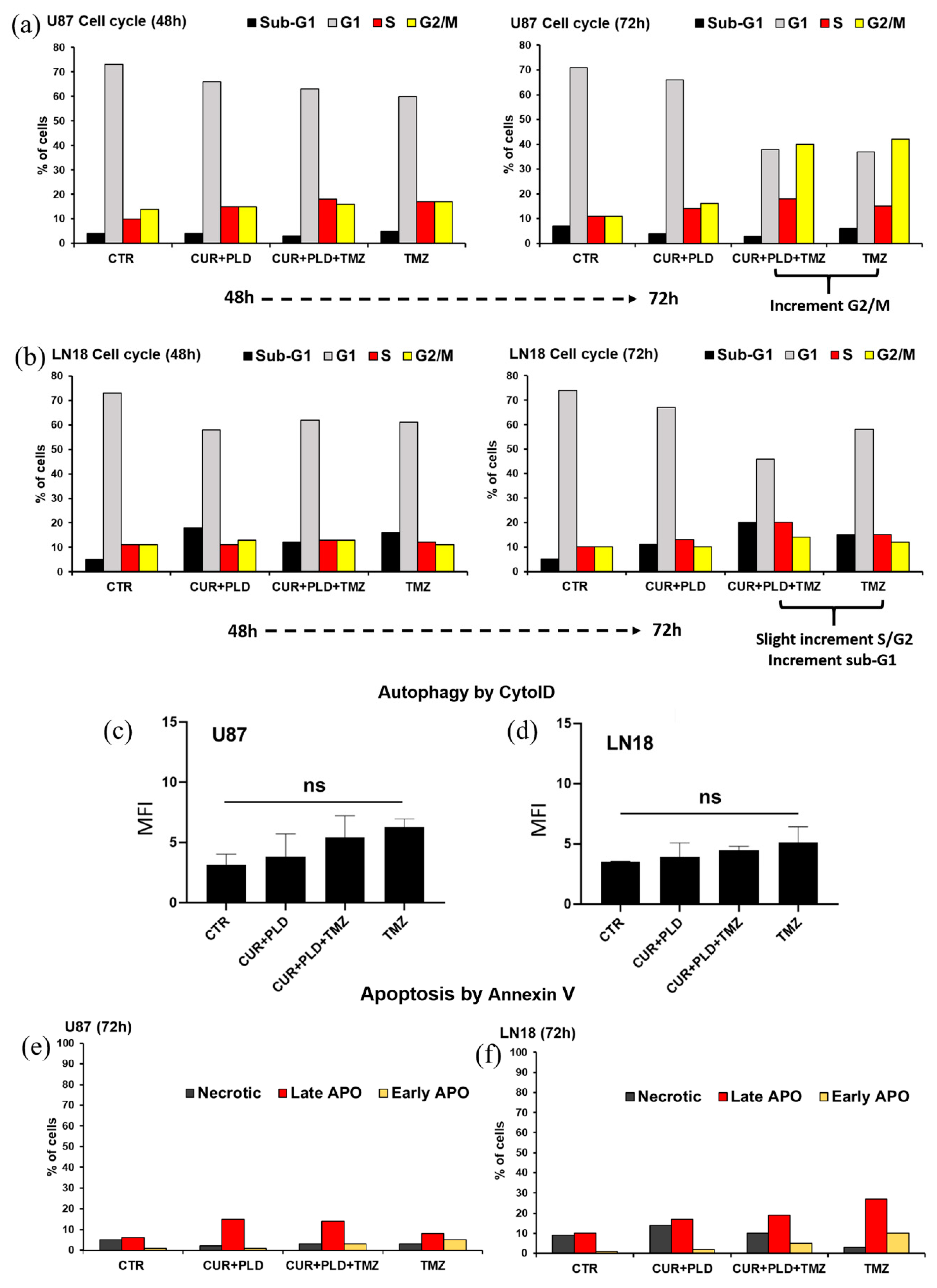
2.4. Pretreatment with CUR+PLD Induced a Cell Cycle Arrest between the S and G2/M Phases
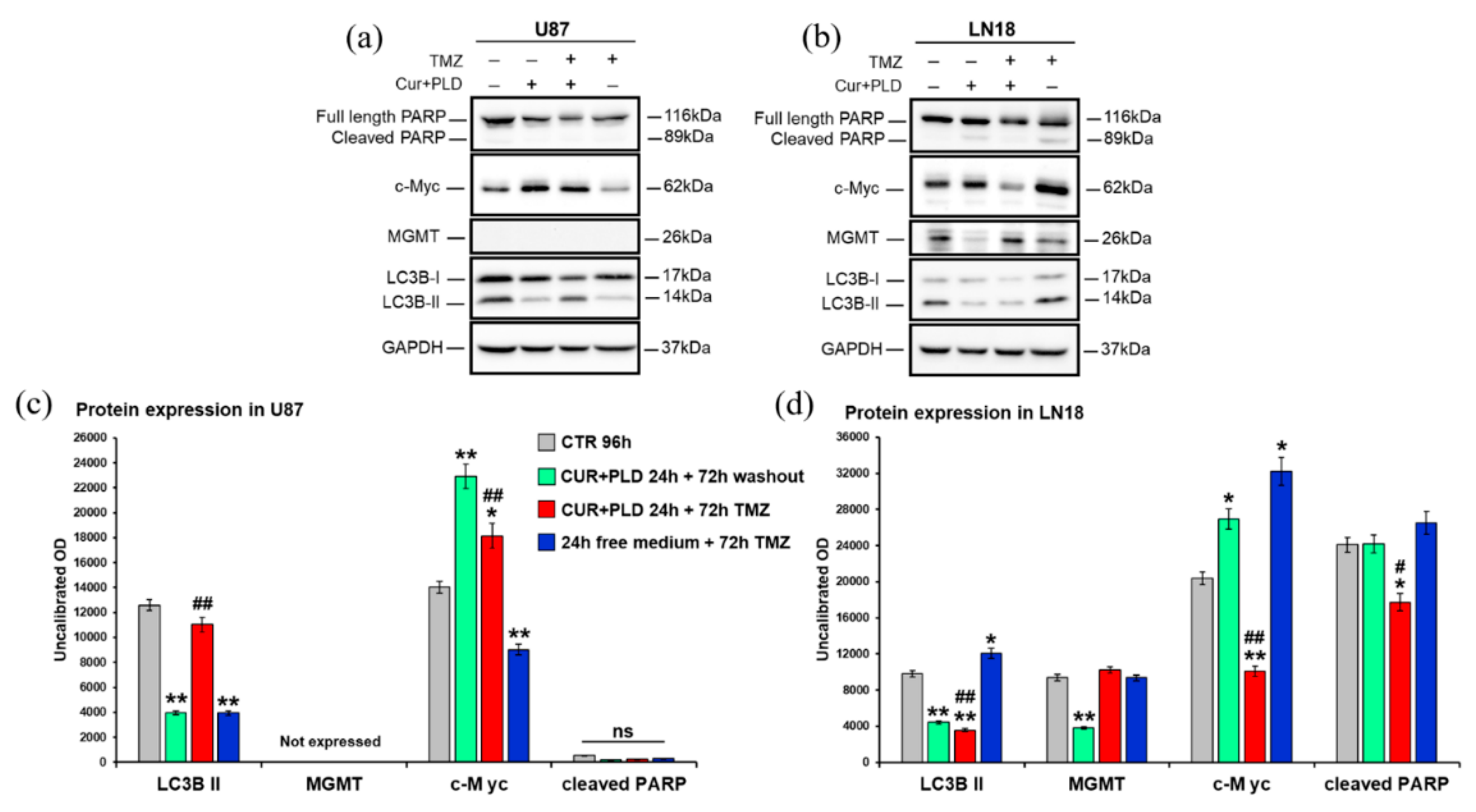
2.5. Effects of the Synergic Combination of CUR+PLD on the expression of MGMT and Proteins Related to Cell Proliferation, Apoptosis, or Autophagy
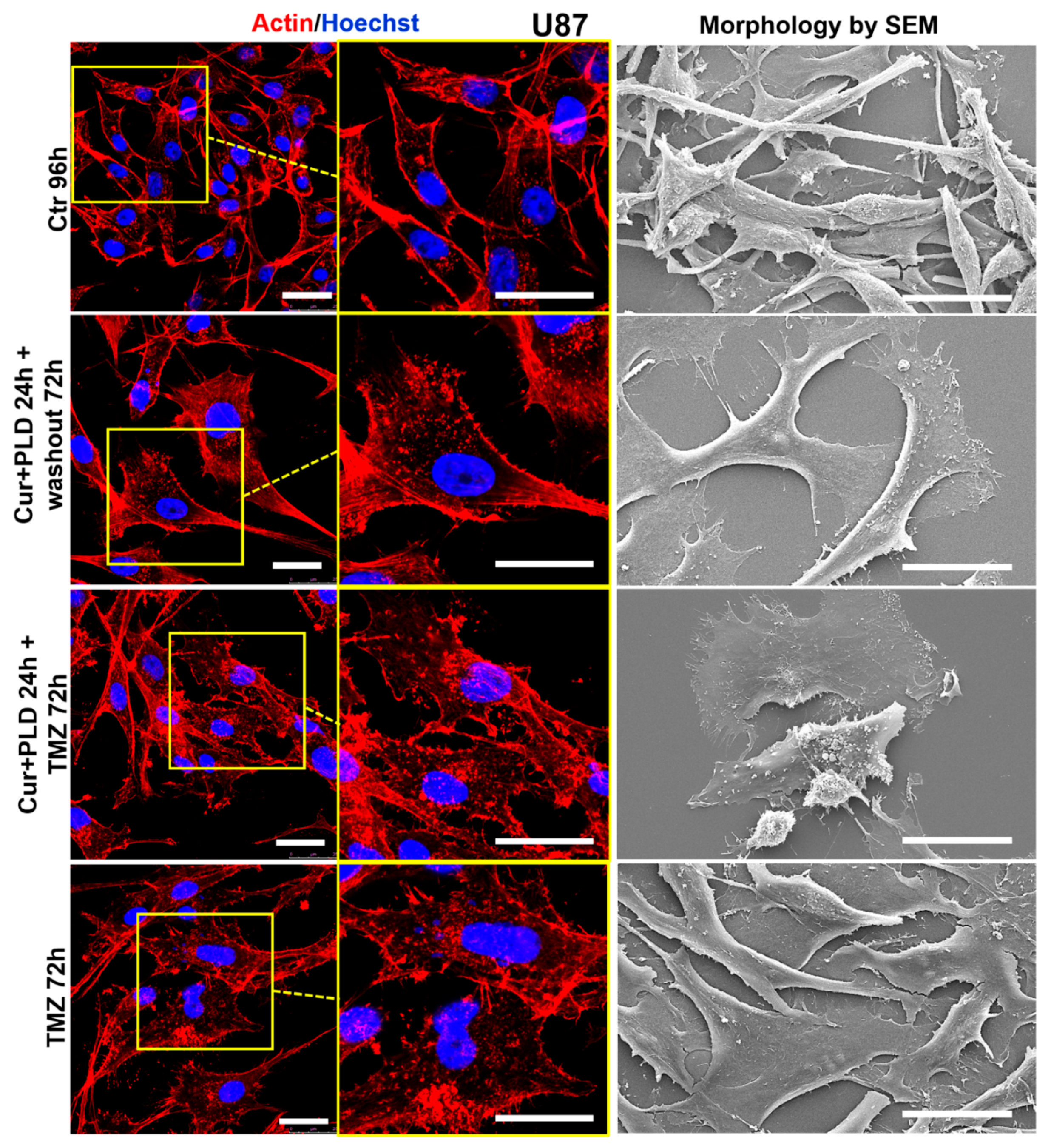
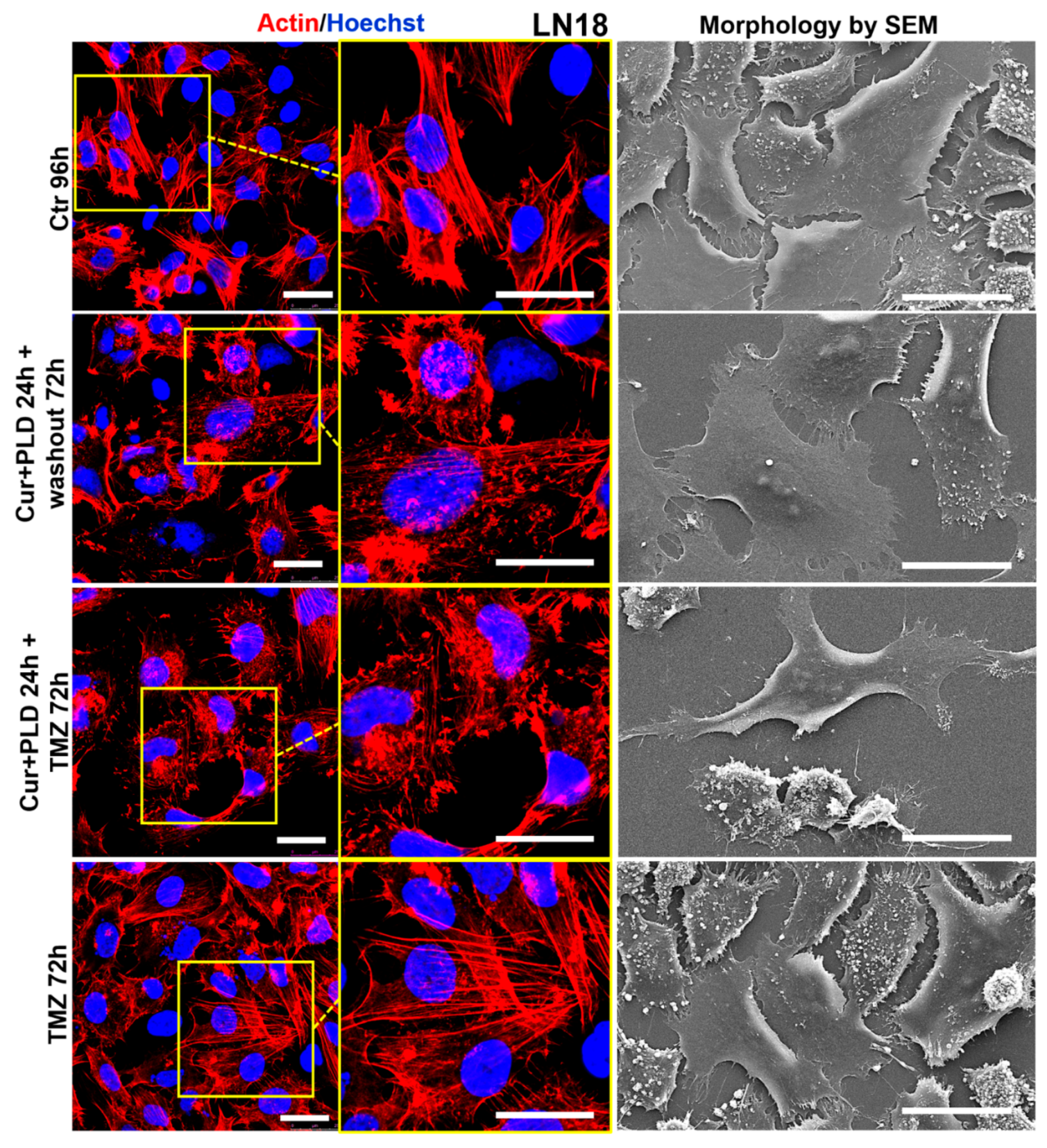
2.6. Pretreatment with CUR+PLD Alters the Actin Network and Ultrastructural Morphology of U87 and LN18 Cells
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Cultures
4.3. Cell Treatments and Cell Viability Analyses
4.4. Evaluation of Cell Morphology
4.5. Western Blot (WB) Analysis
4.6. Confocal Laser Scanning Microscopy (CLSM)
4.7. Cytofluorimetric Analyses of Cell Cycle, Apoptosis, and Autophagy
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Esteller, M.; Garcia-Foncillas, J.; Andion, E.; Goodman, S. N.; Hidalgo, O. F.; Vanaclocha, V.; Baylin, S. B.; Herman, J. G., Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. The New England journal of medicine 2000, 343 (19), 1350-4. [CrossRef]
- Hegi, M.E.; Diserens, A.-C.; Godard, S.; Dietrich, P.-Y.; Regli, L.; Ostermann, S.; Otten, P.; Van Melle, G.; de Tribolet, N.; Stupp, R. Clinical Trial Substantiates the Predictive Value of O-6-Methylguanine-DNA Methyltransferase Promoter Methylation in Glioblastoma Patients Treated with Temozolomide. Clinical cancer research: an official journal of the American Association for Cancer Research 2004, 10, 1871–1874. [CrossRef]
- Tan, A.C.; Ashley, D.M.; López, G.Y.; Malinzak, M.; Friedman, H.S.; Khasraw, M. Management of glioblastoma: State of the art and future directions. CA Cancer J. Clin. 2020, 70, 299–312. [CrossRef]
- Sferrazza G, C. M., Brusotti G, Pierimarchi P, Temporini C, Serafino A, Calleri E, Nature-derived compounds modulating Wnt/β-catenin pathway: a preventive and therapeutic opportunity in neoplastic diseases. Acta Pharmaceutica Sinica B 2020, 10 (10), 1814-1834. [CrossRef]
- Pulido-Moran, M.; Moreno-Fernandez, J.; Ramirez-Tortosa, C.; Ramirez-Tortosa, M., Curcumin and Health. Molecules 2016, 21 (3), 264. [CrossRef]
- Abrahams, S.; Haylett, W.L.; Johnson, G.; Carr, J.A.; Bardien, S. Antioxidant effects of curcumin in models of neurodegeneration, aging, oxidative and nitrosative stress: A review. Neuroscience 2019, 406, 1–21. [CrossRef]
- Kahkhaie, K.R.; Mirhosseini, A.; Aliabadi, A.; Mohammadi, A.; Mousavi, M.J.; Haftcheshmeh, S.M.; Sathyapalan, T.; Sahebkar, A. Curcumin: a modulator of inflammatory signaling pathways in the immune system. Inflammopharmacology 2019, 27, 885–900. [CrossRef]
- Ferreira, N.; Saraiva, M.J.; Almeida, M.R. Uncovering the Neuroprotective Mechanisms of Curcumin on Transthyretin Amyloidosis. Int. J. Mol. Sci. 2019, 20, 1287. [CrossRef]
- Prasad, S.; Tyagi, A.K. Curcumin and its analogues: a potential natural compound against HIV infection and AIDS. Food Funct. 2015, 6, 3412–3419. [CrossRef]
- Zhang, D.; Yang, Y.; Yao, B.; Hu, T.; Ma, Z.; Shi, W.; Ye, Y. Curcumin inhibits Aspergillus flavus infection and aflatoxin production possibly by inducing ROS burst. Food Res. Int. 2023, 167, 112646. [CrossRef]
- Willenbacher, E.; Khan, S.Z.; Mujica, S.C.A.; Trapani, D.; Hussain, S.; Wolf, D.; Willenbacher, W.; Spizzo, G.; Seeber, A. Curcumin: New Insights into an Ancient Ingredient against Cancer. Int. J. Mol. Sci. 2019, 20, 1808. [CrossRef]
- Luthra, P.M.; Lal, N. Prospective of curcumin, a pleiotropic signalling molecule from Curcuma longa in the treatment of Glioblastoma. Eur. J. Med. Chem. 2016, 109, 23–35. [CrossRef]
- Dhandapani, K. M.; Mahesh, V. B.; Brann, D. W., Curcumin suppresses growth and chemoresistance of human glioblastoma cells via AP-1 and NFkappaB transcription factors. Journal of neurochemistry 2007, 102 (2), 522-38. [CrossRef]
- Du, Q.-H.; Peng, C.; Zhang, H. Polydatin: A review of pharmacology and pharmacokinetics. Pharm. Biol. 2013, 51, 1347–1354. [CrossRef]
- Ye, P.; Wu, H.; Jiang, Y.; Xiao, X.; Song, D.; Xu, N.; Ma, X.; Zeng, J.; Guo, Y. Old dog, new tricks: Polydatin as a multitarget agent for current diseases. Phytotherapy Res. 2021, 36, 214–230. [CrossRef]
- Fabris, S.; Momo, F.; Ravagnan, G.; Stevanato, R. Antioxidant properties of resveratrol and piceid on lipid peroxidation in micelles and monolamellar liposomes. Biophys. Chem. 2008, 135, 76–83. [CrossRef]
- Li, Z.; Chen, X.; Liu, G.; Li, J.; Zhang, J.; Cao, Y.; Miao, J. Antioxidant Activity and Mechanism of Resveratrol and Polydatin Isolated from Mulberry (Morus alba L.). Molecules 2021, 26, 7574. [CrossRef]
- Lanzilli, G.; Cottarelli, A.; Nicotera, G.; Guida, S.; Ravagnan, G.; Fuggetta, M.P. Anti-inflammatory Effect of Resveratrol and Polydatin by In Vitro IL-17 Modulation. Inflammation 2011, 35, 240–248. [CrossRef]
- Ravagnan, G.; De Filippis, A.; Carteni, M.; De Maria, S.; Cozza, V.; Petrazzuolo, M.; Tufano, M. A.; Donnarumma, G., Polydatin, a natural precursor of resveratrol, induces beta-defensin production and reduces inflammatory response. Inflammation 2013, 36 (1), 26-34. [CrossRef]
- Fuggetta, M.; Mattivi, F. The immunomodulating activities of resveratrol glucosides in humans.. Recent Patents Food, Nutr. Agric. 2011, 3, 81–90. [CrossRef]
- De Maria, S.; Scognamiglio, I.; Lombardi, A.; Amodio, N.; Caraglia, M.; Cartenì, M.; Ravagnan, G.; Stiuso, P. Polydatin, a natural precursor of resveratrol, induces cell cycle arrest and differentiation of human colorectal Caco-2 cell. J. Transl. Med. 2013, 11, 264–264. [CrossRef]
- Jin, Y.; Zhan, X.; Zhang, B.; Chen, Y.; Liu, C.; Yu, L., Polydatin Exerts an Antitumor Effect Through Regulating the miR-382/PD-L1 Axis in Colorectal Cancer. Cancer Biother Radiopharm 2020, 35 (2), 83-91. [CrossRef]
- De Gregorio, A.; Krasnowska, E.K.; Zonfrillo, M.; Ravagnan, G.; Bordignon, V.; Bonmassar, E.; Fuggetta, M.P. Influence of Polydatin on the Tumor Microenvironment In Vitro: Studies with a Colon Cancer Cell Model. Int. J. Mol. Sci. 2022, 23, 8442. [CrossRef]
- Chen, Y.; Niu, J.; Li, L.; Li, Z.; Jiang, J.; Zhu, M.; Dong, T.; Zhang, J.; Shi, C.; Xu, P.; Lu, Y.; Jiang, Y.; Liu, P.; Chen, W., Polydatin executes anticancer effects against glioblastoma multiforme by inhibiting the EGFR-AKT/ERK1/2/STAT3-SOX2/Snail signaling pathway. Life sciences 2020, 258, 118158. [CrossRef]
- Klinger, N.V.; Mittal, S. Therapeutic Potential of Curcumin for the Treatment of Brain Tumors. Oxidative Med. Cell. Longev. 2016, 2016, 9324085. [CrossRef]
- DU, Q.; Hu, B.; An, H.-M.; Shen, K.-P.; Xu, L.; Deng, S.; Wei, M.-M. Synergistic anticancer effects of curcumin and resveratrol in Hepa1-6 hepatocellular carcinoma cells. Oncol. Rep. 2013, 29, 1851–1858. [CrossRef]
- Majumdar, A.P.N.; Banerjee, S.; Nautiyal, J.; Patel, B.B.; Patel, V.; Du, J.; Yu, Y.; Elliott, A.A.; Levi, E.; Sarkar, F.H. Curcumin Synergizes With Resveratrol to Inhibit Colon Cancer. Nutr. Cancer 2009, 61, 544–553. [CrossRef]
- Wang, P.; Wu, Q.; Peng, Q.; Kang, Z.; Xiao, S.; Zheng, P.; Li, J.; Chen, Y. Comparative analysis of the molecular mechanism of inhibiting proliferation and migration in cervical cancer HeLa cell by curcumin and resveratrol. Nat. Prod. Res. 2023, 37, 4032–4037. [CrossRef]
- Focaccetti, C.; Palumbo, C.; Benvenuto, M.; Carrano, R.; Melaiu, O.; Nardozi, D.; Angiolini, V.; Lucarini, V.; Kërpi, B.; Masuelli, L.; et al. The Combination of Bioavailable Concentrations of Curcumin and Resveratrol Shapes Immune Responses While Retaining the Ability to Reduce Cancer Cell Survival. Int. J. Mol. Sci. 2023, 25, 232. [CrossRef]
- Muhanmode, Y.; Wen, M.K.; Maitinuri, A.; Shen, G. Curcumin and resveratrol inhibit chemoresistance in cisplatin-resistant epithelial ovarian cancer cells via targeting P13K pathway. Hum. Exp. Toxicol. 2022, 41. [CrossRef]
- Ravagnan, G.; Bonucci, M. Composition Comprising Resveratrolosides and Curcumins. WO2020026185-A1, February 2020, 2020.
- Bordin, D.L.; Lima, M.; Lenz, G.; Saffi, J.; Meira, L.B.; Mésange, P.; Soares, D.G.; Larsen, A.K.; Escargueil, A.E.; Henriques, J.A. DNA alkylation damage and autophagy induction. Mutat. Res. Mol. Mech. Mutagen. 2013, 753, 91–99. [CrossRef]
- Pollard, T.D.; Cooper, J.A. Actin, a Central Player in Cell Shape and Movement. Science 2009, 326, 1208–1212. [CrossRef]
- Kitambi, S.S.; Masoumi, S.; Harishankar, A.; Gracias, A.; Bachinger, F.; Fufa, T.; Chandrasekar, G.; Gaunitz, F.; Walfridsson, J. Understanding cytoskeleton regulators in glioblastoma multiforme for therapy design. Drug Des. Dev. Ther. 2016, ume 10, 2881–2897. [CrossRef]
- Avci, N. G.; Ebrahimzadeh-Pustchi, S.; Akay, Y. M.; Esquenazi, Y.; Tandon, N.; Zhu, J. J.; Akay, M., NF-kappaB inhibitor with Temozolomide results in significant apoptosis in glioblastoma via the NF-kappaB(p65) and actin cytoskeleton regulatory pathways. Scientific reports 2020, 10 (1), 13352. [CrossRef]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.-P.; Li, S.; Chen, Y.-M.; Li, H.-B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8, 515. [CrossRef]
- Kunnumakkara, A.B.; Bordoloi, D.; Padmavathi, G.; Monisha, J.; Roy, N.K.; Prasad, S.; Aggarwal, B.B. Curcumin, the golden nutraceutical: multitargeting for multiple chronic diseases. Br. J. Pharmacol. 2017, 174, 1325–1348. [CrossRef]
- Kotecha, R.; Takami, A.; Espinoza, J.L. Dietary phytochemicals and cancer chemoprevention: a review of the clinical evidence. Oncotarget 2016, 7, 52517–52529. [CrossRef]
- Cai, J.; Qiao, Y.; Chen, L.; Lu, Y.; Zheng, D. Regulation of the Notch signaling pathway by natural products for cancer therapy. J. Nutr. Biochem. 2024, 123, 109483. [CrossRef]
- Chen, X. J.; Shen, Y. S.; He, M. C.; Yang, F.; Yang, P.; Pang, F. X.; He, W.; Cao, Y. M.; Wei, Q. S., Polydatin promotes the osteogenic differentiation of human bone mesenchymal stem cells by activating the BMP2-Wnt/beta-catenin signaling pathway. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 2019, 112, 108746. [CrossRef]
- Filippi-Chiela, E.C.; Thomé, M.P.; e Silva, M.M.B.; Pelegrini, A.L.; Ledur, P.F.; Garicochea, B.; Zamin, L.L.; Lenz, G. Resveratrol abrogates the Temozolomide-induced G2 arrest leading to mitotic catastrophe and reinforces the Temozolomide-induced senescence in glioma cells. BMC Cancer 2013, 13, 147–147. [CrossRef]
- Lin, C.-J.; Lee, C.-C.; Shih, Y.-L.; Lin, T.-Y.; Wang, S.-H.; Lin, Y.-F.; Shih, C.-M. Resveratrol enhances the therapeutic effect of temozolomide against malignant glioma in vitro and in vivo by inhibiting autophagy. Free. Radic. Biol. Med. 2011, 52, 377–391. [CrossRef]
- Holy, J. Curcumin inhibits cell motility and alters microfilament organization and function in prostate cancer cells. Cell Motil. Cytoskelet. 2004, 58, 253–268. [CrossRef]
- Dhar, G.; Chakravarty, D.; Hazra, J.; Dhar, J.; Poddar, A.; Pal, M.; Chakrabarti, P.; Surolia, A.; Bhattacharyya, B. Actin–Curcumin Interaction: Insights into the Mechanism of Actin Polymerization Inhibition. Biochemistry 2015, 54, 1132–1143. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



