Submitted:
05 August 2024
Posted:
05 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Prescriptions and Chemicals
2.2. Cell Culture and Treatment
2.2. Cell Viability Assay
2.3. Nitrite Determination
2.4. Western Blot
2.5. Immunofluorescence Assay
2.6. Quantitative Real-Time Polymerase Chain Reaction
2.7. ELISA Analysis
2.8. Acquisition of Potential Active Ingredients and Targets of SST and Isolating Long-Covid DEGs from Online Database
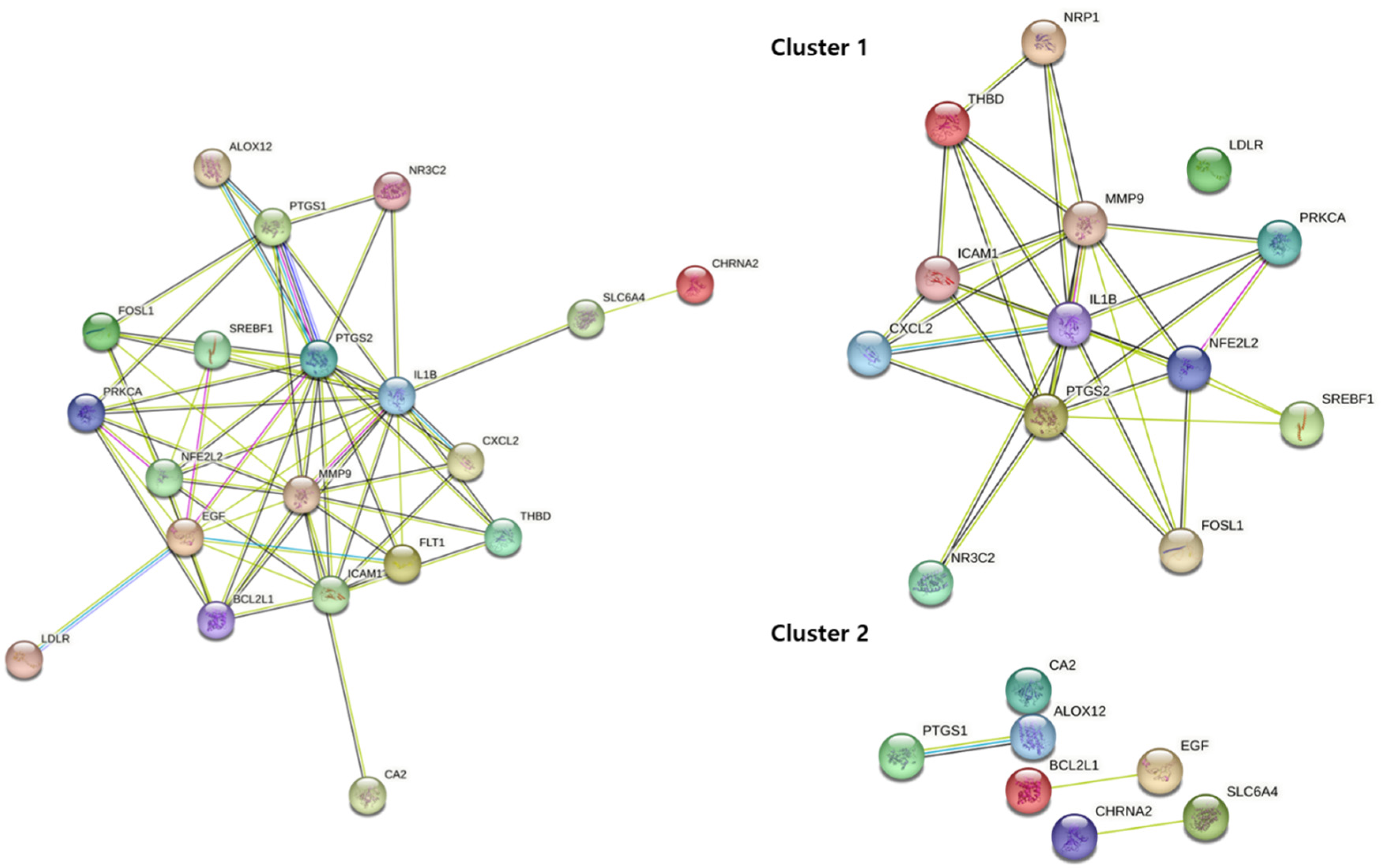
2.9. Protein-Protein Interaction (PPI) Network Construction
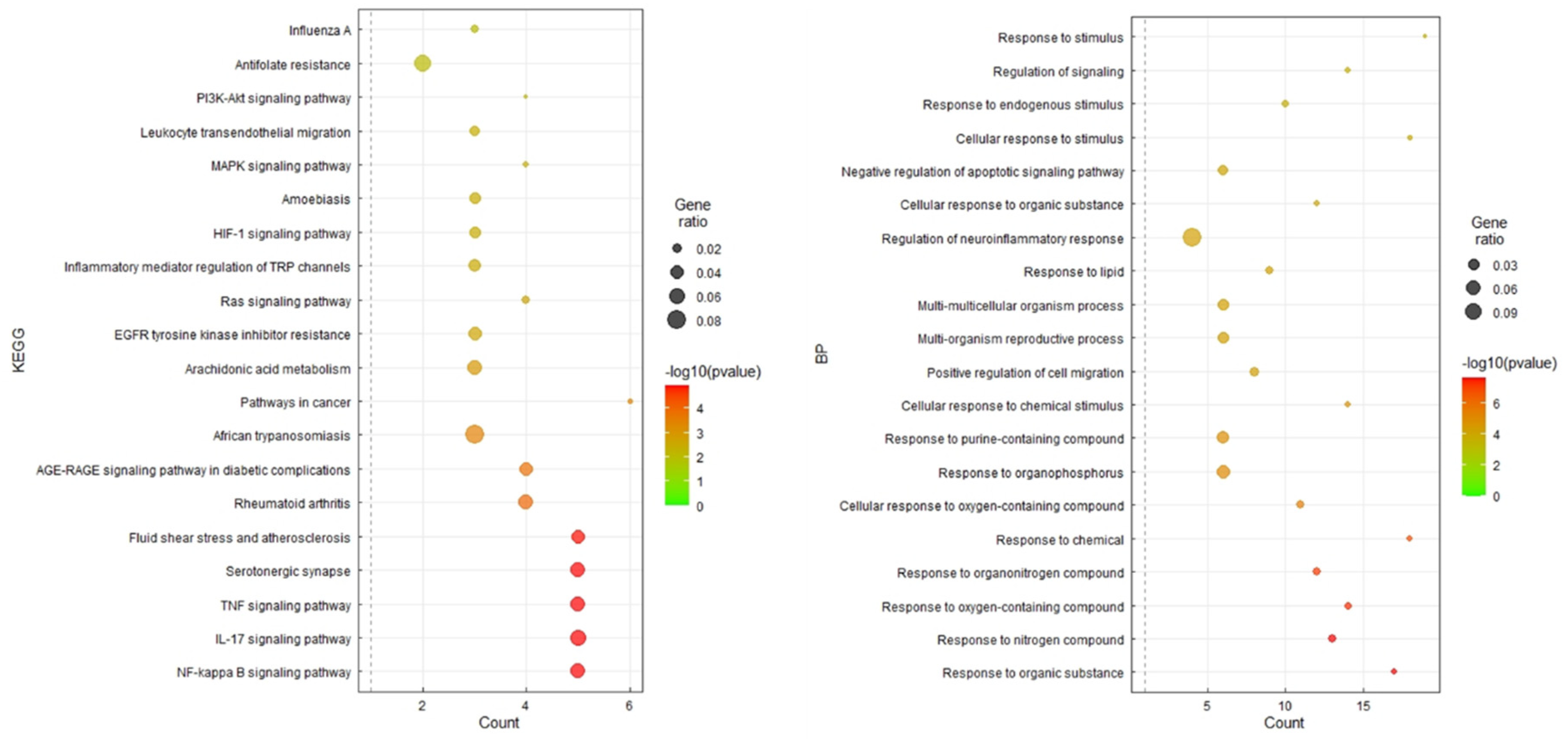
2.10. Gene Ontology (GO) Enrichment and KEGG Pathway Analysis
3. Results
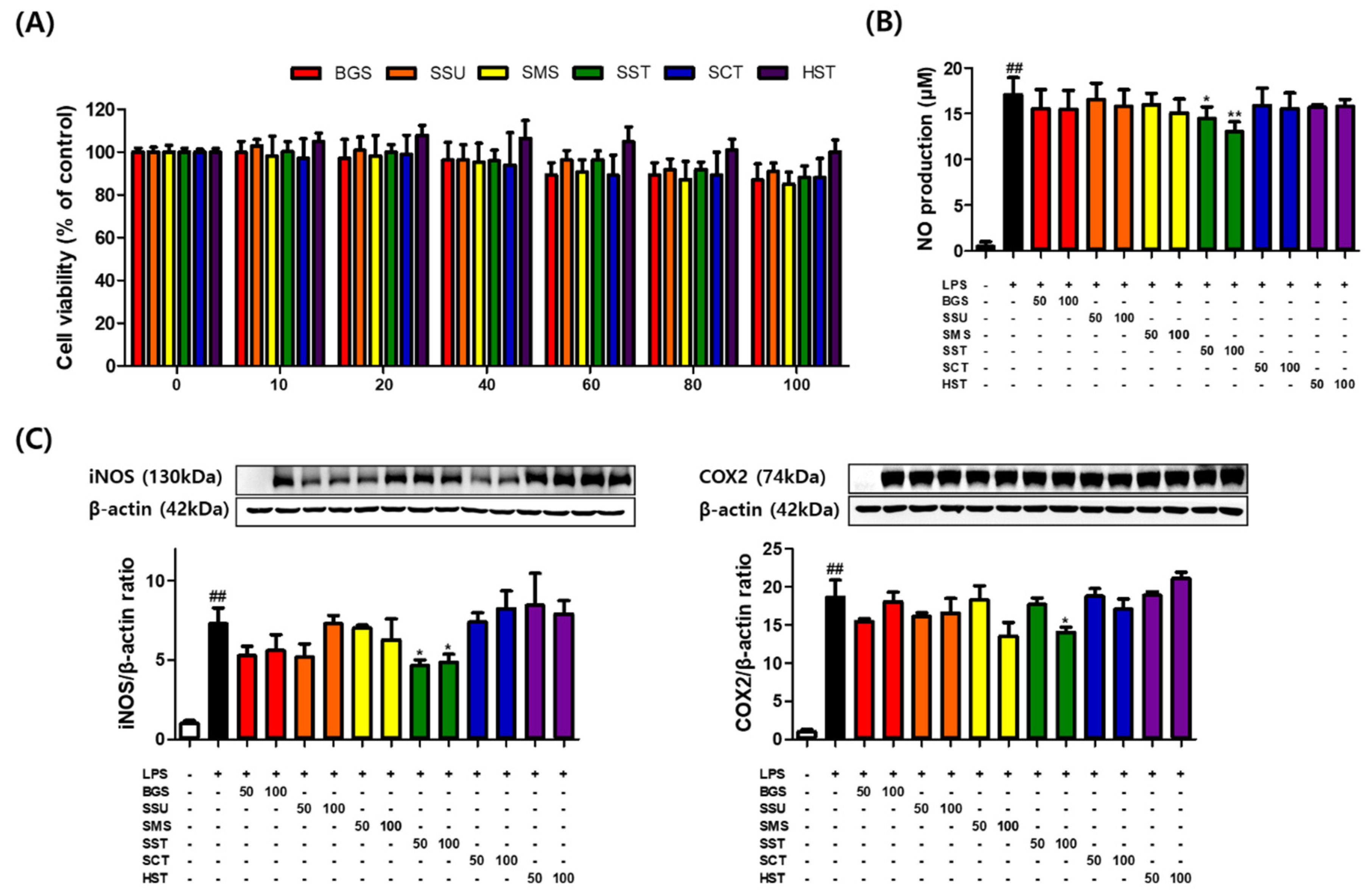
3.1. Effects of Prescriptions on RAW264.7 Cell Viability
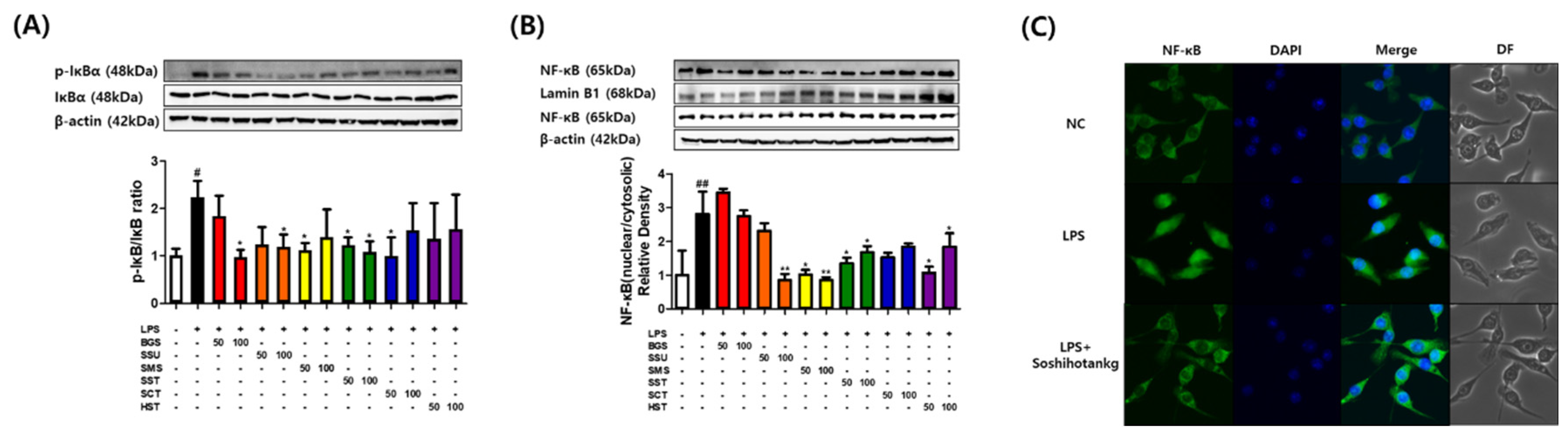
3.2. Effects of Six TKM Prescriptions on the Nuclear Translocation of NF-κB in LPS-Stimulated Macrophages
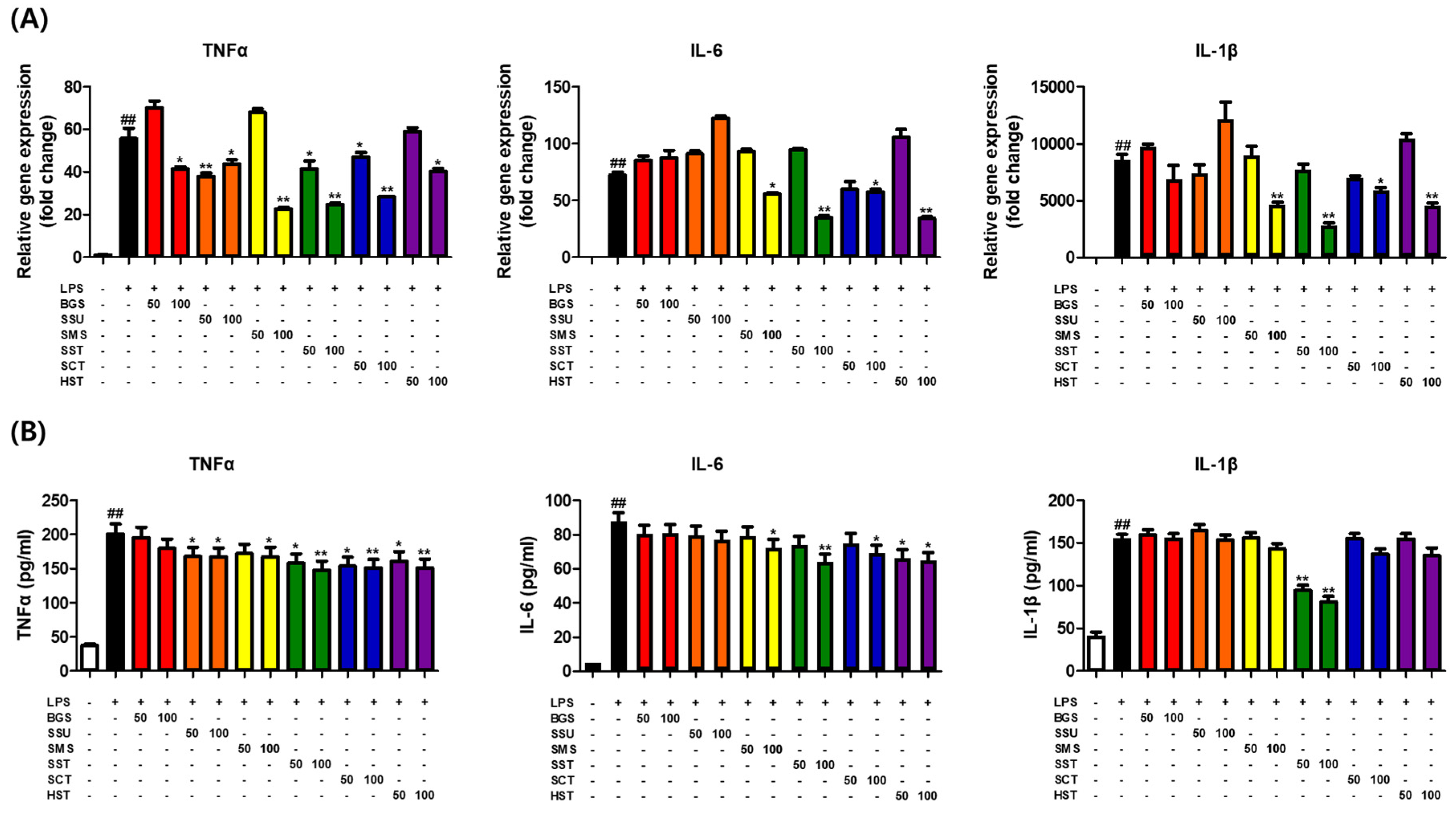
3.3. Effects of Six TKM Prescriptions on Pro-Inflammatory Cytokines Levels in LPS-Stimulated Macrophages
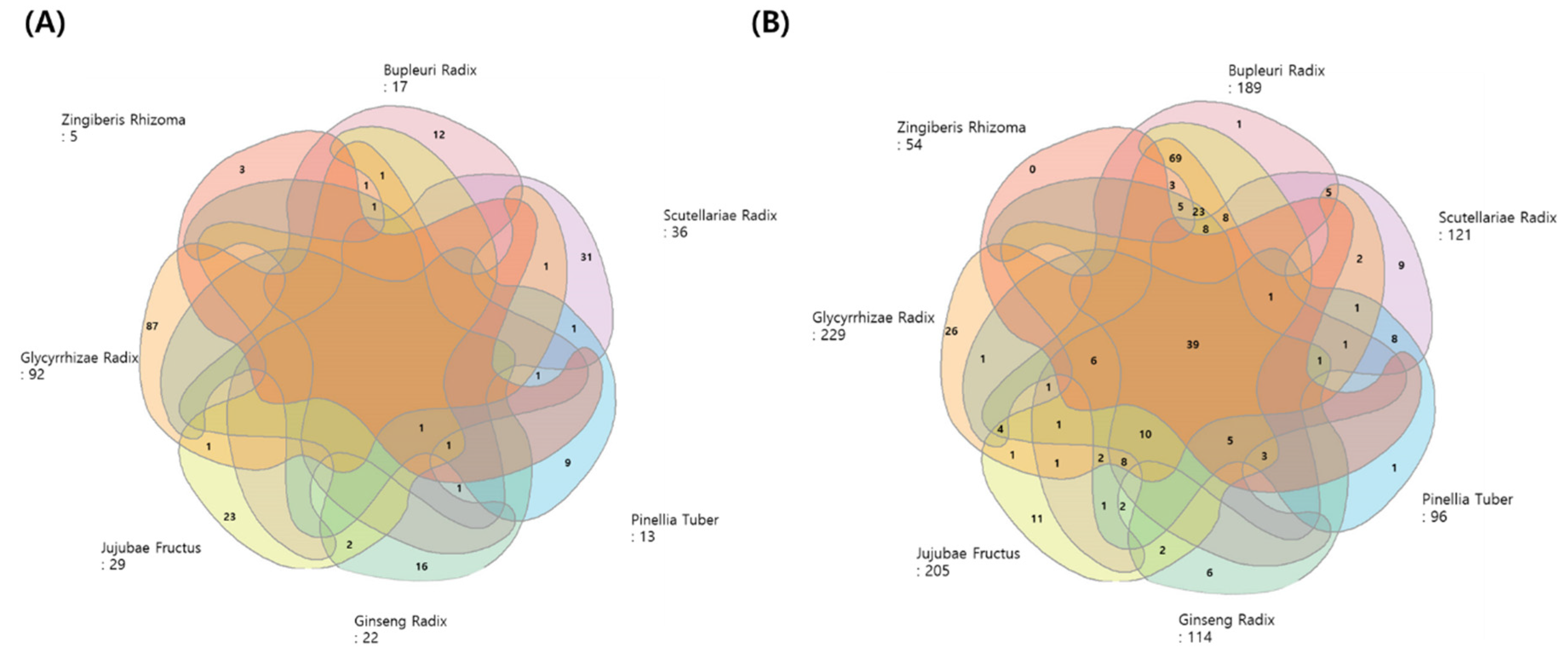
3.4. Active Compound Screening and Key Targets of SST
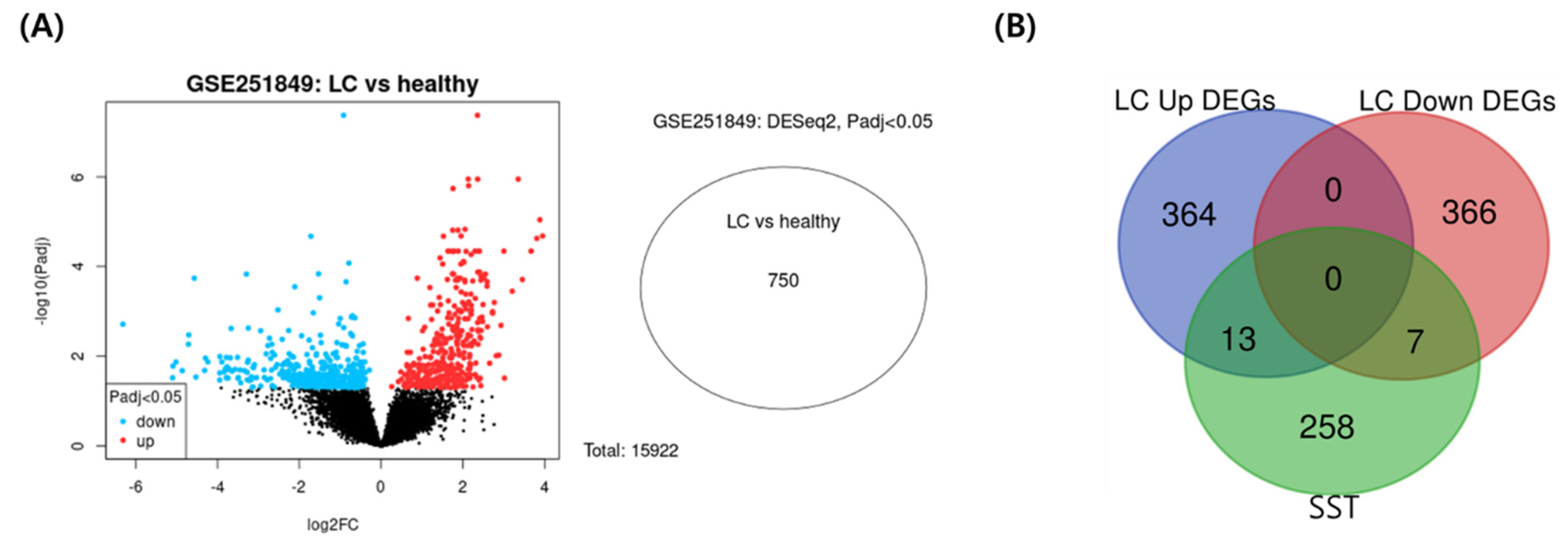
3.5. Identification of COVID-19 Associated-Targets
3.6. Identification of COVID-19 Associated-Targets
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ali, S.A.; Baloch, M.; Ahmed, N.; Ali, A.A.; Iqbal, A. The outbreak of Coronavirus Disease 2019 (COVID-19)—An emerging global health threat. Journal of infection and public health 2020, 13, 644–646. [Google Scholar]
- Yong, S.J.; Liu, S. Proposed subtypes of post-COVID-19 syndrome (or long-COVID) and their respective potential therapies. Reviews in medical virology 2022, 32, e2315. [Google Scholar] [CrossRef]
- Raveendran, A.; Jayadevan, R.; Sashidharan, S. Long COVID: an overview. Diabetes & Metabolic Syndrome: Clinical Research & Reviews 2021, 15, 869–875. [Google Scholar]
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Long COVID: major findings, mechanisms and recommendations. Nature Reviews Microbiology 2023, 21, 133–146. [Google Scholar] [CrossRef]
- Nehme, M.; Chappuis, F.; Kaiser, L.; Assal, F.; Guessous, I. The prevalence, severity, and impact of post-COVID persistent fatigue, post-exertional malaise, and chronic fatigue syndrome. Journal of General Internal Medicine 2023, 38, 835–839. [Google Scholar] [CrossRef]
- Mueller, M.R.; Ganesh, R.; Hurt, R.T.; Beckman, T.J. Post-COVID conditions. In Proceedings of Mayo Clinic Proceedings; pp. 1071–1078.
- Alonso-Domínguez, J.; Gallego-Rodríguez, M.; Martínez-Barros, I.; Calderón-Cruz, B.; Leiro-Fernández, V.; Pérez-González, A.; Poveda, E. High levels of IL-1β, TNF-α and MIP-1α one month after the onset of the acute SARS-CoV-2 infection, predictors of post COVID-19 in hospitalized patients. Microorganisms 2023, 11, 2396. [Google Scholar] [CrossRef]
- Patterson, B.K.; Francisco, E.B.; Yogendra, R.; Long, E.; Pise, A.; Rodrigues, H.; Herrera, M.; Hekmati, S.; Mora, J. Persistence of SARS CoV-2 S1 protein in CD16+ monocytes in post-acute sequelae of COVID-19 (PASC) up to 15 months post-infection. Frontiers in immunology 2022, 12, 746021. [Google Scholar] [CrossRef]
- Weidenbusch, M.; Anders, H.-J. Tissue microenvironments define and get reinforced by macrophage phenotypes in homeostasis or during inflammation, repair and fibrosis. Journal of innate immunity 2012, 4, 463–477. [Google Scholar] [CrossRef]
- Tang, J.; Xu, L.; Zeng, Y.; Gong, F. Effect of gut microbiota on LPS-induced acute lung injury by regulating the TLR4/NF-kB signaling pathway. International immunopharmacology 2021, 91, 107272. [Google Scholar] [CrossRef]
- Gao, L.-N.; Cui, Y.-L.; Wang, Q.-S.; Wang, S.-X. Amelioration of Danhong injection on the lipopolysaccharide-stimulated systemic acute inflammatory reaction via multi-target strategy. Journal of ethnopharmacology 2013, 149, 772–782. [Google Scholar] [CrossRef]
- Fernando, M.R.; Reyes, J.L.; Iannuzzi, J.; Leung, G.; McKay, D.M. The pro-inflammatory cytokine, interleukin-6, enhances the polarization of alternatively activated macrophages. PloS one 2014, 9, e94188. [Google Scholar] [CrossRef] [PubMed]
- Reis, P.A.; de Albuquerque, C.F.G.; Gutierrez, T.; Silva, A.R.; de Castro Faria Neto, H.C. Role of nitric oxide synthase in the function of the central nervous system under normal and infectious conditions. Nitric Oxide Synthase–Simple Enzyme-Complex Roles. London: InTech 2017, 55-70.
- Pahan, K.; Sheikh, F.G.; Namboodiri, A.; Singh, I. Lovastatin and phenylacetate inhibit the induction of nitric oxide synthase and cytokines in rat primary astrocytes, microglia, and macrophages. The Journal of clinical investigation 1997, 100, 2671–2679. [Google Scholar] [CrossRef] [PubMed]
- Rajakariar, R.; Yaqoob, M.M.; Gilroy, D.W. COX-2 in inflammation and resolution. Molecular interventions 2006, 6, 199. [Google Scholar] [CrossRef]
- Al-Harbi, N.O.; Imam, F.; Al-Harbi, M.M.; Ansari, M.A.; Zoheir, K.M.; Korashy, H.M.; Sayed-Ahmed, M.M.; Attia, S.M.; Shabanah, O.A.; Ahmad, S.F. Dexamethasone attenuates LPS-induced acute lung injury through inhibition of NF-κB, COX-2, and pro-inflammatory mediators. Immunological Investigations 2016, 45, 349–369. [Google Scholar] [CrossRef] [PubMed]
- Millar, M.W.; Fazal, F.; Rahman, A. Therapeutic targeting of NF-κB in acute lung injury: A double-edged sword. Cells 2022, 11, 3317. [Google Scholar] [CrossRef] [PubMed]
- Moine, P.; McIntyre, R.; Schwartz, M.D.; Kaneko, D.; Shenkar, R.; Le Tulzo, Y.; Moore, E.E.; Abraham, E. NF-κB regulatory mechanisms in alveolar macrophages from patients with acute respiratory distress syndrome. Shock 2000, 13, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Singleton, K.D.; Beckey, V.E.; Wischmeyer, P.E. Glutamine prevents activation of NF-κB and stress kinase pathways, attenuates inflammatory cytokine release, and prevents acute respiratory distress syndrome (ARDS) following sepsis. Shock 2005, 24, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Tung, Y.-T.; Wei, C.-H.; Yen, C.-C.; Lee, P.-Y.; Ware, L.B.; Huang, H.-E.; Chen, W.; Chen, C.-M. Aspirin attenuates hyperoxia-induced acute respiratory distress syndrome (ARDS) by suppressing pulmonary inflammation via the NF-κB signaling pathway. Frontiers in Pharmacology 2022, 12, 793107. [Google Scholar] [CrossRef] [PubMed]
- Abraham, E. NF-κB activation. Critical Care Medicine 2000, 28, N100–N104. [Google Scholar] [CrossRef]
- Xin, W.; Zi-Yi, W.; Zheng, J.-H.; Shao, L. TCM network pharmacology: a new trend towards combining computational, experimental and clinical approaches. Chinese journal of natural medicines 2021, 19, 1–11. [Google Scholar]
- Ru, J.; Li, P.; Wang, J.; Zhou, W.; Li, B.; Huang, C.; Li, P.; Guo, Z.; Tao, W.; Yang, Y. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. Journal of cheminformatics 2014, 6, 1–6. [Google Scholar] [CrossRef]
- Wang, Y. Network Pharmacology Approaches for Understanding Traditional Chinese Medicine. University of Helsinki, Finland, 2021.
- Chun, S.-C.; Jee, S.Y.; Lee, S.G.; Park, S.J.; Lee, J.R.; Kim, S.C. Anti-inflammatory activity of the methanol extract of moutan cortex in LPS-activated Raw264. 7 cells. Evidence-based Complementary and Alternative Medicine: eCAM 2007, 4, 327. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.M.; Nam, B.; Paudel, S.B.; Nam, J.-W.; Han, A.-R.; Jeong, H.G.; Jin, C.H. 9-Hydroxy-isoegomaketone inhibits LPS-induced NO and inflammatory cytokine production in RAW264. 7 cells. Molecular Medicine Reports 2021, 23, 1–1. [Google Scholar] [CrossRef]
- Xu, J.; Zhao, Y.; Aisa, H.A. Anti-inflammatory effect of pomegranate flower in lipopolysaccharide (LPS)-stimulated RAW264. 7 macrophages. Pharmaceutical Biology 2017, 55, 2095–2101. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.-S.; Shin, P.-G.; Lee, J.-H.; Kim, G.-D. The regulatory effect of veratric acid on NO production in LPS-stimulated RAW264. 7 macrophage cells. Cellular immunology 2012, 280, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhao, W.; Zhang, X.; Chen, X. Neocryptotanshinone inhibits lipopolysaccharide-induced inflammation in RAW264. 7 macrophages by suppression of NF-κB and iNOS signaling pathways. Acta Pharmaceutica Sinica B 2015, 5, 323–329. [Google Scholar] [CrossRef]
- Guo, C.; Yang, L.; Luo, J.; Zhang, C.; Xia, Y.; Ma, T.; Kong, L. Sophoraflavanone G from Sophora alopecuroides inhibits lipopolysaccharide-induced inflammation in RAW264. 7 cells by targeting PI3K/Akt, JAK/STAT and Nrf2/HO-1 pathways. International Immunopharmacology 2016, 38, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.-K.; Lee, M.-Y.; Yuk, J.-E.; Oh, S.-R.; Chin, Y.-W.; Lee, H.-K.; Ahn, K.-S. Anti-inflammatory effects of methanol extracts of the root of Lilium lancifolium on LPS-stimulated Raw264. 7 cells. Journal of ethnopharmacology 2010, 130, 28–34. [Google Scholar] [CrossRef]
- Greene, C.; Connolly, R.; Brennan, D.; Laffan, A.; O’Keeffe, E.; Zaporojan, L.; O’Callaghan, J.; Thomson, B.; Connolly, E.; Argue, R. Blood–brain barrier disruption and sustained systemic inflammation in individuals with long COVID-associated cognitive impairment. Nature neuroscience 2024, 27, 421–432. [Google Scholar] [CrossRef]
- Ran, J.; Li, H.; Fu, J.; Liu, L.; Xing, Y.; Li, X.; Shen, H.; Chen, Y.; Jiang, X.; Li, Y. Construction and analysis of the protein-protein interaction network related to essential hypertension. BMC systems biology 2013, 7, 1–12. [Google Scholar] [CrossRef]
- Bonnot, T.; Gillard, M.B.; Nagel, D.H. A simple protocol for informative visualization of enriched gene ontology terms. Bio-protocol 2019, e3429–e3429. [Google Scholar] [CrossRef]
- Lim, D.W.; Ahn, J.Y.; Yu, G.R.; Kim, J.E.; Park, W.H.; Lim, D.W.; Ahn, J.Y.; Yu, G.R.; Kim, J.E.; Park, W.H. Study on the distribution in major disease category and frequency of clinical usage of national health insurance herbal prescription based on analysis on KCD8 disease code of indications. Journal of Korean Medicine 2023, 44, 1–15. [Google Scholar] [CrossRef]
- Tahaghoghi-Hajghorbani, S.; Zafari, P.; Masoumi, E.; Rajabinejad, M.; Jafari-Shakib, R.; Hasani, B.; Rafiei, A. The role of dysregulated immune responses in COVID-19 pathogenesis. Virus research 2020, 290, 198197. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, C.; Cassatella, M.A. Neutrophil-derived cytokines involved in physiological and pathological angiogenesis. Angiogenesis, Lymphangiogenesis and Clinical Implications 2014, 99, 123–137. [Google Scholar]
- Sun, S.-C.; Ganchi, P.A.; Ballard, D.W.; Greene, W.C. NF-κB controls expression of inhibitor IκBα: evidence for an inducible autoregulatory pathway. Science 1993, 259, 1912–1915. [Google Scholar] [CrossRef] [PubMed]
- Ratan, Z.A.; Haidere, M.F.; Hong, Y.H.; Park, S.H.; Lee, J.-O.; Lee, J.; Cho, J.Y. Pharmacological potential of ginseng and its major component ginsenosides. Journal of ginseng research 2021, 45, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Christensen, L.P. Ginsenosides: chemistry, biosynthesis, analysis, and potential health effects. Advances in food and nutrition research 2008, 55, 1–99. [Google Scholar]
- You, L.; Cha, S.; Kim, M.-Y.; Cho, J.Y. Ginsenosides are active ingredients in Panax ginseng with immunomodulatory properties from cellular to organismal levels. Journal of ginseng research 2022, 46, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Shahrajabian, M.H.; Sun, W.; Cheng, Q. A review of ginseng species in different regions as a multipurpose herb in traditional Chinese medicine, modern herbology and pharmacological science. Journal of Medicinal Plants Research 2019, 13, 213–226. [Google Scholar]
- He, M.T.; Park, G.; Park, D.H.; Choi, M.; Ku, S.; Go, S.H.; Lee, Y.G.; Song, S.J.; Ahn, C.-W.; Jang, Y.P. So Shiho Tang Reduces Inflammation in Lipopolysaccharide-Induced RAW 264.7 Macrophages and Dextran Sodium Sulfate-Induced Colitis Mice. Biomolecules 2024, 14, 451. [Google Scholar] [CrossRef]
- Ohtake, N.; Nakai, Y.; Yamamoto, M.; Sakakibara, I.; Takeda, S.; Amagaya, S.; Aburada, M. Separation and isolation methods for analysis of the active principles of Sho-saiko-to (SST) oriental medicine. Journal of Chromatography B 2004, 812, 135–148. [Google Scholar] [CrossRef]
- Tran, N.K.S.; Lee, J.H.; Lee, M.J.; Park, J.Y.; Kang, K.S. Multitargeted Herbal Prescription So Shiho Tang: A Scoping Review on Biomarkers for the Evaluation of Therapeutic Effects. Pharmaceuticals 2023, 16, 1371. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.-h.; Choi, Y.-k.; Jeon, C.-y.; Yang, S.-b. The effects of Qingfei Paidu Decoction on coronavirus disease-19: A narrative review. The Journal of Internal Korean Medicine 2020, 41, 424–433. [Google Scholar] [CrossRef]
- Lim, D.-W.; Kim, D.-H.; Yu, G.-R.; Park, W.-H.; Kim, J.-E. Verification of the Potential Targets of the Herbal Prescription Sochehwan for Drug Repurposing Processes as Deduced by Network Pharmacology. Processes 2021, 9, 2034. [Google Scholar] [CrossRef]
- Kim, T.-H.; Yu, G.-R.; Kim, H.; Kim, J.-E.; Lim, D.-W.; Park, W.-H. Network pharmacological analysis of a new herbal combination targeting hyperlipidemia and efficacy validation in vitro. Current Issues in Molecular Biology 2023, 45, 1314–1332. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



