Submitted:
29 July 2024
Posted:
30 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Administration of Adenoviral Vectors and Bleomycin
2.3. Collection of Mouse Specimens and Analysis of Lung Tissues
2.4. Acquisition and Analysis
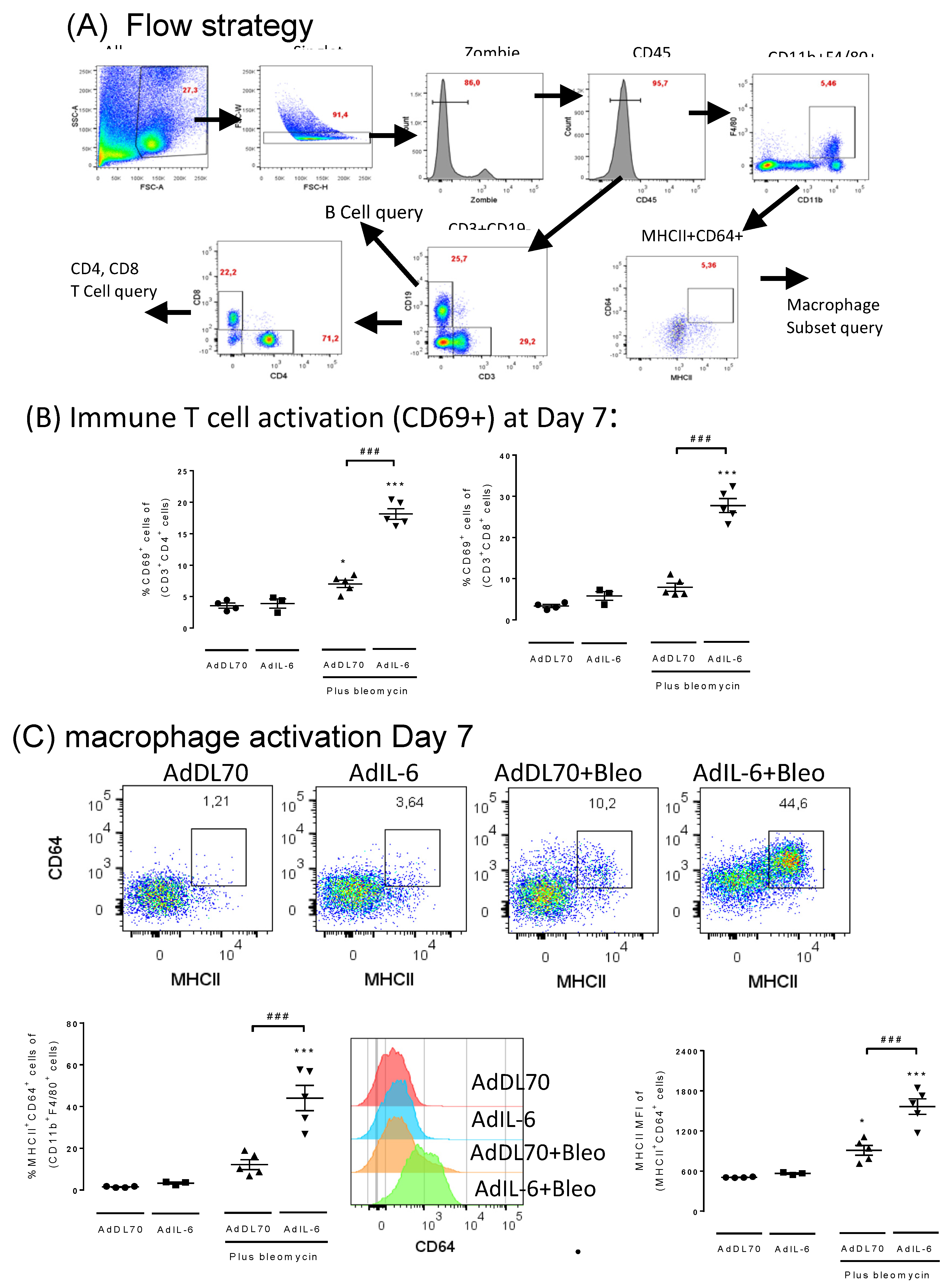
2.5. Flow Cytometry:
2.6. Statistical Analysis
2.7. Bulk RNAseq and Bioinformatic Analysis
2.8. Publicly Available RNA Datasets
3. Results
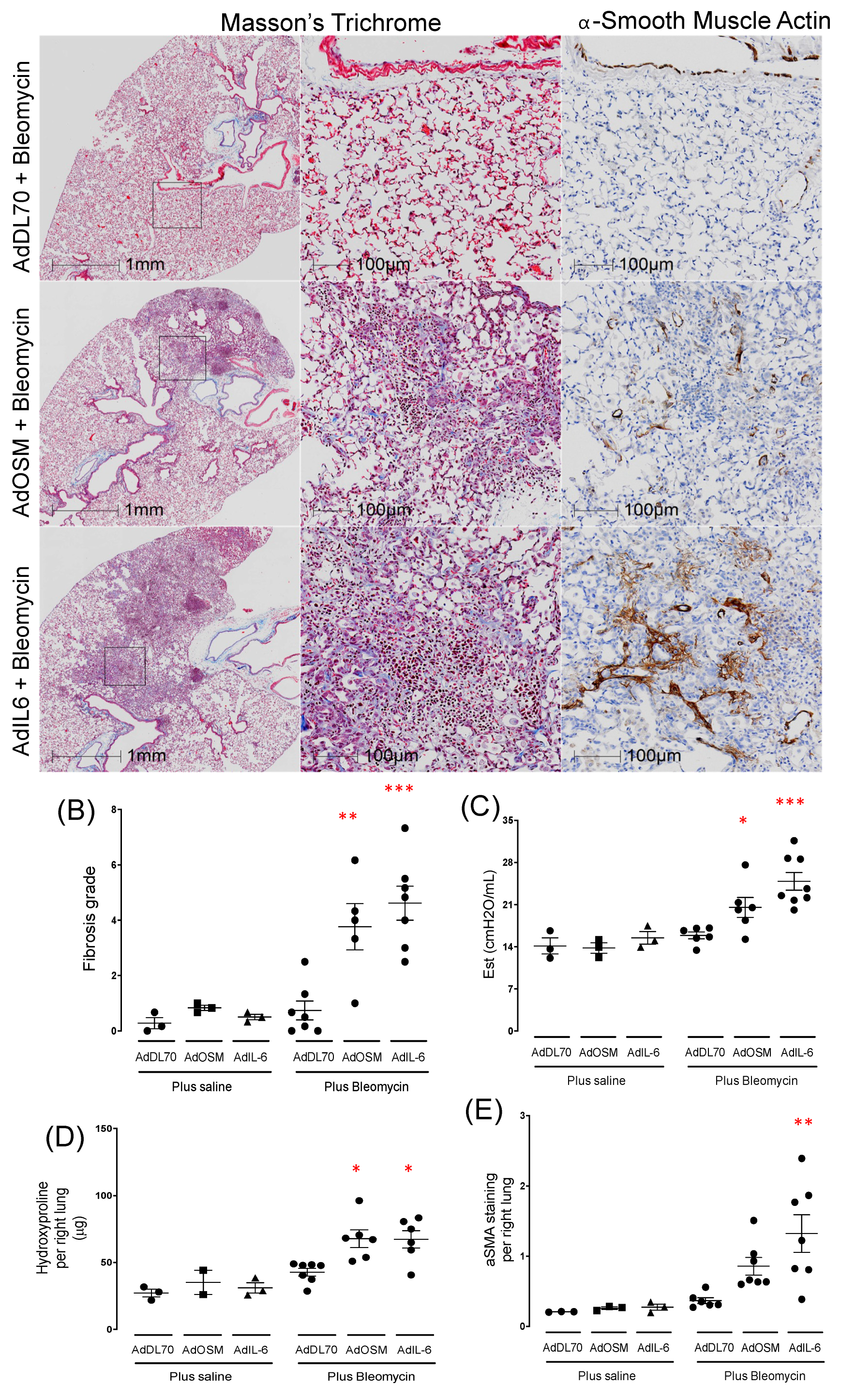
3.1.1. AdIL-6 Combined with Bleomycin Induces Robust Fibrosis at Day 21 in BALB/c Mice
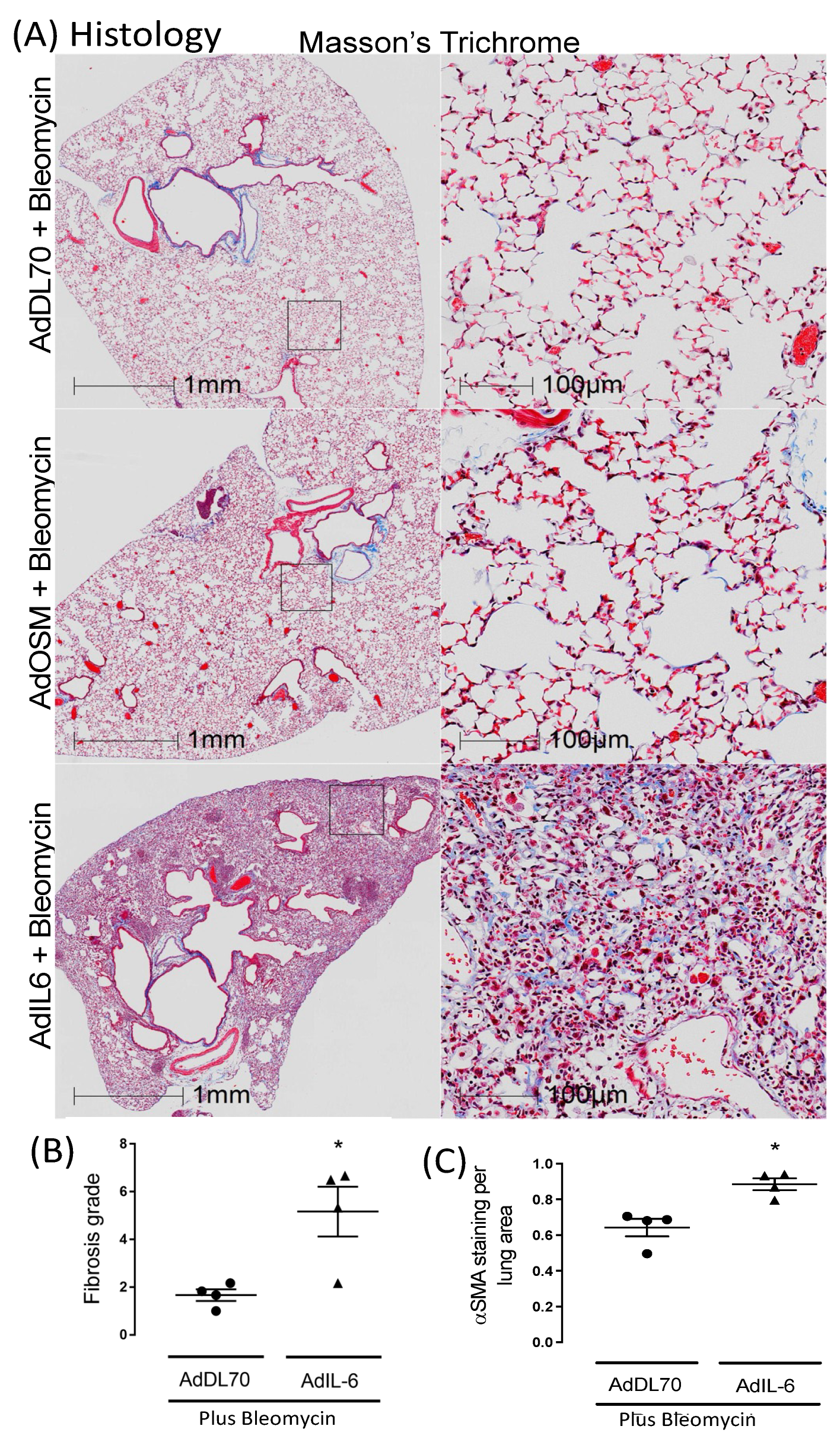
3.1.2. AdIL-6+Bleo Induces Sustained Fibrosis out to Day 50
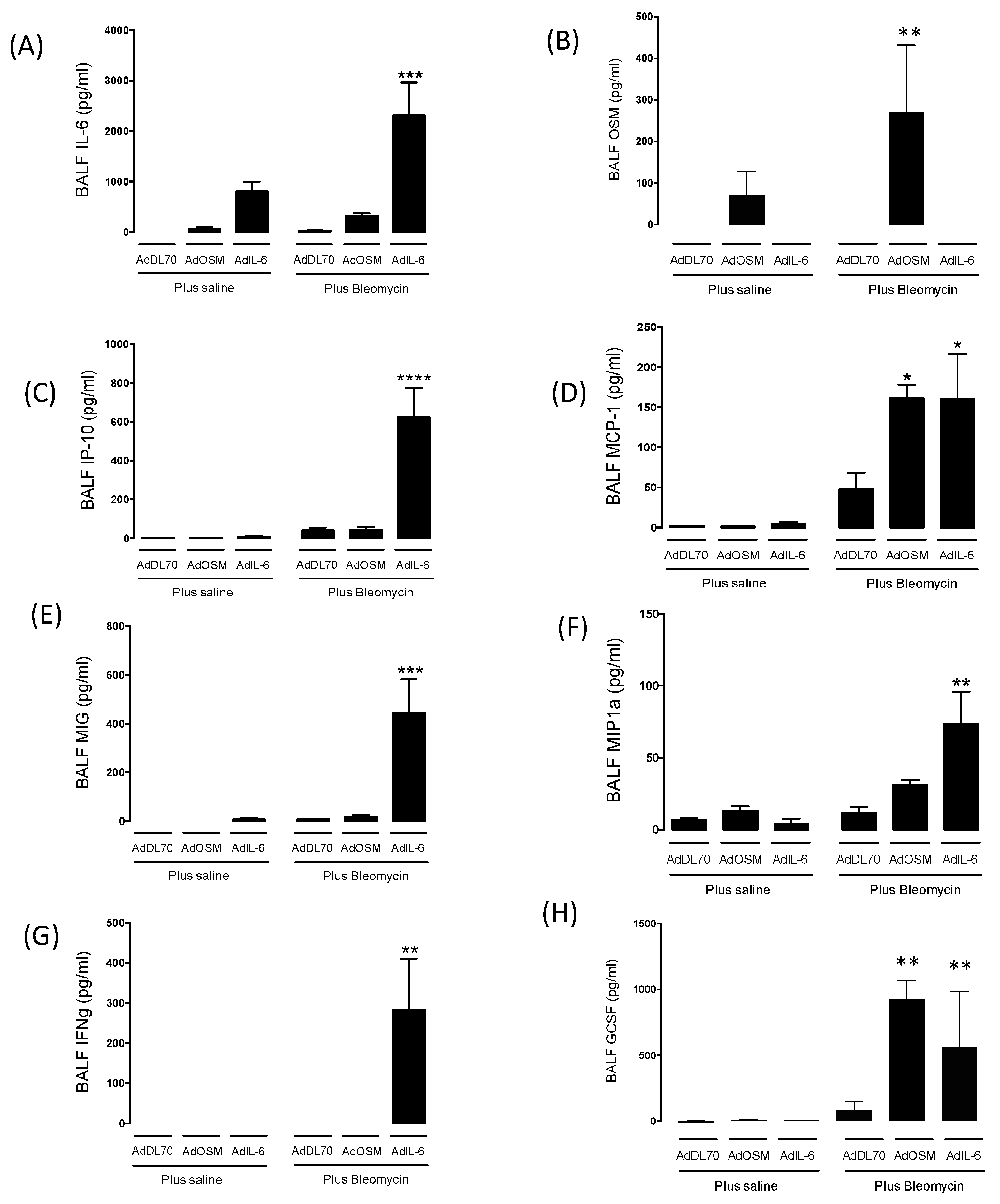
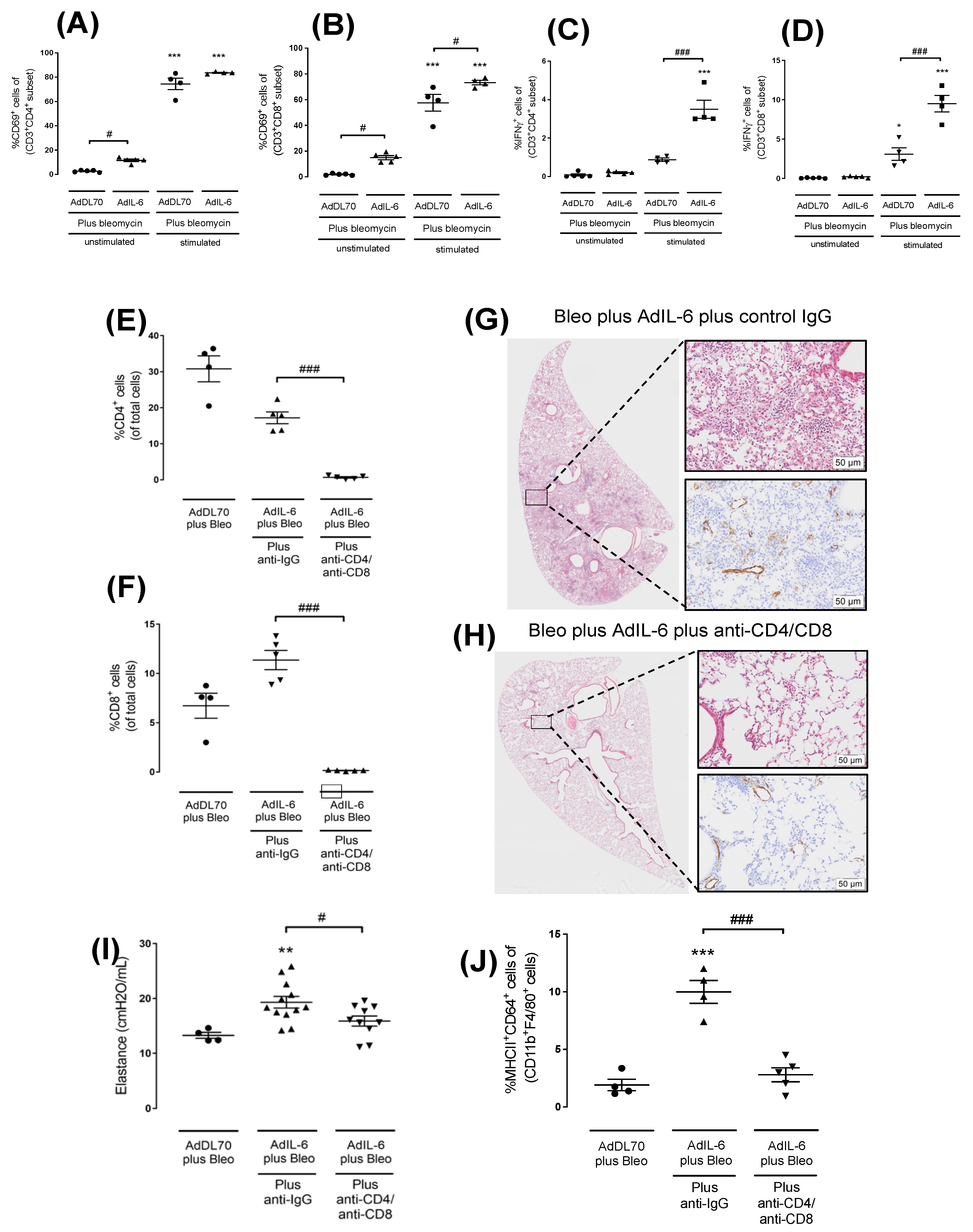
3.2. Cytokine Levels in Broncho-Alveolar Lavage and Flow Cytometry at Day 7
3.3. T-Cell Depleting Antibodies Inhibits Fibrotic Responses at Day 21 in AdIL-6+Bleo BALB/c Mice
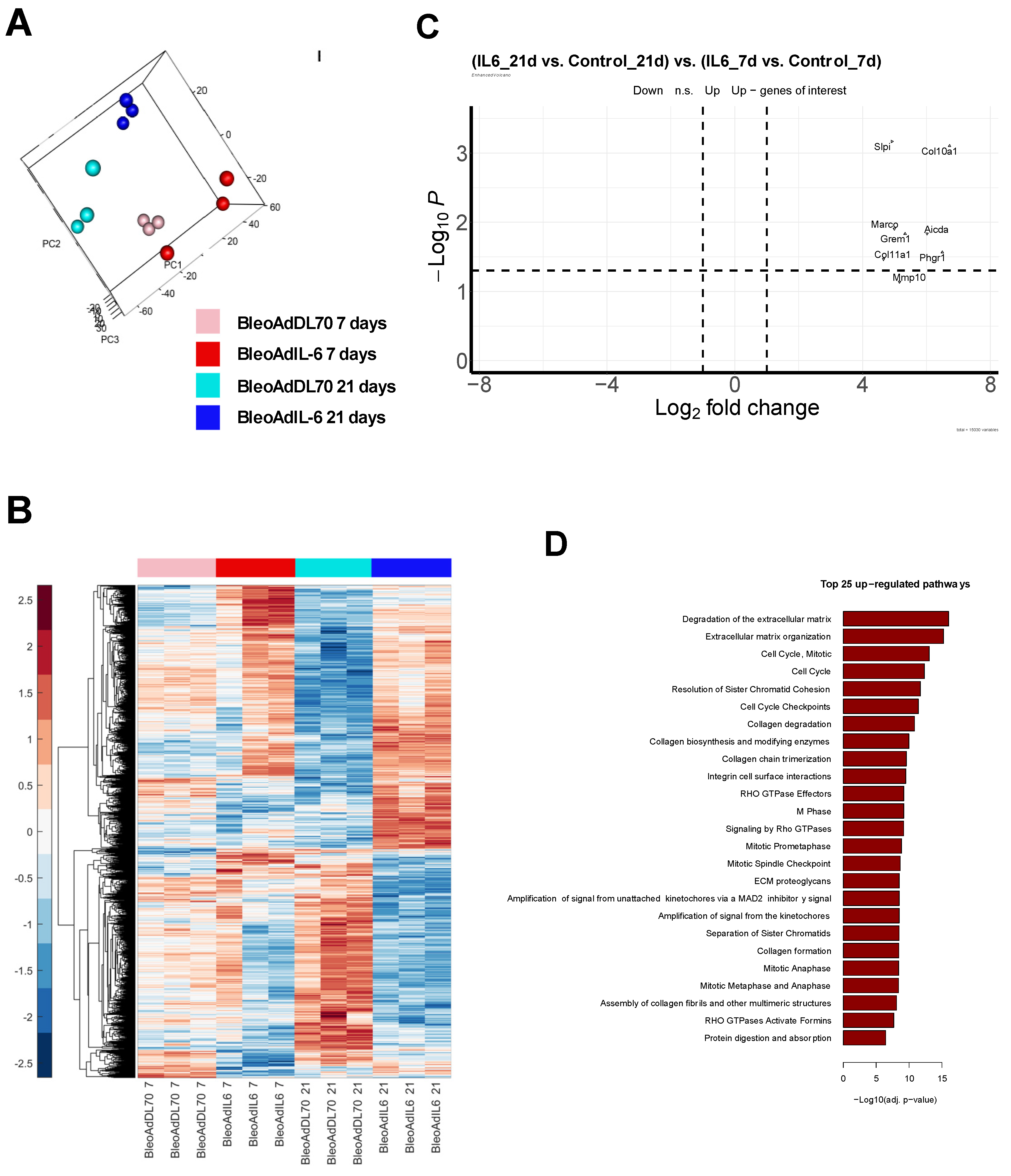
3.4.1. Transcriptome Analysis and Differentially Expressed Genes at Day 7 and 21 of AdIL-6+Bleo Treated BALB/c Mice
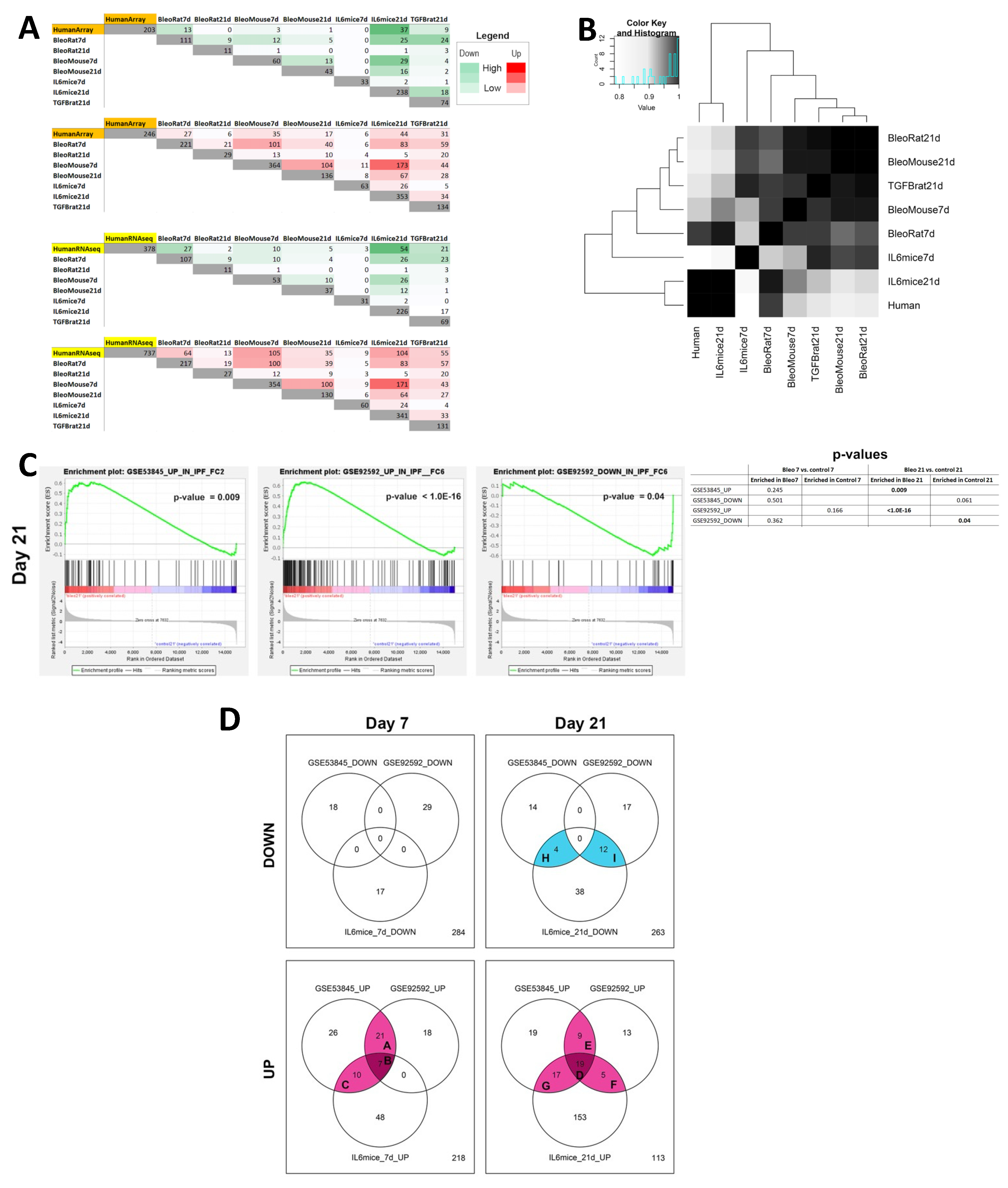
3.4.2. Comparison of Transcriptomes of Mouse Lung Fibrosis Models to Human IPF Transcriptomes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- King, T.E.; Pardo, A.; Selman, M. Idiopathic Pulmonary Fibrosis. The Lancet 2011, 378, 1949–1961. [Google Scholar] [CrossRef]
- Sgalla, G.; Biffi, A.; Richeldi, L. Idiopathic Pulmonary Fibrosis: Diagnosis, Epidemiology and Natural History. Respirology 2016, 21, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, E.B.; Noble, P.W. Idiopathic Pulmonary Fibrosis. Orphanet J Rare Dis 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- Ley, B.; Collard, H.R.; King, T.E. Clinical Course and Prediction of Survival in Idiopathic Pulmonary Fibrosis. Am J Respir Crit Care Med 2011, 183, 431–440. [Google Scholar] [CrossRef] [PubMed]
- du Bois, R.M. An Earlier and More Confident Diagnosis of Idiopathic Pulmonary Fibrosis. European Respiratory Review 2012, 21, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G, Masta S, Meyers D, N. A. American Review of Respiratory Disease. Am Rev Respir Dis 1989, 140, 95–100. [Google Scholar]
- Pardo, A.; Selman, M. Idiopathic Pulmonary Fibrosis: New Insights in Its Pathogenesis; 2002; Vol. 34;
- King, T.E.; Pardo, A.; Selman, M. Idiopathic Pulmonary Fibrosis. The Lancet 2011, 378, 1949–1961. [Google Scholar] [CrossRef] [PubMed]
- Willis, B.C.; Liebler, J.M.; Luby-Phelps, K.; Nicholson, A.G.; Crandall, E.D.; du Bois, R.M.; Borok, Z. Induction of Epithelial-Mesenchymal Transition in Alveolar Epithelial Cells by Transforming Growth Factor-Beta1: Potential Role in Idiopathic Pulmonary Fibrosis. Am J Pathol 2005, 166, 1321–1332. [Google Scholar] [CrossRef] [PubMed]
- Chapman, H.A. Epithelial-Mesenchymal Interactions in Pulmonary Fibrosis. Annu Rev Physiol 2011, 73, 413–435. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, R.J.J.; Knight, D.A.; Richards, C.D.; Prêle, C.M.; Lau, H.L.; Jarnicki, A.G.; Jones, J.; Bozinovski, S.; Vlahos, R.; Thiem, S.; et al. Genetic Partitioning of Interleukin-6 Signalling in Mice Dissociates Stat3 from Smad3-Mediated Lung Fibrosis. EMBO Mol Med 2012, 4, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.; Botelho, F.M.; Rodrigues, R.M.; Richards, C.D. Oncostatin M Overexpression Induces Matrix Deposition, STAT3 Activation, and SMAD1 Dysregulation in Lungs of Fibrosis-Resistant BALB/c Mice. Laboratory Investigation 2014, 94, 1003–1016. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, L.; Albera, C.; Bradford, W.Z.; Costabel, U.; du Bois, R.M.; Fagan, E.A.; Fishman, R.S.; Glaspole, I.; Glassberg, M.K.; King Jr, T.E.; et al. Safety of Pirfenidone in Patients with Idiopathic Pulmonary Fibrosis: Integrated Analysis of Cumulative Data from 5 Clinical Trials. BMJ Open Respir Res 2016, 3, e000105–e000105. [Google Scholar] [CrossRef] [PubMed]
- Richeldi, L.; du Bois, R.M.; Raghu, G.; Azuma, A.; Brown, K.K.; Costabel, U.; Cottin, V.; Flaherty, K.R.; Hansell, D.M.; Inoue, Y.; et al. Efficacy and Safety of Nintedanib in Idiopathic Pulmonary Fibrosis. New England Journal of Medicine 2014, 370, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Wollin, L.; Wex, E.; Pautsch, A.; Schnapp, G.; Hostettler, K.E.; Stowasser, S.; Kolb, M. Mode of Action of Nintedanib in the Treatment of Idiopathic Pulmonary Fibrosis. Eur Respir J 2015, 45, 1434–1445. [Google Scholar] [CrossRef]
- Raghu, G.; Selman, M. Nintedanib and Pirfenidone-New Antifibrotic Treatments Indicated for Idiopathic Pulmonary Fibrosis. Am J Respir Crit Care Med 2015, 191, 252–254. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, J.; Rubio, G.A.; Limper, A.H.; Williams, K.; Elliot, S.J.; Ninou, I.; Aidinis, V.; Tzouvelekis, A.; Glassberg, M.K. Exploring Animal Models That Resemble Idiopathic Pulmonary Fibrosis. Front Med (Lausanne) 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Moore, B.B.; Hogaboam, C.M. Murine Models of Pulmonary Fibrosis. American Journal of Physiology-Lung Cellular and Molecular Physiology 2008, 294, L152–L160. [Google Scholar] [CrossRef] [PubMed]
- Moeller, A.; Ask, K.; Warburton, D.; Gauldie, J.; Kolb, M. The Bleomycin Animal Model: A Useful Tool to Investigate Treatment Options for Idiopathic Pulmonary Fibrosis? Int J Biochem Cell Biol 2008, 40, 362–382. [Google Scholar] [CrossRef] [PubMed]
- Kolb, P.; Upagupta, C.; Vierhout, M.; Ayaub, E.; Bellaye, P.S.; Gauldie, J.; Shimbori, C.; Inman, M.; Ask, K.; Kolb, M.R.J. The Importance of Interventional Timing in the Bleomycin Model of Pulmonary Fibrosis. Eur Respir J 2020, 55. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.H.; Lazo, J.S. High Dose Continuous Infusion of Bleomycin in Mice: A New Model for Drug-Induced Pulmonary Fibrosis. Journal of Pharmacology and Experimental Therapeutics 1987, 243, 1185, LP – 1194. [Google Scholar] [PubMed]
- Schrier, D.J.; Kunkel, R.G.; Phan, S.H. The Role of Strain Variation in Murine Bleomycin-Lnduced Pulmonary Fibrosis. American Review of Respiratory Disease 1983, 127, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Ask, K.; Labiris, R.; Farkas, L.; Moeller, A.; Froese, A.; Farncombe, T.; McClelland, G.B.; Inman, M.; Gauldie, J.; Kolb, M.R.J. Comparison between Conventional and “Clinical” Assessment of Experimental Lung Fibrosis. J Transl Med 2008, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Ayaub, E.A.; Dubey, A.; Imani, J.; Botelho, F.; Kolb, M.R.J.; Richards, C.D.; Ask, K. Overexpression of OSM and IL-6 Impacts the Polarization of pro-Fibrotic Macrophages and the Development of Bleomycin-Induced Lung Fibrosis. Sci Rep 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Taga, T.; Kishimoto, T. Gp130 AND THE INTERLEUKIN-6 FAMILY OF CYTOKINES. Annu Rev Immunol 1997, 15, 797–819. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. Therapeutic Targeting of the Interleukin-6 Receptor. Annu Rev Pharmacol Toxicol 2012, 52, 199–219. [Google Scholar] [CrossRef] [PubMed]
- Silver, J.S.; Hunter, C.A. Gp130 at the Nexus of Inflammation, Autoimmunity, and Cancer. J Leukoc Biol 2010, 88, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Xing, Z.; Braciak, T.; Jordana, M.; Croitoru, K.; Graham, F.L.; Gauldie, J. Adenovirus-Mediated Cytokine Gene Transfer at Tissue Sites. Overexpression of IL-6 Induces Lymphocytic Hyperplasia in the Lung. The Journal of Immunology 1994, 153, 4059, LP – 4069. [Google Scholar] [CrossRef]
- Mauer, J.; Chaurasia, B.; Goldau, J.; Vogt, M.C.; Ruud, J.; Nguyen, K.D.; Theurich, S.; Hausen, A.C.; Schmitz, J.; Brönneke, H.S.; et al. Signaling by IL-6 Promotes Alternative Activation of Macrophages to Limit Endotoxemia and Obesity-Associated Resistance to Insulin. Nat Immunol 2014, 15, 423–430. [Google Scholar] [CrossRef] [PubMed]
- West, N.R. Coordination of Immune-Stroma Crosstalk by IL-6 Family Cytokines. Front Immunol 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Dienz, O.; Rincon, M. The Effects of IL-6 on CD4 T Cell Responses. Clin Immunol 2009, 130, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Ridgley, L.A.; Anderson, A.E.; Maney, N.J.; Naamane, N.; Skelton, A.J.; Lawson, C.A.; Emery, P.; Isaacs, J.D.; Carmody, R.J.; Pratt, A.G. IL-6 Mediated Transcriptional Programming of Naïve CD4+ T Cells in Early Rheumatoid Arthritis Drives Dysregulated Effector Function. Front Immunol 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Jones, L.L.; Geiger, T.L. IL-6 Promotes T Cell Proliferation and Expansion under Inflammatory Conditions in Association with Low-Level RORγt Expression. The Journal of Immunology 2018, 201, 2934–2946. [Google Scholar] [CrossRef] [PubMed]
- De Lauretis, A.; Sestini, P.; Pantelidis, P.; Hoyles, R.; Hansell, D.M.; Goh, N.S.L.; Zappala, C.J.; Visca, D.; Maher, T.M.; Denton, C.P.; et al. Serum Interleukin 6 Is Predictive of Early Functional Decline and Mortality in Interstitial Lung Disease Associated with Systemic Sclerosis. J Rheumatol 2013, 40, 435, LP – 446. [Google Scholar] [CrossRef] [PubMed]
- Collard, H.R.; Calfee, C.S.; Wolters, P.J.; Song, J.W.; Hong, S.-B.; Brady, S.; Ishizaka, A.; Jones, K.D.; King Jr, T.E.; Matthay, M.A.; et al. Plasma Biomarker Profiles in Acute Exacerbation of Idiopathic Pulmonary Fibrosis. Am J Physiol Lung Cell Mol Physiol 2010, 299, L3–L7. [Google Scholar] [CrossRef]
- Wong, S.; Botelho, F.M.; Rodrigues, R.M.; Richards, C.D. Oncostatin M Overexpression Induces Matrix Deposition, STAT3 Activation, and SMAD1 Dysregulation in Lungs of Fibrosis-Resistant BALB/c Mice. Laboratory Investigation 2014, 94, 1003–1016. [Google Scholar] [CrossRef]
- Ayaub, E.A.; Dubey, A.; Imani, J.; Botelho, F.; Kolb, M.R.J.; Richards, C.D.; Ask, K. Overexpression of OSM and IL-6 Impacts the Polarization of pro-Fibrotic Macrophages and the Development of Bleomycin-Induced Lung Fibrosis. Sci Rep 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Yao, Y.; Jeyanathan, M.; Haddadi, S.; Barra, N.G.; Vaseghi-Shanjani, M.; Damjanovic, D.; Lai, R.; Afkhami, S.; Chen, Y.; Dvorkin-Gheva, A.; et al. Induction of Autonomous Memory Alveolar Macrophages Requires T Cell Help and Is Critical to Trained Immunity. Cell 2018, 175, 1634–1650.e17. [Google Scholar] [CrossRef]
- Botelho, F.M.; Rangel-Moreno, J.; Fritz, D.; Randall, T.D.; Xing, Z.; Richards, C.D. Pulmonary Expression of Oncostatin M (OSM) Promotes Inducible BALT Formation Independently of IL-6, Despite a Role for IL-6 in OSM-Driven Pulmonary Inflammation. The Journal of Immunology 2013, 191, 1453–1464. [Google Scholar] [CrossRef] [PubMed]
- Mekhael O; Naiel S; Vierhout M; Hayat AI; Revill SD; Abed S; Inman MD; Kolb MRJ; Ask K. Mouse Models of Lung Fibrosis. Methods Mol Biol. 2021, 2299, 291–321. [Google Scholar]
- Botelho, F.M.; Rodrigues, R.; Guerette, J.; Wong, S.; Fritz, D.K.; Richards, C.D. Extracellular Matrix and Fibrocyte Accumulation in BALB/c Mouse Lung upon Transient Overexpression of Oncostatin M. Cells 2019, 8, 126. [Google Scholar] [CrossRef]
- Kim, D.; Pertea, G.; Trapnell, C.; Pimentel, H.; Kelley, R.; Salzberg, S.L. TopHat2: Accurate Alignment of Transcriptomes in the Presence of Insertions, Deletions and Gene Fusions. Genome Biol 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq-A Python Framework to Work with High-Throughput Sequencing Data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, D.J.; Chen, Y.; Smyth, G.K. Differential Expression Analysis of Multifactor RNA-Seq Experiments with Respect to Biological Variation. Nucleic Acids Res 2012, 40, 4288–4297. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. EdgeR: A Bioconductor Package for Differential Expression Analysis of Digital Gene Expression Data. Bioinformatics 2009, 26, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; Oshlack, A. A Scaling Normalization Method for Differential Expression Analysis of RNA-Seq Data; 2010;
- Law, C.W.; Chen, Y.; Shi, W.; Smyth, G.K. Voom: Precision Weights Unlock Linear Model Analysis Tools for RNA-Seq Read Counts; 2014; Vol. 15;
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma Powers Differential Expression Analyses for RNA-Sequencing and Microarray Studies. Nucleic Acids Res 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Benjamini_and_Hochberg_1995.
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene Set Enrichment Analysis: A Knowledge-Based Approach for Interpreting Genome-Wide Expression Profiles; 2005;
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P.; et al. The STRING Database in 2021: Customizable Protein-Protein Networks, and Functional Characterization of User-Uploaded Gene/Measurement Sets. Nucleic Acids Res 2021, 49, D605–D612. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI Gene Expression and Hybridization Array Data Repository; 2002; Vol. 30;
- DePianto, D.J.; Chandriani, S.; Abbas, A.R.; Jia, G.; N’Diaye, E.N.; Caplazi, P.; Kauder, S.E.; Biswas, S.; Karnik, S.K.; Ha, C.; et al. Heterogeneous Gene Expression Signatures Correspond to Distinct Lung Pathologies and Biomarkers of Disease Severity in Idiopathic Pulmonary Fibrosis. Thorax 2015, 70, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Schafer, M.J.; White, T.A.; Iijima, K.; Haak, A.J.; Ligresti, G.; Atkinson, E.J.; Oberg, A.L.; Birch, J.; Salmonowicz, H.; Zhu, Y.; et al. Cellular Senescence Mediates Fibrotic Pulmonary Disease. Nat Commun 2017, 8. [Google Scholar] [CrossRef]
- De Lauretis, A.; Sestini, P.; Pantelidis, P.; Hoyles, R.; Hansell, D.M.; Goh, N.S.L.; Zappala, C.J.; Visca, D.; Maher, T.M.; Denton, C.P.; et al. Serum Interleukin 6 Is Predictive of Early Functional Decline and Mortality in Interstitial Lung Disease Associated with Systemic Sclerosis. J Rheumatol 2013, 40, 435, LP – 446. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Lin, C.J.F.; Furst, D.E.; Goldin, J.; Kim, G.; Kuwana, M.; Allanore, Y.; Matucci-Cerinic, M.; Distler, O.; Shima, Y.; et al. Tocilizumab in Systemic Sclerosis: A Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet Respir Med 2020, 8, 963–974. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Tanaka, K.; Fujita, T.; Umezawa, H.; Amano, H.; Yoshioka, K.; Naito, Y.; Hatano, M.; Kimura, S.; Tatsumi, K.; et al. Bidirectional Role of IL-6 Signal in Pathogenesis of Lung Fibrosis. Respir Res 2015, 16, 99. [Google Scholar] [CrossRef] [PubMed]
- Le, T.-T.T.; Karmouty-Quintana, H.; Melicoff, E.; Le, T.-T.T.; Weng, T.; Chen, N.-Y.; Pedroza, M.; Zhou, Y.; Davies, J.; Philip, K.; et al. Blockade of IL-6 Trans Signaling Attenuates Pulmonary Fibrosis. The Journal of Immunology 2014, 193, 3755–3768. [Google Scholar] [CrossRef] [PubMed]
- Segel, M.J.; Izbicki, G.; Cohen, P.Y.; Or, R.; Christensen, T.G.; Wallach-Dayan, S.B.; Breuer, R. Role of Interferon-in the Evolution of Murine Bleomycin Lung Fibrosis. Am J Physiol Lung Cell Mol Physiol 2003, 285, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.S.; Greenlee, B.M.; Wills-Karp, M.; Moller, D.R. Attenuation of Lung Inflammation and Fibrosis in Interferon-Deficient Mice after Intratracheal Bleomycin; 2001; Vol. 24;
- Vu, T.N.; Chen, X.; Foda, H.D.; Smaldone, G.C.; Hasaneen, N.A. Interferon-Γenhances the Antifibrotic Effects of Pirfenidone by Attenuating IPF Lung Fibroblast Activation and Differentiation. Respir Res 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Brown, K.K.; Bradford, W.Z.; Starko, K.; Noble, P.W.; Schwartz, D.A.; King, T.E. A Placebo-Controlled Trial of Interferon Gamma-1b in Patients with Idiopathic Pulmonary Fibrosis; 2004; Vol. 350;
- Segawa, S.; Goto, D.; Iizuka, A.; Kaneko, S.; Yokosawa, M.; Kondo, Y.; Matsumoto, I.; Sumida, T. The Regulatory Role of Interferon-γ Producing Gamma Delta T Cells via the Suppression of T Helper 17 Cell Activity in Bleomycin-Induced Pulmonary Fibrosis. Clin Exp Immunol 2016, 185, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Monnier, J.; Zabel, B.A. Anti-Asialo GM1 NK Cell Depleting Antibody Does Not Alter the Development of Bleomycin Induced Pulmonary Fibrosis. PLoS One 2014, 9, e99350–e99350. [Google Scholar] [CrossRef] [PubMed]
- El Taweel, M.; Walker, H.; Mekhael, O.; Vierhout, M.; Padwal, M.; Naiel, S.; Patel, H.; Parthasarathy, P.; Imani, J.; Ayaub, E.; et al. Dectin-1 Is a Marker of Alternatively Activated Macrophages and a Therapeutic Target in Interstitial Fibrotic Lung Diseases. In C64. PULMONARY FIBROSIS MODELS AND MECHANISTIC INSIGHTS; American Thoracic Society International Conference Abstracts; American Thoracic Society, 2019; pp. A5403–A5403.
- Choi, J.E.; Lee, S.S.; Sunde, D.A.; Huizar, I.; Haugk, K.L.; Thannickal, V.J.; Vittal, R.; Plymate, S.R.; Schnapp, L.M. Insulin-Like Growth Factor-i Receptor Blockade Improves Outcome in Mouse Model of Lung Injury. Am J Respir Crit Care Med 2009, 179, 212–219. [Google Scholar] [CrossRef]
- Hung, C.F.; Rohani, M.G.; Lee, S. soon; Chen, P.; Schnapp, L.M. Role of IGF-1 Pathway in Lung Fibroblast Activation. Respir Res 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Chen, L.; Song, B.; Cui, Z.; Chen, G.; Yu, Z.; Song, B. Insulin-like Growth Factor-2 (IGF-2) in Fibrosis. Biomolecules 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Waldrep, K.M.; Rodgers, J.I.; Garrett, S.M.; Wolf, B.J.; Feghali-Bostwick, C.A. The Role of SOX9 in IGF-II-Mediated Pulmonary Fibrosis. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



