Submitted:
26 July 2024
Posted:
29 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Search Strategy
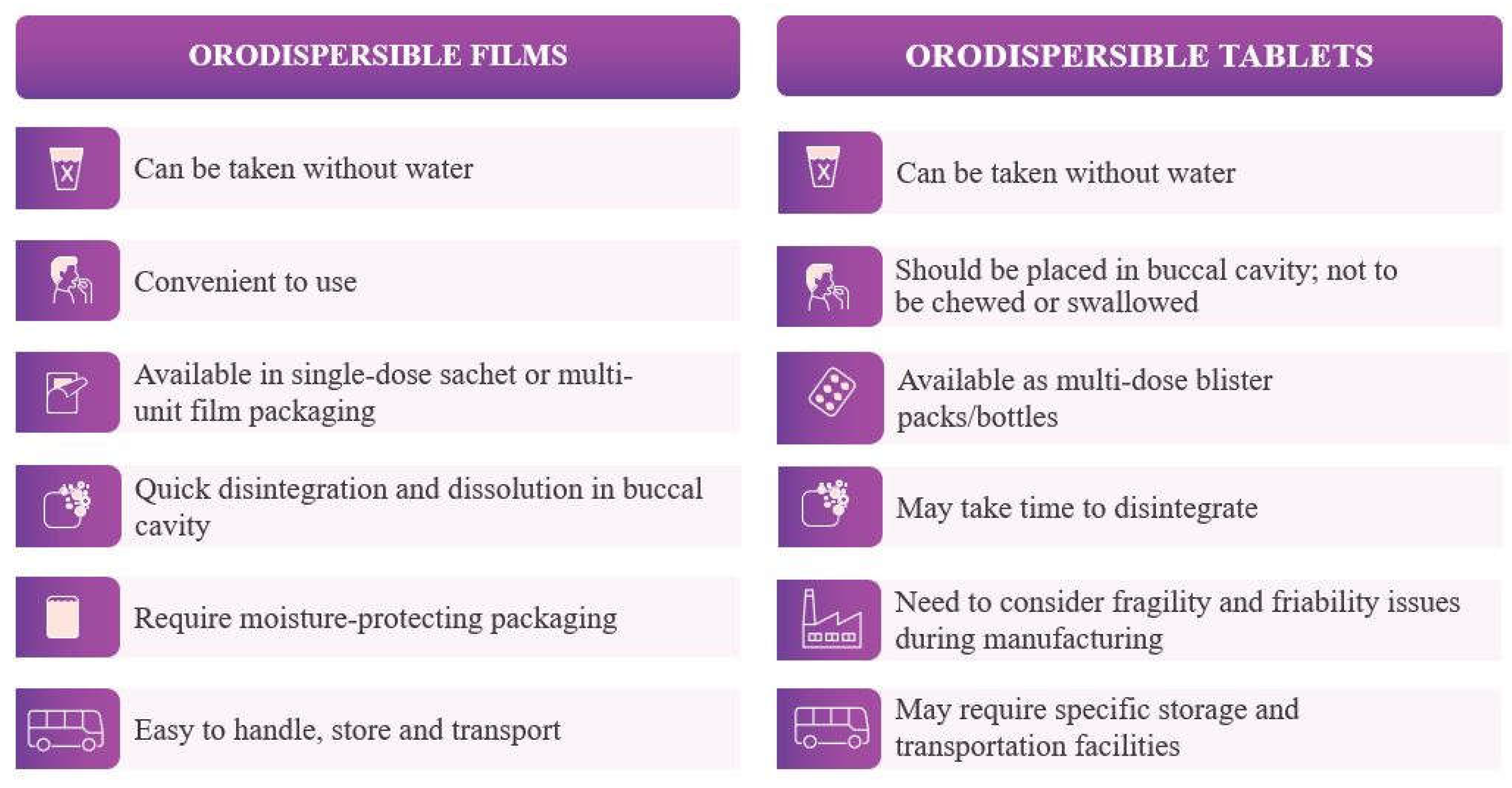
2.1. Orodispersible Dosage Forms
2.2. Manufacturing Methods
2.3. Bioequivalence and Stability Testing
2.4. Stress Test
3. Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Ferlak J, Guzenda W, Osmałek, T. Orodispersible films—Current state of the art, limitations, advances and future perspectives. Pharmaceutics. 2023, 15, 361. [CrossRef] [PubMed]
- Lau, E.T.; Steadman, K.J.; Cichero, J.A.; Nissen, L.M. Dosage form modification and oral drug delivery in older people. Adv. Drug Deliv. Rev. 2018, 135, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Bala, R.; Khanna, S.; Pawar, P.; Arora, S. Orally dissolving strips: A new approach to oral drug delivery system. Int. J. Pharm. Investig. 2013, 3, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.; Nair, A.B.; Boddu, S.H.S.; Gorain, B.; Sreeharsha, N.; Shah, J. An Updated Overview of the Emerging Role of Patch and Film-Based Buccal Delivery Systems. Pharmaceutics 2021, 13, 1206. [Google Scholar] [CrossRef]
- Salawi, A. An Insight into Preparatory Methods and Characterization of Orodispersible Film—A Review. Pharmaceuticals 2022, 15, 844. [Google Scholar] [CrossRef] [PubMed]
- Morath, B.; Sauer, S.; Zaradzki, M.; Wagner, A. Orodispersible films – Recent developments and new applications in drug delivery and therapy. Biochem. Pharmacol. 2022, 200, 115036. [Google Scholar] [CrossRef] [PubMed]
- Jannini, E.A.; Droupy, S. Needs and Expectations of Patients with Erectile Dysfunction: An Update on Pharmacological Innovations in Phosphodiesterase Type 5 Inhibition with Focus on Sildenafil. Sex. Med. 2019, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.S.; Kumar, T.P.; Gowda, D.V. Orodispersible Thin Film: A new patient-centered innovation. J. Drug Deliv. Sci. Technol. 2020, 59, 101843. [Google Scholar] [CrossRef]
- zakar RS, Özakar, E. Current overview of oral thin films. Turkish journal of pharmaceutical sciences. 2021, 18, 111.
- Karki, S.; Kim, H.; Na, S.-J.; Shin, D.; Jo, K.; Lee, J. Thin films as an emerging platform for drug delivery. Asian, J. Pharm. Sci. 2016, 11, 559–574. [Google Scholar] [CrossRef]
- Irfan, M.; Rabel, S.; Bukhtar, Q.; Qadir, M.I.; Jabeen, F.; Khan, A. Orally disintegrating films: A modern expansion in drug delivery system. Saudi Pharm. J. 2015, 24, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Kathpalia, H.; Gupte, A. An Introduction to Fast Dissolving Oral Thin Film Drug Delivery Systems: A Review. Curr. Drug Deliv. 2013, 10, 667–684. [Google Scholar] [CrossRef] [PubMed]
- Borges AF, Silva C, Coelho JF, Simões, S. Oral films: current status and future perspectives: I—galenical development and quality attributes. Journal of Controlled Release. 2015, 206, 1–19.
- Cilurzo, F.; Musazzi, U.M.; Franzé, S.; Selmin, F.; Minghetti, P. Orodispersible dosage forms: biopharmaceutical improvements and regulatory requirements. Drug Discov. Today 2018, 23, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Mahboob MBH, Riaz T, Jamshaid M, Bashir, I., Zulfiqar, S. Oral films: A comprehensive review. International Current Pharmaceutical Journal 2016, 5, 111–117. [Google Scholar] [CrossRef]
- Saini P, Kumar A, Sharma P, Visht, S. Fast disintegrating oral films: A recent trend of drug delivery. Int J Drug Dev Res. 2012, 4, 80–94.
- Desai, P.P.; Date, A.A.; Patravale, V.B. Overcoming poor oral bioavailability using nanoparticle formulations – opportunities and limitations. Drug Discov. Today: Technol. 2012, 9, e87–e95. [Google Scholar] [CrossRef] [PubMed]
- Food, U., Administration, D. Quality by design for ANDAs: an example for immediate-release dosage forms. US Department of Health and Human Service (FDA, Rockville, MD, 2012). 2012.
- Hoffmann, E.M.; Breitenbach, A.; Breitkreutz, J.; Pharm, D.; D., P. Advances in orodispersible films for drug delivery. Expert Opin. Drug Deliv. 2011, 8, 299–316. [Google Scholar] [CrossRef] [PubMed]
- Gijare, C.; Deshpande, A. Orodispersible Films: A Systematic Patent Review. Recent Patents Drug Deliv. Formul. 2018, 12, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Turković, E.; Vasiljević, I.; Drašković, M.; Parojčić, J. Orodispersible films — Pharmaceutical development for improved performance: A review. J. Drug Deliv. Sci. Technol. 2022, 75, 103708. [Google Scholar] [CrossRef]
- Goel, H.; Rai, P.; Rana, V.; Tiwary, A.K. Orally Disintegrating Systems: Innovations in Formulation and Technology. Recent Patents Drug Deliv. Formul. 2008, 2, 258–274. [Google Scholar] [CrossRef] [PubMed]
- Nagar P, Singh K, Chauhan I, Verma M, Yasir M, Khan A, et al. Orally disintegrating tablets: formulation, preparation techniques and evaluation. Journal of Applied Pharmaceutical Science. 2011:35-45.
- Shaw A, Lawrence TE, Yan T, Liu M, Summers N, Daggumati, V., et al. Bioequivalence Studies of Sildenafil Citrate Orodispersible Film Administered With and Without Water versus Viagra® Film-coated Tablets in Healthy Male Subjects. Current Therapeutic Research. 2023:100708.
- Andersson, K.-E. Mechanisms of Penile Erection and Basis for Pharmacological Treatment of Erectile Dysfunction. Pharmacol. Rev. 2011, 63, 811–859. [Google Scholar] [CrossRef] [PubMed]
- Gratzke, C.; Angulo, J.; Chitaley, K.; Dai, Y.-T.; Kim, N.N.; Paick, J.-S.; Simonsen, U.; Ückert, S.; Wespes, E.; Andersson, K.E.; et al. Anatomy, Physiology, and Pathophysiology of Erectile Dysfunction. J. Sex. Med. 2010, 7, 445–475. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Cucinotta, D.; Di Lorenzo, G.; Ferlin, A.; Giagulli, V.A.; Gnessi, L.; Isidori, A.M.; Maiorino, M.I.; Miserendino, P.; Murrone, A.; et al. The Italian Society of Andrology and Sexual Medicine (SIAMS), along with ten other Italian Scientific Societies, guidelines on the diagnosis and management of erectile dysfunction. J. Endocrinol. Investig. 2023, 46, 1241–1274. [Google Scholar] [CrossRef] [PubMed]
- Scaglione, F.; Donde, S.; Hassan, T.A.; Jannini, E.A. Phosphodiesterase Type 5 Inhibitors for the Treatment of Erectile Dysfunction: Pharmacology and Clinical Impact of the Sildenafil Citrate Orodispersible Tablet Formulation. Clin. Ther. 2017, 39, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Jannini, E.A.; Lenzi, A.; Isidori, A.; Fabbri, A. Subclinical Erectile Dysfunction: Proposal for a Novel Taxonomic Category in Sexual Medicine. J. Sex. Med. 2006, 3, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Artom, N.; Pinna, G.; Musso, N.R.; Orlandini, F.; Malasoma, P.; Uccelli, M.; Artom, A.; Rabbia, F.; Pascale, C.; Lantieri, F.; et al. Prevalence of erectile dysfunction in a cohort of Italian hypertensive subjects. Clin. Exp. Hypertens. 2015, 38, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Yafi FA, Jenkins L, Albersen M, Corona G, Isidori AM, Goldfarb, S., et al. Erectile dysfunction. Nature reviews Disease primers. 2016, 2, 1–20.
- Muneer A, Kalsi J, Nazareth I, Arya, M. Erectile dysfunction. BMJ. 2014, 348:129.
- Aytaç; McKinlay, J. B.; Krane, R.J. The likely worldwide increase in erectile dysfunction between 1995 and 2025 and some possible policy consequences. BJU Int. 1999, 84, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Rastrelli, G.; Isidori, A.; Pivonello, R.; Bettocchi, C.; Reisman, Y.; Sforza, A.; Maggi, M. Erectile dysfunction and cardiovascular risk: a review of current findings. Expert Rev. Cardiovasc. Ther. 2020, 18, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Jannini, EA. SM= SM: The interface of systems medicine and sexual medicine for facing non-communicable diseases in a gender-dependent manner. Sexual Medicine Reviews. 2017, 5, 349–64. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Zhang, F.; Zhang, Y.; Wang, W.; Ran, Y.; Wu, C.; Zhu, S.; Qin, F.; Yuan, J. Insights into modifiable risk factors of erectile dysfunction, a wide-angled Mendelian Randomization study. J. Adv. Res. 2024, 58, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Terentes-Printzios, D.; Ioakeimidis, N.; Aznaouridis, K.; Rokkas, K.; Synodinos, A.; Christoforatou, E.; Aggelis, A.; Samentzas, A.; Stefanadis, C. PREDICTION OF CARDIOVASCULAR EVENTS AND ALL-CAUSE MORTALITY WITH ERECTILE DYSFUNCTION: A SYSTEMATIC REVIEW AND META-ANALYSIS OF COHORT STUDIES. Circ. 2012, 59, E2074. [Google Scholar] [CrossRef]
- Yannas D, Sansone A, Jannini EA. The canary in the coal mine. Comment on “Association between cardiometabolic index and erectile dysfunction among US adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey 2001–2004”. International Journal of Impotence Research. 2024:1-2.
- Dolci, S.; Belmonte, A.; Santone, R.; Giorgi, M.; Pellegrini, M.; Carosa, E.; Piccione, E.; Lenzi, A.; Jannini, E.A. Subcellular localization and regulation of type-1C and type-5 phosphodiesterases. Biochem. Biophys. Res. Commun. 2006, 341, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Cesarini, V.; Guida, E.; Campolo, F.; Crescioli, C.; Di Baldassarre, A.; Pisano, C.; Balistreri, C.R.; Ruvolo, G.; Jannini, E.A.; Dolci, S. Type 5 phosphodiesterase (PDE5) and the vascular tree: From embryogenesis to aging and disease. Mech. Ageing Dev. 2020, 190, 111311–111311. [Google Scholar] [CrossRef] [PubMed]
- Mónica FZ, De Nucci, G. Tadalafil for the treatment of benign prostatic hyperplasia. Expert opinion on pharmacotherapy. 2019, 20, 929–37.
- Brant, W. , Lue, T. , Smith, J. Evaluation and management of erectile dysfunction in clinical practice. Journal of Clinical Outcomes Management. 2009, 16, 83–96. [Google Scholar]
- Seftel, A.D. Phosphodiesterase type 5 inhibitor differentiation based on selectivity, pharmacokinetic, and efficacy profiles. Clin. Cardiol. 2004, 27, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.; Hill, S. A comparison of the available phosphodie sterase-5 inhibitors in the treatment of erectile dysfunction: a focus on avanafil. Patient Preference Adherence 2015, ume 9, 1159–1164. [Google Scholar] [CrossRef]
- Hatzimouratidis, K.; Amar, E.; Eardley, I.; Giuliano, F.; Hatzichristou, D.; Montorsi, F.; Vardi, Y.; Wespes, E. Guidelines on Male Sexual Dysfunction: Erectile Dysfunction and Premature Ejaculation. Eur. Urol. 2010, 57, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Medicines.org.uk. Viagra 50 mg film-coated tablets - Summary of Product Characteristics (SmPC) - (emc). [updated 04 May 2023. Available from: https://www.medicines.org.uk/emc/product/7980/smpc#about-medicine.
- Corona, G.; Rastrelli, G.; Burri, A.; Serra, E.; Gianfrilli, D.; Mannucci, E.; Jannini, E.A.; Maggi, M. First-generation phosphodiesterase type 5 inhibitors dropout: a comprehensive review and meta-analysis. Andrology 2016, 4, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Shimoyama, H.; Sasaki, H.; Ogushi, Y.; Niikura, A.; Ota, T.; Ichimura, Y.; Hshimoto, Y.; Kurokawa, I.; Sugishita, H.; Tanifuji, S.; et al. 167 Clinical Analysis on the Pharmaceutical Formulation of VIAGRA OD Film. J. Sex. Med. 2022, 19, S190–S190. [Google Scholar] [CrossRef]
- Radicioni, M.; Castiglioni, C.; Giori, A.; Cupone, I.; Frangione, V.; Rovati, S. Bioequivalence study of a new sildenafil 100 mg orodispersible film compared to the conventional film-coated 100 mg tablet administered to healthy male volunteers. Drug Des. Dev. Ther. 2017, 11, 1183–1192. [Google Scholar] [CrossRef]
- Roh, H.; Son, H.; Lee, D.; Yeon, K.J.; Kim, H.S.; Kim, H.; Park, K. Pharmacokinetic Comparison of an Orally Disintegrating Film Formulation With a Film-Coated Tablet Formulation of Sildenafil in Healthy Korean Subjects: A Randomized, Open-Label, Single-Dose, 2-Period Crossover Study. Clin. Ther. 2013, 35, 205–214. [Google Scholar] [CrossRef]
- Lv, Y.; Luo, B.; LaBadie, R.R.; Zhu, H.; Feng, Y.; Ernst, C.; Crownover, P.H.; Liang, Y.; Zhao, Q. Bioequivalence and Bioavailability of an Orodispersible Tablet of Sildenafil Citrate in Healthy Chinese Male Subjects. Clin. Pharmacol. Drug Dev. 2020, 9, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Maggi, M.; Jannini, E.A. EDEUS, a Real-Life Study on the Users of Phosphodiesterase Type 5 Inhibitors: Prevalence, Perceptions, and Health Care-Seeking Behavior Among European Men With a Focus on 2nd-Generation Avanafil. Sex. Med. 2018, 6, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Jannini, E.A.; Sternbach, N.; Limoncin, E.; Ciocca, G.; Gravina, G.L.; Tripodi, F.; Petruccelli, I.; Keijzer, S.; Isherwood, G.; Wiedemann, B.; et al. Health-Related Characteristics and Unmet Needs of Men with Erectile Dysfunction: A Survey in Five European Countries. J. Sex. Med. 2014, 11, 40–50. [Google Scholar] [CrossRef]
- Jannini, E.A.; Isidori, A.M.; Gravina, G.L.; Aversa, A.; Balercia, G.; Bocchio, M.; Boscaro, M.; Carani, C.; Corona, G.; Fabbri, A.; et al. The ENDOTRIAL Study: A Spontaneous, Open-Label, Randomized, Multicenter, Crossover Study on the Efficacy of Sildenafil, Tadalafil, and Vardenafil in the Treatment of Erectile Dysfunction. J. Sex. Med. 2009, 6, 2547–2560. [Google Scholar] [CrossRef]
- Zucchi, A.; Costantini, E.; Scroppo, F.I.; Silvani, M.; Kopa, Z.; Illiano, E.; Petrillo, M.G.; Cari, L.; Nocentini, G. The first-generation phosphodiesterase 5 inhibitors and their pharmacokinetic issue. Andrology 2019, 7, 804–817. [Google Scholar] [CrossRef]
- Tapolsky GH, Osborne DW. Bioerodable film for delivery of pharmaceutical compounds of mucosal surfaces. Google Patents; 2000.
- Shimoda, H.; Taniguchi, K.; Nishimura, M.; Matsuura, K.; Tsukioka, T.; Yamashita, H.; Inagaki, N.; Hirano, K.; Yamamoto, M.; Kinosada, Y.; et al. Preparation of a fast dissolving oral thin film containing dexamethasone: A possible application to antiemesis during cancer chemotherapy. Eur. J. Pharm. Biopharm. 2009, 73, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Efremov E, Kasatonova E, Mel’nik YI, Nikushina, A. PDE-5 inhibitors: patients’ preferences. Urologiia. 2017,120-6.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Contents | Amount |
| API | 5-30% w/w |
| Water soluble polymer | 45% w/w |
| Plasticizers | 0-20% w/w |
| Surfactants | q.s. |
| Sweetening agents | 3-6% w/w |
| Saliva stimulating agents | 2-6% w/w |
| Fillers, colors, flavors | q.s. |
| Quality Attributes for ODF | Specifications |
| Physical attributes | |
|
Length, width, and thickness allow the film to be convenient for placement on the surface of the tongue. The size of the film is 1 cm × 1 cm, and the thickness is 100 µm. |
|
High tensile strength, high elongation at break, and low Young’s modulus. |
| Identification | Positive for drug |
| Assay | 100% w/w of label claim |
| Content uniformity | Conforms to USP <905> uniformity of dosage units |
| Disintegration time | Not more than 60 seconds |
| Dissolution | Acceptance criteria similar to the conventional immediate-release solid dosage forms |
| Parameter | Specification | Control | Negative | Positive | |||
|---|---|---|---|---|---|---|---|
|
Initial |
60°C 3W |
Initial |
60°C 3W |
Initial |
60°C 3W |
||
| Description | Product is a pale red, film-like oral disintegrating formulation. |
Meets the criteria |
|||||
| Purity (1) Degradation product |
UK-111,868 is ≤ 0.2% |
<0.05%* | 0.17% | <0.05% * | 0.17% | <0.05% * | 0.16% |
| Other individual degradation products is ≤ 0.2% | <0.05%* | <0.05%* | <0.05%* | <0.05%* | <0.05%* | <0.05%* | |
| The total amount of these degradation products is ≤ 0.5% | <0.05%* | 0.17% | <0.05%* | 0 . 17% | <0.05%* | 0 . 16% | |
| Purity (2) Residual solvents |
Ethanol is 1 0 mg/sheet or less | 3.7 mg | 3.9 mg | 5.8 mg | 2.9 mg | 2.6 mg | 2.7 mg |
| Disintegration | After 3 minutes, it has collapsed. | Meets the criteria | |||||
| Dissolution 01 | Dissolution rate over 80% for 45 mm. | 93-101% | 93- 102% | 93-105% | 95-102% | 91-106% | 96- 103% |
| Assay | 95.0-105 .0% of labeled amount | 100.67% | 100.93% | 101.30% | 101.73% | 102.22% | 101.28% |
| Water content | - | 1.943% | 1.679% | 1.798% | 2.036% | 1.795% | 1.672% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




