Submitted:
23 July 2024
Posted:
24 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
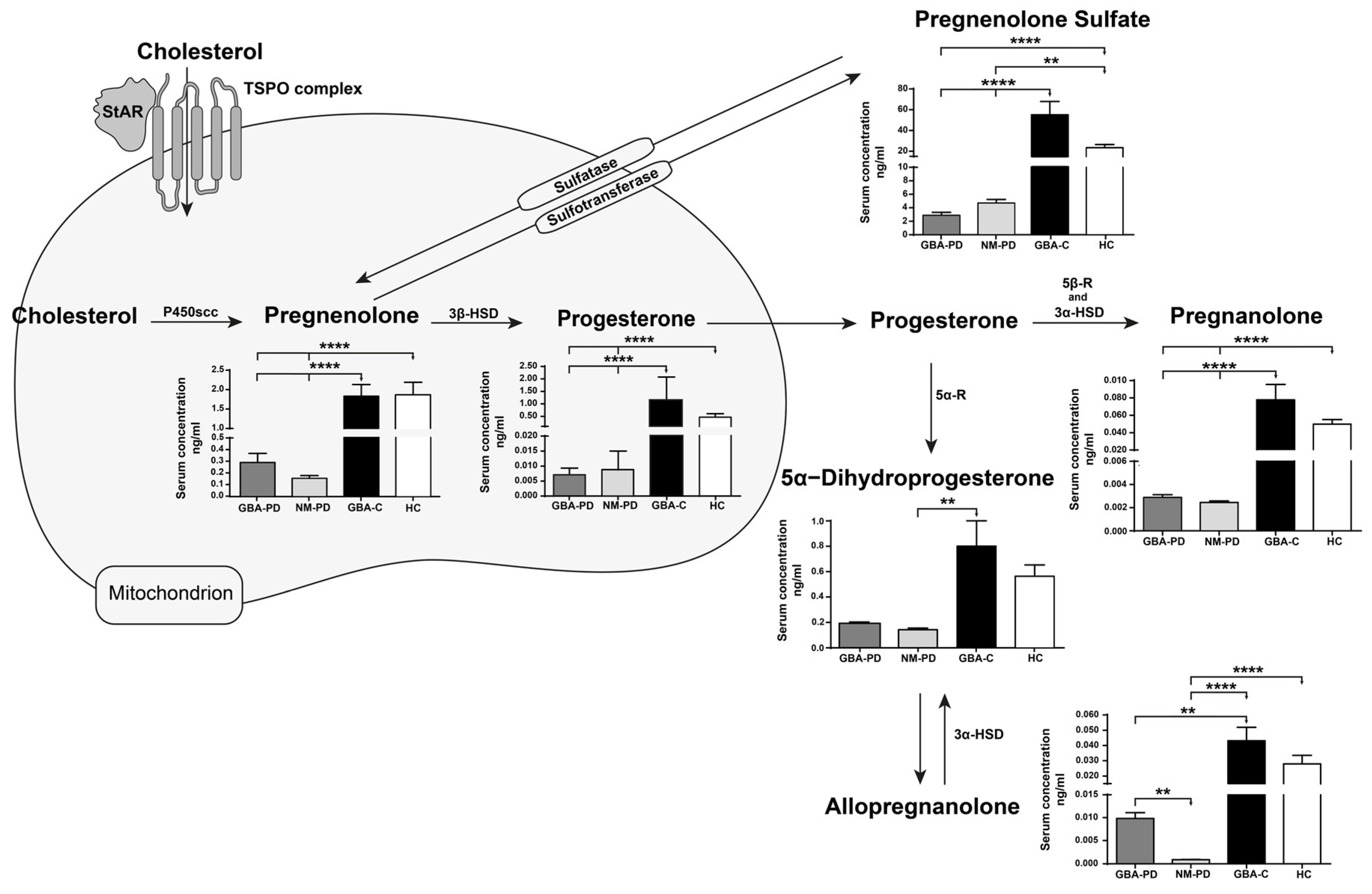
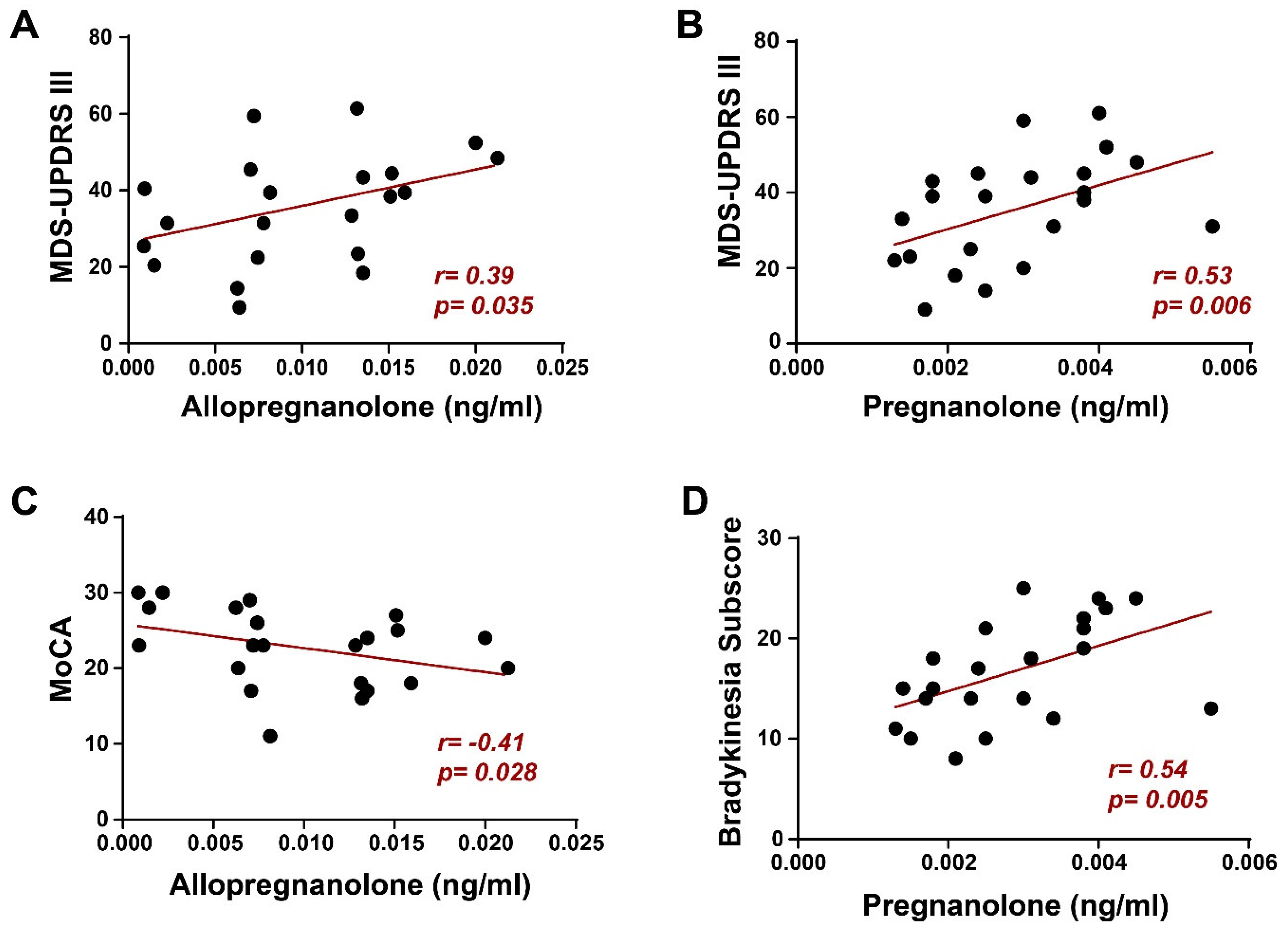
2. Results
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Clinical Assessment
4.3. Quantitative Analysis of NSs in Serum by Liquid Chromatography-Electrospray Tandem Mass Spectrometry (LC-MS/MS)
4.3.1. Chemicals and Reagents
4.3.2. Standard Solutions
4.3.3. Sample Preparation
4.3.4. LC-MS/MS Analysis
4.4. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kalia LV, Lang AE. Parkinson’s disease. Lancet Lond Engl. 2015;386:896–912.
- Riboldi, G.M.; Di Fonzo, A.B. GBA, Gaucher Disease, and Parkinson’s Disease: From Genetic to Clinic to New Therapeutic Approaches. Cells 2019, 8, 364. [CrossRef]
- Cavallieri F, Cury RG, Guimarães T, et al. Recent Advances in the Treatment of Genetic Forms of Parkinson’s Disease: Hype or Hope? Cells. 2023;12:764.
- Billingsley, K.J.; Ding, J.; Jerez, P.A.; Illarionova, A.; Levine, K.; Grenn, F.P.; Makarious, M.B.; Moore, A.; Vitale, D.; Reed, X.; et al. Genome-Wide Analysis of Structural Variants in Parkinson Disease. Ann. Neurol. 2023, 93, 1012–1022. [CrossRef]
- Petrucci S, Ginevrino M, Trezzi I, et al. GBA-Related Parkinson’s Disease: Dissection of Genotype-Phenotype Correlates in a Large Italian Cohort. Mov Disord Off J Mov Disord Soc. 2020;35:2106–2111.
- Menozzi E, Schapira AHV. Exploring the Genotype-Phenotype Correlation in GBA-Parkinson Disease: Clinical Aspects, Biomarkers, and Potential Modifiers. Front Neurol. 2021;12:694764.
- Cilia R, Tunesi S, Marotta G, et al. Survival and dementia in GBA-associated Parkinson’s disease: The mutation matters. Ann Neurol. 2016;80:662–673.
- Yılmaz, C.; Karali, K.; Fodelianaki, G.; Gravanis, A.; Chavakis, T.; Charalampopoulos, I.; Alexaki, V.I. Neurosteroids as regulators of neuroinflammation. Front. Neuroendocrinol. 2019, 55, 100788. [CrossRef]
- di Michele, F.; Longone, P.; Romeo, E.; Lucchetti, S.; Brusa, L.; Pierantozzi, M.; Bassi, A.; Bernardi, G.; Stanzione, P. Decreased plasma and cerebrospinal fluid content of neuroactive steroids in Parkinson’s disease. Neurol. Sci. 2003, 24, 172–173. [CrossRef]
- di Michele, F.; Luchetti, S.; Bernardi, G.; Romeo, E.; Longone, P. Neurosteroid and neurotransmitter alterations in Parkinson’s disease. Front. Neuroendocr. 2013, 34, 132–142. [CrossRef]
- Luchetti, S.; Liere, P.; Pianos, A.; Verwer, R.W.; Sluiter, A.; Huitinga, I.; Schumacher, M.; Swaab, D.F.; Mason, M.R. Disease stage-dependent changes in brain levels and neuroprotective effects of neuroactive steroids in Parkinson's disease. Neurobiol. Dis. 2023, 183, 106169. [CrossRef]
- Mullin, S.; Stokholm, M.G.; Hughes, D.; Mehta, A.; Parbo, P.; Hinz, R.; Pavese, N.; Brooks, D.J.; Schapira, A.H. Brain Microglial Activation Increased in Glucocerebrosidase (GBA) Mutation Carriers without Parkinson's disease. Mov. Disord. 2021, 36, 774–779. [CrossRef]
- Compagnone, N.A.; Mellon, S.H. Neurosteroids: Biosynthesis and Function of These Novel Neuromodulators. Front. Neuroendocr. 2000, 21, 1–56. [CrossRef]
- Mellon, S.H.; Griffin, L.D. Neurosteroids: biochemistry and clinical significance. Trends Endocrinol. Metab. 2002, 13, 35–43. [CrossRef]
- Frau, R.; Abbiati, F.; Bini, V.; Casti, A.; Caruso, D.; Devoto, P.; Bortolato, M. Targeting neurosteroid synthesis as a therapy for schizophrenia-related alterations induced by early psychosocial stress. Schizophr. Res. 2015, 168, 640–648. [CrossRef]
- Rupprecht R, Wetzel CH, Dorostkar M, et al. Translocator protein (18kDa) TSPO: a new diagnostic or therapeutic target for stress-related disorders? Mol Psychiatry. 2022;27:2918–2926.
- Walton, N.L.; Antonoudiou, P.; Maguire, J.L. Neurosteroid influence on affective tone. Neurosci. Biobehav. Rev. 2023, 152, 105327. [CrossRef]
- Frau, R.; Traccis, F.; Concas, L.; Cadeddu, R.; Mosher, L.J.; Nordkild, P.; Gaikwad, N.W.; Bortolato, M. Prefrontal allopregnanolone synergizes with D1 receptor activation to disrupt sensorimotor gating in male Sprague-Dawley rats. Psychopharmacology 2023, 240, 1359–1372. [CrossRef]
- Zoetmulder, M.; Biernat, H.B.; Nikolic, M.; Korbo, L.; Friberg, L.; Jennum, P.J. Prepulse Inhibition is Associated with Attention, Processing Speed, and 123I-FP-CIT SPECT in Parkinson's Disease. J. Park. Dis. 2014, 4, 77–87. [CrossRef]
- Lipari, N.; Centner, A.; Glinski, J.; Cohen, S.; Manfredsson, F.P.; Bishop, C. Characterizing the relationship between L-DOPA-induced-dyskinesia and psychosis-like behaviors in a bilateral rat model of Parkinson's disease. Neurobiol. Dis. 2023, 176, 105965. [CrossRef]
- Skrahina, V.; Gaber, H.; Vollstedt, E.; Förster, T.M.; Usnich, T.; Curado, F.; Brüggemann, N.; Paul, J.; Bogdanovic, X.; Zülbahar, S.; et al. The Rostock International Parkinson's Disease (ROPAD) Study: Protocol and Initial Findings. Mov. Disord. 2021, 36, 1005–1010. [CrossRef]
- Grisanti, S.; Fraternali, A.; Cavallieri, F.; Fioravanti, V.; Casali, M.; Toschi, G.; Ferri, L.; Sabadini, R.; Zedde, M.; Salomone, G.; et al. Author response for "Quantitative dopamine transporter imaging assessment in Parkinson's disease patients carrying GBA gene mutations compared with idiopathic PD patients: A case-control study". 2023. [CrossRef]
- Grisanti, S.; Ferri, L.; Cavallieri, F.; Fioravanti, V.; Vincenzi, C.; Toschi, G.; Grisendi, I.; Sabadini, R.; Paul, J.J.; Bauer, P.; et al. Increased Stroke Risk in Patients with Parkinson's Disease with LRRK2 Mutations. Mov. Disord. 2022, 37, 1117–1118. [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310:2191–2194.
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-sponsored revision of the Unified Parkinson's Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [CrossRef]
- Goetz CG, Poewe W, Rascol O, et al. Movement Disorder Society Task Force report on the Hoehn and Yahr staging scale: status and recommendations. Mov Disord Off J Mov Disord Soc. 2004;19:1020–1028.
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool For Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [CrossRef]
- Jost, S.T.; Kaldenbach, M.; Antonini, A.; Martinez-Martin, P.; Timmermann, L.; Odin, P.; Katzenschlager, R.; Borgohain, R.; Fasano, A.; Stocchi, F.; et al. Levodopa Dose Equivalency in Parkinson's Disease: Updated Systematic Review and Proposals. Mov. Disord. 2023, 38, 1236–1252. [CrossRef]
- Trivisano, M.; Lucchi, C.; Rustichelli, C.; Terracciano, A.; Cusmai, R.; Ubertini, G.M.; Giannone, G.; Bertini, E.S.; Vigevano, F.; Gecz, J.; et al. Reduced steroidogenesis in patients with PCDH19-female limited epilepsy. Epilepsia 2017, 58, e91–e95. [CrossRef]
- Meletti, S.; Lucchi, C.; Monti, G.; Giovannini, G.; Bedin, R.; Trenti, T.; Rustichelli, C.; Biagini, G. Decreased allopregnanolone levels in cerebrospinal fluid obtained during status epilepticus. Epilepsia 2017, 58, e16–e20. [CrossRef]
- Meletti, S.; Lucchi, C.; Monti, G.; Giovannini, G.; Bedin, R.; Trenti, T.; Rustichelli, C.; Biagini, G. Low levels of progesterone and derivatives in cerebrospinal fluid of patients affected by status epilepticus. J. Neurochem. 2018, 147, 275–284. [CrossRef]
- Brunialti E, Villa A, Mekhaeil M, et al. Inhibition of microglial β-glucocerebrosidase hampers the microglia-mediated antioxidant and protective response in neurons. J Neuroinflammation. 2021;18:220.
- Genazzani AR, Petraglia F, Bernardi F, et al. Circulating Levels of Allopregnanolone in Humans: Gender, Age, and Endocrine Influences. J Clin Endocrinol Metab. 1998;83:2099–2103.
- Bali, A.; Jaggi, A.S. Multifunctional aspects of allopregnanolone in stress and related disorders. Prog. Neuro-Psychopharmacology Biol. Psychiatry 2014, 48, 64–78. [CrossRef]
- Mosher, L.J.; Cadeddu, R.; Yen, S.; Staudinger, J.L.; Traccis, F.; Fowler, S.C.; Maguire, J.L.; Bortolato, M. Allopregnanolone is required for prepulse inhibition deficits induced by D1 dopamine receptor activation. Psychoneuroendocrinology 2019, 108, 53–61. [CrossRef]
- Darbra, S.; Mòdol, L.; Pallarès, M. Allopregnanolone infused into the dorsal (CA1) hippocampus increases prepulse inhibition of startle response in Wistar rats. Psychoneuroendocrinology 2012, 37, 581–585. [CrossRef]
- Zetsen, S.P.; Schellekens, A.F.; Paling, E.P.; Kan, C.C.; Kessels, R.P. Cognitive Functioning in Long-Term Benzodiazepine Users. Eur. Addict. Res. 2022, 28, 377–381. [CrossRef]
- Penninx, B.W.J.H.; Pine, D.S.; A Holmes, E.; Reif, A. Benzodiazepines for the long-term treatment of anxiety disorders? – Authors' reply. Lancet 2021, 398, 120–120. [CrossRef]
- de Gage, S.B.; Moride, Y.; Ducruet, T.; Kurth, T.; Verdoux, H.; Tournier, M.; Pariente, A.; Bégaud, B. Benzodiazepine use and risk of Alzheimer's disease: case-control study. BMJ 2014, 349, g5205–g5205. [CrossRef]
- Leng, Y.; Stone, K.L.; Yaffe, K. Race Differences in the Association Between Sleep Medication Use and Risk of Dementia. J. Alzheimer's Dis. 2023, 91, 1133–1139. [CrossRef]
- Bäckström T, Turkmen S, Das R, Doverskog M, Blackburn TP. The GABA system, a new target for medications against cognitive impairment—Associated with neuroactive steroids. J Intern Med. 2023;294:281–294.
- del Ser, T.; Zea, M.-A.; Valentí, M.; Olazarán, J.; López-Álvarez, J.; Rebollo-Vázquez, A.; Ávila-Villanueva, M.; Frades, B.; Medina, M.; A Fernández-Blázquez, M. Effects of commonly prescribed drugs on cognition and mild cognitive impairment in healthy elderly people. J. Psychopharmacol. 2019, 33, 965–974. [CrossRef]
| VARIABLE |
GBA-PD (n= 22) |
NM -PD (n= 22) |
GBA-C (n= 14) |
NM-HC (n= 15) |
| No. (%); mean [SD]; median {range} | ||||
| Age | 63.68 [8.43]; 63.50 {46.00-79.00} |
63.05 [8.59]; 62.50 {46.00-79.00} |
49.36 [12.58]; 47.50 {30.00-73.00}*°§ |
60.60 [11.77]; 62.00 {41.00-78.00} |
| Sex, male | 11/22 (50%) | 11/22 (50%) | 8/14 (57.10%) | 4/15 (26.67%) |
| LEDD | 704.32 [546.05]; 740.00 {.00-2297.00} |
693.68 [318.02]; 655.00 {157.00-1250.00} |
NA | NA |
| H&Y | 2.34 [.49]; 2.50 {1.00-3.00} |
2.45 [.50]; 2.50 {2.00-4.00} |
NA | NA |
| Disease duration | 7.77 [4.38]; 6.50 {2.00-17.00} |
7.41 [3.31]; 7.50 {3.00-15.00} |
NA | NA |
| MDS-UPDRS part-I score | 13.36 [7.42]; 13.00 {3.00-26.00} |
10.23 [6.04]; 9.50 {.00-22.00} |
2.21 [3.01]; 1.00 {.00-9.00}*° |
3.60 [2.29]; 4.00 {.00-8.00}*° |
|
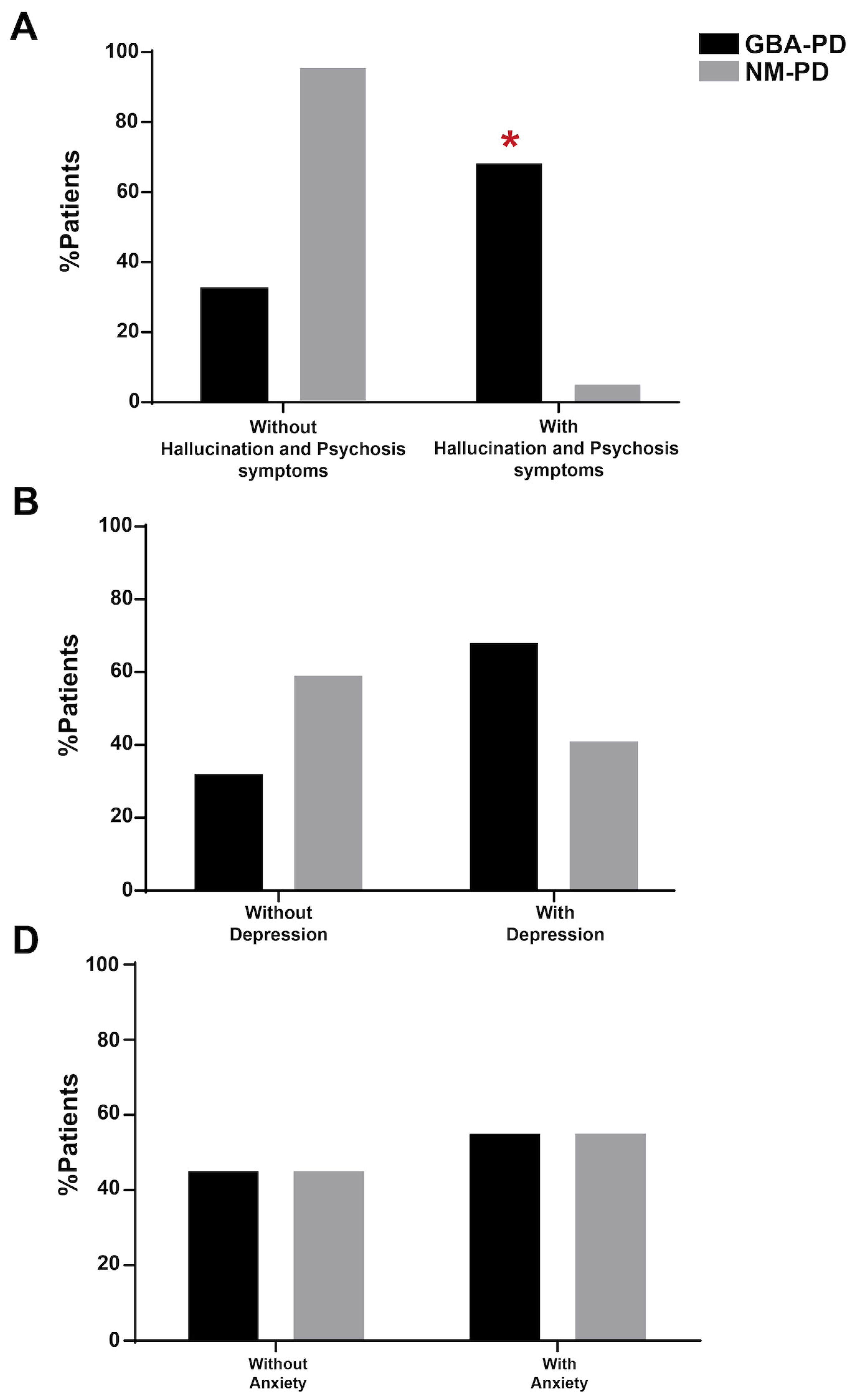
MDS-UPDRS item 1.2 (hallucinations and psychosis) Present Not present |
15/22 (68.2%) 7/22 (31.8%) ç |
1/22 (4.5%) 21/22 (95.5%) |
0/14 (.00%) 14/14 (100.00%) |
0/15 (.00%) 15/15 (100.00%) |
|
MDS-UPDRS item 1.3 (depressed mood) Present Not present |
15/22 (68.2%) 7/22 (31.8%) |
8/22 (36.4%) 13/22 (63.7%) |
3/14 (21.40%) 11/14 (78.60%) |
0/15 (.00%) 15/15 (100.00%) |
|
MDS-UPDRS item 1.4 (anxious mood) Present Not present |
12/22 (54.5%) 10/22 (45.5%) |
12/22 (54.5%) 10/22 (45.5%) |
3/14 (21.40%) 11/14 (78.60%) |
0/15 (.00%) 15/15 (100.00%) |
| MDS-UPDRS part-II score | 17.00 [10.52]; 15.00 {2.00-39.00} |
10.50 [5.68]; 11.50 {.00-21.00}^ |
.29 [.469]; .00 {.00-1.00}*° |
.80 [1.424]; .00 {.00-5.00}*° |
| MDS-UPDRS part-III score | 36.00 [13.22]; 38.50 {14.00-61.00} |
29.68 [11.77]; 28.00 {13.00-59.00} |
.00 [.00]; .00 {.00-.00}*° |
.33 [.724]; .00 {00.-2.00}*° |
| MDS-UPDRS part-IV score | 4.95 [4.89]; 3.00 {.00-14.00} |
3.36 [3.69]; 3.00 {.00-12.00} |
.00 [.00]; .00 {.00-.00}*° |
.00 [.00]; .00 {.00-.00}*° |
| Tremor Subscore | 4.05 [3.34]; 3.50 {.00-11.00} |
3.27 [3.74]; 2.00 {.00-16.00} |
.00 [.00]; .00 {.00-.00}*° |
.27 [.704]; .00 {.00-2.00}*° |
| Bradykinesia subscore | 17.86 [5.52]; 17.00 {8.00-26.00} |
14.95 [5.54]; 14.00 {6.00-22.00} |
.00 [.00]; .00 {.00-.00}*° |
.00 [.00]; .00 {.00-.00}*° |
| Rigidity subscore | 5.27 [2.89]; 5.00 {1.00-12.00} |
4.64 [2.88]; 4.00 {.00-11.00} |
.00 [.00]; .00 {.00-.00}*° |
00 [.00]; .00 {.00-.00}*° |
| PIGD subscore | 7.14 [4.70]; 7.00 {.00-19.00} |
4.68 [3.34]; 4.00 {.00-15.00} |
.00 [.00]; .00 {.00-.00}*° |
00 [.00]; .00 {.00-.00}*° |
| Gait subscore | 2.59 [1.81]; 2.00 {.00-7.00} |
1.73 [1.42]; 1.00 {.00-6.00} |
.00 [.00]; .00 {.00-.00}*° |
.07 [.258]; .00 {.00-1.00}*° |
| Fluctuations subscore | 3.55 [3.40]; 3.00 {.00-10.00} |
2.68 [2.80]; 2.50 {.00-8.00} |
.00 [.00]; .00 {.00-.00}*° |
00 [.00]; .00 {.00-.00}*° |
| Dyskinesia subscore | .62 [1.79]; .00 {.00-4.00} |
.68 [1.32]; .00 {.00-4.00} |
.00 [.00]; .00 {.00-.00}*° |
00 [.00]; .00 {.00-.00}*° |
| MoCA | 22.73 [5.10]; 23.00 {11.00-30.00} |
21.82 [5.43]; 24.00 {12.00-28.00} |
29.79 [.802]; 30.00 {27.00-30.00}*° |
29.40 [.986]; 30.00 {27.00-30.00}*° |
|
Motor phenotype Akineto-rigid Tremor |
13/22 (59.10%) 9/22 (40.90%) |
9/22 (40.90%) 13/22 (59.10%) |
NA | NA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




