Submitted:
21 July 2024
Posted:
24 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Various Combinations of LW/O/E Injections:
2.3. Analytical Method Development Using High-Performance Liquid Chromatography (HPLC)
2.4. Stability of Injectable Formulations
2.5. Preparation of Optimized Formulations
2.6. Viscosity Determination
2.7. Injectability Test
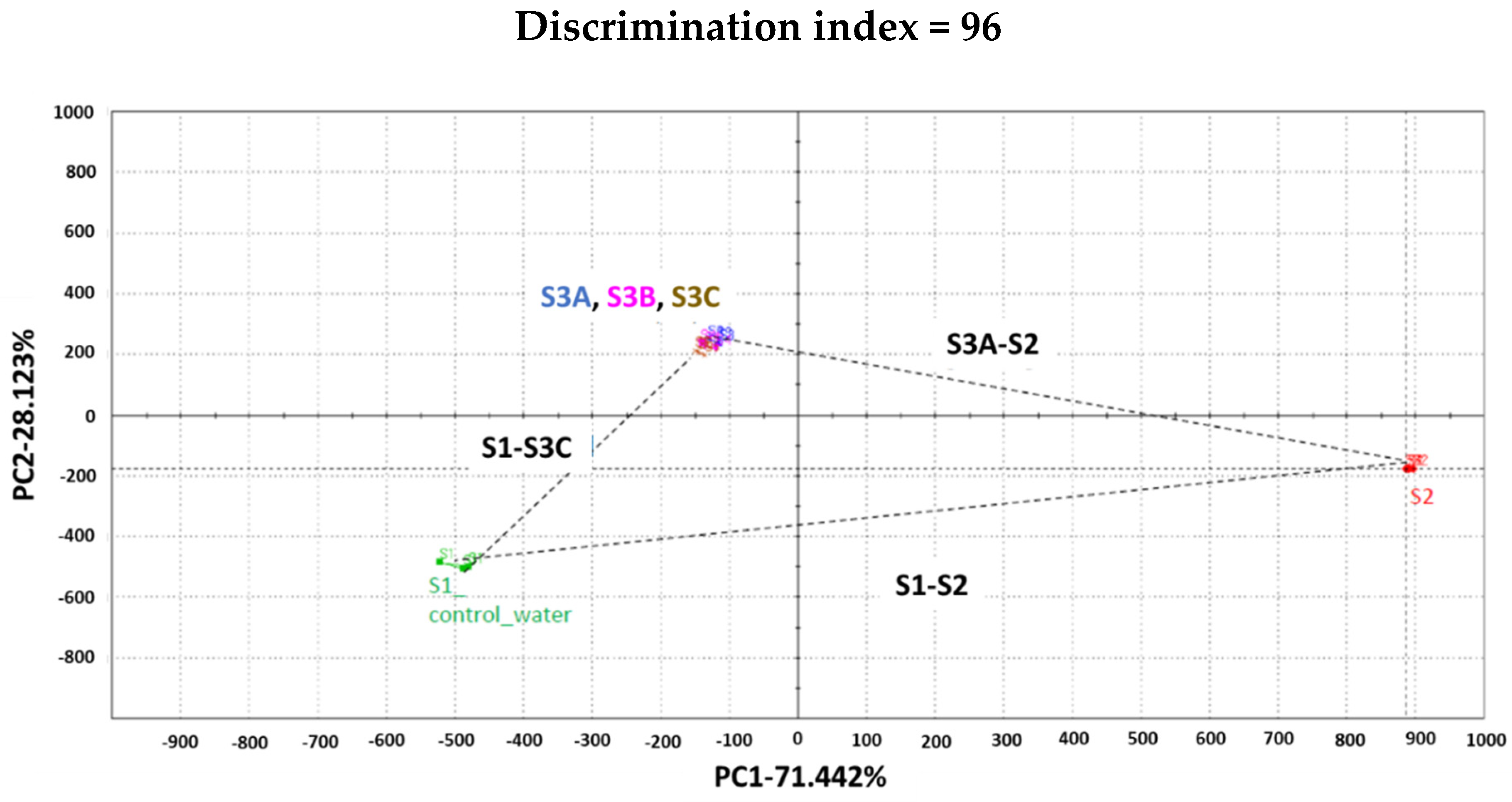
2.8. Taste Analysis Using ETongue
2.9. Local Toxicity and Efficacy Studies
2.9.1. Animals
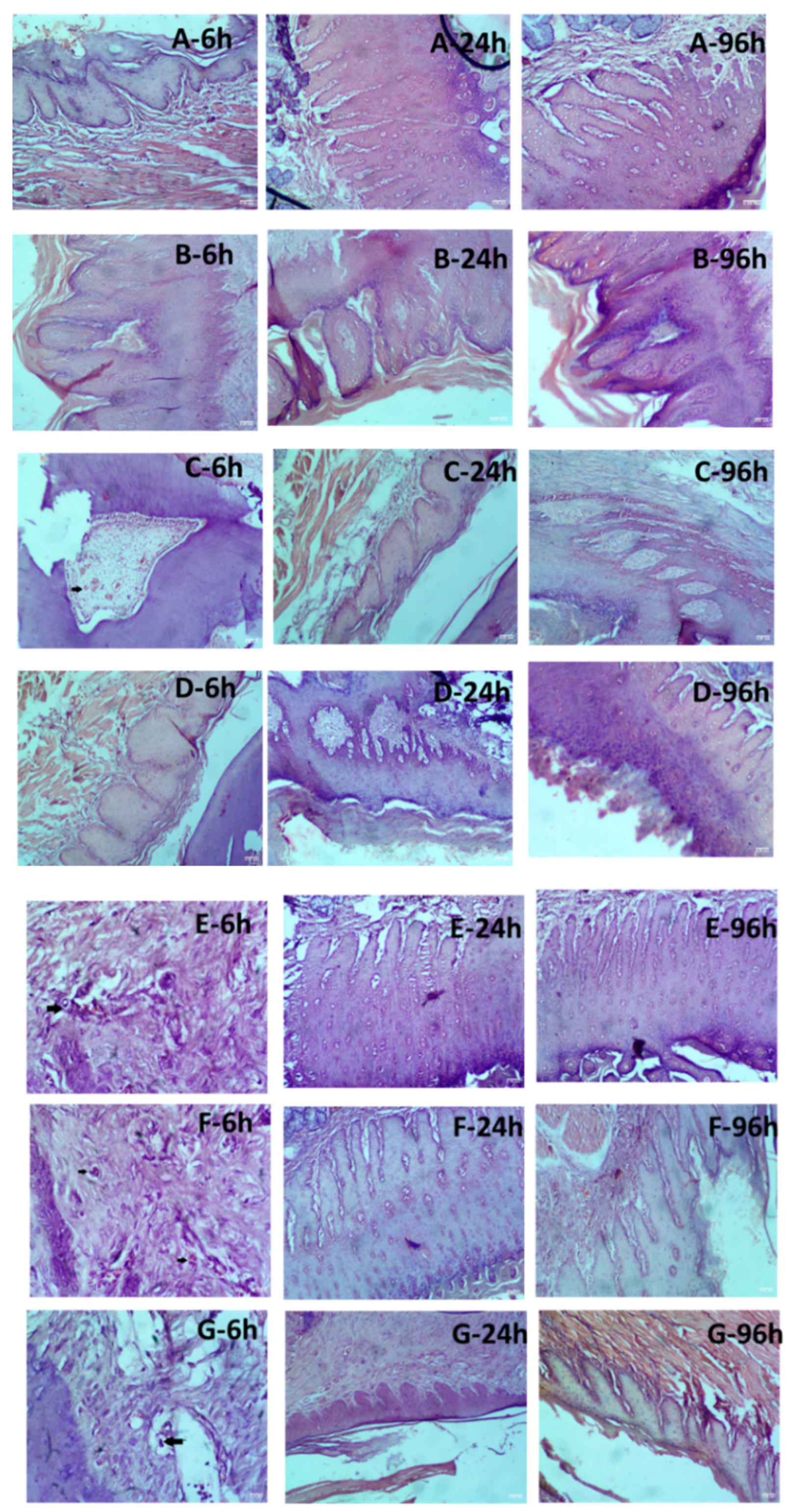
2.9.2. Local Toxicity of LW/O/E Formulations In Sprague Dawley Rats
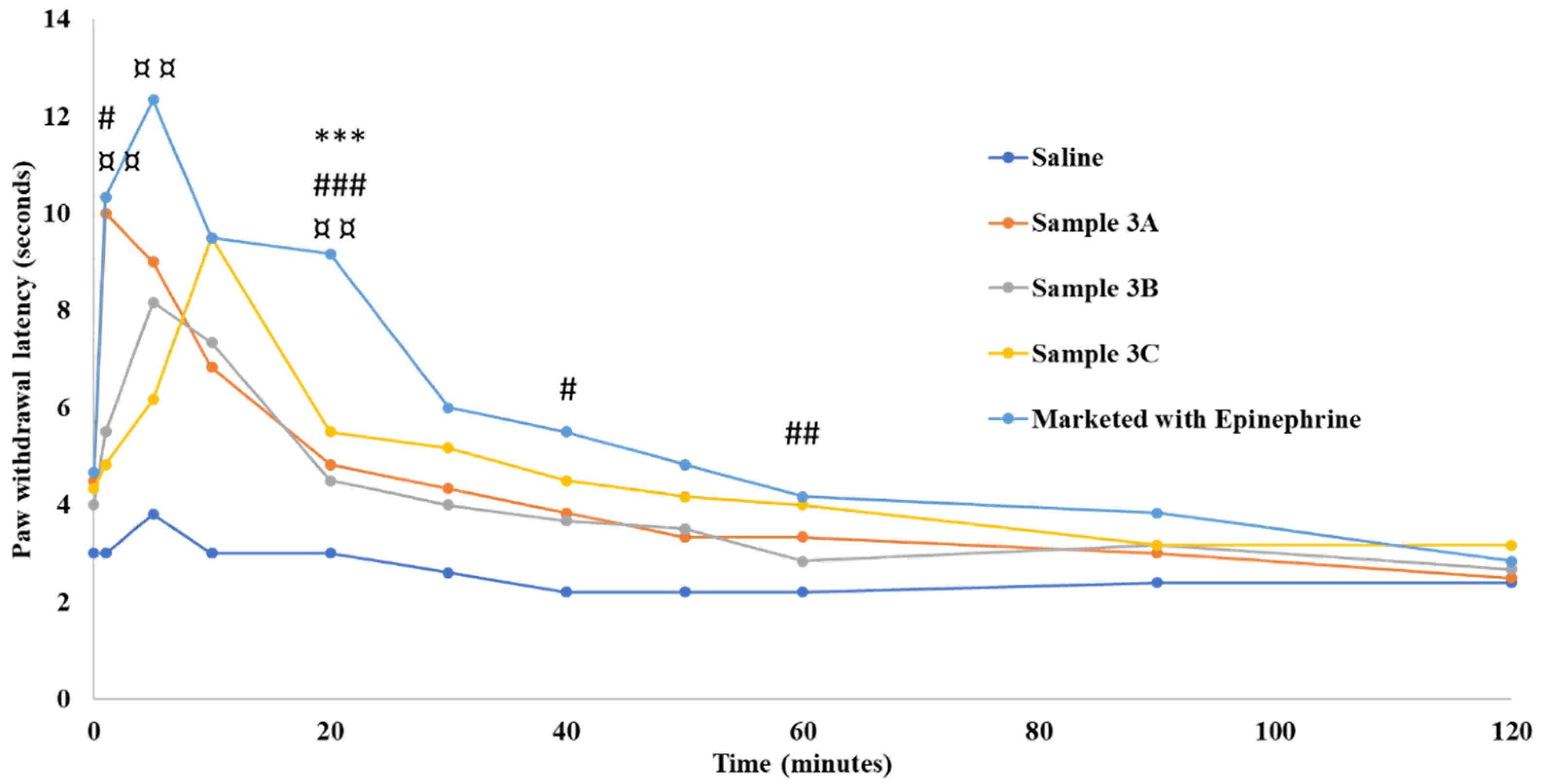
2.9.3. Efficacy Studies of LW/O/E Formulations In Sprague Dawley Rats
2.9.4. Radiant Heat Tail-Flick Latency (RHTFL) Test
2.9.5. Radiant Heat Hot Plate (HP) Test
2.10. Statistical Analysis
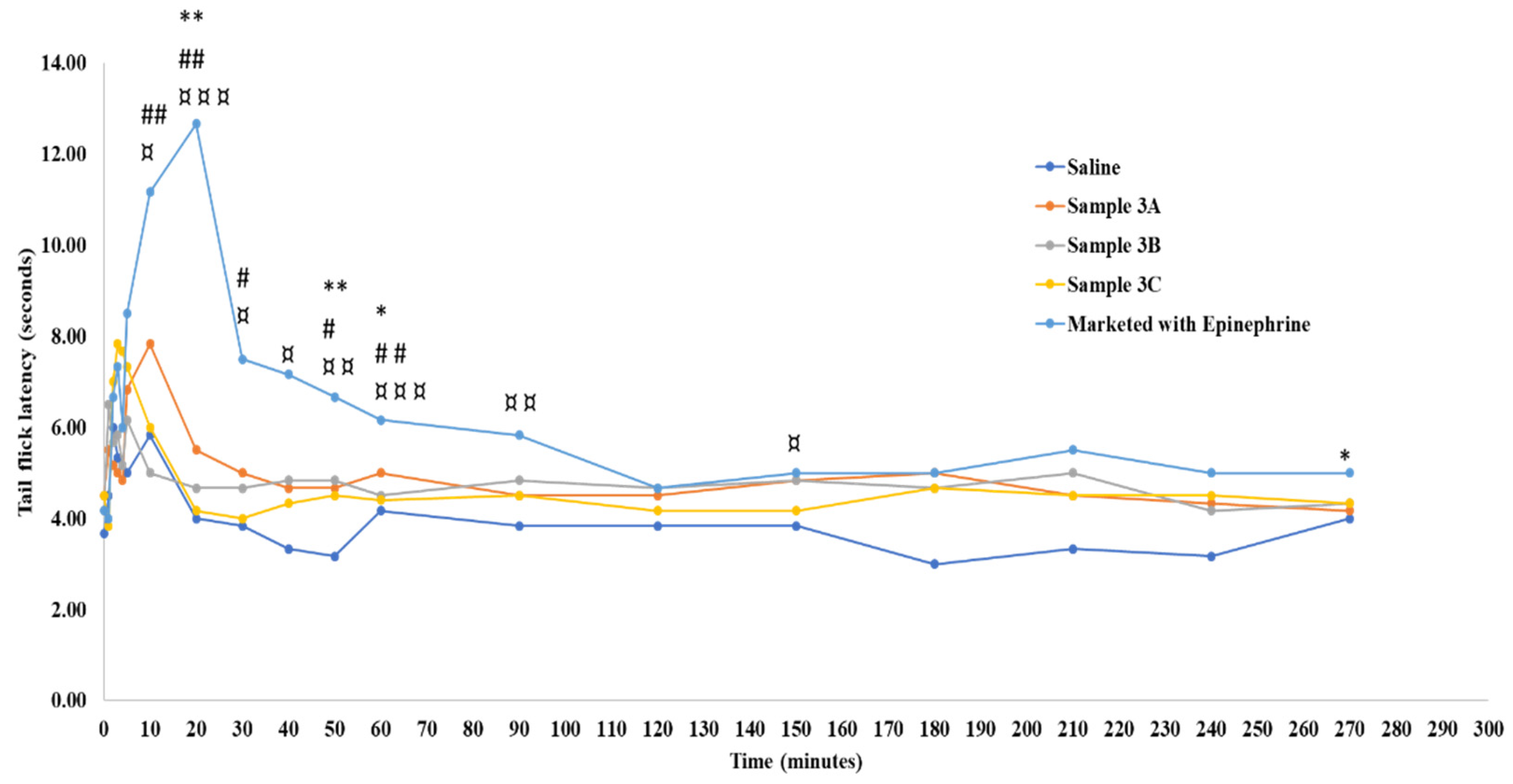
3. Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Huang, D., Wun, E., & Stern, A. (2011). Current treatments and advances in pain and anxiety management. In Dental Clinics of North America (Vol. 55, Issue 3). [CrossRef]
- Crawford, S., Niessen, L., Wong, S., & Dowling, E. (2005). Quantification of patient fears regarding dental injections and patient perceptions of a local noninjectable anesthetic gel. Compendium of Continuing Education in Dentistry (Jamesburg, N.J. : 1995), 26(2 Suppl 1).
- Kohli, K., Ngan, P., Crout, R., & Linscott, C. C. (2001). A survey of local and topical anesthesia use by pediatric dentists in the United States. Pediatric Dentistry, 23(3).
- Gaffar, B. O., Alagl, A. S., & Al-Ansari, A. A. (2014). The prevalence, causes, and relativity of dental anxiety in adult patients to irregular dental visits. Saudi Medical Journal, 35(6).
- Malamed, S. (2019). Handbook of Local Anesthesia-Mosby. Elsevier.
- Löfgren N, Wilson L. Studies on local anesthetics: Xylocaine, a new synthetic drug. 1948: Stockholm.
- Santamaria, C. M., Woodruff, A., Yang, R., & Kohane, D. S. (2017). Drug delivery systems for prolonged duration local anesthesia. In Materials Today (Vol. 20, Issue 1). [CrossRef]
- Kim, H., Hwang, K., Yun, S. M., & Kim, D. J. (2020). Usage of Epinephrine Mixed With Lidocaine in Plastic Surgery. Journal of Craniofacial Surgery, 31(3). [CrossRef]
- Kaufman, E., Goharian, S., & Katz, Y. (2000). Adverse reactions triggered by dental local anesthetics: a clinical survey. Anesthesia Progress, 47(4).
- Arora, G., Degala, S., & Dasukil, S. (2019). Efficacy of buffered local anaesthetics in head and neck infections. British Journal of Oral and Maxillofacial Surgery, 57(9). [CrossRef]
- Malamed, S. F., Tavana, S., & Falkel, M. (2013). Faster onset and more comfortable injection with alkalinized 2% lidocaine with epinephrine 1:100,000. Compendium of Continuing Education in Dentistry (Jamesburg, N.J. : 1995), 34 Spec No 1.
- Vent, A.; Surber, C.; Johansen, N.T.G.; Figueiredo, V.; Schönbächler, G.; Imhof, L.; Buset, C.; Hafner, J. Buffered lidocaine 1%/epinephrine 1:100,000 with sodium bicarbonate (sodium hydrogen carbonate) in a 3:1 ratio is less painful than a 9:1 ratio: A double-blind, randomized, placebo-controlled, crossover trial. J. Am. Acad. Dermatol. 2020, 83, 159–165, . [CrossRef]
- Donnelly, R.F.; Pascuet, E.; Garceau, D.; Vaillancourt, R. Buffered Lidocaine Hydrochloride Solution With and Without Epinephrine: Stability in Polypropylene Syringes. Can. J. Hosp. Pharm. 2009, 62, 375–380, . [CrossRef]
- Harbert, H. Counteracting the bitter taste of local anesthetic. J. Am. Dent. Assoc. 2001, 132, 1418–1419, . [CrossRef]
- Mennella, J.A.; Spector, A.C.; Reed, D.R.; Coldwell, S.E. The bad taste of medicines: Overview of basic research on bitter taste. Clin. Ther. 2013, 35, 1225–1246, . [CrossRef]
- Cram, A.; Breitkreutz, J.; Dessetbrethes, S.; Nunn, T.; Tuleu, C. Challenges of developing palatable oral paediatric formulations. Int. J. Pharm. 2009, 365, 1–3, . [CrossRef]
- White, S.A.; Goldhill, D.R. Is Hartmann's the solution?. Anaesthesia 1997, 52, 422–427, . [CrossRef]
- Singh S, Kerndt CC, Davis D. Ringer’s Lactate. [Updated 2023 Aug 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK500033/.
- Uzbelger Feldman D. Improved Local Anesthetic Solution for Dental and Contrast Media Use. PCT Application No. PCT/US15/10002. January 2014.
- Ito, E.; Ichinohe, T.; Shibukawa, Y.; Aida, H.; Kaneko, Y. Anesthetic duration of lidocaine with 10% dextran is comparable to lidocaine with 1:160 000 epinephrine after intraosseous injection in the rabbit. Oral Surgery, Oral Med. Oral Pathol. Oral Radiol. Endodontology 2007, 104, e26–e31, . [CrossRef]
- Wojtalewicz, S.; Vizmeg, J.; Erickson, S.; Lade, C.; Shea, J.; Sant, H.; Magda, J.; Gale, B.; Agarwal, J.; Davis, B. Evaluating the influence of particle morphology and density on the viscosity and injectability of a novel long-acting local anesthetic suspension. J. Biomater. Appl. 2022, 37, 724–736, . [CrossRef]
- Kanbay, M.; Yilmaz, S.; Dincer, N.; Ortiz, A.; A Sag, A.; Covic, A.; Sánchez-Lozada, L.G.; A Lanaspa, M.; I Cherney, D.Z.; Johnson, R.J.; et al. Antidiuretic Hormone and Serum Osmolarity Physiology and Related Outcomes: What Is Old, What Is New, and What Is Unknown?. J. Clin. Endocrinol. Metab. 2019, 104, 5406–5420, . [CrossRef]
- Vincent Fleury, Hany Charara, Richard Luscan, Laurent Schwartz. Effect of hyperosmolarity and hypoosmolarity on vascular remodeling : either way is a wrong way. 2021. ⟨hal-03449963⟩.
- Wei, Y.; Nedley, M.P.; Bhaduri, S.B.; Bredzinski, X.; Boddu, S.H.S. Masking the Bitter Taste of Injectable Lidocaine HCl Formulation for Dental Procedures. Aaps Pharmscitech 2014, 16, 455–465, . [CrossRef]
- Kirschenbaum, H.L.; Aronoff, W.; Perentesis, G.P.; Plitz, G.W.; Cutie, A.J. Stability and compatibility of lidocaine hydrochloride with selected large-volume parenterals and drug additives. Am. J. Heal. Pharm. 1982, 39, 1013–1015, . [CrossRef]
- Hocking, G.; Wildsmith, J.A.W. Intrathecal drug spread. Br. J. Anaesth. 2004, 93, 568–578, . [CrossRef]
- Hauser, R. A., & Hauser, M. A. (2011). Evidence-Based Use of Dextrose Prolotherapy for Musculoskeletal Pain: A Scientific Literature Review. Journal of Prolotherapy, 3(4).
- Rachid, O.; Rawas-Qalaji, M.; Simons, K.J. Epinephrine in Anaphylaxis: Preclinical Study of Pharmacokinetics after Sublingual Administration of Taste-Masked Tablets for Potential Pediatric Use. Pharmaceutics 2018, 10, 24, . [CrossRef]
- Ogawa, T.; Nakamura, T.; Tsuji, E.; Miyanaga, Y.; Nakagawa, H.; Hirabayashi, H.; Uchida, T. The Combination Effect of L-Arginine and NaCl on Bitterness Suppression of Amino Acid Solutions. Chem. Pharm. Bull. 2004, 52, 172–177, . [CrossRef]
- Agresti, C.; Tu, Z.; Ng, C.; Yang, Y.; Liang, J.F. Specific interactions between diphenhydramine and α-helical poly(glutamic acid) – A new ion-pairing complex for taste masking and pH-controlled diphenhydramine release. Eur. J. Pharm. Biopharm. 2008, 70, 226–233, . [CrossRef]
- Ghanchi, S. D., & Dhawale, S. C. (2011). Taste masking technologies of pharmaceuticals. In Research Journal of Pharmacy and Technology (Vol. 4, Issue 10).
- Bitner, D.P.; Feldman, D.U.; Axx, K.; Albandar, J.M. Description and evaluation of an intraoral cervical plexus anesthetic technique. Clin. Anat. 2015, 28, 608–613, . [CrossRef]
- Boddu, S.H.S.; Tukaramrao, D.B.; Al-Tabakha, M.M.; Ashames, A.; Neupane, R.; Babu, R.J.; Renukuntla, J.; Tiwari, A.K. Evaluation of Cytotoxicity and Taste-Masking Effect of Selected Flavors on Dental Lidocaine HCl Injection. Pharmaceuticals 2020, 13, 353, . [CrossRef]
- Uzbelger Feldman, D. Local anesthetic solution for dental and/or contrast media use. US Patent App. 16/545,263. July 2020.
- Neocaine, LLC. Anesthetic Formulations Method Trade Secret. Bernstein Blockchain Certificate ID: 7df611f0-7aca-486e-8d1c-7cf373b9bdbf. Feb 13, 2024.
- Bhusal, P.; Sharma, M.; Harrison, J.; Procter, G.; Andrews, G.; Jones, D.S.; Hill, A.G.; Svirskis, D. Development, Validation and Application of a Stability Indicating HPLC Method to Quantify Lidocaine from Polyethylene-co-Vinyl Acetate (EVA) Matrices and Biological Fluids. J. Chromatogr. Sci. 2017, 55, 832–838, . [CrossRef]
- United States Food and Drug Administration. 1959. Appraisal of the Safety of Chemicals in Foods, Drugs, and Cosmetics. Austin, Tex: Association of Food & Drug Officials of the United States.
- Cassens, J.; Prudic, A.; Ruether, F.; Sadowski, G. Solubility of Pharmaceuticals and Their Salts As a Function of pH. Ind. Eng. Chem. Res. 2013, 52, 2721–2731, . [CrossRef]
- Lidocaine Hydrochloride: AHFS 24:04.04.08”. ASHP® Injectable Drug Information™. Bethesda MD, USA: ASHP, 2021. . Web. 12 Jul. 2024.
- Martins, S.I.; Jongen, W.M.; van Boekel, M.A. A review of Maillard reaction in food and implications to kinetic modelling. Trends Food Sci. Technol. 2000, 11, 364–373, . [CrossRef]
- Cilurzo, F.; Selmin, F.; Minghetti, P.; Adami, M.; Bertoni, E.; Lauria, S.; Montanari, L. Injectability Evaluation: An Open Issue. Aaps Pharmscitech 2011, 12, 604–609, . [CrossRef]
- McPherson, J.S.; Dixon, S.A.; Townsend, R.; Vandewalle, K.S. Effect of Needle Design on Pain From Dental Local Anesthetic Injections. Anesthesia Prog. 2015, 62, 2–7, . [CrossRef]
- Rył, A.; Owczarz, P. Injectability of Thermosensitive, Low-Concentrated Chitosan Colloids as Flow Phenomenon through the Capillary under High Shear Rate Conditions. Polymers 2020, 12, 2260, . [CrossRef]
- Schwarzenbach, F.; Berteau, C.; Filipe-Santos, O.; Wang, T.; Rojas, H.; Granger, C. Evaluation of the impact of viscosity, injection volume, and injection flow rate on subcutaneous injection tolerance. Med Devices: Évid. Res. 2015, 8, 473–484, . [CrossRef]
- Usach, I.; Martinez, R.; Festini, T.; Peris, J.-E. Subcutaneous Injection of Drugs: Literature Review of Factors Influencing Pain Sensation at the Injection Site. Adv. Ther. 2019, 36, 2986–2996, . [CrossRef]
- Sasahara, T.; Yayama, K.; Tahara, T.; Onoe, H.; Okamoto, H. Na+/H+ exchanger inhibitor augments hyperosmolarity-induced vasoconstriction by enhancing actin polymerization. Vasc. Pharmacol. 2013, 59, 120–126, . [CrossRef]
- Ün, I.; Kurt, A.H.; Büyükafşar, K. Hyperosmolar glucose induces vasoconstriction through Rho/Rho-kinase pathway in the rat aorta. Fundam. Clin. Pharmacol. 2011, 27, 244–251, . [CrossRef]
- Schnuelle, M.L.; Hopster, K.; Toribio, R.E.; Hurcombe, S.D. Effects of 7.2% hypertonic saline solution on cardiovascular parameters and endogenous arginine vasopressin secretion in euvolemic isoflurane-anesthetized horses. Am. J. Veter- Res. 2022, 83, . [CrossRef]
- Bader, J.D.; Bonito, A.J.; Shugars, D.A. A systematic review of cardiovascular effects of epinephrine on hypertensive dental patients. Oral Surgery, Oral Med. Oral Pathol. Oral Radiol. Endodontology 2002, 93, 647–653, . [CrossRef]


| Ingredients | Sample 3A | Sample 3B | Sample 3C |
|---|---|---|---|
| Lidocaine hydrochloride monohydrate (g) | 5.332 | 5.065 | 3.999 |
| Sodium saccharine (g) | 0.225 | 0.225 | 0.263 |
| Dextrose (g) | 5 | 5.25 | 6 |
| Glutamic acid (g) | 1.25 | 1.25 | 1.25 |
| Glycine (g) | 0.5 | 0.5 | 0.5 |
| Citric acid (g) | 0.25 | 0.25 | 0.25 |
| L-arginine | 0.5 | 0.5 | 0.5 |
| Water for injection (ml) | 50 | 50 | 50 |
| 10 N Sodium hydroxide (to adjust pH 6.7-7.0) | 1.3 | 1.4 | 1.3 |
| Lactated Ringers (ml) | QS 250 | QS 250 | QS 250 |
| Osmolality (mOsm/kg) | 590-610 | 590-610 | 590-610 |
| Sample | Viscosity (cps) (mean value ± SD) n=3 |
10 ml syringe with 27G needle (Newtons) | 1 ml syringe with 30G needle (Newtons) |
|---|---|---|---|
| Marketed LW/E | 1.142 ± 0.006 | 19.45±0.35 | 2.12±0.16 |
| Sample 3A | 1.157 ± 0.006 | 19.20±0.01 | 1.68±0.01 |
| Sample 3B | 1.167 ± 0.012 | 19.85±0.07 | 2.78±0.16 |
| Sample 3C | 1.213 ± 0.008 | 20.13±0.23 | 3.87±0.53 |
| Code | SD | %RSD |
|---|---|---|
| S1-Water | 12.58 | 1.57 |
| S2-Marketed LW/E | 11.49 | 1.36 |
| S3A-Sample 3A | 5.06 | 0.58 |
| S3B-Sample 3B | 4.75 | 0.78 |
| S3C-Sample 3C | 4.43 | 0.53 |
| Erythema and Eschar Formation | Edema Formation | Histology | |||||||
| Samples/Time points | 6 h | 24 h | 96 h | 6 h | 24 h | 96 h | 6 h | 24 h | 96 h |
| 0.9% Saline solution | 0 | 0 | 0 | 0 | 0 | 0 | 1.0±0.0 | 1.0±0.0 | 1.0±0.0 |
| Marketed LW/E | 0 | 0 | 0 | 0 | 0 | 0 | 1.0±0.0 | 1.0±0.0 | 1.0±0.0 |
| Dextrose 5% and Sodium Chloride 0.45% Injection (406 mOsm/L) | 1.0±0.71 | 0 | 0 | 0 | 0 | 0 | 1.2±0.5 | 1.0±0.0 | 1.0±0.0 |
| Dextrose 10% w/v Solution (505 mOsm/L) | 1.6±0.6 | 0 | 0 | 0 | 0 | 0 | 1.6±0.55 | 1.0±0.0 | 1.0±0.0 |
| Sample 3A | 1.4±0.6* | 0.6±0.5 | 0 | 0.6±0.5 | 0 | 0 | 1.0±0.5*** | 1.0±0.0 | 1.0±0.0 |
| Sample 3B | 1.4±0.9* | 0 | 0 | 0.8±0.5 | 0 | 0 | 2.0±0.7*** | 1.2+0.5 | 1.2+0.5 |
| Sample 3C | 1.6±0.6** | 0 | 0 | 0.6±0.6 | 0 | 0 | 1.4±0.6 | 1.2+0.5 | 1.2+0.5 |
| ns: no significant difference, *p < 0.05 moderate difference; **p < 0.01 strong difference; ****p < 0.001 very strong difference | |||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




