Submitted:
23 July 2024
Posted:
23 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Recuitment of Participants and Eligibility Criteria
2.3. Data Collection, Research Instruments, and Procedures
2.3.1. Collection of Clinical and Demographic Data
2.3.2. SARS-CoV-2 Testing
2.3.3. Enzyme-Linked Immunosorbent Assays (ELISA) for Anti-SARS-CoV-2 IgM and IgG
2.4. Statistical Analysis
3. Results
3.1. Clinical and Demographic Characteristics
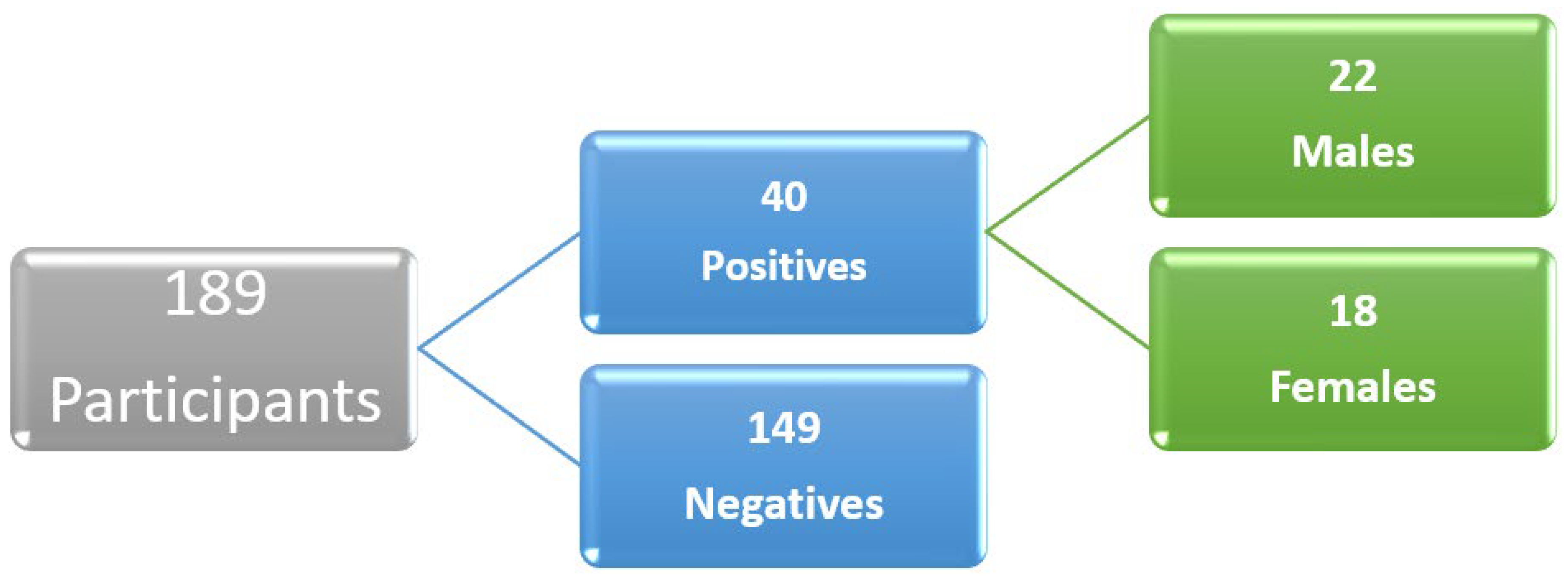
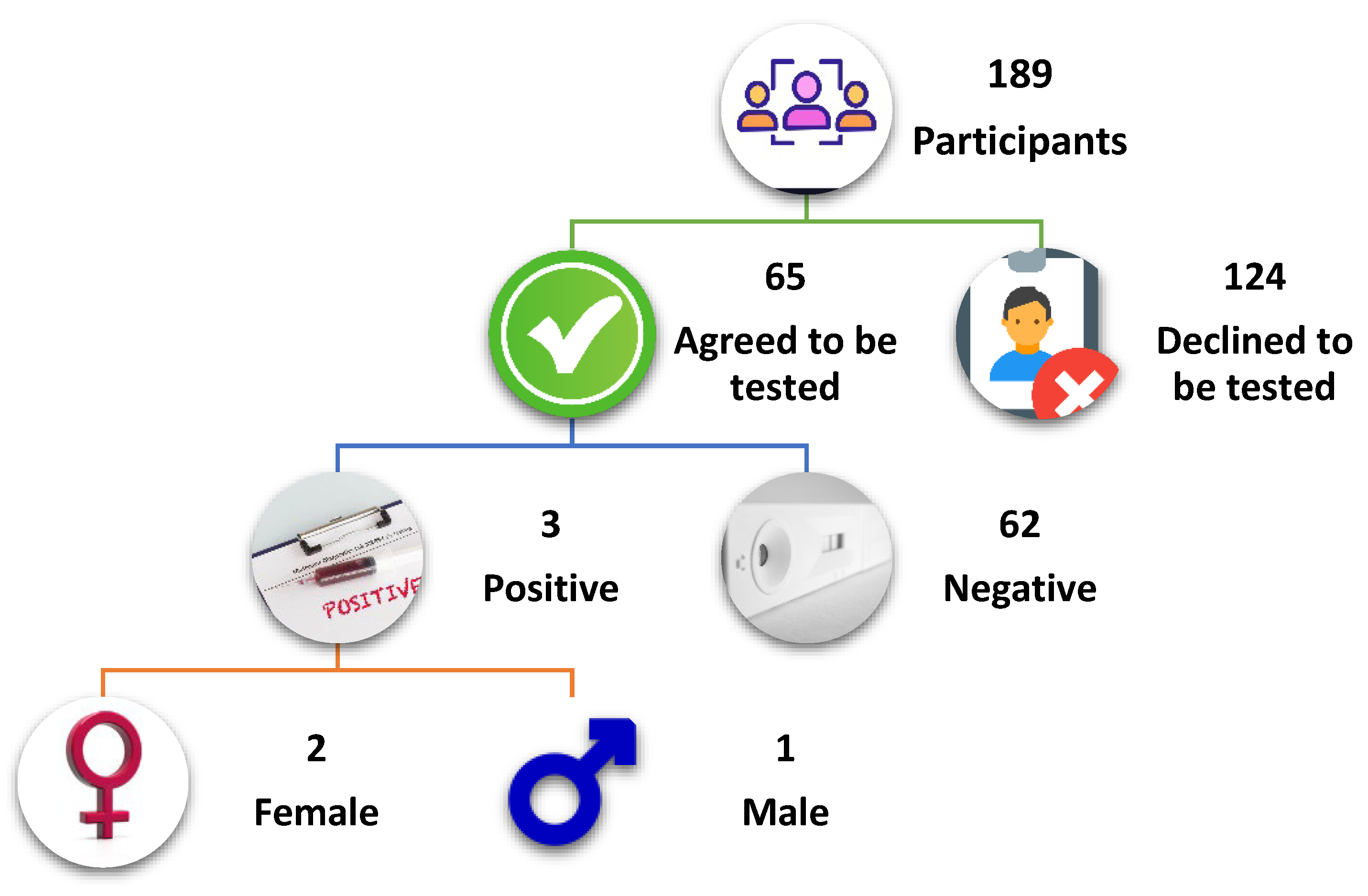
3.2. Previous and Current Status of COVID-19
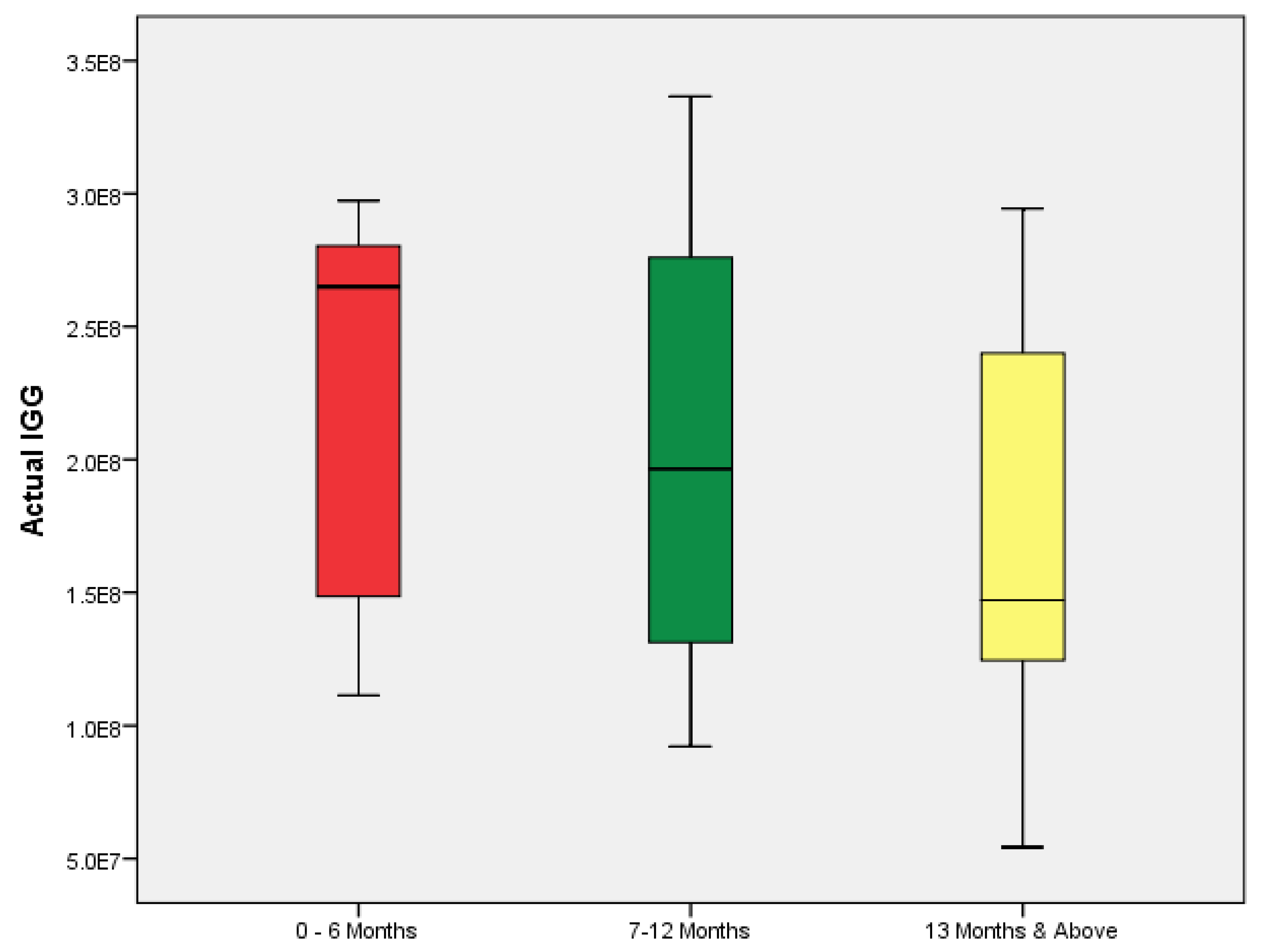
3.3. Seroprevalence of Anti-SARS-CoV-2 IgM and IgG and Comparison of IgG Titers
3.5. COVID-19 Vaccine Hesitancy
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Worldometer Kenya COVID - Coronavirus Statistics - Worldometer. Available online: https://www.worldometers.info/coronavirus/country/kenya/ (accessed on 4 July 2024).
- Bwire, G.; Ario, A.R.; Eyu, P.; Ocom, F.; Wamala, J.F.; Kusi, K.A.; Ndeketa, L.; Jambo, K.C.; Wanyenze, R.K.; Talisuna, A.O. The COVID-19 Pandemic in the African Continent. BMC Med. 2022, 20, 167. [Google Scholar] [CrossRef] [PubMed]
- Mohammad Hossein, Z.J.; Mehran, D. Seroprevalence of Anti-SARS-CoV-2 IgG and IgM Antibodies among Government Employees in Iran. Arch. Razi Inst. 2023, 78, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Chen, L.; Li, F.; He, A. Long-Term Evaluation of the Seroprevalence of SARS-CoV-2 IgG and IgM Antibodies in Recovered Patients: A Meta-Analysis. BMC Infect. Dis. 2023, 23, 444. [Google Scholar] [CrossRef]
- Adetifa, I.M.O.; Uyoga, S.; Gitonga, J.N.; Mugo, D.; Otiende, M.; Nyagwange, J.; Karanja, H.K.; Tuju, J.; Wanjiku, P.; Aman, R.; et al. Temporal Trends of SARS-CoV-2 Seroprevalence during the First Wave of the COVID-19 Epidemic in Kenya. Nat. Commun. 2021, 12, 3966. [Google Scholar] [CrossRef] [PubMed]
- Etyang, A.O.; Adetifa, I.; Omore, R.; Misore, T.; Ziraba, A.K.; Ng’oda, M.A.; Gitau, E.; Gitonga, J.; Mugo, D.; Kutima, B.; et al. SARS-CoV-2 Seroprevalence in Three Kenyan Health and Demographic Surveillance Sites, December 2020-May 2021. PLOS Glob. Public Health 2022, 2, e0000883. [Google Scholar] [CrossRef] [PubMed]
- Kagucia, E.W.; Ziraba, A.K.; Nyagwange, J.; Kutima, B.; Kimani, M.; Akech, D.; Ng’oda, M.; Sigilai, A.; Mugo, D.; Karanja, H.; et al. SARS-CoV-2 Seroprevalence and Implications for Population Immunity: Evidence from Two Health and Demographic Surveillance System Sites in Kenya, February-December 2022. Influenza Other Respir. Viruses 2023, 17, e13173. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Statement on the Fifteenth Meeting of the IHR (2005) Emergency Committee on the COVID-19 Pandemic. 2005. Available online: https://www.who.int/news/item/05-05-2023-statement-on-the-fifteenth-meeting-of-the-international-health-regulations-.
- Orangi, S.; Mbuthia, D.; Chondo, E.; Ngunu, C.; Kabia, E.; Ojal, J.; Barasa, E. A Qualitative Inquiry on Drivers of COVID-19 Vaccine Hesitancy among Adults in Kenya. PLOS Glob. Public Health 2024, 4, e0002986. [Google Scholar] [CrossRef]
- Khamadi, S.A.; Opanda, S.; Symekher, S.L.; Limbaso, S.K.; Langat, S.; Cirindi, J.K.; Mwangi, M.; Mwikwabe, N.; Okeyo, S.; Koskei, E.; et al. Whole-Genome Sequence Analysis Reveals the Circulation of Multiple SARS-CoV-2 Variants of Concern in Nairobi and Neighboring Counties, Kenya between March and July 2021. Virol. J. 2022, 19, 178. [Google Scholar] [CrossRef]
- Gerretsen, P.; Kim, J.; Caravaggio, F.; Quilty, L.; Sanches, M.; Wells, S.; Brown, E.E.; Agic, B.; Pollock, B.G.; Graff-Guerrero, A. Individual Determinants of COVID-19 Vaccine Hesitancy. PLoS ONE 2021, 16, e0258462. [Google Scholar] [CrossRef]
- Manirakiza, A.; Malaka, C.; Mossoro-Kpinde, H.D.; Yambiyo, B.M.; Mossoro-Kpinde, C.D.; Fandema, E.; Yakola, C.N.; Doyama-Woza, R.; Kangale-Wando, I.M.; Komba, J.E.K.; et al. Seroprevalence of Anti-SARS-CoV-2 Antibodies before and after Implementation of Anti-COVID-19 Vaccination among Hospital Staff in Bangui, Central African Republic. PLOS Glob. Public Health 2023, 3, e0001497. [Google Scholar] [CrossRef]
- Sorel, B.K.D.; Didiane, Y.M.; Samuel, M.M.; Pierre, F.C.S.; Caroline, D.T.N.; Richard, N.; Nicolas, N.Y. Seroprevalence of SARS-CoV-2 Antibodies among the University Population of Ngaoundere-Cameroon.
- Ruttoh, V.K.; Kipkoech, B.; Tonui, R.; John, R.O.; Mwangi, I.N.; Gicho, R.W.; Kamau, S.W.; Njihia, M.W.; Symekher, S.L.; Mwanzia, T.M.; et al. Seroprevalence of Anti-SARS-Cov-2 Antibodies among Patients Visiting Hospital-Based Sentinel Sites in the Rift Valley Region, Kenya 2024.
- Karl, T.; Schuster, A.; Stangassinger, L.M.; Stiboller, T.; Cadamuro, J.; Oostingh, G.J. Factors Affecting SARS-CoV-2 IgG Production after Vaccination and/or Disease: A Large-Scale Seroprevalence Study. Vaccines 2023, 11, 1615. [Google Scholar] [CrossRef] [PubMed]
- Bullock, J.L.; Hickey, T.E.; Kemp, T.J.; Metz, J.; Loftus, S.; Haynesworth, K.; Castro, N.; Luke, B.T.; Lowy, D.R.; Pinto, L.A. Longitudinal Assessment of BNT162b2- and mRNA-1273-Induced Anti-SARS-CoV-2 Spike IgG Levels and Avidity Following Three Doses of Vaccination. Vaccines 2024, 12, 516. [Google Scholar] [CrossRef] [PubMed]
- Pegu, A.; O’Connell, S.E.; Schmidt, S.D.; O’Dell, S.; Talana, C.A.; Lai, L.; Albert, J.; Anderson, E.; Bennett, H.; Corbett, K.S.; et al. Durability of mRNA-1273 Vaccine-Induced Antibodies against SARS-CoV-2 Variants. Science 2021, 373, 1372–1377. [Google Scholar] [CrossRef]
- Collier, A.-R.Y.; Yu, J.; McMahan, K.; Liu, J.; Chandrashekar, A.; Maron, J.S.; Atyeo, C.; Martinez, D.R.; Ansel, J.L.; Aguayo, R.; et al. Differential Kinetics of Immune Responses Elicited by Covid-19 Vaccines. N. Engl. J. Med. 2021, 385, 2010–2012. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, N.; Sasaki, A.; Kadowaki, T.; Mitsuhashi, T.; Takao, S.; Yorifuji, T. Longitudinal Antibody Dynamics after COVID-19 Vaccine Boosters Based on Prior Infection Status and Booster Doses. Sci. Rep. 2024, 14, 4564. [Google Scholar] [CrossRef] [PubMed]
- Kislaya, I.; Machado, A.; Magalhães, S.; Rodrigues, A.P.; Franco, R.; Leite, P.P.; Dias, C.M.; Nunes, B. COVID-19 mRNA Vaccine Effectiveness (Second and First Booster Dose) against Hospitalisation and Death during Omicron BA.5 Circulation: Cohort Study Based on Electronic Health Records, Portugal, May to July 2022. Eurosurveillance 2022, 27, 2200697. [Google Scholar] [CrossRef] [PubMed]
- Mattiuzzi, C.; Lippi, G. Efficacy of the Second COVID-19 Vaccine Booster Dose in the Elderly. Vaccines 2023, 11, 213. [Google Scholar] [CrossRef] [PubMed]
- Biedrzycka, B.W.; Bieńkowska, A.; Smolińska-Fijołek, E.; Biedrzycki, G.; Dorf, J. The Influence of Two Priming Doses of Different Anti-COVID-19 Vaccines on the Production of Anti-SARS-CoV-2 Antibodies After the Administration of the Pfizer/BioNTech Booster. Infect. Drug Resist. 2022, 15, 7811–7821. [Google Scholar] [CrossRef]
- Osur, J.; Muinga, E.; Carter, J.; Kuria, S.; Hussein, S.; Ireri, E.M. COVID-19 Vaccine Hesitancy: Vaccination Intention and Attitudes of Community Health Volunteers in Kenya. PLOS Glob. Public Health 2022, 2. [Google Scholar] [CrossRef]
- Rajshekhar, N.; Pinchoff, J.; Boyer, C.B.; Barasa, E.; Abuya, T.; Muluve, E.; Mwanga, D.; Mbushi, F.; Austrian, K. Exploring COVID-19 Vaccine Hesitancy and Uptake in Nairobi’s Urban Informal Settlements: An Unsupervised Machine Learning Analysis of a Longitudinal Prospective Cohort Study from 2021 to 2022. BMJ Open 2023, 13, e071032. [Google Scholar] [CrossRef]
- CDC Archive: COVID-19 Vaccination and Case Trends by Age Group, United States | Data | Centers for Disease Control and Prevention. Available online: https://data.cdc.gov/Vaccinations/Archive-COVID-19-Vaccination-and-Case-Trends-by-Ag/gxj9-t96f/about_data (accessed on 4 July 2024).
- Kim, S.; Willis, E.; Wehlage, S.; Scheffer-Wentz, H.; Dulitz, M. COVID-19 Vaccine Hesitancy and Short-Term and Long-Term Intentions among Unvaccinated Young Adults: A Mixed-Method Approach. BMC Public Health 2022, 22, 2030. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Chisty, M.A.; Alam, M.A.; Sakib, M.S.; Quader, M.A.; Shobuj, I.A.; Halim, M.A.; Rahman, F. Knowledge, Attitude, and Hesitancy towards COVID-19 Vaccine among University Students of Bangladesh. PLOS ONE 2022, 17, e0270684. [Google Scholar] [CrossRef] [PubMed]
| Variables | Frequency (N) | Percentages (%) | |
|---|---|---|---|
| Sex | Male | 93 | 49.2% |
| Female | 96 | 50.8% | |
| Age (years) | 18–19 | 30 | 15.9% |
| 20–29 | 135 | 71.4% | |
| 30–39 | 10 | 5.3% | |
| 40–49 | 9 | 4.8% | |
| ≥50 | 5 | 2.6% | |
| Level of education | College/University | 189 | 100% |
| Current occupation | Teaching staff | 10 | 5.3% |
| Non-teaching staff | 9 | 4.8% | |
| Students | 170 | 89.9% | |
| Vaccination status | Vaccinated | 107 | 56.6% |
| Not vaccinated | 82 | 43.4% |
| Variable | Frequency (N) | No. of positive | Prevalence | |
|---|---|---|---|---|
| IgM | ||||
| Overall | All participants | 189 | 24 | 12.7% |
| Sex | Male | 93 | 9 | 9.7% |
| Female | 96 | 15 | 15.6% | |
| Age (years) | 18–19 | 30 | 0 | 0.0% |
| 20–29 | 135 | 24 | 17.8% | |
| 30–39 | 10 | 0 | 0.0% | |
| 40–49 | 9 | 0 | 0.0% | |
| ≥50 | 5 | 0 | 0.0% | |
| Current occupation | Teaching staff | 10 | 0 | 0.0% |
| Non-teaching staff | 9 | 0 | 0.0% | |
| Students | 170 | 24 | 14.1% | |
| Vaccination status | Vaccinated | 107 | 22 | 20.6% |
| Not vaccinated | 82 | 2 | 2.4% | |
| IgG | ||||
| Overall | All participants | 189 | 166 | 87.8% |
| Sex | Male | 93 | 77 | 83.8% |
| Female | 96 | 89 | 92.7% | |
| Age (years) | 18–19 | 30 | 24 | 80.0% |
| 20–29 | 135 | 120 | 88.9% | |
| 30–39 | 10 | 10 | 100% | |
| 40–49 | 9 | 9 | 100% | |
| ≥50 | 5 | 5 | 100% | |
| Current occupation | Teaching staff | 10 | 10 | 100% |
| Non-teaching staff | 9 | 9 | 100% | |
| Students | 170 | 147 | 86.5% | |
| Vaccination status | Vaccinated | 107 | 105 | 98.1% |
| Not vaccinated | 82 | 61 | 74.4% |
| Variables | Number (N) |
Median x103 Units/mL |
IQR x103 Units/mL |
p | |
|---|---|---|---|---|---|
| Sex | Male | 93 | 145245.00 | 139330.00 | 0.024 U=3616 |
| Female | 96 | 180350.00 | 132730.00 | ||
| Age (years) | 18–19 | 30 | 160422.64 | 149457.50 | 0.123 H=7.260 |
| 20–29 | 135 | 146147.50 | 136992.50 | ||
| 30–39 | 10 | 184650.00 | 100855.00 | ||
| 40–49 | 9 | 266950.00 | 70400.00 | ||
| ≥50 | 5 | 201200.00 | 6546.55 | ||
| Vaccination status | Vaccinated | 107 | 189050.00 | 137355.00 | 0.001 U=2817.5 |
| Not vaccinated | 82 | 133120.00 | 139817.50 |
| Variables | Number (N) |
Median x103 Units/mL |
IQR x103 Units/mL |
P -Value | |
|---|---|---|---|---|---|
| Type of Vaccine | Moderna | 21 | 137295.00 | 117290.00 | 0.170, H=5.027 |
| AstraZeneca | 41 | 232150.00 | 110650.00 | ||
| Pfizer | 16 | 144145.00 | 149617.50 | ||
| Johnson & Johnson | 24 | 175772.50 | 139305.00 | ||
| Booster Doses | Received Booster Doses | 9 | 1747.63 | 4338.70 | 0.758 U=1087 |
| Did Not Receive Booster Doses | 78 | 2020.00 | 30185.71 |
| 3Cs model | Theme | Frequency* (%) |
Examples of Participants’ Responses | |
|---|---|---|---|---|
| Confidence | Health concerns/side effects | 15 (19.7%) | “Fear of adverse reactions (blood clotting)” “I had health concerns/suspicions about the vaccines side effect” |
|
| Lack of Information | 14 (18.4%) | “I have not been infected” “I had never experienced any COVID-related signs, so I thought my immune system is strong enough hence there was no need to introduce any vaccines or drugs” |
||
| Mistrust | 17 (22.4) | “I doubted the effectiveness of the vaccine” “I thought the vaccine was approved too fast” “I see no point in getting vaccinated, especially with something that is experimental (has not gone through the proper testing process)” |
||
| Religious and cultural reasons | 3 (3.9) | “religious and cultural reasons” | ||
| Convenience | Vaccine unavailability | 2 (2.6) | “Just missed out” “Had no access to the vaccine” |
|
| Lack of time | 3 (3.9) | “I couldn't spare time to obtain the vaccine” | ||
| Complacency | No reason | 11 (14.5) | “No any specific reason” | |
| Lack of interest | 9 (11.8) | “I didn’t feel the need to” | ||
| Procrastination | 2 (2.6) | “I just kept on postponing” |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




