Submitted:
20 July 2024
Posted:
22 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
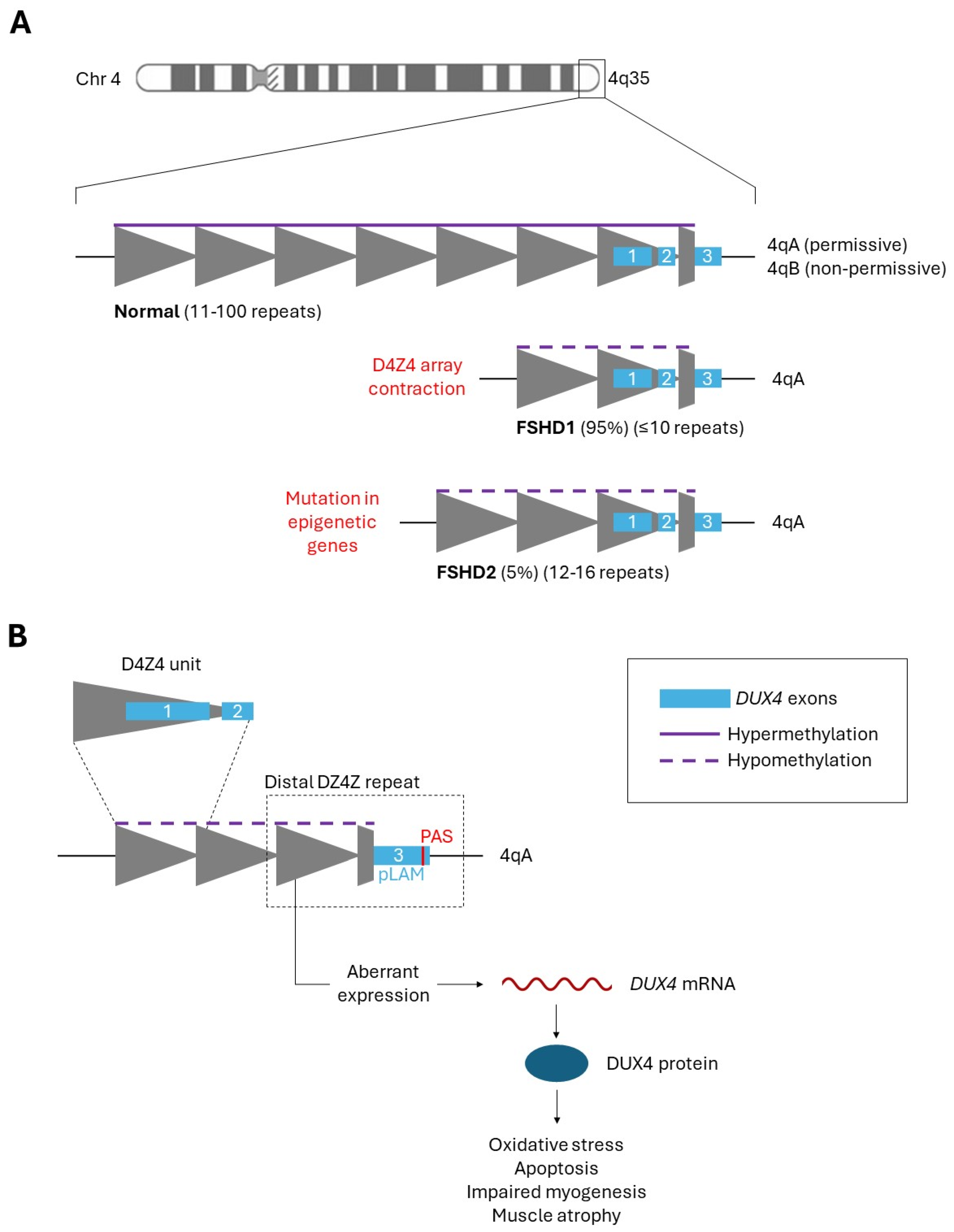
1.1. FSHD Overview
1.2. DUX4 Is the Central Cause of FSHD
2. Oligonucleotide Therapies Targeting DUX4
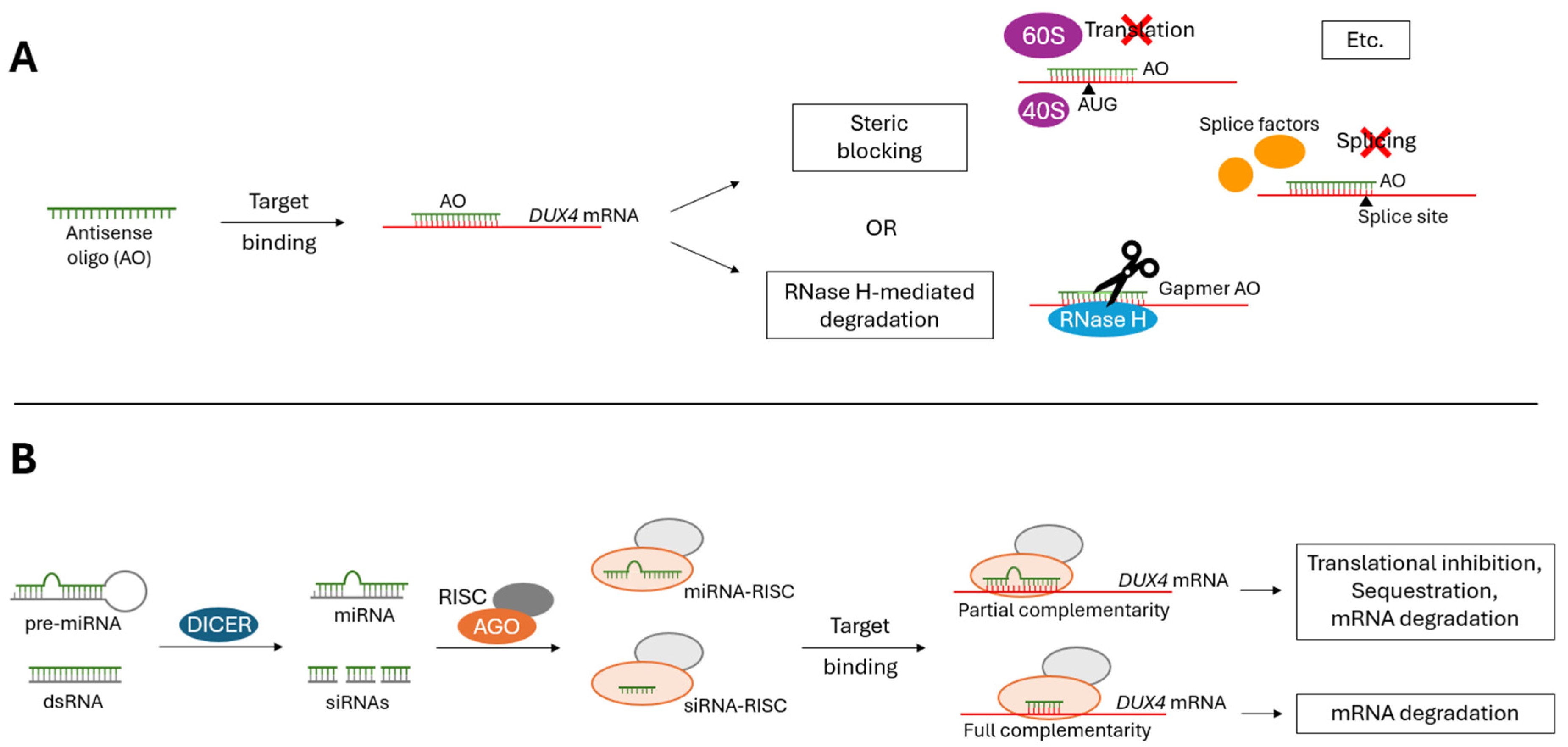
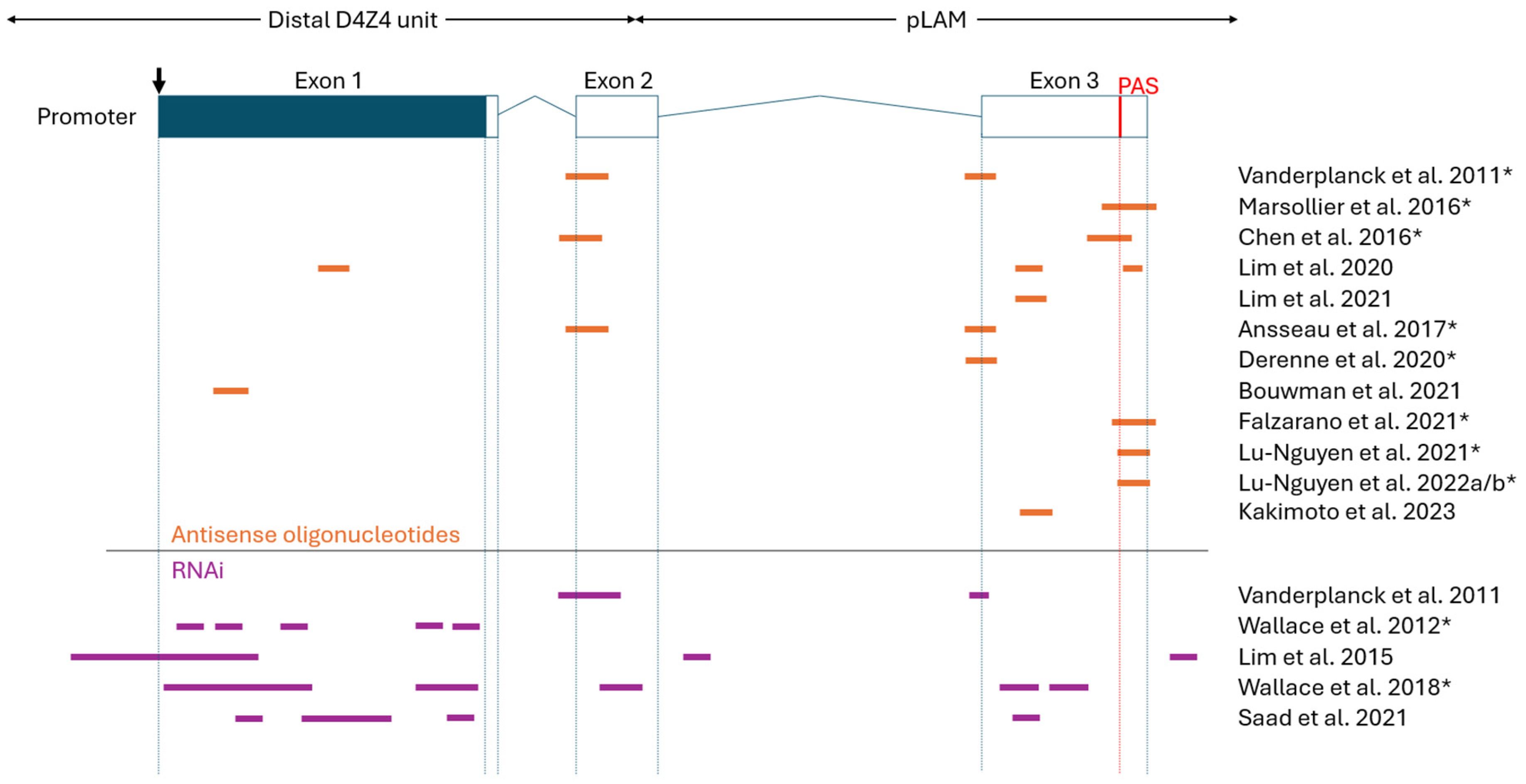
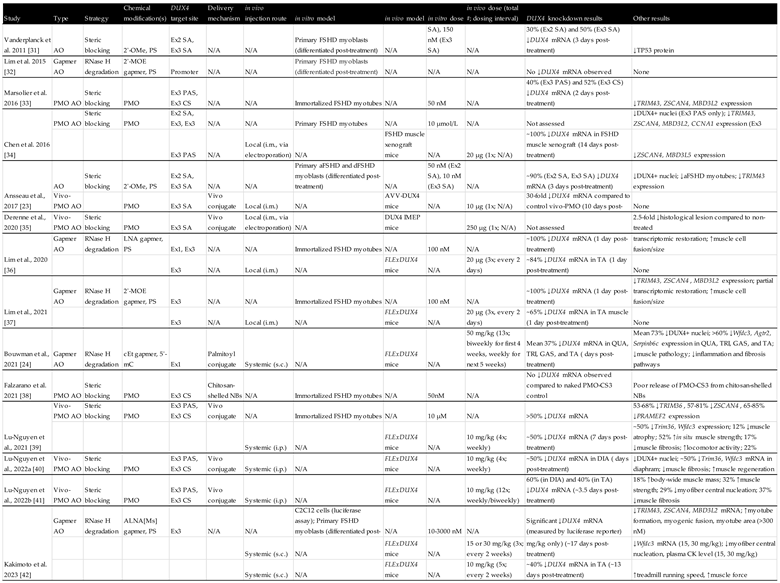
2.1. Antisense Oligonucleotides (AOs)
2.2. RNA Interference (RNAi)
2.3. Other
2.3.1. CRISPR/dCas9 Transcriptional Repression
2.3.2. DNA Aptamers
2.3.3. dsDNA Decoy Trapping
2.3.4. U7-snRNA pre-mRNA Inhibition
3. Further Considerations
3.1. Advantages of Antisense Approaches
3.2. Disadvantages of Antisense Approaches
3.3. Early-Stage Clinical Trials for Select FSHD Therapies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, L.H.; Tawil, R. Facioscapulohumeral Dystrophy. Curr Neurol Neurosci Rep 2016, 16, 66. [Google Scholar] [CrossRef] [PubMed]
- Mostacciuolo, M.; Pastorello, E.; Vazza, G.; Miorin, M.; Angelini, C.; Tomelleri, G.; Galluzzi, G.; Trevisan, C. Facioscapulohumeral Muscular Dystrophy: Epidemiological and Molecular Study in a North-east Italian Population Sample. Clinical Genetics 2009, 75, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Deenen, J.C.W.; Arnts, H.; Van Der Maarel, S.M.; Padberg, G.W.; Verschuuren, J.J.G.M.; Bakker, E.; Weinreich, S.S.; Verbeek, A.L.M.; Van Engelen, B.G.M. Population-Based Incidence and Prevalence of Facioscapulohumeral Dystrophy. Neurology 2014, 83, 1056–1059. [Google Scholar] [CrossRef] [PubMed]
- Mul, K.; Lassche, S.; Voermans, N.C.; Padberg, G.W.; Horlings, C.G.; Van Engelen, B.G. What’s in a Name? The Clinical Features of Facioscapulohumeral Muscular Dystrophy. Pract Neurol 2016, 16, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, A.S.; Astudillo Moncayo, O.M.; Mosquera, J.; Muyolema Arce, V.E.; Gallegos, C.; Ortiz, J.F.; Andrade, A.F.; Oña, S.; Buj, M.J. Treatment of Facioscapulohumeral Muscular Dystrophy (FSHD): A Systematic Review. Cureus 2023. [Google Scholar] [CrossRef] [PubMed]
- Lemmers, R.J.L.F.; Van Der Vliet, P.J.; Klooster, R.; Sacconi, S.; Camaño, P.; Dauwerse, J.G.; Snider, L.; Straasheijm, K.R.; Jan Van Ommen, G.; Padberg, G.W.; et al. A Unifying Genetic Model for Facioscapulohumeral Muscular Dystrophy. Science 2010, 329, 1650–1653. [Google Scholar] [CrossRef] [PubMed]
- Lemmers, R.J.L.F.; Tawil, R.; Petek, L.M.; Balog, J.; Block, G.J.; Santen, G.W.E.; Amell, A.M.; Van Der Vliet, P.J.; Almomani, R.; Straasheijm, K.R.; et al. Digenic Inheritance of an SMCHD1 Mutation and an FSHD-Permissive D4Z4 Allele Causes Facioscapulohumeral Muscular Dystrophy Type 2. Nat Genet 2012, 44, 1370–1374. [Google Scholar] [CrossRef]
- Van den Boogaard, M.L.; Lemmers, R.J.L.F.; Balog, J.; Wohlgemuth, M.; Auranen, M.; Mitsuhashi, S.; van der Vliet, P.J.; Straasheijm, K.R.; van den Akker, R.F.P.; Kriek, M.; et al. Mutations in DNMT3B Modify Epigenetic Repression of the D4Z4 Repeat and the Penetrance of Facioscapulohumeral Dystrophy. The American Journal of Human Genetics 2016, 98, 1020–1029. [Google Scholar] [CrossRef]
- Hamanaka, K.; Šikrová, D.; Mitsuhashi, S.; Masuda, H.; Sekiguchi, Y.; Sugiyama, A.; Shibuya, K.; Lemmers, R.J.L.F.; Goossens, R.; Ogawa, M.; et al. Homozygous Nonsense Variant in LRIF1 Associated with Facioscapulohumeral Muscular Dystrophy. Neurology 2020, 94. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, P.G.; Doráis, J.A.; Grow, E.J.; Whiddon, J.L.; Lim, J.-W.; Wike, C.L.; Weaver, B.D.; Pflueger, C.; Emery, B.R.; Wilcox, A.L.; et al. Conserved Roles for Murine DUX and Human DUX4 in Activating Cleavage Stage Genes and MERVL/HERVL Retrotransposons. 2017.
- De Iaco, A.; Planet, E.; Coluccio, A.; Verp, S.; Duc, J.; Trono, D. DUX-Family Transcription Factors Regulate Zygotic Genome Activation in Placental Mammals. Nat Genet 2017, 49, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Snider, L.; Geng, L.N.; Lemmers, R.J.L.F.; Kyba, M.; Ware, C.B.; Nelson, A.M.; Tawil, R.; Filippova, G.N.; Van Der Maarel, S.M.; Tapscott, S.J.; et al. Facioscapulohumeral Dystrophy: Incomplete Suppression of a Retrotransposed Gene. PLoS Genet 2010, 6, e1001181. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Chadwick, B.P. Influence of Repressive Histone and DNA Methylation upon D4Z4 Transcription in Non-Myogenic Cells. PLoS ONE 2016, 11, e0160022. [Google Scholar] [CrossRef] [PubMed]
- Himeda, C.L.; Jones, P.L. The Genetics and Epigenetics of Facioscapulohumeral Muscular Dystrophy. Annu. Rev. Genom. Hum. Genet. 2019, 20, 265–291. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.R.Q.; Nguyen, Q.; Yokota, T. DUX4 Signalling in the Pathogenesis of Facioscapulohumeral Muscular Dystrophy. IJMS 2020, 21, 729. [Google Scholar] [CrossRef] [PubMed]
- Mocciaro, E.; Runfola, V.; Ghezzi, P.; Pannese, M.; Gabellini, D. DUX4 Role in Normal Physiology and in FSHD Muscular Dystrophy. Cells 2021, 10, 3322. [Google Scholar] [CrossRef] [PubMed]
- Geng, L.N.; Yao, Z.; Snider, L.; Fong, A.P.; Cech, J.N.; Young, J.M.; van der Maarel, S.M.; Ruzzo, W.L.; Gentleman, R.C.; Tawil, R.; et al. DUX4 Activates Germline Genes, Retroelements, and Immune Mediators: Implications for Facioscapulohumeral Dystrophy. Developmental Cell 2012, 22, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Snider, L.; Balog, J.; Lemmers, R.J.L.F.; Van Der Maarel, S.M.; Tawil, R.; Tapscott, S.J. DUX4-Induced Gene Expression Is the Major Molecular Signature in FSHD Skeletal Muscle. Human Molecular Genetics 2014, 23, 5342–5352. [Google Scholar] [CrossRef]
- Stephenson, M.L.; Zamecnik, P.C. Inhibition of Rous Sarcoma Viral RNA Translation by a Specific Oligodeoxyribonucleotide. Proc. Natl. Acad. Sci. U.S.A. 1978, 75, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Dias, N.; Stein, C.A. Antisense Oligonucleotides: Basic Concepts and Mechanisms. Molecular Cancer Therapeutics 2002, 1, 347–355. [Google Scholar]
- Khvorova, A.; Watts, J.K. The Chemical Evolution of Oligonucleotide Therapies of Clinical Utility. Nat Biotechnol 2017, 35, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.J. Antisense Oligonucleotides. In Introduction to Basics of Pharmacology and Toxicology; Raj, G.M., Raveendran, R., Eds.; Springer Singapore: Singapore, 2019; pp. 407–411. ISBN 978-981-329-778-4. [Google Scholar]
- Ansseau, E.; Vanderplanck, C.; Wauters, A.; Harper, S.; Coppée, F.; Belayew, A. Antisense Oligonucleotides Used to Target the DUX4 mRNA as Therapeutic Approaches in FaciosScapuloHumeral Muscular Dystrophy (FSHD). Genes 2017, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Bouwman, L.F.; Den Hamer, B.; Van Den Heuvel, A.; Franken, M.; Jackson, M.; Dwyer, C.A.; Tapscott, S.J.; Rigo, F.; Van Der Maarel, S.M.; De Greef, J.C. Systemic Delivery of a DUX4-Targeting Antisense Oligonucleotide to Treat Facioscapulohumeral Muscular Dystrophy. Molecular Therapy - Nucleic Acids 2021, 26, 813–827. [Google Scholar] [CrossRef] [PubMed]
- Monia, B.P.; Lesnik, E.A.; Gonzalez, C.; Lima, W.F.; McGee, D.; Guinosso, C.J.; Kawasaki, A.M.; Cook, P.D.; Freier, S.M. Evaluation of 2‘-Modified Oligonucleotides Containing 2‘-Deoxy Gaps as Antisense Inhibitors of Gene Expression. Journal of Biological Chemistry 1993, 268, 14514–14522. [Google Scholar] [CrossRef] [PubMed]
- Crooke, S.T.; Baker, B.F.; Crooke, R.M.; Liang, X. Antisense Technology: An Overview and Prospectus. Nat Rev Drug Discov 2021, 20, 427–453. [Google Scholar] [CrossRef] [PubMed]
- Minshull, J.; Hunt, T. The Use of Single-Stranded DNA and RNase H to Promote Quantitative ‘Hybrid Arrest of Translation’ of mRNA/DNA Hybrids in Reticulocyte Lysate Cell-Free Translations. Nucl Acids Res 1986, 14, 6433–6451. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Oda, Y.; Iwai, S.; Inoue, H.; Ohtsuka, E.; Kanaya, S.; Kimura, S.; Katsuda, C.; Katayanagi, K.; Morikawa, K. How Does RNase H Recognize a DNA.RNA Hybrid? Proc. Natl. Acad. Sci. U.S.A. 1991, 88, 11535–11539. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.J.; Norrbom, M.; Chun, S.; Bennett, C.F.; Rigo, F. Nonsense-Mediated Decay as a Terminating Mechanism for Antisense Oligonucleotides. Nucleic Acids Research 2014, 42, 5871–5879. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Nichols, J.G.; Hsu, C.-W.; Vickers, T.A.; Crooke, S.T. mRNA Levels Can Be Reduced by Antisense Oligonucleotides via No-Go Decay Pathway. Nucleic Acids Research 2019, 47, 6900–6916. [Google Scholar] [CrossRef] [PubMed]
- Vanderplanck, C.; Ansseau, E.; Charron, S.; Stricwant, N.; Tassin, A.; Laoudj-Chenivesse, D.; Wilton, S.D.; Coppée, F.; Belayew, A. The FSHD Atrophic Myotube Phenotype Is Caused by DUX4 Expression. PLoS ONE 2011, 6, e26820. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.-W.; Snider, L.; Yao, Z.; Tawil, R.; Van Der Maarel, S.M.; Rigo, F.; Bennett, C.F.; Filippova, G.N.; Tapscott, S.J. DICER/AGO-Dependent Epigenetic Silencing of D4Z4 Repeats Enhanced by Exogenous siRNA Suggests Mechanisms and Therapies for FSHD. Hum. Mol. Genet. 2015, 24, 4817–4828. [Google Scholar] [CrossRef] [PubMed]
- Marsollier, A.-C.; Ciszewski, L.; Mariot, V.; Popplewell, L.; Voit, T.; Dickson, G.; Dumonceaux, J. Antisense Targeting of 3′ End Elements Involved in DUX4 mRNA Processing Is an Efficient Therapeutic Strategy for Facioscapulohumeral Dystrophy: A New Gene-Silencing Approach. Hum. Mol. Genet. 2016, 25, 1468–1478. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.C.; King, O.D.; Zhang, Y.; Clayton, N.P.; Spencer, C.; Wentworth, B.M.; Emerson, C.P.; Wagner, K.R. Morpholino-Mediated Knockdown of DUX4 Toward Facioscapulohumeral Muscular Dystrophy Therapeutics. Molecular Therapy 2016, 24, 1405–1411. [Google Scholar] [CrossRef] [PubMed]
- Derenne, A.; Tassin, A.; Nguyen, T.H.; De Roeck, E.; Jenart, V.; Ansseau, E.; Belayew, A.; Coppée, F.; Declèves, A.-E.; Legrand, A. Induction of a Local Muscular Dystrophy Using Electroporation in Vivo: An Easy Tool for Screening Therapeutics. Sci Rep 2020, 10, 11301. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.R.Q.; Maruyama, R.; Echigoya, Y.; Nguyen, Q.; Zhang, A.; Khawaja, H.; Sen Chandra, S.; Jones, T.; Jones, P.; Chen, Y.-W.; et al. Inhibition of DUX4 Expression with Antisense LNA Gapmers as a Therapy for Facioscapulohumeral Muscular Dystrophy. Proc. Natl. Acad. Sci. U.S.A. 2020, 117, 16509–16515. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.R.Q.; Bittel, A.; Maruyama, R.; Echigoya, Y.; Nguyen, Q.; Huang, Y.; Dzierlega, K.; Zhang, A.; Chen, Y.-W.; Yokota, T. DUX4 Transcript Knockdown with Antisense 2′-O-Methoxyethyl Gapmers for the Treatment of Facioscapulohumeral Muscular Dystrophy. Molecular Therapy 2021, 29, 848–858. [Google Scholar] [CrossRef] [PubMed]
- Falzarano, M.S.; Argenziano, M.; Marsollier, A.C.; Mariot, V.; Rossi, D.; Selvatici, R.; Dumonceaux, J.; Cavalli, R.; Ferlini, A. Chitosan-Shelled Nanobubbles Irreversibly Encapsulate Morpholino Conjugate Antisense Oligonucleotides and Are Ineffective for Phosphorodiamidate Morpholino-Mediated Gene Silencing of DUX4. Nucleic Acid Therapeutics 2021, 31, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Lu-Nguyen, N.; Malerba, A.; Herath, S.; Dickson, G.; Popplewell, L. Systemic Antisense Therapeutics Inhibiting DUX4 Expression Ameliorates FSHD-like Pathology in an FSHD Mouse Model. Human Molecular Genetics 2021, 30, 1398–1412. [Google Scholar] [CrossRef] [PubMed]
- Lu-Nguyen, N.; Malerba, A.; Antoni Pineda, M.; Dickson, G.; Popplewell, L. Improving Molecular and Histopathology in Diaphragm Muscle of the Double Transgenic ACTA1-MCM/FLExDUX4 Mouse Model of FSHD with Systemic Antisense Therapy. Human Gene Therapy 2022, 33, 923–935. [Google Scholar] [CrossRef] [PubMed]
- Lu-Nguyen, N.; Dickson, G.; Malerba, A.; Popplewell, L. Long-Term Systemic Treatment of a Mouse Model Displaying Chronic FSHD-like Pathology with Antisense Therapeutics That Inhibit DUX4 Expression. Biomedicines 2022, 10, 1623. [Google Scholar] [CrossRef] [PubMed]
- Kakimoto, T.; Ogasawara, A.; Ishikawa, K.; Kurita, T.; Yoshida, K.; Harada, S.; Nonaka, T.; Inoue, Y.; Uchida, K.; Tateoka, T.; et al. A Systemically Administered Unconjugated Antisense Oligonucleotide Targeting DUX4 Improves Muscular Injury and Motor Function in FSHD Model Mice. Biomedicines 2023, 11, 2339. [Google Scholar] [CrossRef] [PubMed]
- Stadler, G.; Chen, J.C.; Wagner, K.; Robin, J.D.; Shay, J.W.; Emerson, C.P.; Wright, W.E. Establishment of Clonal Myogenic Cell Lines from Severely Affected Dystrophic Muscles - CDK4 Maintains the Myogenic Population. Skeletal Muscle 2011, 1, 12. [Google Scholar] [CrossRef] [PubMed]
- Krom, Y.D.; Dumonceaux, J.; Mamchaoui, K.; Den Hamer, B.; Mariot, V.; Negroni, E.; Geng, L.N.; Martin, N.; Tawil, R.; Tapscott, S.J.; et al. Generation of Isogenic D4Z4 Contracted and Noncontracted Immortal Muscle Cell Clones from a Mosaic Patient. The American Journal of Pathology 2012, 181, 1387–1401. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.; Jones, P.L. A Cre-Inducible DUX4 Transgenic Mouse Model for Investigating Facioscapulohumeral Muscular Dystrophy. PLoS ONE 2018, 13, e0192657. [Google Scholar] [CrossRef] [PubMed]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and Specific Genetic Interference by Double-Stranded RNA In. Nature 1998, 391. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.C.; Doudna, J.A. Molecular Mechanisms of RNA Interference. Annu. Rev. Biophys. 2013, 42, 217–239. [Google Scholar] [CrossRef] [PubMed]
- Wallace, L.M.; Liu, J.; Domire, J.S.; Garwick-Coppens, S.E.; Guckes, S.M.; Mendell, J.R.; Flanigan, K.M.; Harper, S.Q. RNA Interference Inhibits DUX4-Induced Muscle Toxicity In Vivo: Implications for a Targeted FSHD Therapy. Molecular Therapy 2012, 20, 1417–1423. [Google Scholar] [CrossRef] [PubMed]
- Wallace, L.M.; Saad, N.Y.; Pyne, N.K.; Fowler, A.M.; Eidahl, J.O.; Domire, J.S.; Griffin, D.A.; Herman, A.C.; Sahenk, Z.; Rodino-Klapac, L.R.; et al. Pre-Clinical Safety and Off-Target Studies to Support Translation of AAV-Mediated RNAi Therapy for FSHD. Molecular Therapy - Methods & Clinical Development 2018, 8, 121–130. [Google Scholar] [CrossRef]
- Saad, N.Y.; Al-Kharsan, M.; Garwick-Coppens, S.E.; Chermahini, G.A.; Harper, M.A.; Palo, A.; Boudreau, R.L.; Harper, S.Q. Human miRNA miR-675 Inhibits DUX4 Expression and May Be Exploited as a Potential Treatment for Facioscapulohumeral Muscular Dystrophy. Nat Commun 2021, 12, 7128. [Google Scholar] [CrossRef] [PubMed]
- Arrowhead Pharmaceuticals, Inc. Arrowhead Presents Preclinical Data on ARO-DUX4 at FSHD Society International Research Congress. 2021. Available online: https://ir.arrowheadpharma.com/news-releases/news-release-details/arrowhead-presents-preclinical-data-aro-dux4-fshd-society (accessed on 29 June 2024).
- Jagannathan, S.; De Greef, J.C.; Hayward, L.J.; Yokomori, K.; Gabellini, D.; Mul, K.; Sacconi, S.; Arjomand, J.; Kinoshita, J.; Harper, S.Q. Meeting Report: The 2021 FSHD International Research Congress. Skeletal Muscle 2022, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Mariot, V.; Sidlauskaite, E.; Gall, L.L.; Corbex, E.; Dumonceaux, J. VP. 61 An AAV-shRNA DUX4-Based Therapy to Treat Facioscapulohumeral Muscular Dystrophy (FSHD). Neuromuscular Disorders 2022, 32, S107. [Google Scholar] [CrossRef]
- Malecova, B.; Sala, D.; Melikian, G.M.; Erdogan, G.; Johns, R.; Young, J.; Ventre, E.; Gatto, S.; Onorato, M.; Pavlicek, A.; et al. DUX4 siRNA Optimization for the Development of an Antibody-Oligonucleotide Conjugate (AOC) for the Treatment of FSHD (P17-13.009). Neurology 2022, 98, 1776. [Google Scholar] [CrossRef]
- Avidity Biosciences, Inc. Targeting DUX4 for Silencing with AOC for the Treatment of FSHD. 2024. Available online: https://www.aviditybiosciences.com/wp-content/uploads/2024/03/MDA-2024-FSHD-RNASeq_v6.0_STC.pdf (accessed on 29 June 2024).
- Dyne Therapeutics, Inc. Dyne Therapeutics Presents New Preclinical Data for its Facioscapulohumeral Muscular Dystrophy Program During the FSHD Society International Research Congress. 2024. Available online: https://investors.dyne-tx.com/news-releases/news-release-details/dyne-therapeutics-presents-new-preclinical-data-its (accessed on 14 July 2024).
- Dyne Therapeutics, Inc. The FORCETM platform achieves robust and durable DUX4 suppression and functional benefit in FSHD mouse models. 2024. Available online: https://www.dyne-tx.com/wp-content/uploads/Natoli_FSHDIRC_June2024.pdf (accessed on 14 July 2024).
- Wallace, L.M.; Garwick, S.E.; Mei, W.; Belayew, A.; Coppee, F.; Ladner, K.J.; Guttridge, D.; Yang, J.; Harper, S.Q. DUX4, a Candidate Gene for Facioscapulohumeral Muscular Dystrophy, Causes P53-dependent Myopathy in Vivo. Annals of Neurology 2011, 69, 540–552. [Google Scholar] [CrossRef] [PubMed]
- Larson, M.H.; Gilbert, L.A.; Wang, X.; Lim, W.A.; Weissman, J.S.; Qi, L.S. CRISPR Interference (CRISPRi) for Sequence-Specific Control of Gene Expression. Nat Protoc 2013, 8, 2180–2196. [Google Scholar] [CrossRef] [PubMed]
- Himeda, C.L.; Jones, T.I.; Jones, P.L. Targeted Epigenetic Repression by CRISPR/dSaCas9 Suppresses Pathogenic DUX4-Fl Expression in FSHD. Molecular Therapy - Methods & Clinical Development 2021, 20, 298–311. [Google Scholar] [CrossRef]
- Himeda, C.L.; Jones, T.I.; Jones, P.L. CRISPR/dCas9-Mediated Transcriptional Inhibition Ameliorates the Epigenetic Dysregulation at D4Z4 and Represses DUX4-Fl in FSH Muscular Dystrophy. Molecular Therapy 2016, 24, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Himeda, C.L.; Jones, T.I.; Virbasius, C.-M.; Zhu, L.J.; Green, M.R.; Jones, P.L. Identification of Epigenetic Regulators of DUX4-Fl for Targeted Therapy of Facioscapulohumeral Muscular Dystrophy. Molecular Therapy 2018, 26, 1797–1807. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Chadwick, B.P. CRISPR Mediated Targeting of DUX4 Distal Regulatory Element Represses DUX4 Target Genes Dysregulated in Facioscapulohumeral Muscular Dystrophy. Sci Rep 2021, 11, 12598. [Google Scholar] [CrossRef] [PubMed]
- Sasaki-Honda, M.; Jonouchi, T.; Arai, M.; He, J.; Okita, K.; Sakurai, S.; Yamamoto, T.; Sakurai, H. Hit-and-Run Silencing of Endogenous DUX4 by Targeting DNA Hypomethylation on D4Z4 Repeats in Facioscapulohumeral Muscular Dystrophy. bioRxiv, 2022; (preprint). [Google Scholar]
- Rashnonejad, A.; Amini-Chermahini, G.; Taylor, N.; Fowler, A.; Kraus, E.; King, O.; Harper, S. FP. 29 AAV-CRISPR-Cas13 Gene Therapy for FSHD: DUX4 Gene Silencing Efficacy and Immune Responses to Cas13b Protein. Neuromuscular Disorders 2022, 32, S103–S104. [Google Scholar] [CrossRef]
- Saljoughian, N.; Rizzotto, L.; Sezgin, Y.; Faraji, H.; Wallace, L.; Kararoudi, M.N.; Palmieri, D.; Harper, S.P. 144 Developing Cas13-ADAR-Mediated DUX4 mRNA Editing as a Prospective Therapy for FSHD. Neuromuscular Disorders 2022, 32, S106. [Google Scholar] [CrossRef]
- Di Mauro, V.; Lauta, F.C.; Modica, J.; Appleton, S.L.; De Franciscis, V.; Catalucci, D. Diagnostic and Therapeutic Aptamers. JACC: Basic to Translational Science 2024, 9, 260–277. [Google Scholar] [CrossRef] [PubMed]
- Klingler, C.; Ashley, J.; Shi, K.; Stiefvater, A.; Kyba, M.; Sinnreich, M.; Aihara, H.; Kinter, J. DNA Aptamers against the DUX4 Protein Reveal Novel Therapeutic Implications for FSHD. FASEB j. 2020, 34, 4573–4590. [Google Scholar] [CrossRef] [PubMed]
- Mariot, V.; Joubert, R.; Marsollier, A.-C.; Hourdé, C.; Voit, T.; Dumonceaux, J. A Deoxyribonucleic Acid Decoy Trapping DUX4 for the Treatment of Facioscapulohumeral Muscular Dystrophy. Molecular Therapy - Nucleic Acids 2020, 22, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Rashnonejad, A.; Amini-Chermahini, G.; Taylor, N.K.; Wein, N.; Harper, S.Q. Designed U7 snRNAs Inhibit DUX4 Expression and Improve FSHD-Associated Outcomes in DUX4 Overexpressing Cells and FSHD Patient Myotubes. Molecular Therapy - Nucleic Acids 2021, 23, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.C.; Langer, R.; Wood, M.J.A. Advances in Oligonucleotide Drug Delivery. Nat Rev Drug Discov 2020, 19, 673–694. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, M.; Ashizawa, A.T. The Challenges and Strategies of Antisense Oligonucleotide Drug Delivery. Biomedicines 2021, 9, 433. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Zhang, M.; Huang, Y. Three ‘E’ Challenges for siRNA Drug Development. Trends in Molecular Medicine 2024, 30, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Li, Y.; Yu, W.; Fu, Y.; Zhang, J.; Cao, H. Recent Progress of Small Interfering RNA Delivery on the Market and Clinical Stage. Mol. Pharmaceutics 2024, 21, 2081–2096. [Google Scholar] [CrossRef] [PubMed]
- Study of ARO-DUX4 in Adult Patients With Facioscapulohumeral Muscular Dystrophy Type 1. Available online: https://clinicaltrials.gov/study/NCT06131983 (accessed on 6 July 2024).
- Halseth, A.; Ackermann, E.; Brandt, T.; Chen, C.; Cho, H.; Stahl, M.; DiTrapani, K.; Hughes, S.; Tawil, R.; Statland, J. P51 Phase 1/2 Study to Evaluate AOC 1020 for Adult Patients with Facioscapulohumeral Muscular Dystrophy: FORTITUDE Trial Design. Neuromuscular Disorders 2023, 33, S71. [Google Scholar] [CrossRef]
- Phase 1/2 Study of AOC 1020 in Adults With Facioscapulohumeral Muscular Dystrophy (FSHD) (FORTITUDE). Available online: https://clinicaltrials.gov/study/NCT05747924 (accessed on 6 July 2024).


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




