Submitted:
18 July 2024
Posted:
18 July 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Bruton Tyrosine Kinase Protein
History of BTK Discovery
Molecular Family, Biochemistry and Activation of BTK
BTK in the B-Cell Receptor Signaling Pathway
The Role of BTK in B-Cell Malignancies
Covalent BTK Inhibitors in Clinical Practice
Resistance Mechanisms to Covalent BTK Inhibitors
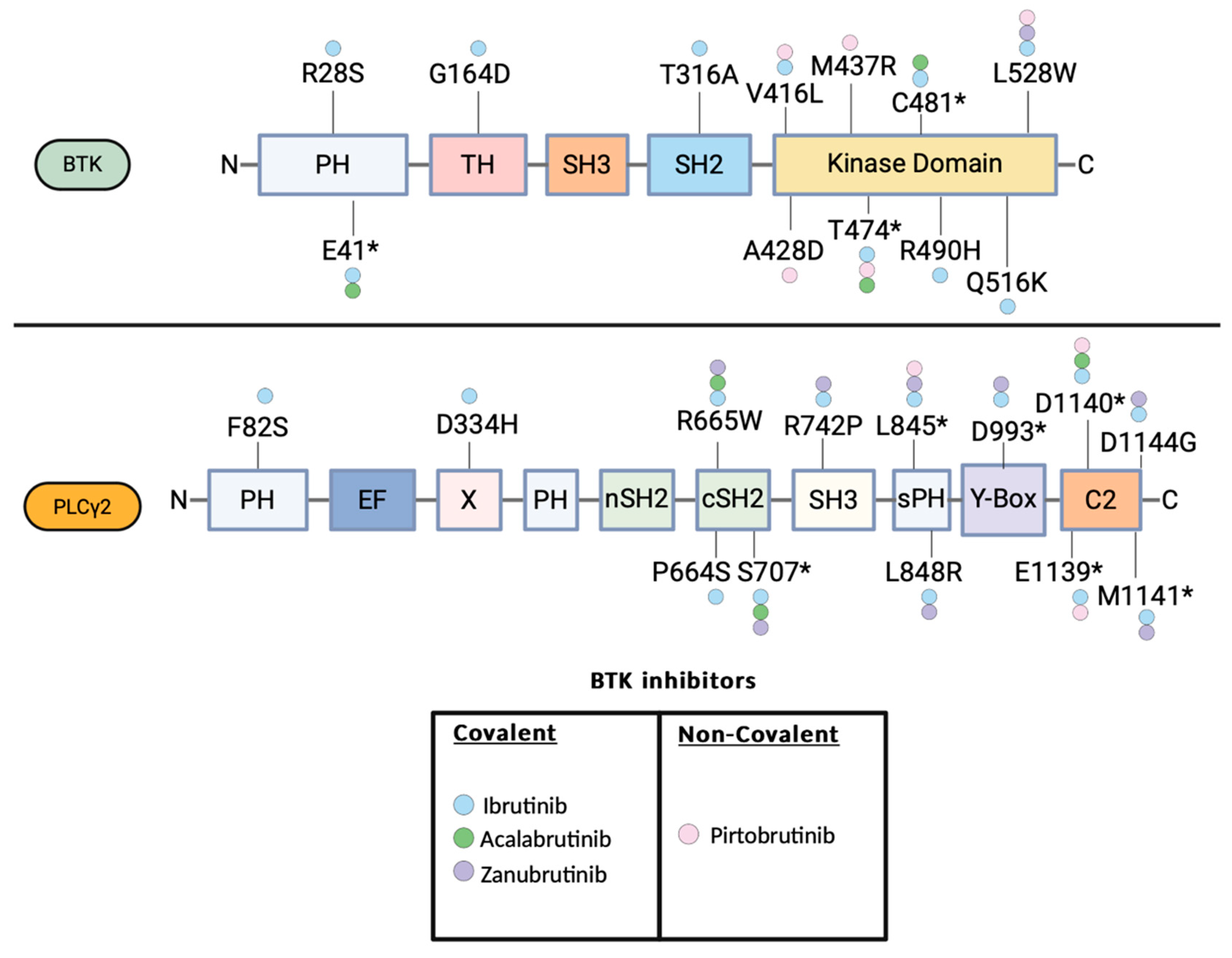
Noncovalent BTK Inhibitors: Clinical Data and Emergence of Resistance
BTK Degraders: The Future of BTK Targeted Therapies
Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Teras, L.R.; DeSantis, C.E.; Cerhan, J.R.; Morton, L.M.; Jemal, A.; Flowers, C.R. 2016 US Lymphoid Malignancy Statistics by World Health Organization Subtypes. CA Cancer J Clin 2016, 66, 443–459. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.; Xanthopoulos, C.; Kostareli, E. The Role of Bruton’s Tyrosine Kinase in the Immune System and Disease. Immunology 2021, 164, 722–736. [Google Scholar] [CrossRef]
- Pan, Z.; Scheerens, H.; Li, S.-J.; Schultz, B.E.; Sprengeler, P.A.; Burrill, L.C.; Mendonca, R.V.; Sweeney, M.D.; Scott, K.C.K.; Grothaus, P.G.; et al. Discovery of Selective Irreversible Inhibitors for Bruton’s Tyrosine Kinase. ChemMedChem 2007, 2, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Quinquenel, A.; Fornecker, L.-M.; Letestu, R.; Ysebaert, L.; Fleury, C.; Lazarian, G.; Dilhuydy, M.-S.; Nollet, D.; Guieze, R.; Feugier, P.; et al. Prevalence of BTK and PLCG2 Mutations in a Real-Life CLL Cohort Still on Ibrutinib after 3 Years: A FILO Group Study. Blood 2019, 134, 641–644. [Google Scholar] [CrossRef]
- Wang, M.; Shah, N.N.; Alencar, A.J.; Gerson, J.N.; Patel, M.R.; Fakhri, B.; Jurczak, W.; Tan, X.N.; Lewis, K.; Fenske, T.S.; et al. Pirtobrutinib, A Next Generation, Highly Selective, Non-Covalent BTK Inhibitor in Previously Treated Mantle Cell Lymphoma: Updated Results from the Phase 1/2 BRUIN Study. Blood 2021, 138, 381. [Google Scholar] [CrossRef]
- Mato, A.R.; Shah, N.N.; Jurczak, W.; Cheah, C.Y.; Pagel, J.M.; Woyach, J.A.; Fakhri, B.; Eyre, T.A.; Lamanna, N.; Patel, M.R.; et al. Pirtobrutinib in Relapsed or Refractory B-Cell Malignancies (BRUIN): A Phase 1/2 Study. The Lancet 2021, 397, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Montoya, S.; Bourcier, J.; Noviski, M.; Lu, H.; Thompson, M.C.; Chirino, A.; Jahn, J.; Sondhi, A.K.; Gajewski, S.; Tan, Y.S.M.; et al. Kinase-Impaired BTK Mutations Are Susceptible to Clinical-Stage BTK and IKZF1/3 Degrader NX-2127. Science 2024, 383, eadi5798. [Google Scholar] [CrossRef] [PubMed]
- Bruton, O.C. Agammaglobulinemia. Pediatrics 1952, 9, 722–728. [Google Scholar] [CrossRef]
- Vetrie, D.; Vorechovský, I.; Sideras, P.; Holland, J.; Davies, A.; Flinter, F.; Hammarström, L.; Kinnon, C.; Levinsky, R.; Bobrow, M. The Gene Involved in X-Linked Agammaglobulinaemia Is a Member of the Src Family of Protein-Tyrosine Kinases. Nature 1993, 361, 226–233. [Google Scholar] [CrossRef]
- Tsukada, S.; Saffran, D.C.; Rawlings, D.J.; Parolini, O.; Allen, R.C.; Klisak, I.; Sparkes, R.S.; Kubagawa, H.; Mohandas, T.; Quan, S. Deficient Expression of a B Cell Cytoplasmic Tyrosine Kinase in Human X-Linked Agammaglobulinemia. Cell 1993, 72, 279–290. [Google Scholar] [CrossRef]
- Khan, W.N.; Alt, F.W.; Gerstein, R.M.; Malynn, B.A.; Larsson, I.; Rathbun, G.; Davidson, L.; Müller, S.; Kantor, A.B.; Herzenberg, L.A. Defective B Cell Development and Function in Btk-Deficient Mice. Immunity 1995, 3, 283–299. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.J.; Yu, L.; Bäckesjö, C.-M.; Vargas, L.; Faryal, R.; Aints, A.; Christensson, B.; Berglöf, A.; Vihinen, M.; Nore, B.F.; et al. Bruton’s Tyrosine Kinase (Btk): Function, Regulation, and Transformation with Special Emphasis on the PH Domain. Immunol Rev 2009, 228, 58–73. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, D.J.; Scharenberg, A.M.; Park, H.; Wahl, M.I.; Lin, S.; Kato, R.M.; Fluckiger, A.C.; Witte, O.N.; Kinet, J.P. Activation of BTK by a Phosphorylation Mechanism Initiated by SRC Family Kinases. Science 1996, 271, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Wahl, M.I.; Afar, D.E.; Turck, C.W.; Rawlings, D.J.; Tam, C.; Scharenberg, A.M.; Kinet, J.P.; Witte, O.N. Regulation of Btk Function by a Major Autophosphorylation Site within the SH3 Domain. Immunity 1996, 4, 515–525. [Google Scholar] [CrossRef]
- Estupiñán, H.Y.; Bouderlique, T.; He, C.; Berglöf, A.; Cappelleri, A.; Frengen, N.; Zain, R.; Karlsson, M.C.I.; Månsson, R.; Smith, C.I.E. In BTK, Phosphorylated Y223 in the SH3 Domain Mirrors Catalytic Activity, but Does Not Influence Biological Function. Blood Adv 2024, 8, 1981–1990. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.P.; Kühn, R.; Rajewsky, K. In Vivo Ablation of Surface Immunoglobulin on Mature B Cells by Inducible Gene Targeting Results in Rapid Cell Death. Cell 1997, 90, 1073–1083. [Google Scholar] [CrossRef] [PubMed]
- Rolli, V.; Gallwitz, M.; Wossning, T.; Flemming, A.; Schamel, W.W.A.; Zürn, C.; Reth, M. Amplification of B Cell Antigen Receptor Signaling by a Syk/ITAM Positive Feedback Loop. Mol Cell 2002, 10, 1057–1069. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Sekiya, F.; Poulin, B.; Bae, Y.S.; Rhee, S.G. Mechanism of B-Cell Receptor-Induced Phosphorylation and Activation of Phospholipase C-Gamma2. Mol Cell Biol 2004, 24, 9986–9999. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, U.D.; Zhang, K.; Teutsch, M.; Sen, R.; Wortis, H.H. Bruton’s Tyrosine Kinase Links the B Cell Receptor to Nuclear Factor kappaB Activation. J Exp Med 2000, 191, 1735–1744. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.E.; Ngo, V.N.; Lenz, G.; Tolar, P.; Young, R.M.; Romesser, P.B.; Kohlhammer, H.; Lamy, L.; Zhao, H.; Yang, Y.; et al. Chronic Active B-Cell-Receptor Signalling in Diffuse Large B-Cell Lymphoma. Nature 2010, 463, 88–92. [Google Scholar] [CrossRef]
- Honigberg, L.A.; Smith, A.M.; Sirisawad, M.; Verner, E.; Loury, D.; Chang, B.; Li, S.; Pan, Z.; Thamm, D.H.; Miller, R.A.; et al. The Bruton Tyrosine Kinase Inhibitor PCI-32765 Blocks B-Cell Activation and Is Efficacious in Models of Autoimmune Disease and B-Cell Malignancy. Proc Natl Acad Sci U S A 2010, 107, 13075–13080. [Google Scholar] [CrossRef] [PubMed]
- Herman, S.E.M.; Gordon, A.L.; Hertlein, E.; Ramanunni, A.; Zhang, X.; Jaglowski, S.; Flynn, J.; Jones, J.; Blum, K.A.; Buggy, J.J.; et al. Bruton Tyrosine Kinase Represents a Promising Therapeutic Target for Treatment of Chronic Lymphocytic Leukemia and Is Effectively Targeted by PCI-32765. Blood 2011, 117, 6287–6296. [Google Scholar] [CrossRef] [PubMed]
- Advani, R.H.; Buggy, J.J.; Sharman, J.P.; Smith, S.M.; Boyd, T.E.; Grant, B.; Kolibaba, K.S.; Furman, R.R.; Rodriguez, S.; Chang, B.Y.; et al. Bruton Tyrosine Kinase Inhibitor Ibrutinib (PCI-32765) Has Significant Activity in Patients with Relapsed/Refractory B-Cell Malignancies. J Clin Oncol 2013, 31, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.L.; Rule, S.; Martin, P.; Goy, A.; Auer, R.; Kahl, B.S.; Jurczak, W.; Advani, R.H.; Romaguera, J.E.; Williams, M.E.; et al. Targeting BTK with Ibrutinib in Relapsed or Refractory Mantle-Cell Lymphoma. N Engl J Med 2013, 369, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.L.; Blum, K.A.; Martin, P.; Goy, A.; Auer, R.; Kahl, B.S.; Jurczak, W.; Advani, R.H.; Romaguera, J.E.; Williams, M.E.; et al. Long-Term Follow-up of MCL Patients Treated with Single-Agent Ibrutinib: Updated Safety and Efficacy Results. Blood 2015, 126, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Byrd John, C.; Brown Jennifer, R.; O’Brien Susan; Barrientos Jacqueline C. ; Kay Neil E.; Reddy Nishitha M.; Coutre Steven; Tam Constantine S.; Mulligan Stephen P.; Jaeger Ulrich; et al. Ibrutinib versus Ofatumumab in Previously Treated Chronic Lymphoid Leukemia. New England Journal of Medicine 2014, 371, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Munir, T.; Brown, J.R.; O’Brien, S.; Barrientos, J.C.; Barr, P.M.; Reddy, N.M.; Coutre, S.; Tam, C.S.; Mulligan, S.P.; Jaeger, U.; et al. Final Analysis from RESONATE: Up to Six Years of Follow-up on Ibrutinib in Patients with Previously Treated Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma. Am J Hematol 2019, 94, 1353–1363. [Google Scholar] [CrossRef]
- Treon, S.P.; Tripsas, C.K.; Meid, K.; Warren, D.; Varma, G.; Green, R.; Argyropoulos, K.V.; Yang, G.; Cao, Y.; Xu, L.; et al. Ibrutinib in Previously Treated Waldenström’s Macroglobulinemia. N Engl J Med 2015, 372, 1430–1440. [Google Scholar] [CrossRef]
- IMBRUVICA® (Ibrutinib) Official Patient Website. Available online: https://www.imbruvica.com/ (accessed on 8 July 2024).
- Wang, M.; Rule, S.; Zinzani, P.L.; Goy, A.; Casasnovas, O.; Smith, S.D.; Damaj, G.; Doorduijn, J.; Lamy, T.; Morschhauser, F.; et al. Acalabrutinib in Relapsed or Refractory Mantle Cell Lymphoma (ACE-LY-004): A Single-Arm, Multicentre, Phase 2 Trial. Lancet 2018, 391, 659–667. [Google Scholar] [CrossRef]
- Wang, M.; Rule, S.; Zinzani, P.L.; Goy, A.H.; Casasnovas, R.-O.; Smith, S.D.; Damaj, G.L.; Doorduijn, J.K.; Lamy, T.; Morschhauser, F.; et al. Acalabrutinib Monotherapy in Patients with Relapsed/Refractory Mantle Cell Lymphoma: Long-Term Efficacy and Safety Results from a Phase 2 Study. Blood 2020, 136, 38–39. [Google Scholar] [CrossRef]
- Sharman, J.P.; Egyed, M.; Jurczak, W.; Skarbnik, A.; Pagel, J.M.; Flinn, I.W.; Kamdar, M.; Munir, T.; Walewska, R.; Corbett, G.; et al. Acalabrutinib with or without Obinutuzumab versus Chlorambucil and Obinutuzmab for Treatment-Naive Chronic Lymphocytic Leukaemia (ELEVATE TN): A Randomised, Controlled, Phase 3 Trial. Lancet 2020, 395, 1278–1291. [Google Scholar] [CrossRef]
- Ghia, P.; Pluta, A.; Wach, M.; Lysak, D.; Kozak, T.; Simkovic, M.; Kaplan, P.; Kraychok, I.; Illes, A.; de la Serna, J.; et al. ASCEND: Phase III, Randomized Trial of Acalabrutinib Versus Idelalisib Plus Rituximab or Bendamustine Plus Rituximab in Relapsed or Refractory Chronic Lymphocytic Leukemia. J Clin Oncol 2020, 38, 2849–2861. [Google Scholar] [CrossRef]
- Song, Y.; Zhou, K.; Zou, D.; Zhou, J.; Hu, J.; Yang, H.; Zhang, H.; Ji, J.; Xu, W.; Jin, J.; et al. Zanubrutinib in Relapsed/Refractory Mantle Cell Lymphoma: Long-Term Efficacy and Safety Results from a Phase 2 Study. Blood 2022, 139, 3148–3158. [Google Scholar] [CrossRef]
- Phillips, T.; Chan, H.; Tam, C.S.; Tedeschi, A.; Johnston, P.; Oh, S.Y.; Opat, S.; Eom, H.-S.; Allewelt, H.; Stern, J.C.; et al. Zanubrutinib Monotherapy in Relapsed/Refractory Indolent Non-Hodgkin Lymphoma. Blood Adv 2022, 6, 3472–3479. [Google Scholar] [CrossRef]
- Research, C. for D.E. and FDA Approves Zanubrutinib for Waldenström’s Macroglobulinemia. FDA 2021.
- Dimopoulos, M.A.; Opat, S.; D’Sa, S.; Jurczak, W.; Lee, H.-P.; Cull, G.; Owen, R.G.; Marlton, P.; Wahlin, B.E.; Garcia-Sanz, R.; et al. Zanubrutinib Versus Ibrutinib in Symptomatic Waldenström Macroglobulinemia: Final Analysis From the Randomized Phase III ASPEN Study. J Clin Oncol 2023, 41, 5099–5106. [Google Scholar] [CrossRef]
- Research, C. for D.E. and FDA Grants Accelerated Approval to Zanubrutinib for Marginal Zone Lymphoma. FDA 2022. [Google Scholar]
- Opat, S.; Tedeschi, A.; Linton, K.; McKay, P.; Hu, B.; Chan, H.; Jin, J.; Sobieraj-Teague, M.; Zinzani, P.L.; Coleman, M.; et al. The MAGNOLIA Trial: Zanubrutinib, a Next-Generation Bruton Tyrosine Kinase Inhibitor, Demonstrates Safety and Efficacy in Relapsed/Refractory Marginal Zone Lymphoma. Clin Cancer Res 2021, 27, 6323–6332. [Google Scholar] [CrossRef] [PubMed]
- Tam, C.S.; Brown, J.R.; Kahl, B.S.; Ghia, P.; Giannopoulos, K.; Jurczak, W.; Šimkovič, M.; Shadman, M.; Österborg, A.; Laurenti, L.; et al. Zanubrutinib versus Bendamustine and Rituximab in Untreated Chronic Lymphocytic Leukaemia and Small Lymphocytic Lymphoma (SEQUOIA): A Randomised, Controlled, Phase 3 Trial. Lancet Oncol 2022, 23, 1031–1043. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; Eichhorst, B.; Hillmen, P.; Jurczak, W.; Kaźmierczak, M.; Lamanna, N.; O’Brien, S.M.; Tam, C.S.; Qiu, L.; Zhou, K.; et al. Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. N Engl J Med 2023, 388, 319–332. [Google Scholar] [CrossRef]
- Zinzani, P.L.; Mayer, J.; Flowers, C.R.; Bijou, F.; De Oliveira, A.C.; Song, Y.; Zhang, Q.; Merli, M.; Bouabdallah, K.; Ganly, P.; et al. ROSEWOOD: A Phase II Randomized Study of Zanubrutinib Plus Obinutuzumab Versus Obinutuzumab Monotherapy in Patients With Relapsed or Refractory Follicular Lymphoma. J Clin Oncol 2023, 41, 5107–5117. [Google Scholar] [CrossRef]
- Burger, J.A.; Tedeschi, A.; Barr, P.M.; Robak, T.; Owen, C.; Ghia, P.; Bairey, O.; Hillmen, P.; Bartlett, N.L.; Li, J.; et al. Ibrutinib as Initial Therapy for Patients with Chronic Lymphocytic Leukemia. N Engl J Med 2015, 373, 2425–2437. [Google Scholar] [CrossRef]
- M, A.; Sr, J.; G, P. Richter’s Syndrome (RS): Emerging Therapeutic Horizons. Journal of Oncology Research and Therapy 2024. [Google Scholar]
- Woyach, J.A. Patterns of Resistance to B Cell-Receptor Pathway Antagonists in Chronic Lymphocytic Leukemia and Strategies for Management. Hematology Am Soc Hematol Educ Program 2015, 2015, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Ahn, I.E.; Underbayev, C.; Albitar, A.; Herman, S.E.M.; Tian, X.; Maric, I.; Arthur, D.C.; Wake, L.; Pittaluga, S.; Yuan, C.M.; et al. Clonal Evolution Leading to Ibrutinib Resistance in Chronic Lymphocytic Leukemia. Blood 2017, 129, 1469–1479. [Google Scholar] [CrossRef]
- Tsushima, T.; Sato, N.; Guo, Y.-M.; Nakamura, H.; Kunisada, K.; Chi, S.-G.; Akie, K.; Takahashi, Y.; Nakamura, S.; Shimada, K.; et al. Richter Transformation Acquiring PLCG2 Mutation during Bruton Tyrosine Kinase Inhibitors Treatment. EJHaem 2024, 5, 642–645. [Google Scholar] [CrossRef] [PubMed]
- Blombery, P.; Thompson, E.R.; Lew, T.E.; Tiong, I.S.; Bennett, R.; Cheah, C.Y.; Lewis, K.L.; Handunnetti, S.M.; Tang, C.P.S.; Roberts, A.; et al. Enrichment of BTK Leu528Trp Mutations in Patients with CLL on Zanubrutinib: Potential for Pirtobrutinib Cross-Resistance. Blood Adv 2022, 6, 5589–5592. [Google Scholar] [CrossRef]
- Jones, D.; Woyach, J.A.; Zhao, W.; Caruthers, S.; Tu, H.; Coleman, J.; Byrd, J.C.; Johnson, A.J.; Lozanski, G. PLCG2 C2 Domain Mutations Co-Occur with BTK and PLCG2 Resistance Mutations in Chronic Lymphocytic Leukemia Undergoing Ibrutinib Treatment. Leukemia 2017, 31, 1645–1647. [Google Scholar] [CrossRef]
- Sedlarikova, L.; Petrackova, A.; Papajik, T.; Turcsanyi, P.; Kriegova, E. Resistance-Associated Mutations in Chronic Lymphocytic Leukemia Patients Treated With Novel Agents. Front Oncol 2020, 10, 894. [Google Scholar] [CrossRef]
- Maddocks, K.J.; Ruppert, A.S.; Lozanski, G.; Heerema, N.A.; Zhao, W.; Abruzzo, L.; Lozanski, A.; Davis, M.; Gordon, A.; Smith, L.L.; et al. Etiology of Ibrutinib Therapy Discontinuation and Outcomes in Patients With Chronic Lymphocytic Leukemia. JAMA Oncol 2015, 1, 80–87. [Google Scholar] [CrossRef]
- Stephens, D.M.; Byrd, J.C. Resistance to Bruton Tyrosine Kinase Inhibitors: The Achilles Heel of Their Success Story in Lymphoid Malignancies. Blood 2021, 138, 1099–1109. [Google Scholar] [CrossRef]
- Brown, J.; Mashima, K.; Fernandes, S.; Naeem, A.; Shupe, S.; Fardoun, R.; Davids, M. Mutations Detected in Real World Clinical Sequencing during BTK Inhibitor Treatment in CLL. Res Sq 2024. [Google Scholar] [CrossRef]
- Woyach, J.A.; Furman, R.R.; Liu, T.-M.; Ozer, H.G.; Zapatka, M.; Ruppert, A.S.; Xue, L.; Li, D.H.-H.; Steggerda, S.M.; Versele, M.; et al. Resistance Mechanisms for the Bruton’s Tyrosine Kinase Inhibitor Ibrutinib. N Engl J Med 2014, 370, 2286–2294. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.A.; Landau, D.A.; Taylor-Weiner, A.; Bozic, I.; Zhang, H.; Sarosiek, K.; Wang, L.; Stewart, C.; Fan, J.; Hoellenriegel, J.; et al. Clonal Evolution in Patients with Chronic Lymphocytic Leukaemia Developing Resistance to BTK Inhibition. Nat Commun 2016, 7, 11589. [Google Scholar] [CrossRef] [PubMed]
- Woyach, J.A.; Jones, D.; Jurczak, W.; Robak, T.; Illes, A.; Kater, A.P.; Ghia, P.; Byrd, J.C.; Seymour, J.F.; Long, S.; et al. Mutational Profile of Previously Treated Chronic Lymphocytic Leukemia Patients Progressing on Acalabrutinib or Ibrutinib. Blood, 2024. [Google Scholar] [CrossRef]
- Tatarczuch, M.; Waltham, M.; Shortt, J.; Polekhina, G.; Hawkes, E.A.; Ho, S.-J.; Trotman, J.; Brasacchio, D.; Co, M.; Li, J.; et al. Molecular Associations of Response to the New-Generation BTK Inhibitor Zanubrutinib in Marginal Zone Lymphoma. Blood Adv 2023, 7, 3531–3539. [Google Scholar] [CrossRef]
- Woolston, D.W.; Lee, N.D.; Shadman, M.; Latorre-Esteves, E.; Tee, X.R.; Fredrickson, J.; Kohrn, B.F.; Ujjani, C.; Eckel, A.; Till, B.; et al. Ultra-Deep Mutational Landscape in Chronic Lymphocytic Leukemia Uncovers Dynamics of Resistance to Targeted Therapies. Haematologica 2024, 109, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-M.; Woyach, J.A.; Zhong, Y.; Lozanski, A.; Lozanski, G.; Dong, S.; Strattan, E.; Lehman, A.; Zhang, X.; Jones, J.A.; et al. Hypermorphic Mutation of Phospholipase C, Γ2 Acquired in Ibrutinib-Resistant CLL Confers BTK Independency upon B-Cell Receptor Activation. Blood 2015, 126, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Walliser, C.; Hermkes, E.; Schade, A.; Wiese, S.; Deinzer, J.; Zapatka, M.; Désiré, L.; Mertens, D.; Stilgenbauer, S.; Gierschik, P. The Phospholipase Cγ2 Mutants R665W and L845F Identified in Ibrutinib-Resistant Chronic Lymphocytic Leukemia Patients Are Hypersensitive to the Rho GTPase Rac2 Protein. J Biol Chem 2016, 291, 22136–22148. [Google Scholar] [CrossRef] [PubMed]
- Landau, D.A.; Sun, C.; Rosebrock, D.; Herman, S.E.M.; Fein, J.; Sivina, M.; Underbayev, C.; Liu, D.; Hoellenriegel, J.; Ravichandran, S.; et al. The Evolutionary Landscape of Chronic Lymphocytic Leukemia Treated with Ibrutinib Targeted Therapy. Nat Commun 2017, 8, 2185. [Google Scholar] [CrossRef] [PubMed]
- Hill, H.A.; Qi, X.; Jain, P.; Nomie, K.; Wang, Y.; Zhou, S.; Wang, M.L. Genetic Mutations and Features of Mantle Cell Lymphoma: A Systematic Review and Meta-Analysis. Blood Adv 2020, 4, 2927–2938. [Google Scholar] [CrossRef]
- Rahal, R.; Frick, M.; Romero, R.; Korn, J.M.; Kridel, R.; Chan, F.C.; Meissner, B.; Bhang, H.; Ruddy, D.; Kauffmann, A.; et al. Pharmacological and Genomic Profiling Identifies NF-κB-Targeted Treatment Strategies for Mantle Cell Lymphoma. Nat Med 2014, 20, 87–92. [Google Scholar] [CrossRef]
- Nakhoda, S.; Vistarop, A.; Wang, Y.L. Resistance to Bruton Tyrosine Kinase Inhibition in Chronic Lymphocytic Leukaemia and Non-Hodgkin Lymphoma. Br J Haematol 2023, 200, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Reiff, S.D.; Mantel, R.; Smith, L.L.; Greene, J.T.; Muhowski, E.M.; Fabian, C.A.; Goettl, V.M.; Tran, M.; Harrington, B.K.; Rogers, K.A.; et al. The BTK Inhibitor ARQ 531 Targets Ibrutinib-Resistant CLL and Richter Transformation. Cancer Discov 2018, 8, 1300–1315. [Google Scholar] [CrossRef]
- Mato, A.R.; Woyach, J.A.; Brown, J.R.; Ghia, P.; Patel, K.; Eyre, T.A.; Munir, T.; Lech-Maranda, E.; Lamanna, N.; Tam, C.S.; et al. Pirtobrutinib after a Covalent BTK Inhibitor in Chronic Lymphocytic Leukemia. N Engl J Med 2023, 389, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Woyach, J.A.; Stephens, D.M.; Flinn, I.W.; Bhat, S.A.; Savage, R.E.; Chai, F.; Eathiraj, S.; Reiff, S.D.; Muhowski, E.M.; Granlund, L.; et al. First-in-Human Study of the Reversible BTK Inhibitor Nemtabrutinib in Patients with Relapsed/Refractory Chronic Lymphocytic Leukemia and B-Cell Non–Hodgkin Lymphoma. Cancer Discovery 2024, 14, 66–75. [Google Scholar] [CrossRef]
- Tadmor, T.; Eyre, T.A.; Benjamini, O.; Chaudhry, A.; Shen, J.; Leng, S.; Farooqui, M.Z.H.; Lavie, D. BELLWAVE-011: Phase 3 Randomized Trial of Nemtabrutinib versus Ibrutinib or Acalabrutinib in Untreated Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. JCO 2024, 42, TPS7088–TPS7088. [Google Scholar] [CrossRef]
- Wang, E.; Mi, X.; Thompson, M.C.; Montoya, S.; Notti, R.Q.; Afaghani, J.; Durham, B.H.; Penson, A.; Witkowski, M.T.; Lu, S.X.; et al. Mechanisms of Resistance to Noncovalent Bruton’s Tyrosine Kinase Inhibitors. N Engl J Med 2022, 386, 735–743. [Google Scholar] [CrossRef] [PubMed]
- On-Target BTK Mutations Promote Resistance to Noncovalent BTK Inhibitors. Cancer Discovery 2022, 12, 1179. [CrossRef]
- Dobrovolsky, D.; Wang, E.S.; Morrow, S.; Leahy, C.; Faust, T.; Nowak, R.P.; Donovan, K.A.; Yang, G.; Li, Z.; Fischer, E.S.; et al. Bruton Tyrosine Kinase Degradation as a Therapeutic Strategy for Cancer. Blood 2019, 133, 952–961. [Google Scholar] [CrossRef]
- Robbins, D.W.; Noviski, M.A.; Tan, Y.S.; Konst, Z.A.; Kelly, A.; Auger, P.; Brathaban, N.; Cass, R.; Chan, M.L.; Cherala, G.; et al. Discovery and Preclinical Pharmacology of NX-2127, an Orally Bioavailable Degrader of Bruton’s Tyrosine Kinase with Immunomodulatory Activity for the Treatment of Patients with B Cell Malignancies. J Med Chem 2024, 67, 2321–2336. [Google Scholar] [CrossRef]
- Danilov, A.; Tees, M.T.; Patel, K.; Wierda, W.G.; Patel, M.; Flinn, I.W.; Latif, T.; Ai, W.; Thompson, M.C.; Wang, M.L.; et al. A First-in-Human Phase 1 Trial of NX-2127, a First-in-Class Bruton’s Tyrosine Kinase (BTK) Dual-Targeted Protein Degrader with Immunomodulatory Activity, in Patients with Relapsed/Refractory B Cell Malignancies. Blood 2023, 142, 4463. [Google Scholar] [CrossRef]
- Searle, E. Initial Findings from a First-in-Human Phase 1a/b Trial of NX-5948, a Selective Bruton’s Tyrosine Kinase (BTK) Degrader, in Patients with Relapsed/Refractory B Cell Malignancies; ASH, 2023 December 11. [Google Scholar]
- Seymour, J.F.; Cheah, C.Y.; Parrondo, R.; Thompson, M.C.; Stevens, D.A.; Lasica, M.; Wang, M.L.; Kumar, A.; Trotman, J.; Alwan, M.; et al. First Results from a Phase 1, First-in-Human Study of the Bruton’s Tyrosine Kinase (BTK) Degrader Bgb-16673 in Patients (Pts) with Relapsed or Refractory (R/R) B-Cell Malignancies (BGB-16673-101). Blood 2023, 142, 4401. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




