Submitted:
12 July 2024
Posted:
15 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
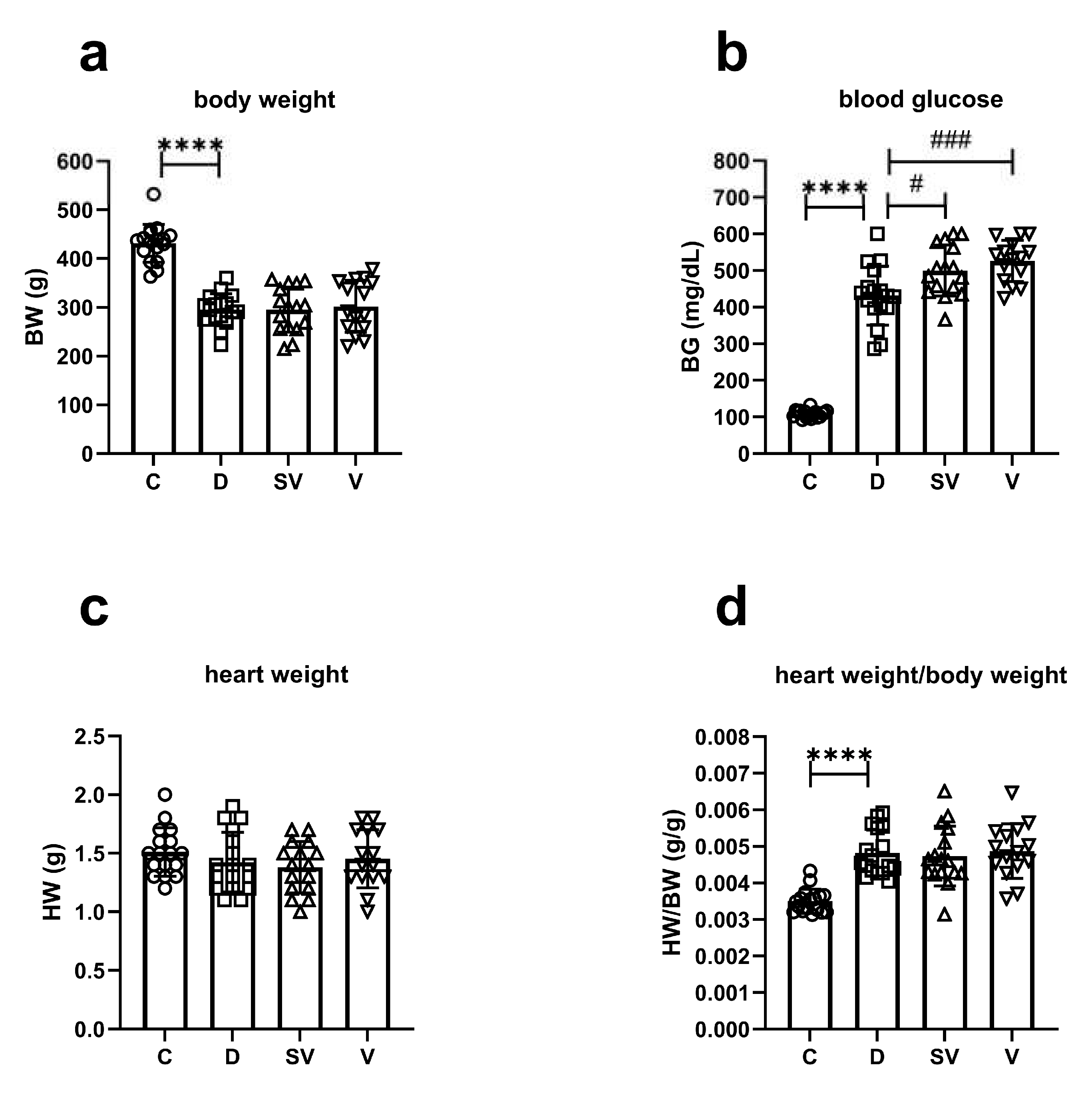
2.1. General Characteristics of Animals
2.2. In Vivo Cardiac Parameters
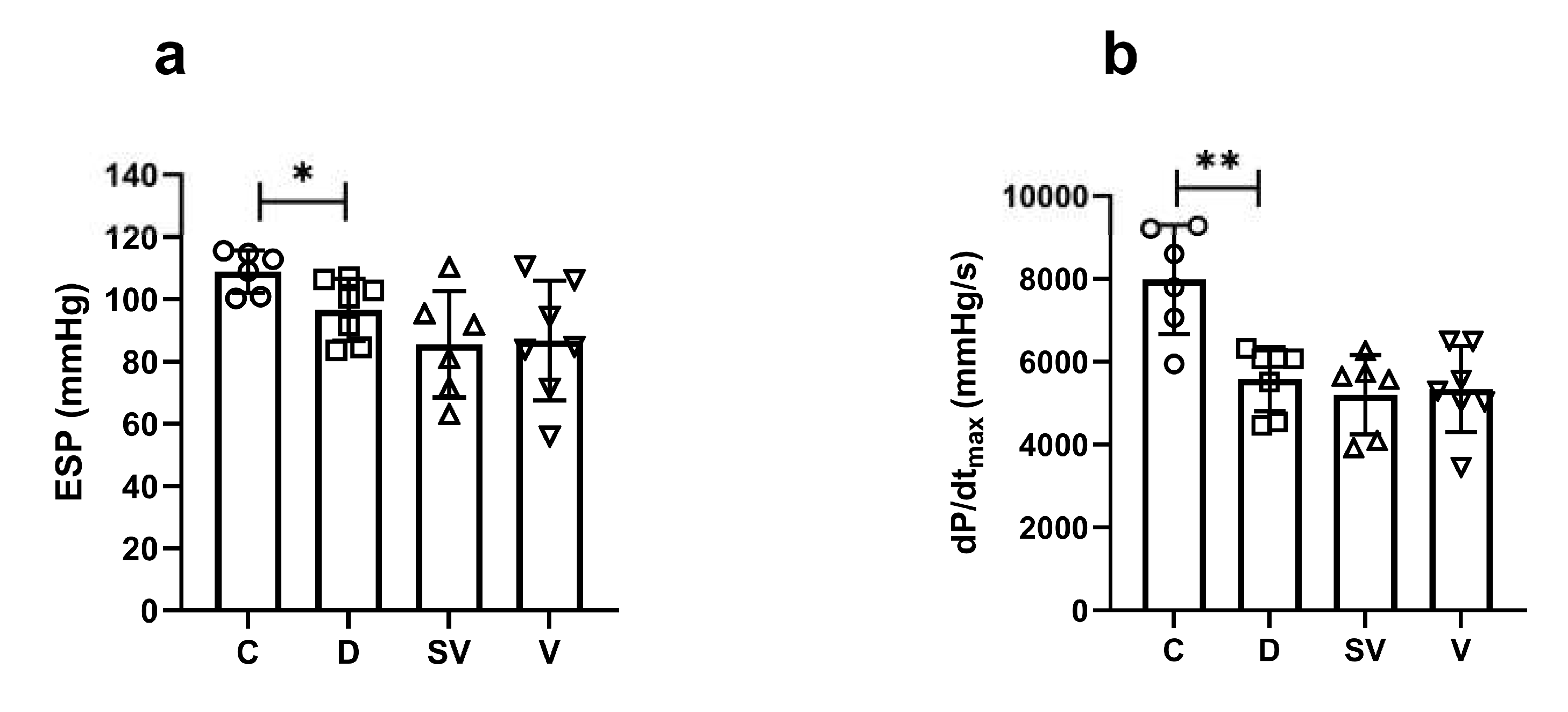
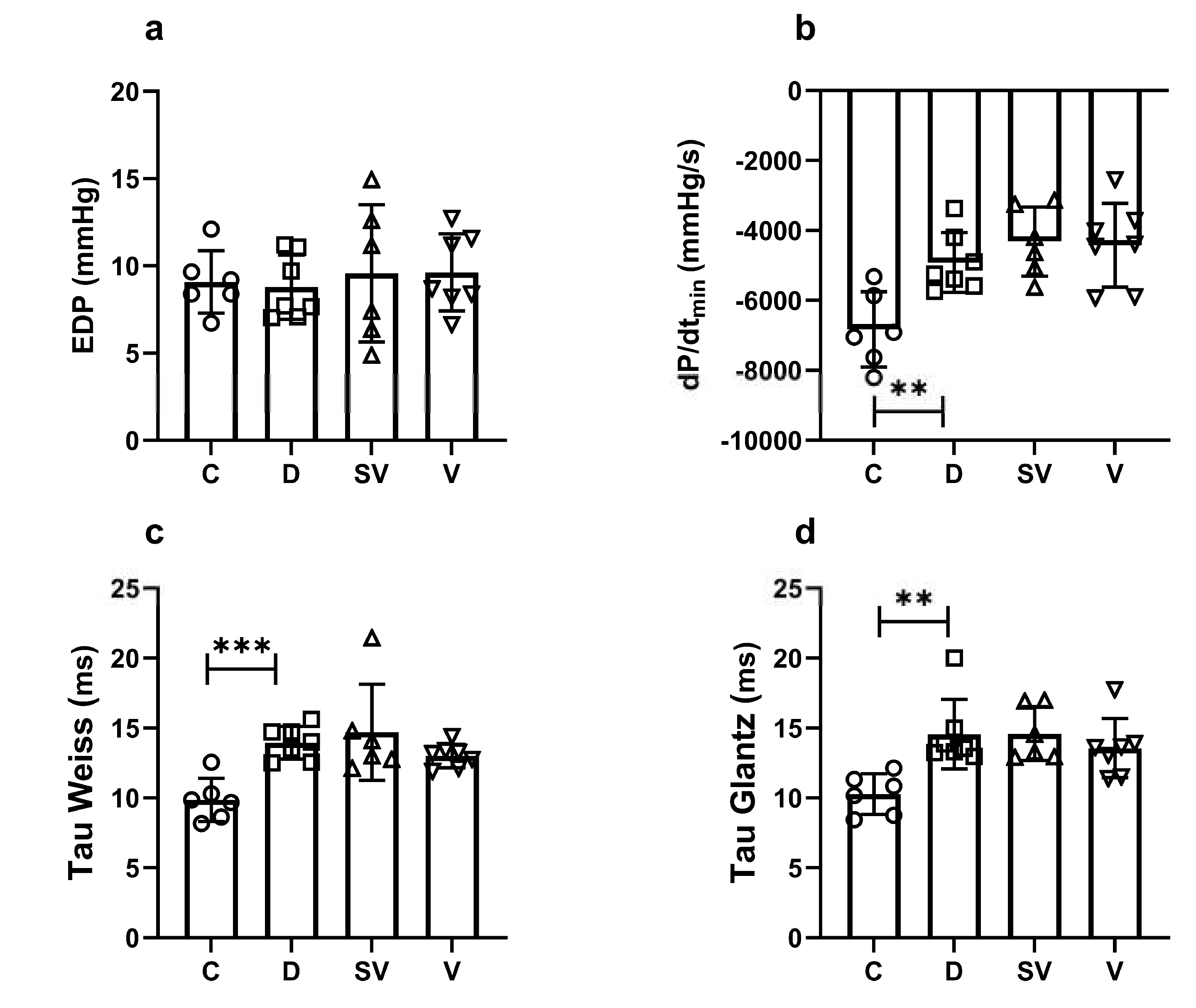
2.2.1. In Vivo Pressure-Volume (PV) Loop Analysis
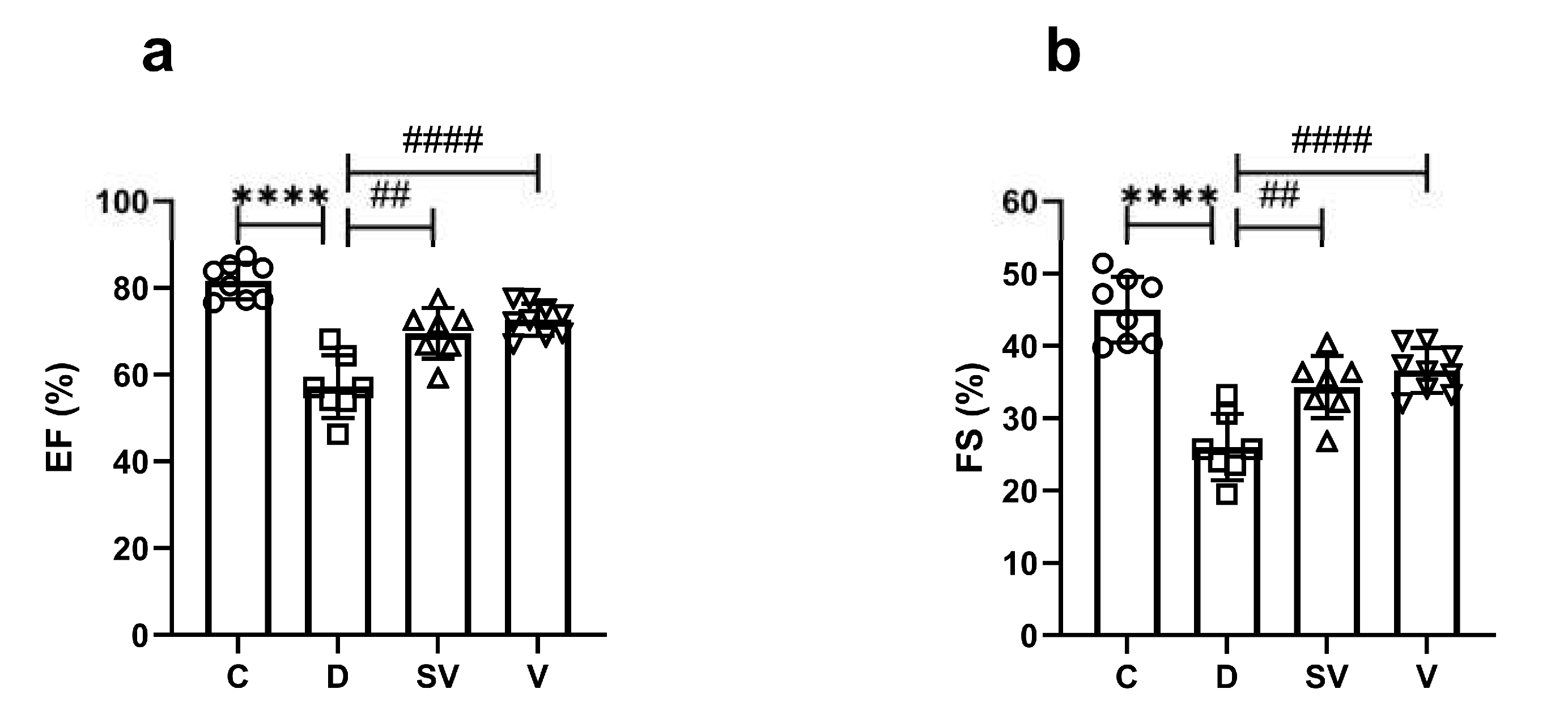
2.2.2. In Vivo Echocardiography Analysis
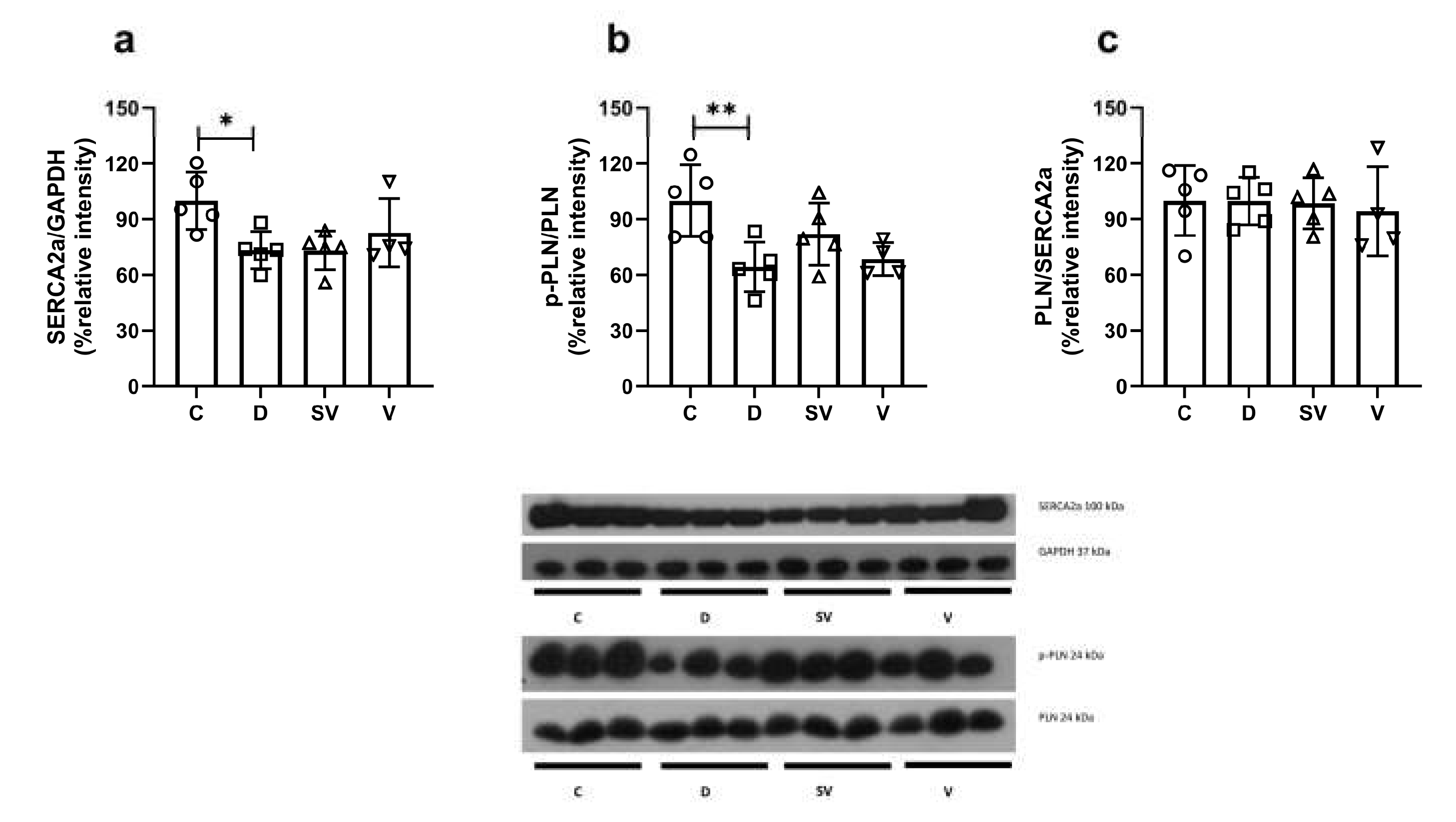
2.4. Protein Expression of Diastolic Components
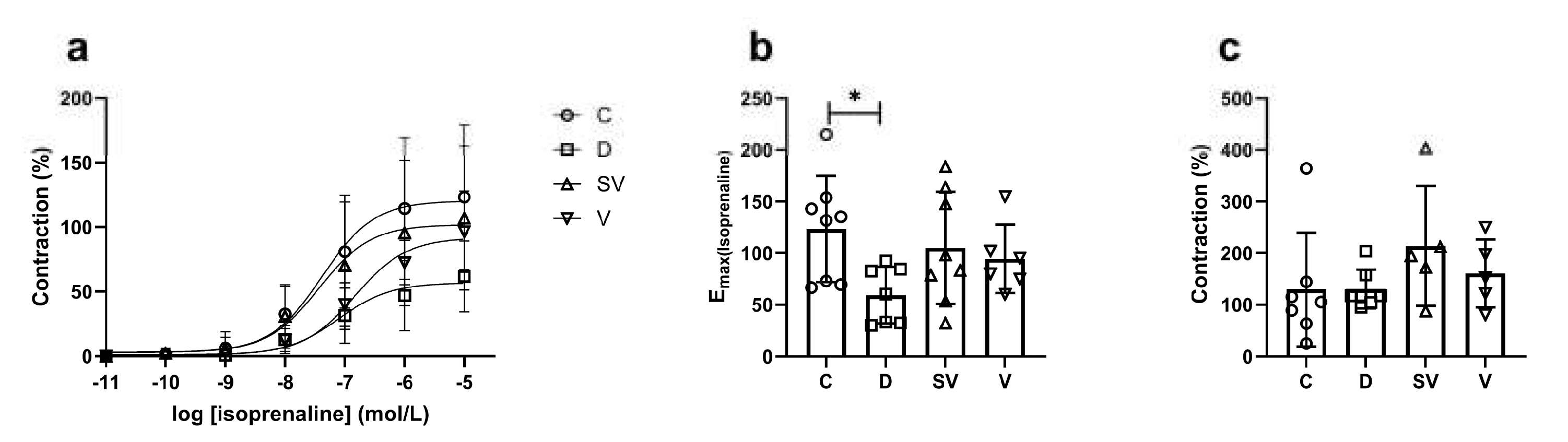
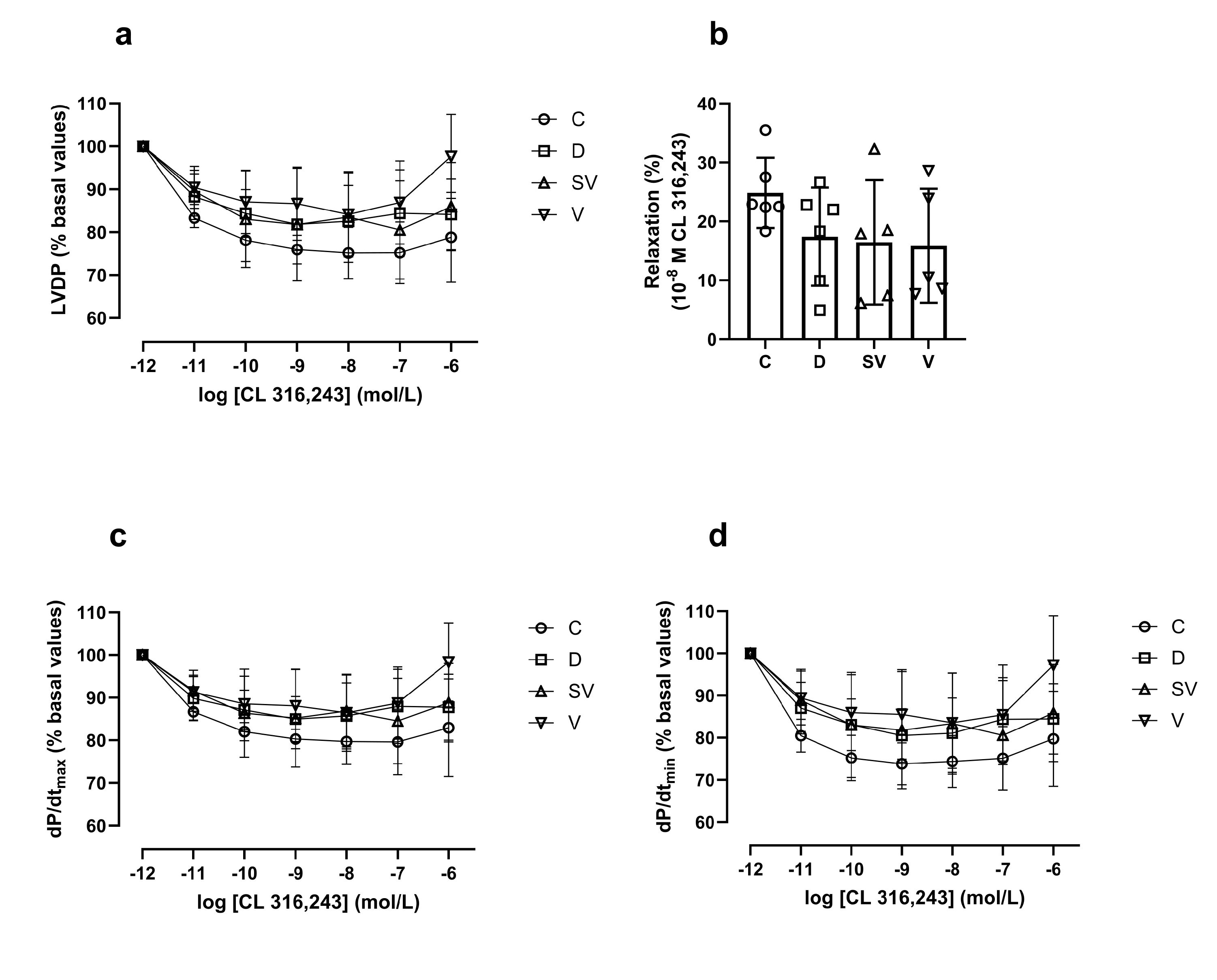
3. Discussion
3.1. Critique of Study Design and Experimental Model
3.2. Cardiac Hypertrophy
3.3. Cardiac Hemodynamic Parameters
3.5. Protein Expression of Diastolic Components
4. Materials and Methods
4.1. Animals and the Study Protocol
4.2. Oral Glucose Tolerance Test (OGTT)
4.3. In Vivo PV Loop Analysis
4.4. In Vivo Echocardiography Experiment
4.5. In Vitro Papillary Muscle Experiment
4.6. In Vitro Langendorff Heart Preparation Experiment
4.7. Western Blot Experiments
4.8. Data Analysis
4.9. Chemicals
5. Conclusions
- The age of the animals was considered as a baseline characteristic rather than blood glucose levels at the time of randomized group allocation. Therefore, it was not possible to interpret whether sacubitril/valsartan or valsartan affected glycemic control in the present study.
- Interpretation of the statistical significance data in some experiments was difficult with large standard deviations. In addition, we were not able to increase the sample size due to compliance with ethical constraints and the limited number of animals in the study.
- Cardiac β-AR subtype mRNA and/or protein expression levels could not be measured due to the non-specific binding capacity of the antibodies [83] and for economic reasons.
- The possible involvement of components contributing to the β-AR signaling pathways could not be investigated in the present study.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FDA. Center for Drug Evaluation and Research. 2015. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/207620Orig1s000OtherR.pdf.
- Mangiafico, S.; Costello-Boerrigter, L.C.; Andersen, I.A.; Cataliotti, A.; Burnett Jr, J.C. Neutral endopeptidase inhibition and the natriuretic peptide system: an evolving strategy in cardiovascular therapeutics. European heart journal 2013, 34, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.C.; Brunner, H.R.; Foster, C.; Huo, Y. Angiotensin II type 1 receptor antagonists in animal models of vascular, cardiac, metabolic and renal disease. Pharmacology & therapeutics 2016, 164, 1–81. [Google Scholar] [CrossRef]
- McMurray, J.J.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K. Angiotensin–neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014, 371, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhou, R.; Lu, C.; Chen, Q.; Xu, T.; Li, D. Effects of the angiotensin-receptor neprilysin inhibitor on cardiac reverse remodeling: meta-analysis. Journal of the American Heart Association 2019, 8, e012272. [Google Scholar] [CrossRef] [PubMed]
- Badreldin, H.A.; Aldosari, N.; Alnashwan, L.; Almutairi, T.; Yousif, N.; Alsulaiman, K.; Aljuhani, O.; Hafiz, A.; Alshaya, O. What the near Future Holds for Sacubitril/Valsartan: A Summary of Major Ongoing Studies. Journal of Cardiovascular Development and Disease 2022, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- IDF. International Diabetes Federation. IDF Diabetes Atlas 2019, 9. [Google Scholar]
- Haas, A.V.; McDonnell, M.E. Pathogenesis of cardiovascular disease in diabetes. Endocrinology and Metabolism Clinics 2018, 47, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Palomer, X.; Pizarro-Delgado, J.; Vazquez-Carrera, M. Emerging Actors in Diabetic Cardiomyopathy: Heartbreaker Biomarkers or Therapeutic Targets? Trends Pharmacol Sci 2018, 39, 452–467. [Google Scholar] [CrossRef] [PubMed]
- Jia, G.; DeMarco, V.G.; Sowers, J.R. Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nature Reviews Endocrinology 2016, 12, 144–153. [Google Scholar] [CrossRef]
- Kenny, H.C.; Abel, E.D. Heart failure in type 2 diabetes mellitus: impact of glucose-lowering agents, heart failure therapies, and novel therapeutic strategies. Circulation research 2019, 124, 121–141. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. European heart journal 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Larson, M.G.; Levy, D.; Benjamin, E.J.; Leip, E.P.; Wilson, P.W.; Vasan, R.S. Impact of obesity on plasma natriuretic peptide levels. Circulation 2004, 109, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Plante, E.; Menaouar, A.; Danalache, B.A.; Broderick, T.L.; Jankowski, M.; Gutkowska, J. Treatment with brain natriuretic peptide prevents the development of cardiac dysfunction in obese diabetic db/db mice. Diabetologia 2014, 57, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Lazo, M.; Young, J.H.; Brancati, F.L.; Coresh, J.; Whelton, S.; Ndumele, C.E.; Hoogeveen, R.; Ballantyne, C.M.; Selvin, E. NH2-terminal pro–brain natriuretic peptide and risk of diabetes. Diabetes 2013, 62, 3189–3193. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Wang, D.; Bi, H.; Zhang, H. The role of natriuretic peptides in diabetes and its complications. Biomedicine & Pharmacotherapy 2016, 84, 1826–1832. [Google Scholar] [CrossRef]
- Moro, C. Targeting cardiac natriuretic peptides in the therapy of diabetes and obesity. Expert opinion on therapeutic targets 2016, 20, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Varma, U.; Koutsifeli, P.; Benson, V.; Mellor, K.; Delbridge, L. Molecular mechanisms of cardiac pathology in diabetes–Experimental insights. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 2018, 1864, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Erdogan, B.R.; Michel, M.C.; Arioglu-Inan, E. Expression and signaling of β-adrenoceptor subtypes in the diabetic heart. Cells 2020, 9, 2548. [Google Scholar] [CrossRef]
- Iyngkaran, P.; Anavekar, N.; Majoni, W.; Thomas, M.C. The role and management of sympathetic overactivity in cardiovascular and renal complications of diabetes. Diabetes & metabolism 2013, 39, 290–298. [Google Scholar] [CrossRef]
- Amour, J.; Loyer, X.; Le Guen, M.; Mabrouk, N.; David, J.-S.; Camors, E.; Carusio, N.; Vivien, B.; Andriantsitohaina, R.; Heymes, C. Altered Contractile Response due to Increased β3-Adrenoceptor Stimulation in Diabetic CardiomyopathyThe Role of Nitric Oxide Synthase 1–derived Nitric Oxide. Anesthesiology: The Journal of the American Society of Anesthesiologists 2007, 107, 452–460. [Google Scholar] [CrossRef]
- Carillion, A.; Feldman, S.; Na, N.; Biais, M.; Carpentier, W.; Birenbaum, A.; Cagnard, N.; Loyer, X.; Bonnefont-Rousselot, D.; Hatem, S. Atorvastatin reduces β-Adrenergic dysfunction in rats with diabetic cardiomyopathy. PLoS One 2017, 12, e0180103. [Google Scholar] [CrossRef] [PubMed]
- Haley, J.M.; Thackeray, J.T.; Thorn, S.L.; DaSilva, J.N. Cardiac β-adrenoceptor expression is reduced in Zucker diabetic fatty rats as type-2 diabetes progresses. Plos one 2015, 10, e0127581. [Google Scholar] [CrossRef] [PubMed]
- Bidasee, K.R.; Zheng, H.; Shao, C.-H.; Parbhu, S.K.; Rozanski, G.J.; Patel, K.P. Exercise training initiated after the onset of diabetes preserves myocardial function: effects on expression of β-adrenoceptors. Journal of applied physiology 2008, 105, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Kayki-Mutlu, G.; Arioglu-Inan, E.; Ozakca, I.; Ozcelikay, A.T.; Altan, V.M. beta3-Adrenoceptor-mediated responses in diabetic rat heart. Gen Physiol Biophys 2014, 33, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Okatan, E.N.; Tuncay, E.; Hafez, G.; Turan, B. Profiling of cardiac β-adrenoceptor subtypes in the cardiac left ventricle of rats with metabolic syndrome: Comparison with streptozotocin-induced diabetic rats. Canadian Journal of Physiology and Pharmacology 2015, 93, 517–525. [Google Scholar] [CrossRef]
- Gauthier, C.; Langin, D.; Balligand, J.L. Beta3-adrenoceptors in the cardiovascular system. Trends Pharmacol Sci 2000, 21, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Hartupee, J.; Mann, D.L. Neurohormonal activation in heart failure with reduced ejection fraction. Nature Reviews Cardiology 2017, 14, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Seferovic, J.P.; Claggett, B.; Seidelmann, S.B.; Seely, E.W.; Packer, M.; Zile, M.R.; Rouleau, J.L.; Swedberg, K.; Lefkowitz, M.; Shi, V.C. Effect of sacubitril/valsartan versus enalapril on glycaemic control in patients with heart failure and diabetes: a post-hoc analysis from the PARADIGM-HF trial. The lancet Diabetes & endocrinology 2017, 5, 333–340. [Google Scholar] [CrossRef]
- Bunsawat, K.; Ratchford, S.M.; Alpenglow, J.K.; Stehlik, J.; Smith, A.S.; Richardson, R.S.; Wray, D.W. Sympathoinhibitory effect of sacubitril-valsartan in heart failure with reduced ejection fraction: A pilot study. Autonomic Neuroscience 2021, 235, 102834. [Google Scholar] [CrossRef]
- Suematsu, Y.; Miura, S.i.; Goto, M.; Matsuo, Y.; Arimura, T.; Kuwano, T.; Imaizumi, S.; Iwata, A.; Yahiro, E.; Saku, K. LCZ696, an angiotensin receptor–neprilysin inhibitor, improves cardiac function with the attenuation of fibrosis in heart failure with reduced ejection fraction in streptozotocin-induced diabetic mice. European journal of heart failure 2016, 18, 386–393. [Google Scholar] [CrossRef]
- Ai, J.; Shuai, Z.; Tang, K.; Li, Z.; Zou, L.; Liu, M. Sacubitril/valsartan alleviates myocardial fibrosis in diabetic cardiomyopathy rats. Hellenic journal of cardiology: HJC= Hellenike kardiologike epitheorese 2021, 62, 389–391. [Google Scholar] [CrossRef] [PubMed]
- Ge, Q.; Zhao, L.; Ren, X.-M.; Ye, P.; Hu, Z.-Y. LCZ696, an angiotensin receptor-neprilysin inhibitor, ameliorates diabetic cardiomyopathy by inhibiting inflammation, oxidative stress and apoptosis. Experimental Biology and Medicine 2019, 244, 1028–1039. [Google Scholar] [CrossRef] [PubMed]
- Belali, O.M.; Ahmed, M.M.; Mohany, M.; Belali, T.M.; Alotaibi, M.M.; Al-Hoshani, A.; Al-Rejaie, S.S. LCZ696 Protects against Diabetic Cardiomyopathy-Induced Myocardial Inflammation, ER Stress, and Apoptosis through Inhibiting AGEs/NF-κB and PERK/CHOP Signaling Pathways. International Journal of Molecular Sciences 2022, 23, 1288. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Tu, H.; Zheng, H.; Zhang, L.; Tran, T.P.; Muelleman, R.L.; Li, Y.-L. Alterations of calcium channels and cell excitability in intracardiac ganglion neurons from type 2 diabetic rats. American Journal of Physiology-Cell Physiology 2012, 302, C1119–C1127. [Google Scholar] [CrossRef]
- Liu, X.-Y.; Liu, F.-C.; Deng, C.-Y.; Zhang, M.-Z.; Yang, M.; Xiao, D.-Z.; Lin, Q.-X.; Cai, S.-T.; Kuang, S.-J.; Chen, J. Left ventricular deformation associated with cardiomyocyte Ca 2+ transients delay in early stage of low-dose of STZ and high-fat diet induced type 2 diabetic rats. BMC cardiovascular disorders 2016, 16, 1–12. [Google Scholar] [CrossRef]
- Davidson, E.P.; Coppey, L.J.; Shevalye, H.; Obrosov, A.; Yorek, M.A. Vascular and neural complications in type 2 diabetic rats: improvement by sacubitril/valsartan greater than valsartan alone. Diabetes 2018, 67, 1616–1626. [Google Scholar] [CrossRef]
- Liu, H.-j.; Zhang, C.-y.; Song, F.; Xiao, T.; Meng, J.; Zhang, Q.; Liang, C.-l.; Li, S.; Wang, J.; Zhang, B. A novel partial agonist of peroxisome proliferator-activated receptor γ with excellent effect on insulin resistance and type 2 diabetes. Journal of Pharmacology and Experimental Therapeutics 2015, 353, 573–581. [Google Scholar] [CrossRef]
- Sahin, K.; Tuzcu, M.; Orhan, C.; Sahin, N.; Kucuk, O.; Ozercan, I.H.; Juturu, V.; Komorowski, J.R. Anti-diabetic activity of chromium picolinate and biotin in rats with type 2 diabetes induced by high-fat diet and streptozotocin. British Journal of Nutrition 2013, 110, 197–205. [Google Scholar] [CrossRef]
- Yesilyurt, Z.E.; Erdogan, B.R.; Karaomerlioglu, I.; Muderrisoglu, A.E.; Michel, M.C.; Arioglu-Inan, E. Urinary bladder weight and function in a rat model of mild hyperglycemia and its treatment with dapagliflozin. Frontiers in pharmacology 2019, 10, 911. [Google Scholar] [CrossRef]
- Abo-Khookh, A.M.; Ghoneim, H.A.; Abdelaziz, R.R.; Nader, M.A.; Shawky, N.M. The dual inhibitor Sacubitril-valsartan ameliorate high-fat high-fructose-induced metabolic disorders in rats superiorly compared to valsartan only. Journal of Pharmacy and Pharmacology 2023, 75, 846–858. [Google Scholar] [CrossRef]
- Selvaraj, S.; Claggett, B.L.; Packer, M.; Zannad, F.; Anand, I.S.; Pieske, B.; Zhao, Z.; Shi, V.C.; Lefkowitz, M.P.; McMurray, J.J. Effects of sacubitril/valsartan on serum lipids in heart failure with preserved ejection fraction. Journal of the American Heart Association 2021, 10, e022069. [Google Scholar] [CrossRef]
- Alqahtani, F.; Mohany, M.; Alasmari, A.F.; Alanazi, A.Z.; Belali, O.M.; Ahmed, M.M.; Al-Rejaie, S.S. Angiotensin II receptor Neprilysin inhibitor (LCZ696) compared to Valsartan attenuates Hepatotoxicity in STZ-induced hyperglycemic rats. International Journal of Medical Sciences 2020, 17, 3098. [Google Scholar] [CrossRef] [PubMed]
- Habibi, J.; Aroor, A.R.; Das, N.A.; Manrique-Acevedo, C.M.; Johnson, M.S.; Hayden, M.R.; Nistala, R.; Wiedmeyer, C.; Chandrasekar, B.; DeMarco, V.G. The combination of a neprilysin inhibitor (sacubitril) and angiotensin-II receptor blocker (valsartan) attenuates glomerular and tubular injury in the Zucker Obese rat. Cardiovascular diabetology 2019, 18, 1–17. [Google Scholar] [CrossRef]
- Myakala, K.; Jones, B.A.; Wang, X.X.; Levi, M. Sacubitril/valsartan treatment has differential effects in modulating diabetic kidney disease in db/db mice and KKAy mice compared with valsartan treatment. American Journal of Physiology-Renal Physiology 2021, 320, F1133–F1151. [Google Scholar] [CrossRef] [PubMed]
- Negishi, K. Echocardiographic feature of diabetic cardiomyopathy: where are we now? Cardiovascular diagnosis and therapy 2018, 8, 47. [Google Scholar] [CrossRef]
- Rubler, S.; Dlugash, J.; Yuceoglu, Y.Z.; Kumral, T.; Branwood, A.W.; Grishman, A. New type of cardiomyopathy associated with diabetic glomerulosclerosis. The American journal of cardiology 1972, 30, 595–602. [Google Scholar] [CrossRef]
- Ge, Q.; Zhao, L.; Liu, C.; Ren, X.; Yu, Y.-h.; Pan, C.; Hu, Z. LCZ696, an Angiotensin Receptor-Neprilysin Inhibitor, Improves Cardiac Hypertrophy and Fibrosis and Cardiac Lymphatic Remodeling in Transverse Aortic Constriction Model Mice. BioMed Research International 2020, 2020. [Google Scholar] [CrossRef]
- Kusaka, H.; Sueta, D.; Koibuchi, N.; Hasegawa, Y.; Nakagawa, T.; Lin, B.; Ogawa, H.; Kim-Mitsuyama, S. LCZ696, angiotensin II receptor-neprilysin inhibitor, ameliorates high-salt-induced hypertension and cardiovascular injury more than valsartan alone. American journal of hypertension 2015, 28, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Suematsu, Y.; Jing, W.; Nunes, A.; Kashyap, M.L.; Khazaeli, M.; Vaziri, N.D.; Moradi, H. LCZ696 (sacubitril/valsartan), an angiotensin-receptor neprilysin inhibitor, attenuates cardiac hypertrophy, fibrosis, and vasculopathy in a rat model of chronic kidney disease. Journal of cardiac failure 2018, 24, 266–275. [Google Scholar] [CrossRef]
- Miyoshi, T.; Nakamura, K.; Miura, D.; Yoshida, M.; Saito, Y.; Akagi, S.; Ohno, Y.; Kondo, M.; Ito, H. Effect of LCZ696, a dual angiotensin receptor neprilysin inhibitor, on isoproterenol-induced cardiac hypertrophy, fibrosis, and hemodynamic change in rats. Cardiology journal 2019, 26, 575–583. [Google Scholar] [CrossRef]
- Aroor, A.R.; Mummidi, S.; Lopez-Alvarenga, J.C.; Das, N.; Habibi, J.; Jia, G.; Lastra, G.; Chandrasekar, B.; DeMarco, V.G. Sacubitril/valsartan inhibits obesity-associated diastolic dysfunction through suppression of ventricular-vascular stiffness. Cardiovascular Diabetology 2021, 20, 1–18. [Google Scholar] [CrossRef]
- Werner, R.A.; Eissler, C.; Hayakawa, N.; Arias-Loza, P.; Wakabayashi, H.; Javadi, M.S.; Chen, X.; Shinaji, T.; Lapa, C.; Pelzer, T. Left Ventricular Diastolic Dysfunction in a Rat Model of Diabetic Cardiomyopathy using ECG-gated 18 F-FDG PET. Scientific reports 2018, 8, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wang, K.; Wang, W.; Wen, Z.; Wang, P.; Liu, L.; Wang, D.W. Glucagon-like peptide-1 ameliorates cardiac lipotoxicity in diabetic cardiomyopathy via the PPAR α pathway. Aging Cell 2018, 17, e12763. [Google Scholar] [CrossRef] [PubMed]
- Ye, F.; Li, H.; Chen, X.; Wang, Y.; Lin, W.; Chen, H.; Huang, S.; Han, S.; Guan, F.; Huang, Z. Efficacy and safety of sacubitril valsartan in treating heart failure with midrange ejection fraction after acute myocardial infarction in diabetic patients. Medicine 2022, 101. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-C.; Wo, H.-T.; Lee, H.-L.; Lin, S.-F.; Chu, Y.; Wen, M.-S.; Chou, C.-C. Sacubitril/valsartan therapy ameliorates ventricular tachyarrhythmia inducibility in a rabbit myocardial infarction model. Journal of cardiac failure 2020, 26, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Pericas, P.; Mas-Lladó, C.; Ramis-Barceló, M.F.; Valadrón, I.; Noris Mora, M.; Pasamar Márquez, L.; González Colino, R.; Forteza Albertí, J.F.; Peral Disdier, V.; Rossello, X. Impact of Sacubitril–Valsartan Treatment on Diastolic Function in Patients with Heart Failure and Reduced Ejection Fraction. High Blood Pressure & Cardiovascular Prevention 2021, 28, 167–175. [Google Scholar] [CrossRef]
- Ganesananthan, S.; Shah, N.; Shah, P.; Elsayed, H.; Phillips, J.; Parkes, A.; Morgan, A.; Yousef, Z. Real-world treatment switching to sacubitril/valsartan in patients with heart failure with reduced ejection fraction: A cohort study. Open Heart 2020, 7, e001305. [Google Scholar] [CrossRef] [PubMed]
- Martens, P.; Beliën, H.; Dupont, M.; Vandervoort, P.; Mullens, W. The reverse remodeling response to sacubitril/valsartan therapy in heart failure with reduced ejection fraction. Cardiovascular therapeutics 2018, 36, e12435. [Google Scholar] [CrossRef] [PubMed]
- von Lueder, T.G.; Wang, B.H.; Kompa, A.R.; Huang, L.; Webb, R.; Jordaan, P.; Atar, D.; Krum, H. Angiotensin receptor neprilysin inhibitor LCZ696 attenuates cardiac remodeling and dysfunction after myocardial infarction by reducing cardiac fibrosis and hypertrophy. Circulation: Heart Failure 2015, 8, 71–78. [Google Scholar] [CrossRef]
- Nordén, E.S.; Bendiksen, B.A.; Andresen, H.; Bergo, K.K.; Espe, E.K.; Hasic, A.; Hauge-Iversen, I.M.; Veras, I.; Hussain, R.I.; Sjaastad, I. Sacubitril/valsartan ameliorates cardiac hypertrophy and preserves diastolic function in cardiac pressure overload. ESC heart failure 2021, 8, 918–927. [Google Scholar] [CrossRef]
- Arioglu-Inan, E.; Ozakca, I.; Kayki-Mutlu, G.; Sepici-Dincel, A.; Altan, V.M. The role of insulin–thyroid hormone interaction on β-adrenoceptor-mediated cardiac responses. European journal of pharmacology 2013, 718, 533–543. [Google Scholar] [CrossRef]
- Dinçer, Ü.D.; Onay, A.; Arı, N.; Özçelikay, A.T.; Altan, V.M. The effects of diabetes on β-adrenoceptor mediated responsiveness of human and rat atria. Diabetes research and clinical practice 1998, 40, 113–122. [Google Scholar] [CrossRef]
- Dinçer, Ü.D.; Bidasee, K.R.; Güner, Ş.; Tay, A.; Özçelikay, A.T.; Altan, V.M. The effect of diabetes on expression of β1-, β2-, and β3-adrenoreceptors in rat hearts. Diabetes 2001, 50, 455–461. [Google Scholar] [CrossRef]
- Matsuda, N.; Hattori, Y.; Gando, S.; Akaishi, Y.; Kemmotsu, O.; Kanno, M. Diabetes-induced down-regulation of β1-adrenoceptor mRNA expression in rat heart. Biochemical pharmacology 1999, 58, 881–885. [Google Scholar] [CrossRef]
- Fu, Q.; Shi, Q.; West, T.M.; Xiang, Y.K. Cross-talk between insulin signaling and GPCRs. Journal of cardiovascular pharmacology 2017, 70, 74. [Google Scholar] [CrossRef]
- Eiringhaus, J.; Wünsche, C.M.; Tirilomis, P.; Herting, J.; Bork, N.; Nikolaev, V.O.; Hasenfuss, G.; Sossalla, S.; Fischer, T.H. Sacubitrilat reduces pro-arrhythmogenic sarcoplasmic reticulum Ca2+ leak in human ventricular cardiomyocytes of patients with end-stage heart failure. ESC Heart Failure 2020, 7, 2992–3002. [Google Scholar] [CrossRef]
- George, M.S.; Pitt, G.S. The real estate of cardiac signaling: location, location, location. Proceedings of the National Academy of Sciences 2006, 103, 7535–7536. [Google Scholar] [CrossRef]
- Arioglu-Inan; G Kayki-Mutlu; BR Erdogan; AE Muderrisoglu; I Karaomerlioglu; ZE Yesilyurt; S Degirmenci; B Turan; Altan, V. The effects of leptin on cardiac function in streptozotocin diabetic rats. BPS Pharmacology 2017 Abstract Book 2017. [Google Scholar]
- Uyar-Boztas, C.; Arioglu-Inan, E.; Muderrisoglu, A.; Kayki-Mutlu, G.; Erdogan, B.; Yesilyurt, Z.; Karaomerlioglu, I.; Altan, V. The effect of sitagliptin on beta-adrenoceptor-mediated cardiac responses in streptozotocin induced diabetic rats. Diabetes Stoffwech H 2017, 26, 15. [Google Scholar]
- Derkach, K.; Bondareva, V.; Moyseyuk, I.; Shpakov, A. The influence of two-month treatment with bromocryptine on activity of the adenylyl cyclase signaling system in the myocardium and testes of rats with type 2 diabetes mellitus. Tsitologiia 2014, 56, 907–918. [Google Scholar]
- Bers, D.M. Cardiac excitation–contraction coupling. Nature 2002, 415, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Connelly, K.; Kelly, D.J.; Zhang, Y.; Prior, D.L.; Martin, J.; Cox, A.; Thai, K.; Feneley, M.P.; Tsoporis, J.; White, K. Functional, structural and molecular aspects of diastolic heart failure in the diabetic (mRen-2) 27 rat. Cardiovascular research 2007, 76, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Kawase, Y.; Hajjar, R.J. The cardiac sarcoplasmic/endoplasmic reticulum calcium ATPase: a potent target for cardiovascular diseases. Nature Clinical Practice Cardiovascular Medicine 2008, 5, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Netticadan, T.; Temsah, R.M.; Kent, A.; Elimban, V.; Dhalla, N.S. Depressed levels of Ca2+-cycling proteins may underlie sarcoplasmic reticulum dysfunction in the diabetic heart. Diabetes 2001, 50, 2133–2138. [Google Scholar] [CrossRef] [PubMed]
- Watanuki, S.; Matsuda, N.; Sakuraya, F.; Jesmin, S.; Hattori, Y. Protein kinase C modulation of the regulation of sarcoplasmic reticular function by protein kinase A-mediated phospholamban phosphorylation in diabetic rats. British journal of pharmacology 2004, 141, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Müller, O.J.; Lange, M.; Rattunde, H.; Lorenzen, H.-P.; Müller, M.; Frey, N.; Bittner, C.; Simonides, W.; Katus, H.A.; Franz, W.-M. Transgenic rat hearts overexpressing SERCA2a show improved contractility under baseline conditions and pressure overload. Cardiovascular research 2003, 59, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Teshima, Y.; Takahashi, N.; Saikawa, T.; Hara, M.; Yasunaga, S.; Hidaka, S.; Sakata, T. Diminished expression of sarcoplasmic reticulum Ca2+-ATPase and ryanodine sensitive Ca2+ channel mRNA in streptozotocin-induced diabetic rat heart. Journal of molecular and cellular cardiology 2000, 32, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cannell, M.B.; Phillips, A.R.; Cooper, G.J.; Ward, M.-L. Altered calcium homeostasis does not explain the contractile deficit of diabetic cardiomyopathy. Diabetes 2008, 57, 2158–2166. [Google Scholar] [CrossRef] [PubMed]
- Lai, P.; Nikolaev, V.O.; De Jong, K.A. Understanding the role of SERCA2a microdomain remodeling in heart failure induced by obesity and type 2 diabetes. Journal of Cardiovascular Development and Disease 2022, 9, 163. [Google Scholar] [CrossRef]
- Bowe, J.E.; Franklin, Z.J.; Hauge-Evans, A.C.; King, A.J.; Persaud, S.J.; Jones, P.M. Metabolic phenotyping guidelines: assessing glucose homeostasis in rodent models. Journal of endocrinology 2014, 222, G13–G25. [Google Scholar] [CrossRef]
- Mátyás, C.; Kovács, A.; Németh, B.T.; Oláh, A.; Braun, S.; Tokodi, M.; Barta, B.A.; Benke, K.; Ruppert, M.; Lakatos, B.K. Comparison of speckle-tracking echocardiography with invasive hemodynamics for the detection of characteristic cardiac dysfunction in type-1 and type-2 diabetic rat models. Cardiovascular diabetology 2018, 17, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hamdani, N.; van der Velden, J. Lack of specificity of antibodies directed against human beta-adrenergic receptors. Naunyn-Schmiedeberg's archives of pharmacology 2009, 379, 403–407. [Google Scholar] [CrossRef] [PubMed]
| C (n=6) | D (n=7) | SV (n=6) | V (n=7) | |
|---|---|---|---|---|
| SBP (mmHg) | 110.89 ± 18.41 | 103.40 ± 11.69 | 93.54 ± 19.72 | 91.52 ± 20.41 |
| DBP (mmHg) | 79.29 ± 22.43 | 71.94 ± 9.23 | 67.69 ± 16.18 | 65.99 ± 20.57 |
| MAP (mmHg) | 100.36 ± 19.55 | 92.91 ± 10.77 | 84.92 ± 18.24 | 83.01 ± 20.13 |
| HR (beat/min) | 311.13 ± 39.09 | 247.17 ± 20.27** | 256.29 ± 18.74 | 251.35 ± 21.76 |
| EDV (µL) | 370.64 ± 69.67 | 436.66 ± 46.89 | 436.09 ± 76.22 | 411.52 ± 74.01 |
| EDVI (µL/g) | 0.92 ± 0.16 | 1.56 ± 0.29*** | 1.59 ± 0.31 | 1.43 ± 0.29 |
| ESV (µL) | 162.06 ± 37.83 | 200.17 ± 26.07 | 209.34 ± 26.93 | 191.61 ± 22.42 |
| ESVI (µL/g) | 0.40 ± 0.08 | 0.71 ± 0.13*** | 0.77 ± 0.18 | 0.67 ± 0.11 |
| SV (µL) | 208.58 ± 47.93 | 236.49 ± 36.31 | 226.75 ± 60.39 | 219.91 ± 60.81 |
| SVI (µL/g) | 0.52 ± 0.12 | 0.85 ± 0.19** | 0.82 ± 0.18 | 0.76 ± 0.21 |
| C (n=5) | D (n=6) | SV (n=6) | V (n=6) | |
|---|---|---|---|---|
| ESPVR | 0.390 ± 0.156 | 0.449 ± 0.166 | 0.334 ± 0.106 | 0.317 ± 0.132 |
| EDPVR | 0.010 ± 0.002 | 0.007 ± 0.003 | 0.006 ± 0.003 | 0.006 ± 0.003 |
| PRSW | 62.58 ± 9.44 | 53.69 ± 6.83 | 56.86 ± 7.73 | 53.31 ± 3.79 |
| C (n=8) | D (n=7) | SV (n=7) | V (n=9) | |
|---|---|---|---|---|
| IVSd (mm) | 1.91 ± 0.58 | 2.31 ± 0.38 | 1.83 ± 0.25# | 1.78 ± 0.25## |
| IVSId (mm/kg) | 4.30 ± 1.37 | 6.94 ± 1.04** | 5.64 ± 0.65# | 5.73 ± 1.13# |
| LVIDd (mm) | 5.49 ± 0.67 | 5.06 ± 0.91 | 5.21 ± 1.12 | 4.92 ± 0.50 |
| LVIDId (mm/kg) | 12.26 ± 1.55 | 15.33 ± 3.35* | 16.46 ± 5.39 | 15.84 ± 2.52 |
| LVPWd (mm) | 2.35 ± 1.90 | 2.20 ± 0.72 | 1.89 ± 0.29 | 2.01 ± 0.66 |
| LVPWId (mm/kg) | 5.05 ± 3.47 | 6.51 ± 1.65 | 5.82 ± 0.87 | 6.47 ± 2.30 |
| IVSs (mm) | 2.93 ± 0.83 | 2.76 ± 0.51 | 2.17 ± 0.45# | 2.17 ± 0.35# |
| IVSIs (mm/kg) | 6.50 ± 1.69 | 8.31 ± 1.65 | 6.76 ± 1.65 | 6.96 ± 1.37 |
| LVIDs (mm) | 3.01 ± 0.21 | 3.71 ± 0.51** | 3.43 ± 0.78 | 3.10 ± 0.21## |
| LVIDIs (mm/kg) | 6.75 ± 0.72 | 11.27 ± 2.20**** | 10.86 ± 3.80 | 9.96 ± 1.16 |
| LVPWs (mm) | 2.93 ± 0.49 | 2.80 ± 0.66 | 2.56 ± 0.54 | 2.79 ± 0.60 |
| LVPWIs (mm/kg) | 6.57 ± 1.36 | 8.33 ± 1.43* | 7.80 ± 1.09 | 9.00 ± 2.29 |
| CO (mL/dk) | 101.25 ± 39.07 | 52.86 ± 29.84* | 60.14 ± 28.64 | 51.11 ± 13.64 |
| CI (mL/dk.g) | 0.23 ± 0.09 | 0.16 ± 0.09 | 0.19 ± 0.11 | 0.16 ± 0.05 |
| EF (%) | 81.60 ± 4.20 | 57.27 ± 7.21**** | 69.56 ± 5.84## | 72.69 ± 3.72#### |
| FS (%) | 45.01 ± 4.54 | 26.04 ± 4.60**** | 34.34 ± 4.31## | 36.63 ± 3.11#### |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




