Submitted:
11 July 2024
Posted:
15 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
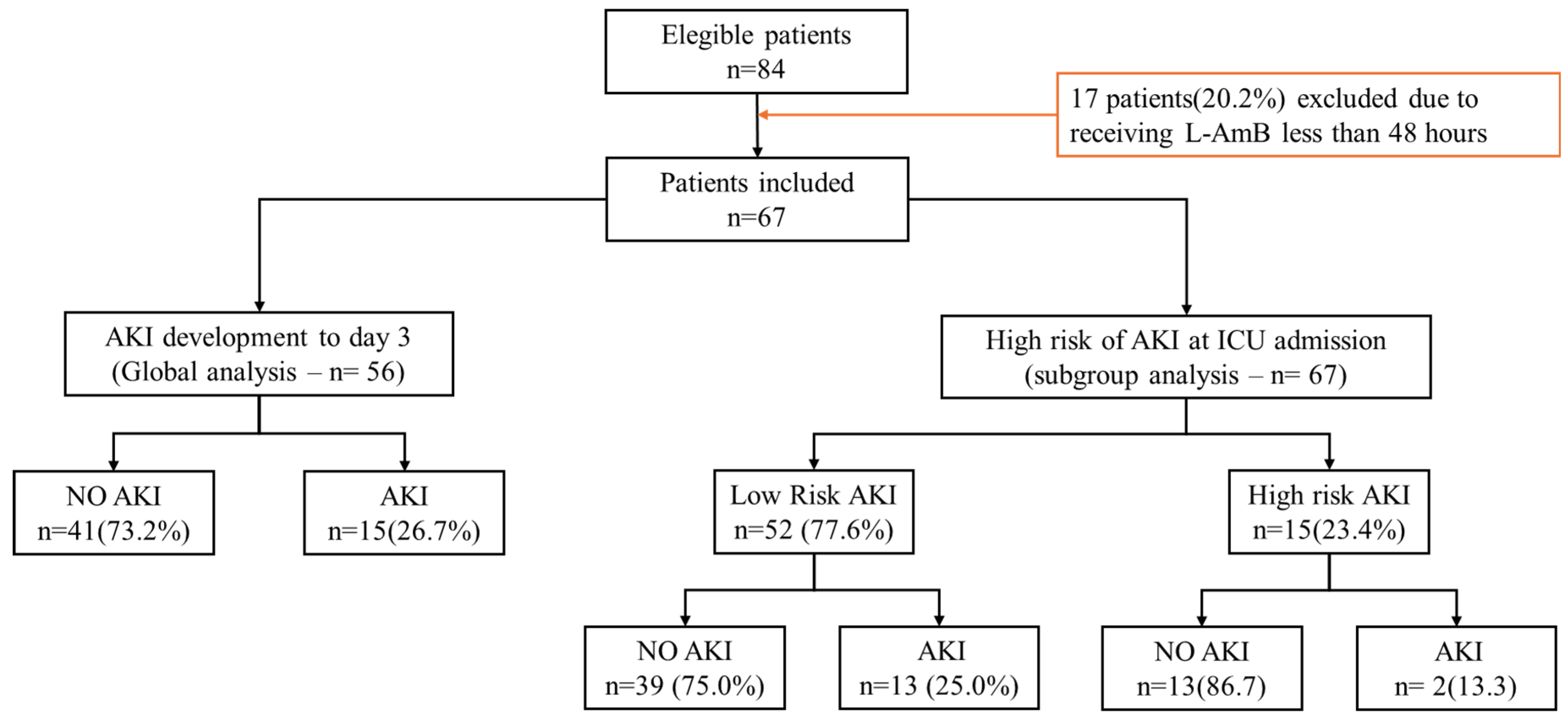
2.1. Whole Population
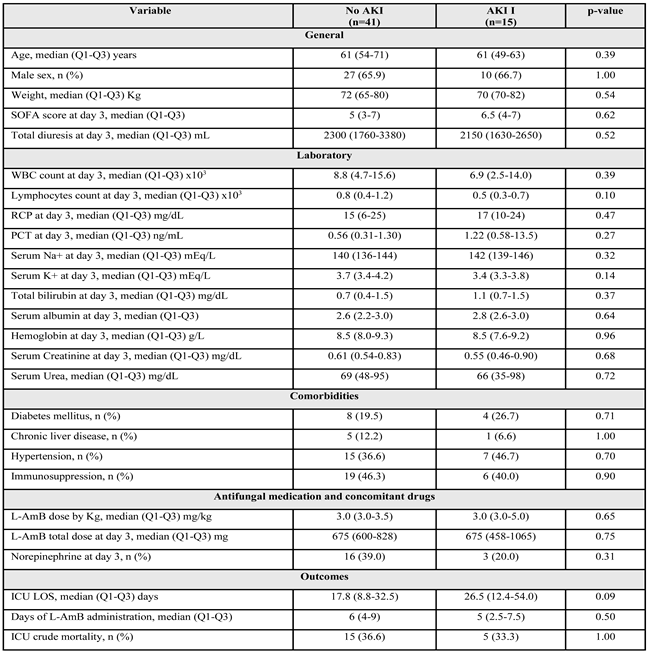
2.2. Development of Renal Dysfunction
2.2. Development of Renal Dysfunction
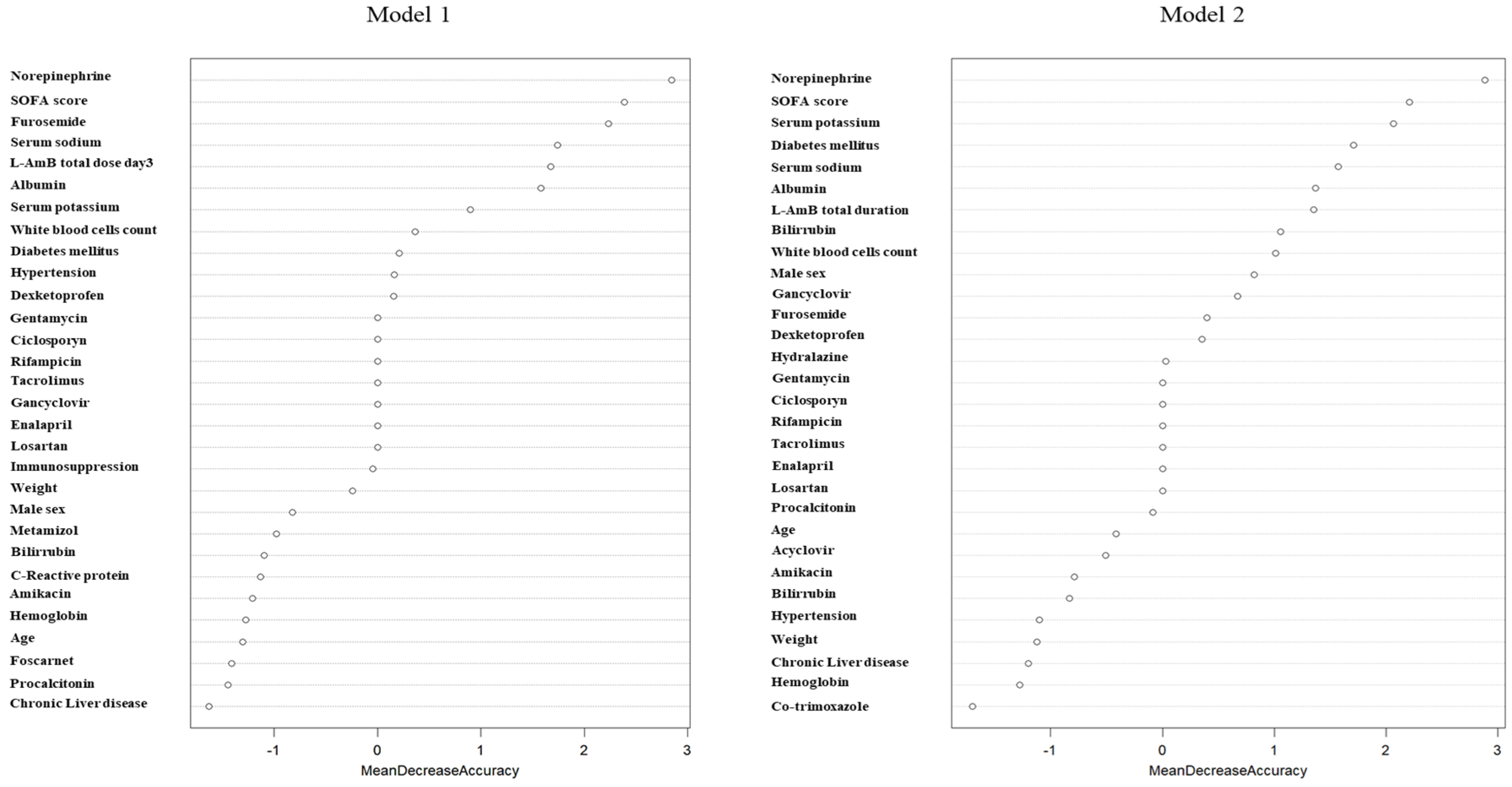
2.3. Factors Associated with AKI Development
2.4. Subgroups Analysis
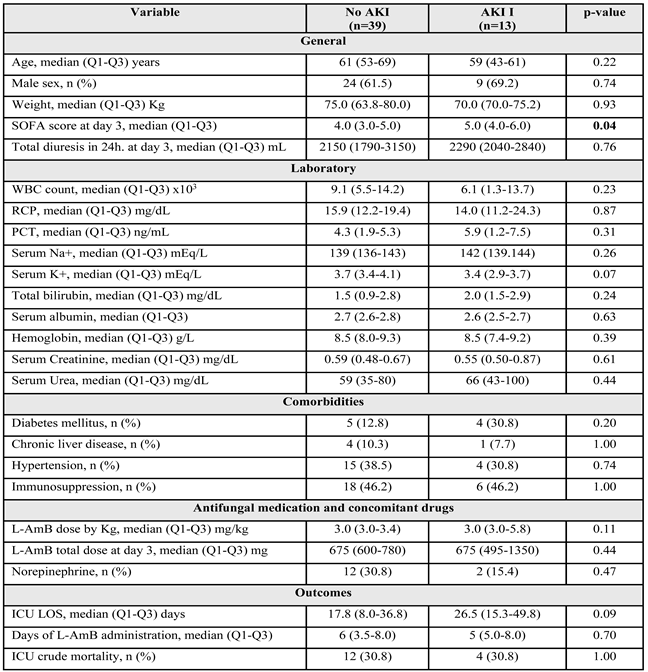
2.5. AKI Development in Patients with “Lower Risk” of AKI at Baseline
2.6. Factors Associated with AKI Development in Patients with Lower Risk of AKI
2.7. AKI Development in Patients with High Risk of AKI
3. Discussion
4. Materials and Methods
4.1. Study Design and Population
- Patients requiring continuous or intermittent renal replacement techniques (CRRT) at the time of beginning L-AmB.
- History of chronic kidney disease [6] with or without need for hemodialysis.
4.2. Subgroup Analysis
4.3. Variables
4.4. Definitions
Baseline Serum Creatinine Value
High Risk of Developing-AKI Subgroup Patients
Post-Treatment Renal Dysfunction Criteria
- AKIN grade I: defined as an absolute increase in serum creatinine value ≥ 0.3 mg/dL or an increase between 50% and 100% of the baseline value at 72h.
- AKIN grade II: defined as an increase in serum creatinine > 100% (and up to 200%) over baseline at 72h.
- AKIN grade III: defined as an increase in serum creatinine > 200% over baseline at 72h, or as an absolute increase in serum creatinine value ≥ 4.0 mg/dL (≥ 354 µmol/L) with an acute increase of at least 0.5 mg/dL (44 µmol/L) or in CRRT.
4.5. Ethical Considerations
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martinez, R. An Update on the Use of Antifungal Agents. J Bras Pneumol 2006, 32, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Klepser, M. The Value of Amphotericin B in the Treatment of Invasive Fungal Infections. J Crit Care 2011, 26, 225.e1–225e10. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Toledo, E.; Jiménez-Delgadillo, A.U.; Manzano-Gayosso, P. Antifúngicos Poliénicos. Mecanismo de Acción y Aplicaciones. Revista de la Facultad de Medicina 2020, 63, 7–17. [Google Scholar] [CrossRef]
- TJ Walsh; RW Finberg; C Arndt; J Hiemenz; C Schwartz; D Bodensteiner; P Pappas; N Seibel; RN Greenberg; S Dummer; et al. Liposomal Amphotericin B for Empirical Therapy in Patients with Persistent Fever and Neutropenia. N Engl J Med 1999, 764–771.
- Deray, G. Amphotericin B Nephrotoxicity. J Antimicrob Chemother 2002, 49, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Kellum, J.A.; Romagnani, P.; Ashuntantang, G.; Ronco, C.; Zarbock, A.; Anders, H.J. Acute Kidney Injury. Nat Rev Dis Primers 2021, 7. [Google Scholar] [CrossRef] [PubMed]
- KDIGO International Section 2: AKI Definition. Kidney Int Suppl (2011) 2012, 2, 19–36. [CrossRef] [PubMed]
- Rocha, P.N.; Kobayashi, C.D.; De Carvalho Almeida, L.; De Oliveira Dos Reis, C.; Santos, B.M.; Glesby, M.J. Incidence, Predictors, and Impact on Hospital Mortality of Amphotericin B Nephrotoxicity Defined Using Newer Acute Kidney Injury Diagnostic Criteria. Antimicrob Agents Chemother 2015, 59, 4759–4769. [Google Scholar] [CrossRef] [PubMed]
- Personett, H.A.; Kayhart, B.M.; Barreto, E.F.; Tosh, P.; Dierkhising, R.; Mara, K.; Leung, N. Renal Recovery Following Liposomal Amphotericin B-Induced Nephrotoxicity. Int J Nephrol 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Takazono, T.; Tashiro, M.; Ota, Y.; Obata, Y.; Wakamura, T.; Miyazaki, T.; Nishino, T.; Izumikawa, K. Factor Analysis of Acute Kidney Injury in Patients Administered Liposomal Amphotericin B in a Real-World Clinical Setting in Japan. Sci Rep 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Lerma, F.; Cruz Soriano, M.; Rodríguez, M.; Catalán, M.; María Llorente, A.; Vidart, N.; Garitacelaya, M.; Maraví, E.; Fernández, E.; Alvarado, F.; et al. Impact of Liposomal Amphotericin B on Renal Function in Critically Ill Patients with Renal Function Impairment. Revista española de Quimioterapia 2012, 25, 206–215. [Google Scholar] [PubMed]
- Ullmann, A.J.; Sanz, M.A.; Tramarin, A.; Barnes, R.A.; Wu, W.; Gerlach, B.A.; Krobot, K.J.; Gerth, W.C. Prospective Study of Amphotericin B Formulations in Immunocompromised Patients in 4 European Countries. Clinical infectious diseases 2006, 43, e29–e38. [Google Scholar] [CrossRef] [PubMed]
- Wingard, J.R.; White, M.H.; Anaissie, E.; Raffalli, J.; Goodman, J.; Arrieta, A. A Randomized, Double-Blind Comparative Trial Evaluating the Safety of Liposomal Amphotericin B versus Amphotericin B Lipid Complex in the Empirical Treatment of Febrile Neutropenia. Clinical Infectious Disease 2000, 31, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Cornely, O.A.; Maertens, J.; Bresnik, M.; Ebrahimi, R.; Ullmann, A.J.; Bouza, E.; Heussel, C.P.; Lorttiolary, O.; Rieger, C.; Boehrne, A.; et al. Liposomal Amphotericin B as Initial Therapy for Invasive Mold Infection: A Randomized Trial Comparing a High-Loading Dose Regimen with Standard Dosing (AmBiLoad Trial). Clinical Infectious Diseases 2007, 44, 1289–1297. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




