Submitted:
08 July 2024
Posted:
10 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
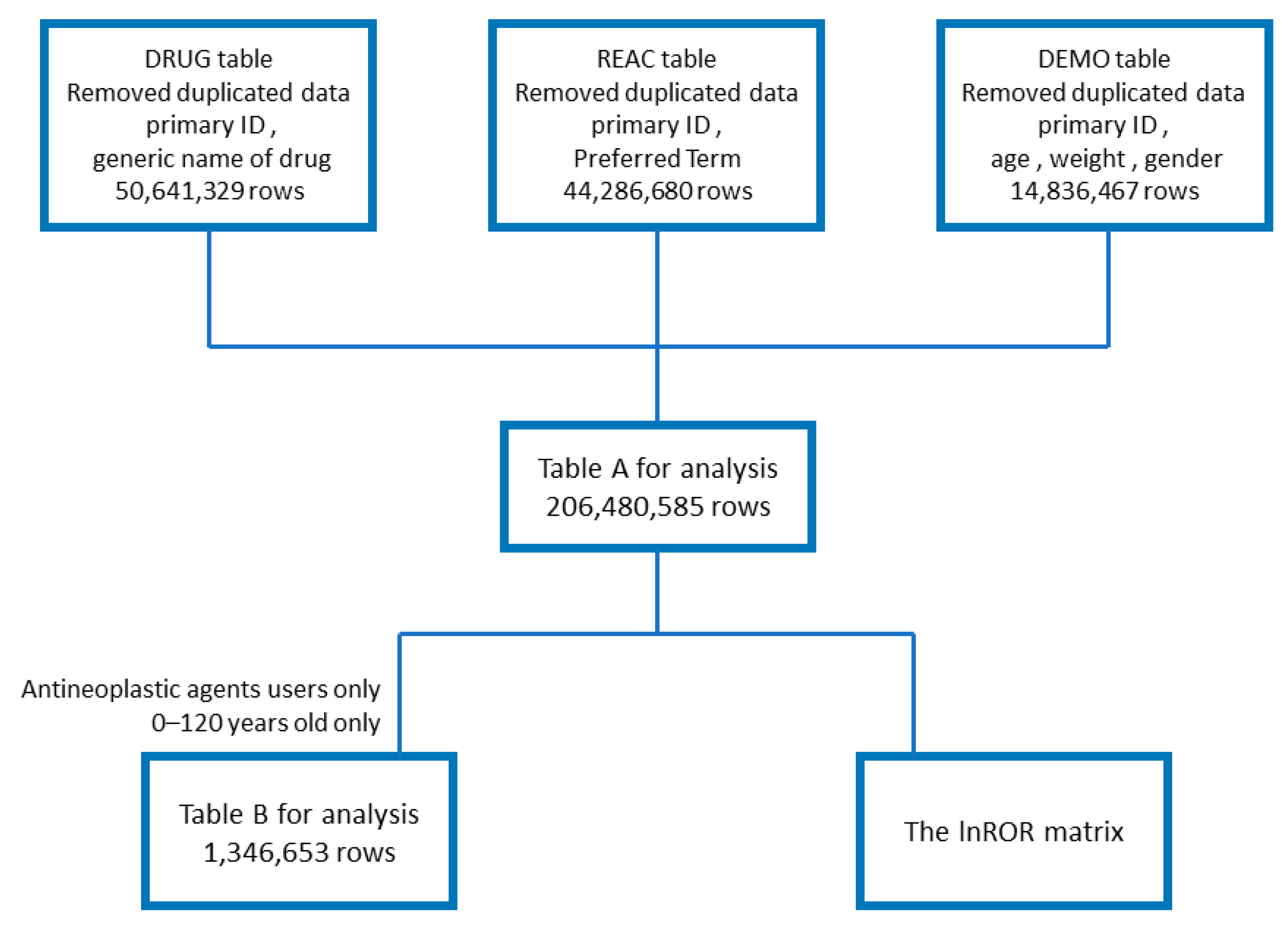
2.1. Creation of Data Tables
2.2. Number of Reports of Taste and Smell Disorders
2.3. Relationships of Patient Age and Gender with Taste and Smell Disorders Induced by Antineoplastic Agents
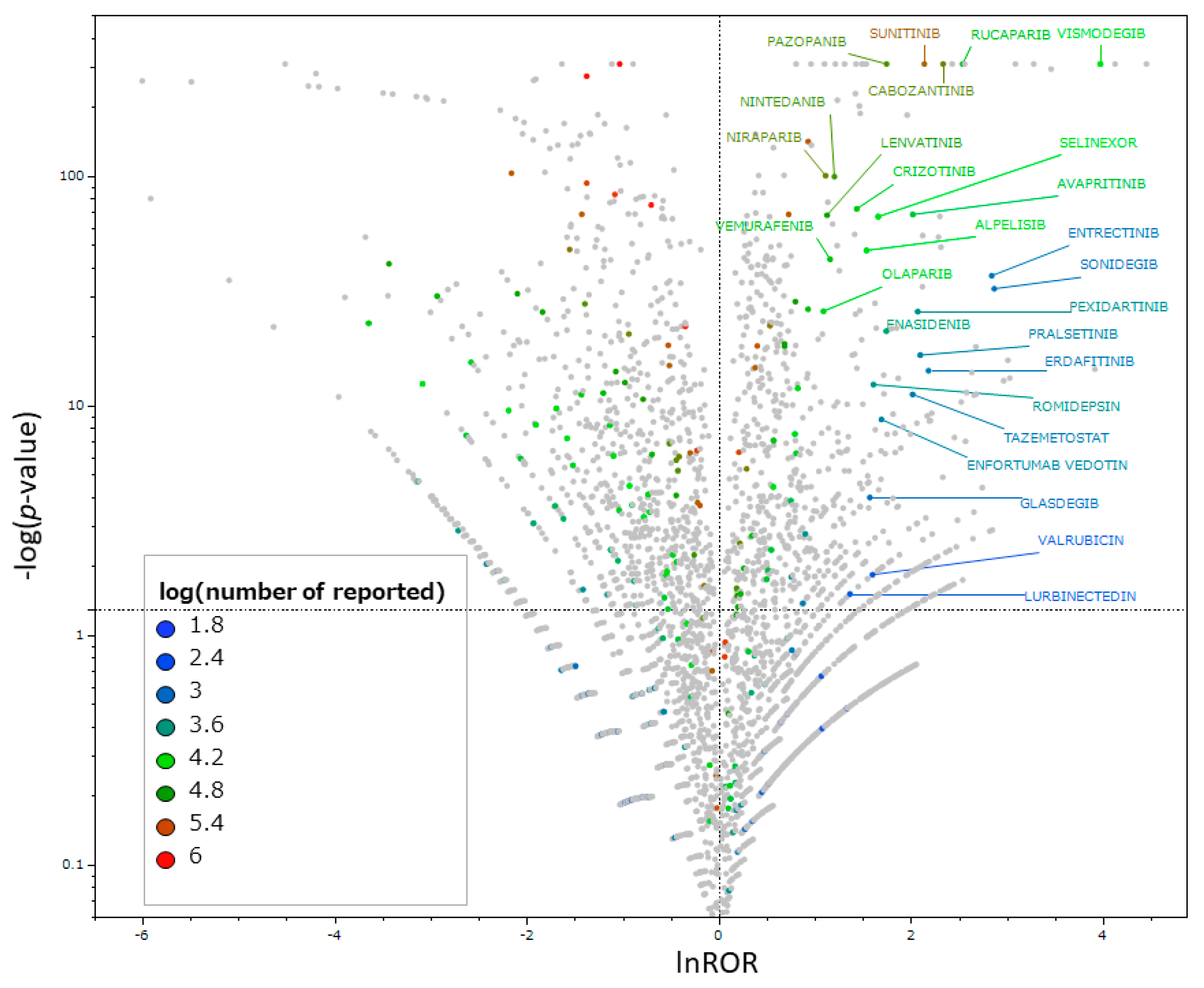
2.4. Identification of Antineoplastic Agents That Induce Taste and Smell Disorders
2.5. Relationships between the Class of Antineoplastic Agents and Taste and Smell Disorders
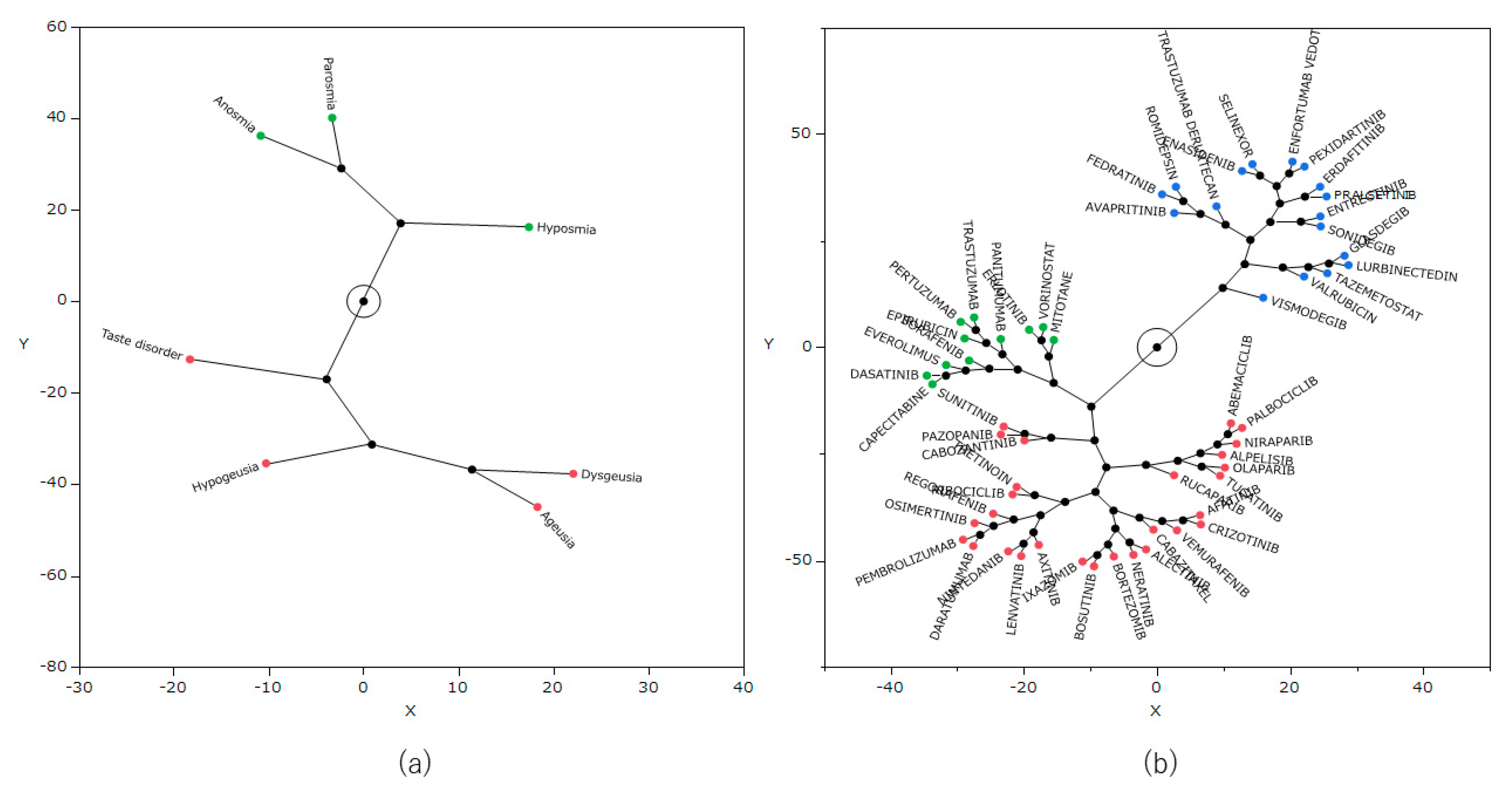
2.6. Hierarchical Cluster Analysis
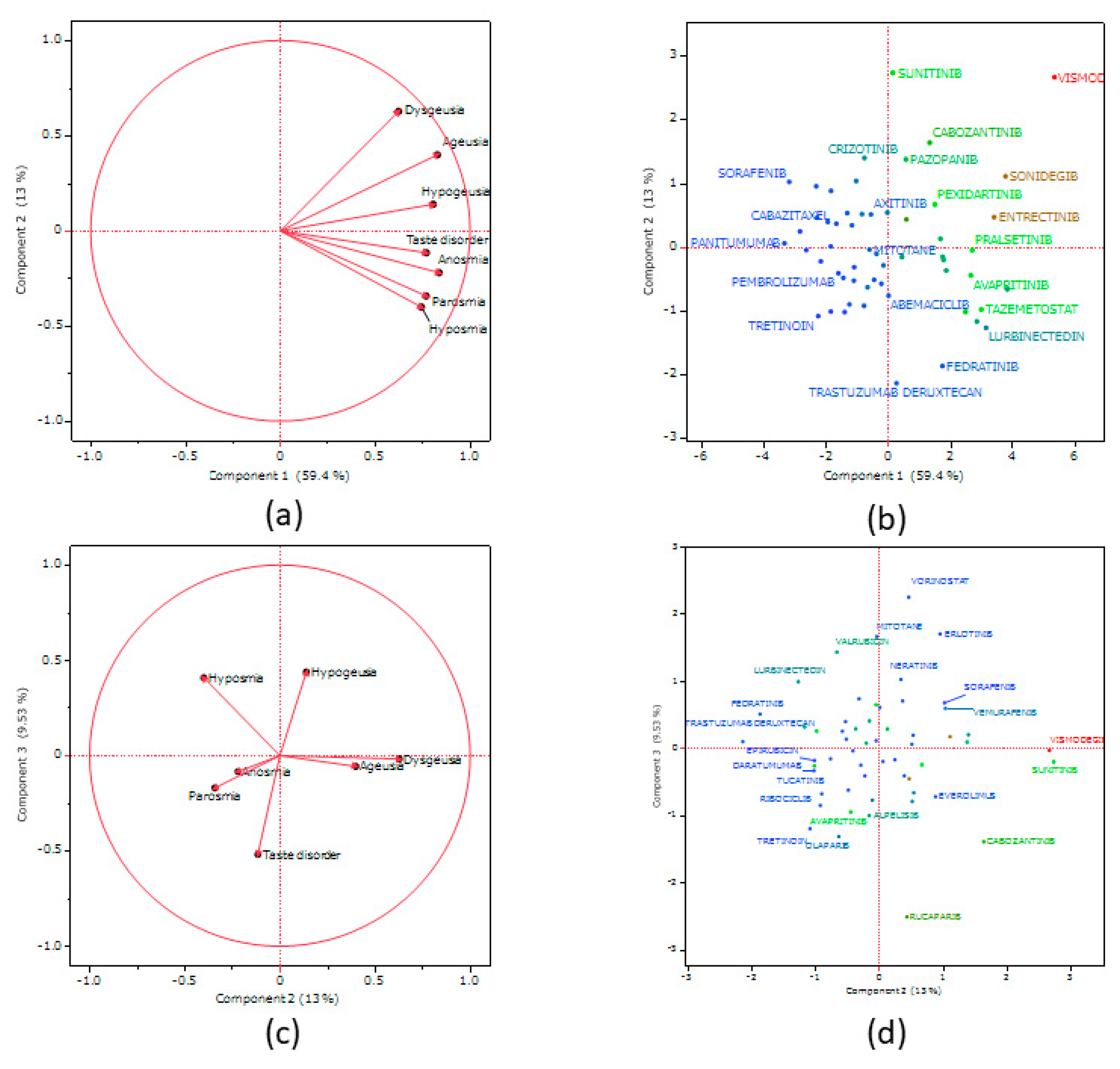
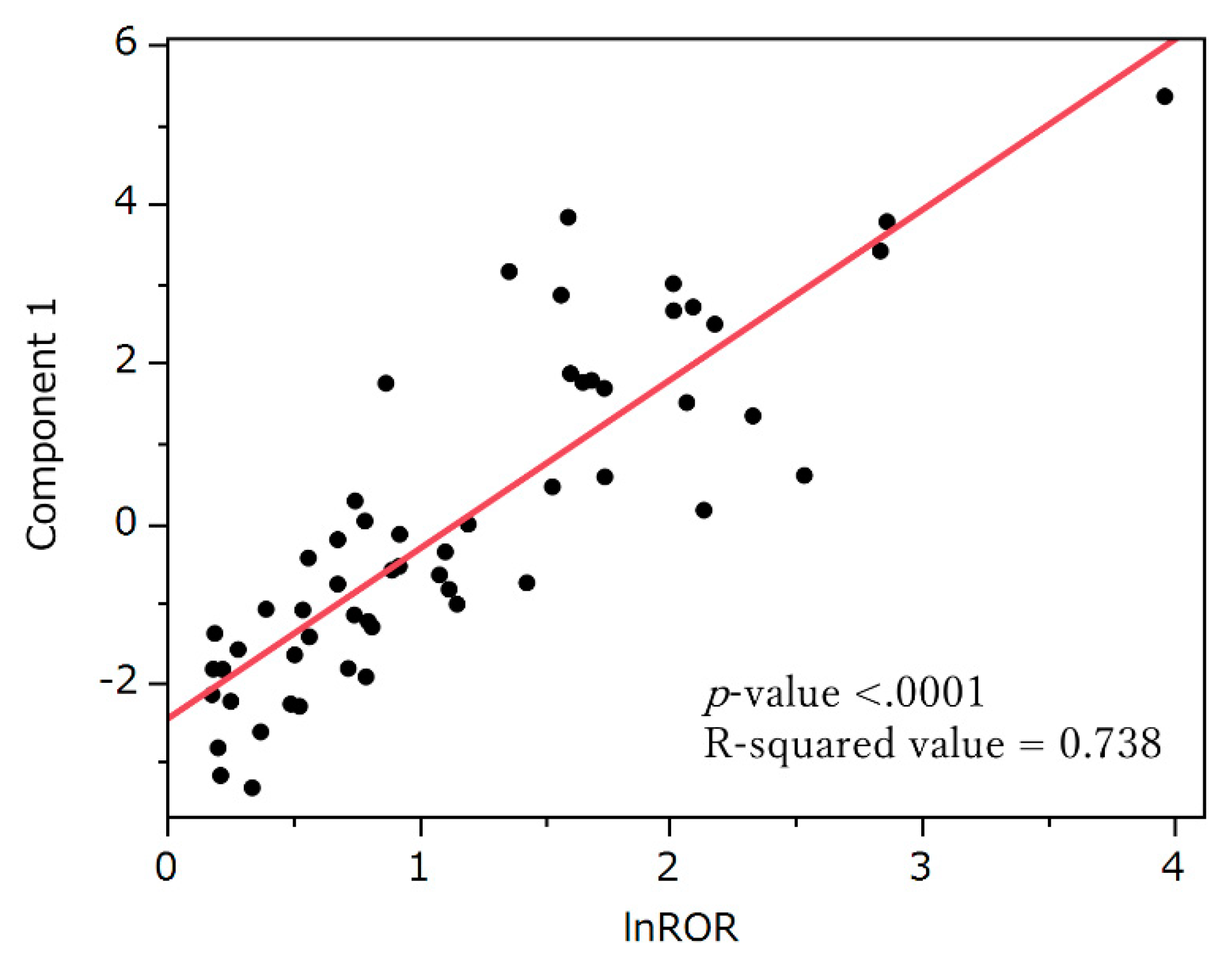
2.7. Principal Component Analysis
3. Discussion
3.1. Number of Reports of Taste and Smell Disorders
3.2. Characteristics of Patients with Taste and Smell Disorders and Antineoplastic Agents
3.3. Relationships of Antineoplastic Agents with Taste and Smell Disorders
3.4. Relationship between the Class of Antineoplastic Agents and Taste and Smell Disorders
3.5. Hierarchical Cluster Analysis
3.6. Principal Component Analysis
3.7. Limitations
4. Materials and Methods
4.1. FAERS Database
4.2. Adverse Event Terms and Drugs for Analysis
4.3. Number of Reports of Taste and Smell Disorders
4.4. Relationships of Taste and Smell Disorders with Age and Genger Among Patients Using Antineoplastic Agents
4.5. Relationships between Antineoplastic Agents and Taste and Smell Disorders
4.6. Relationships of the Class of Antineoplastic Agents with Taste and Smell Disorders
4.7. Creation of Data Tables for Principal Component and Cluster Analyses
4.8. Hierarchical Cluster Analysis
4.9. Principal Component Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cohen, J.; Wakefield, C.E.; Laing, D.G. Smell and Taste Disorders Resulting from Cancer and Chemotherapy. Curr. Pharm. Des. 2016, 22, 2253–2263. [CrossRef]
- Hutton, J.L.; Baracos, V.E.; Wismer, W.V. Chemosensory dysfunction is a primary factor in the evolution of declining nutritional status and quality of life in patients with advanced cancer. J. Pain Symptom Manag. 2007, 33, 156–165. [CrossRef]
- Wickham, R.S.; Rehwaldt, M.; Kefer, C.; Shott, S.; Abbas, K.; Glynn-Tucker, E.; Potter, C.; Blendowski, C. Taste changes experienced by patients receiving chemotherapy. Oncol Nurs Forum. 1999, 26, 697–706.
- Comeau, T.B.; Epstein, J.B.; Migas, C. Taste and smell dysfunction in patients receiving chemotherapy: a review of current knowledge. Support Care Cancer. 2001, 9, 575–580. [CrossRef]
- Brisbois, T.D.; de Kock, I.H.; Watanabe, S.M.; Baracos, V.E.; Wismer, W.V. Characterization of chemosensory alterations in advanced cancer reveals specific chemosensory phenotypes impacting dietary intake and quality of life. J Pain Symptom Manage. 2011, 41, 673–683. [CrossRef]
- Tuccori, M.; Lapi, F.; Testi, A.; Ruggiero, E.; Moretti, U.; Vannacci, A.; Bonaiuti, R.; Antonioli, L.; Fornai, M.; Giustarini, G.; Scollo, C.; Corona, T.; Ferrazin, F.; Sottosanti, L.; Blandizzi, C. Drug-induced taste and smell alterations: a case/non-case evaluation of an italian database of spontaneous adverse drug reaction reporting. Drug Saf. 2011, 34, 849–859. [CrossRef]
- Deems, D.A.; Doty, R.L.; Settle, R.G.; Moore-Gillon, V.; Shaman, P.; Mester, A.F.; Kimmelman, C.P.; Brightman, V.J.; Snow, J.B. Jr. Smell and taste disorders, a study of 750 patients from the University of Pennsylvania Smell and Taste Center. Arch Otolaryngol Head Neck Surg. 1991, 117, 519–528. [CrossRef]
- Goodspeed, R.B.; Gent, J.F.; Catalanotto, F.A. Chemosensory dysfunction. Clinical evaluation results from a taste and smell clinic. Postgrad Med. 1987, 81, 251–257, 260. [CrossRef]
- Buttiron Webber, T.; Briata, I.M.; DeCensi, A.; Cevasco, I.; Paleari, L. Taste and Smell Disorders in Cancer Treatment: Results from an Integrative Rapid Systematic Review. Int J Mol Sci. 2023, 24, 2538. [CrossRef]
- Malta, C.E.N.; de Lima Martins, J.O.; Carlos, A.C.A.M.; Freitas, M.O.; Magalhães, I.A.; de Vasconcelos, H.C.A.; de Lima Silva-Fernandes, I.J.; de Barros Silva, P.G. Risk factors for dysgeusia during chemotherapy for solid tumors: a retrospective cross-sectional study. Support Care Cancer. 2022, 30, 313–325. [CrossRef]
- Amézaga, J.; Alfaro, B.; Ríos, Y.; Larraioz, A.; Ugartemendia, G.; Urruticoechea, A.; Tueros, I. Assessing taste and smell alterations in cancer patients undergoing chemotherapy according to treatment. Support Care Cancer. 2018, 26, 4077–4086. [CrossRef]
- Zabernigg, A.; Gamper, E.M.; Giesinger, J.M.; Rumpold, G.; Kemmler, G.; Gattringer, K.; Sperner-Unterweger, B.; Holzner, B. Taste alterations in cancer patients receiving chemotherapy: a neglected side effect? Oncologist. 2010, 15, 913–920. [CrossRef]
- Eravcı, F.C.; Uçar, G.; Özcan, K.M.; Çolak, M.; Ergün, Y.; Açıkgöz, Y.; Ikincioğulları, A.; Uncu, D.; Dere, H.H. The effect of chemotherapy on olfactory function and mucociliary clearance. Support Care Cancer. 2021, 29, 1635–1641. [CrossRef]
- Pugnaloni, S.; Vignini, A.; Borroni, F.; Sabbatinelli, J.; Alia, S.; Fabri, M.; Taus, M.; Mazzanti, L.; Berardi, R. Modifications of taste sensitivity in cancer patients: a method for the evaluations of dysgeusia. Support Care Cancer. 2020, 28, 1173–1181. [CrossRef]
- Riga, M.; Chelis, L.; Papazi, T.; Danielides, V.; Katotomichelakis, M.; Kakolyris, S. Hyposmia: an underestimated and frequent adverse effect of chemotherapy. Support Care Cancer. 2015, 23, 3053–3058. [CrossRef]
- Ikeda, M.; Ikui, A.; Komiyama, A.; Kobayashi, D.; Tanaka, M. Causative factors of taste disorders in the elderly, and therapeutic effects of zinc. J Laryngol Otol. 2008, 122, 155–160. [CrossRef]
- Ottaviano, G.; Savietto, E.; Scarpa, B.; Bertocco, A.; Maculan, P.; Sergi, G.; Martini, A.; Manzato, E.; Marioni, G. Influence of number of drugs on olfaction in the elderly. Rhinology. 2018, 56, 351–357. [CrossRef]
- Search for Ethical Drug Information on the Website of the Pharmaceuticals and Medical Devices Agency (PMDA). Available online: https://www.pmda.go.jp/PmdaSearch/iyakuSearch/ (accessed on 26 June 2024).
- van der Werf, A.; Rovithi, M.; Langius, J.A.E.; de van der Schueren, M.A.E.; Verheul, H.M.W. Insight in taste alterations during treatment with protein kinase inhibitors. Eur J Cancer. 2017, 86, 125–134. [CrossRef]
- Chen, L.; Xu, P.; Xiao, Q.; Chen, L.; Li, S.; Jian, J.M.; Zhong, Y.B. Sunitinib malate inhibits intestinal tumor development in male ApcMin/+ mice by down-regulating inflammation-related factors with suppressing β-cateinin/c-Myc pathway and re-balancing Bcl-6 and Caspase-3. Int Immunopharmacol. 2021, 90, 107128. [CrossRef]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014, 15, 178–196. [CrossRef]
- Zhang, W.; Zhang, H.; Wang, N.; Zhao, C.; Zhang, H.; Deng, F.; Wu, N.; He, Y.; Chen, X.; Zhang, J.; Wen, S.; Liao, Z.; Zhang, Q.; Zhang, Z.; Liu, W.; Yan, Z.; Luu, H.H.; Haydon, R.C.; Zhou, L.; He, T.C. Modulation of β-catenin signaling by the inhibitors of MAP kinase, tyrosine kinase, and PI3-kinase pathways. Int J Med Sci. 2013, 10, 1888–1898. [CrossRef]
- Barlow, L.A. Progress and renewal in gustation: new insights into taste bud development. Development. 2015, 142, 3620–3629. [CrossRef]
- Miura, H.; Kusakabe, Y.; Sugiyama, C.; Kawamatsu, M.; Ninomiya, Y.; Motoyama, J.; Hino, A. Shh and Ptc are associated with taste bud maintenance in the adult mouse. Mech Dev. 2001, 106, 143–145. [CrossRef]
- Hummel, T.; Whitcroft, K.L.; Rueter, G.; Haehner, A. Intranasal vitamin A is beneficial in post-infectious olfactory loss. Eur Arch Otorhinolaryngol. 2017, 274, 2819–2825. [CrossRef]
- Everitt, B.S.; Landau, S.; Leese, M.; Stahl, D. Cluster Analysis, 5th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011; ISBN 978-0-470-74991-3.
- Jolliffe, I.T.; Cadima, J. Principal component analysis: A review and recent developments. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150202. [CrossRef]
- Maeda, R. JADER from pharmacovigilance point of view. Jpn. J. Pharmacoepidemiol. Yakuzai Ekigaku 2014, 19, 51–56.
- Noguchi, Y.; Tachi, T.; Teramachi, H. Detection algorithms and attentive points of safety signal using spontaneous reporting systems as a clinical data source. Brief. Bioinform. 2021, 22, bbab347. [CrossRef]
- Pariente, A.; Gregoire, F.; Fourrier-Reglat, A.; Haramburu, F.; Moore, N. Impact of safety alerts on measures of disproportionality in spontaneous reporting databases: the notoriety bias. Drug Saf. 2007, 30, 891–898. [CrossRef]
- Hartnell, N.R.; Wilson, J.P. Replication of the Weber effect using postmarketing adverse event reports voluntarily submitted to the United States Food and Drug Administration. Pharmacotherapy. 2004, 24, 743–749. [CrossRef]
- Wang, H.W.; Hochberg, A.M.; Pearson, R.K.; Hauben, M. An experimental investigation of masking in the US FDA adverse event reporting system database. Drug Saf. 2010, 33, 1117–1133. [CrossRef]
- Pariente, A.; Avillach, P.; Salvo, F.; Thiessard, F.; Miremont-Salamé, G.; Fourrier-Reglat, A.; Haramburu, F.; Bégaud, B.; Moore, N.; Association Française des Centres Régionaux de Pharmacovigilance (CRPV). Effect of competition bias in safety signal generation: analysis of a research database of spontaneous reports in France. Drug Saf. 2012, 35, 855–864. [CrossRef]
- Poleksic, A.; Xie, L. Database of adverse events associated with drugs and drug combinations. Sci Rep. 2019, 9, 20025. [CrossRef]
- FDA Adverse Event Reporting System (FAERS). Available online: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-adverse-event-reporting-system-faers (accessed on 26 June, 2024).
- Lumini, A.; Nanni, L. Convolutional Neural Networks for ATC Classification. Curr Pharm Des. 2018, 24, 4007–4012. [CrossRef]
- MedDRA Japanese Maintenance Organization. Available online: https://www.meddra.org/ (accessed on 26 June, 2024).
- Watanabe, H.; Matsushita, Y.; Watanabe, A.; Maeda, T.; Nukui, K.; Ogawa, Y.; Sawa, J.; Maeda, H. Early detection of important safety information. Recent methods for signal detection. Jpn. J. Biomet. 2004, 25, 37–60.
- Ohyama, K.; Sugiura, M. Evaluation of the association between topical prostaglandin F2α analogs and asthma using the JADER database: Comparison with β-blockers. Yakugaku Zasshi. 2018, 138, 559–564. [CrossRef]
- Greenland, S.; Schwartzbaum, J.A.; Finkle, W.D. Problems due to small samples and sparse data in conditional logistic regression analysis. Am. J. Epidemiol. 2000, 151, 531–539. [CrossRef]
- Gravel, C.A.; Douros, A. Considerations on the use of different comparators in pharmacovigilance: A methodological review. Br. J. Clin. Pharmacol. 2023, 89, 2671–2676. [CrossRef]
- Hosoya, R.; Uesawa, Y.; Ishii-Nozawa, R.; Kagaya, H. Analysis of factors associated with hiccups based on the Japanese Adverse Drug Event Report database. PLoS One. 2017, 12, e0172057. [CrossRef]
- Kawabe, A.; Uesawa, Y. Analysis of Corticosteroid-Induced Glaucoma Using the Japanese Adverse Drug Event Reporting Database. Pharmaceuticals. 2023, 16, 948. [CrossRef]
- Okunaka, M.; Kano, D.; Matsui, R.; Kawasaki, T.; Uesawa, Y. Comprehensive Analysis of Chemotherapeutic Agents That Induce Infectious Neutropenia. Pharmaceuticals. 2021, 14, 681. [CrossRef]
- Kan, Y.; Nagai, J.; Uesawa, Y. Evaluation of antibiotic-induced taste and smell disorders using the FDA adverse event reporting system database. Sci Rep. 2021, 11, 9625. [CrossRef]
- Nakao, Y.; Asada, M.; Uesawa, Y. Comprehensive Study of Drug-Induced Pruritus Based on Adverse Drug Reaction Report Database. Pharmaceuticals. 2023, 16, 1500. [CrossRef]
- Chen, J.J.; Wang, S.J.; Tsai, C.A.; Lin, C.J. Selection of differentially expressed genes in microarray data analysis. Pharmacogenomics J. 2007, 7, 212–220. Epub 2006 Aug 29. [CrossRef]
- Van Puijenbroek, E.P.; Bate, A.; Leufkens, H.G.M.; Lindquist, M.; Orre, R.; Egberts, A.C. A Comparison of measures of disproportionality for signal detection in spontaneous reporting systems for adverse drug reactions. Pharmacoepidemiol. Drug. Saf. 2002, 11, 3–10. [CrossRef]

| Number of Reports | Proportion (%) | |
|---|---|---|
| Taste or smell disorders | 19,342 | 100 |
| Taste disorders | 18.764 | 97.0 |
| Taste disorders only | 17,872 | 92.4 |
| Smell disorders | 1470 | 7.6 |
| Smell disorders only | 578 | 3.0 |
| Both taste and smell disorders | 892 | 4.6 |
| Gender | Taste and smell disorders | Non-taste and smell disorders | p (Fisher’s exact test) | ROR | 95% CI | |
| Male | 5391 | 565,885 | 0.569 | 0.990 | 0.955–1.025 | |
| Female | 7164 | 744,229 | ||||
| Age | Taste and smell disorders | Non-taste and smell disorders | p (Fisher’s exact test) | ROR | 95% CI | |
| ≥70 | 4561 | 387,134 | <0.0001 | 1.381 | 1.332–1.432 | |
| <70 | 8077 | 946,863 | ||||
| Medicinal Classification by the ATC Classification | Antineoplastic Agents likely to Induce Taste and Smell Disorders* | Number of all Drugs by Drug Class | Antineoplastic Agents likely to Induce Taste and Smell Disorders/Number of all Drugs by Drug Class (%) |
|---|---|---|---|
| Alkylating agents (L01A) | 1 | 26 | 3.85 |
| Antimetabolites (L01B) | 1 | 19 | 5.26 |
| Plant alkaloids and other natural products (L01C) | 1 | 17 | 5.88 |
| Cytotoxic antibiotics and related substances (L01D) | 2 | 16 | 12.5 |
| Protein kinase inhibitors (L01E) | 29 | 73 | 39.73 |
| Monoclonal antibodies and antibody drug conjugates (L01F) | 7 | 45 | 15.56 |
| Other antineoplastic agents (L01X) | 16 | 65 | 24.62 |
| Medicinal Classification by ATC Classification | a | b | c | d | p | ROR | 95% CII |
|---|---|---|---|---|---|---|---|
| Alkylating agents | 841 | 1,006,708 | 390,456 | 205,082,580 | <0.0001 | 0.439 | 0.410–0.470 |
| Antimetabolites | 3006 | 3,197,865 | 388,291 | 202,891,423 | <0.0001 | 0.491 | 0.474–0.509 |
| Plant alkaloids and other natural products | 1811 | 1,341,772 | 389,486 | 204,747,516 | <0.0001 | 0.710 | 0.677–0.743 |
| Cytotoxic antibiotics and related substances | 428 | 574,924 | 390,69 | 205,514,364 | <0.0001 | 0.391 | 0.356–0.430 |
| Protein kinase inhibitors | 11801 | 2,488,034 | 379,496 | 203,601,254 | <0.0001 | 2.545 | 2.498–2.592 |
| Monoclonal antibodies and antibody drug conjugates | 2989 | 2,153,247 | 388,308 | 203,936,041 | <0.0001 | 0.729 | 0.703–0.756 |
| Other antineoplastic agents | 6575 | 2,2190,02 | 384,722 | 203,870,286 | <0.0001 | 1.570 | 1.532–1.609 |
| Cluster 1 | Cluster 2 | Cluster 3 |
|---|---|---|
| Avapritinib | Capecitabine | Abemaciclib |
| Enasidenib | Dasatinib | Afatinib |
| Enfortumab vedotin | Epirubicin | Alectinib |
| Entrectinib | Erlotinib | Alpelisib |
| Erdafitinib | Everolimus | Axitinib |
| Fedratinib | Mitotane | Bortezomib |
| Glasdegib | Panitumumab | Bosutinib |
| Lurbinectedin | Pertuzumab | Cabazitaxel |
| Pexidartinib | Sorafenib | Cabozantinib |
| Pralsetinib | Trastuzumab | Crizotinib |
| Romidepsin | Vorinostat | Daratumumab |
| Selinexor | Ixazomib | |
| Sonidegib | Lenvatinib | |
| Tazemetostat | Neratinib | |
| Trastuzumab deruxtecan | Nintedanib | |
| Valrubicin | Niraparib | |
| Vismodegib | Olaparib | |
| Osimertinib | ||
| Palbociclib | ||
| Pazopanib | ||
| Pembrolizumab | ||
| Regorafenib | ||
| Ribociclib | ||
| Rucaparib | ||
| Sunitinib | ||
| Tretinoin | ||
| Tucatinib | ||
| Vemurafenib |
| PT_NAME* | PT_CODE | Number of Records |
|---|---|---|
| Dysgeusia | 10013911 | 220,220 |
| Ageusia | 10001480 | 70,503 |
| Taste disorder | 10082490 | 34,849 |
| Anosmia | 10002653 | 30,875 |
| Parosmia | 10034018 | 22,325 |
| Hypogeusia | 10020989 | 6926 |
| Hyposmia | 10050515 | 3547 |
| Hallucination, olfactory | 10019072 | 1371 |
| Olfactory nerve disorder | 10056388 | 320 |
| Hypergeusia | 10069147 | 184 |
| Hallucination, gustatory | 10019071 | 157 |
| Gustometry abnormal | 10064480 | 11 |
| Olfactory test abnormal | 10062927 | 9 |
| Olfactory dysfunction | 10086567 | 0 |
| Taste and Smell Disorders | Non-Taste and Smell Disorders | |
|---|---|---|
| Reports with the suspected drug | a | b |
| All other reports | c | d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




