Submitted:
04 July 2024
Posted:
04 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Isolation and culture of primary human fibroblasts
2.2. Isolation and culture of mouse microglia
2.3. Thioflavin S staining
2.4. Immunofluorescence analysis
2.5. Quantitative real-time PCR (Q-PCR)
2.7. Statistical Analysis
3. Results
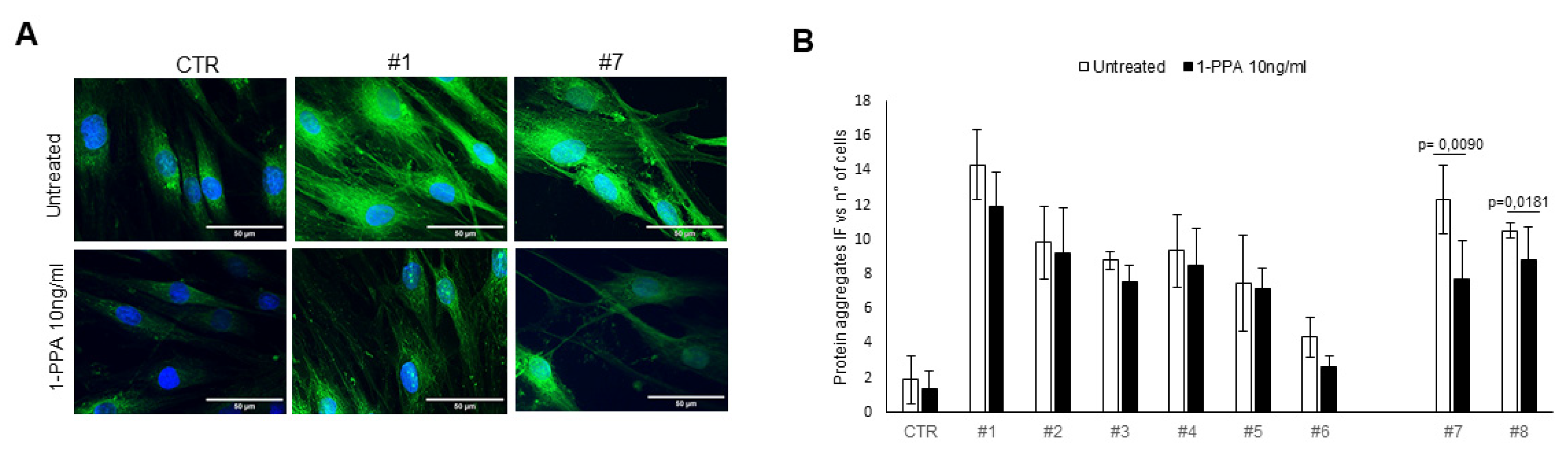
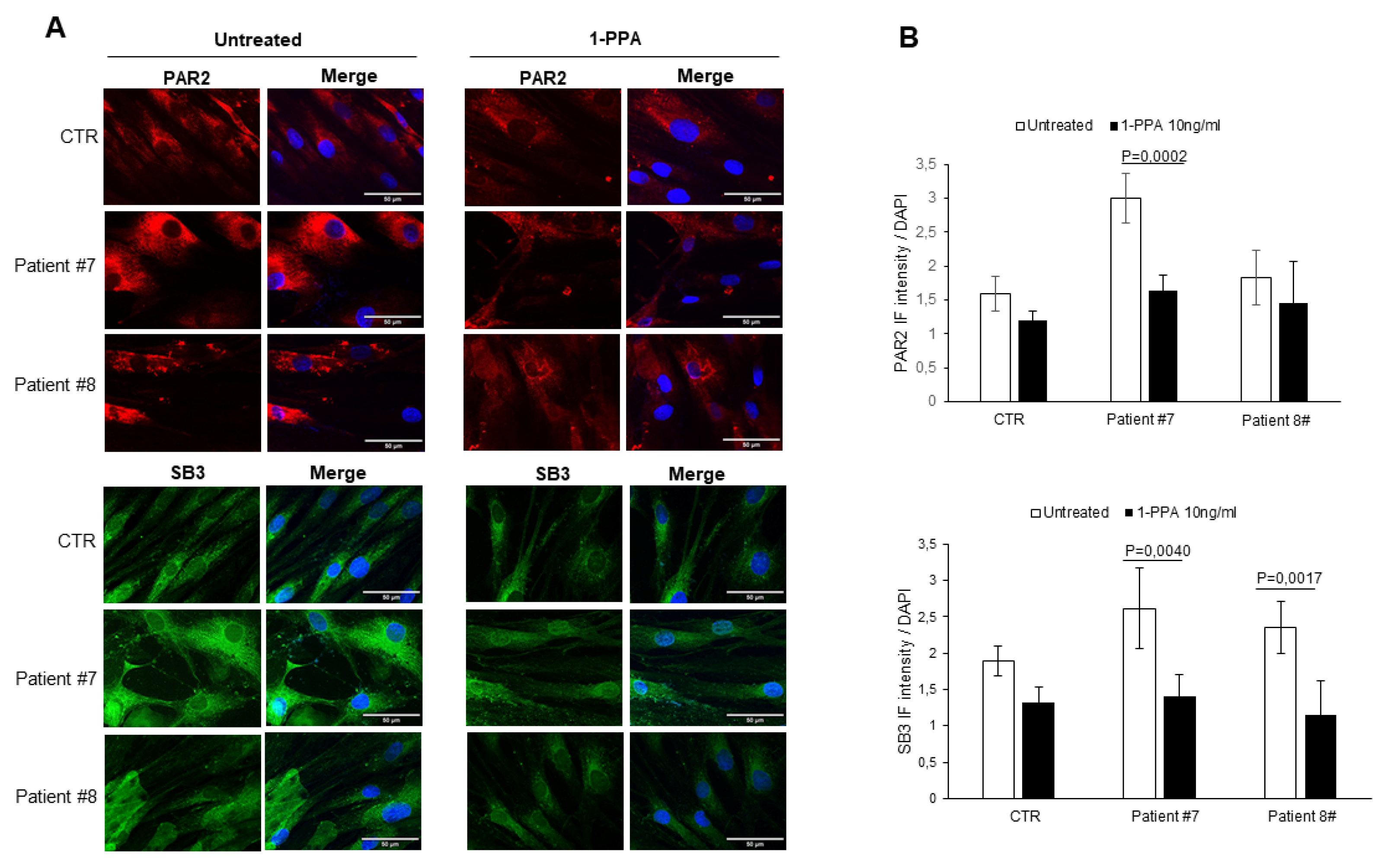
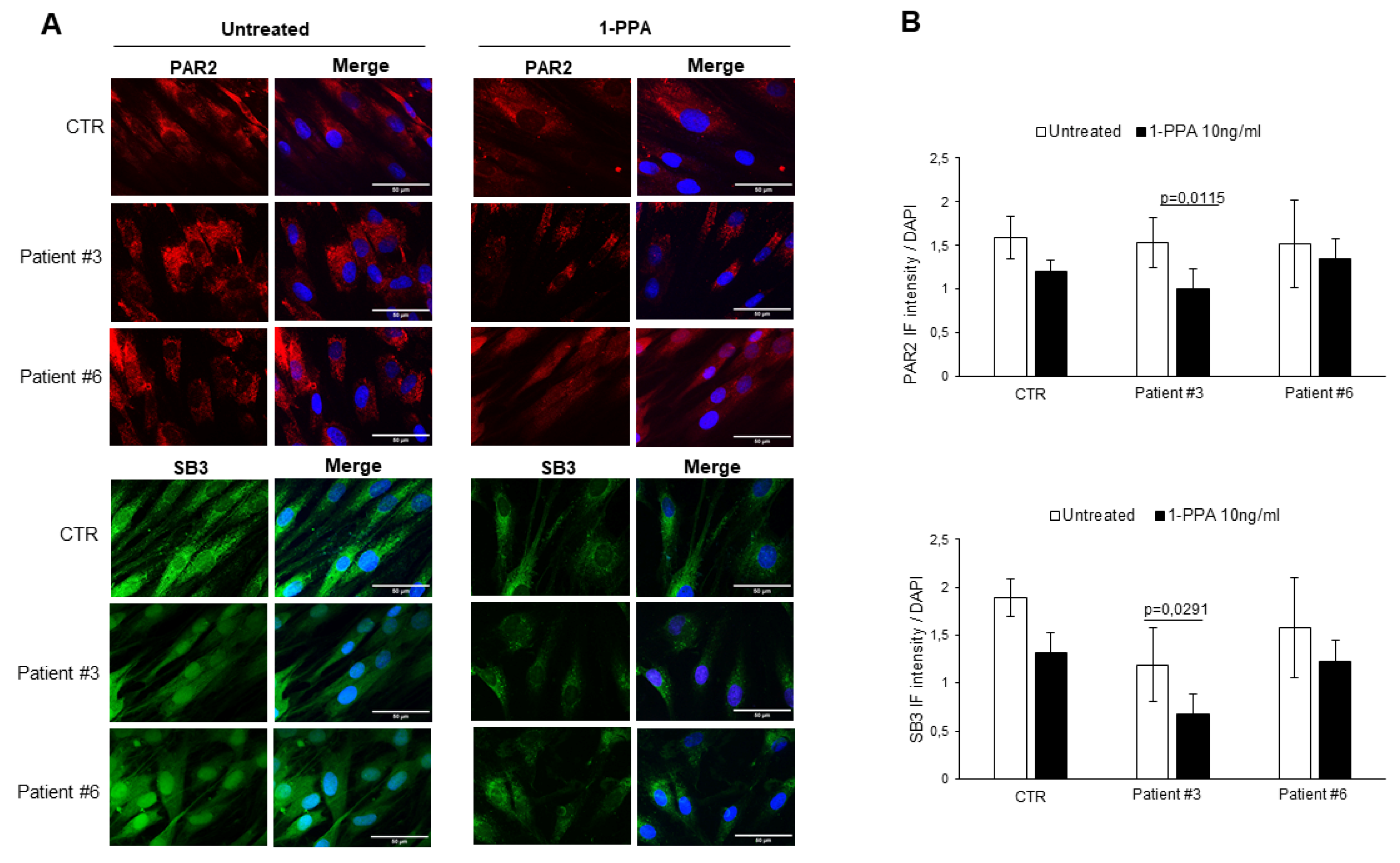
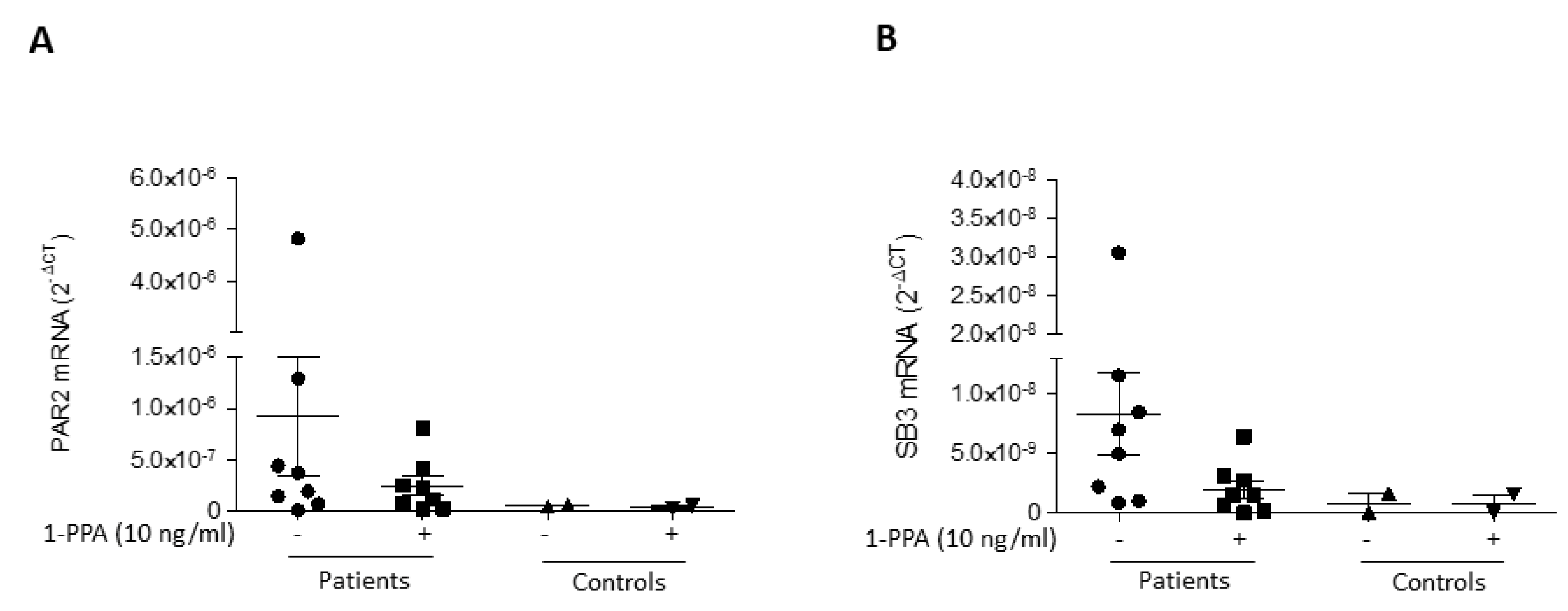
3.1. Human primary fibroblasts
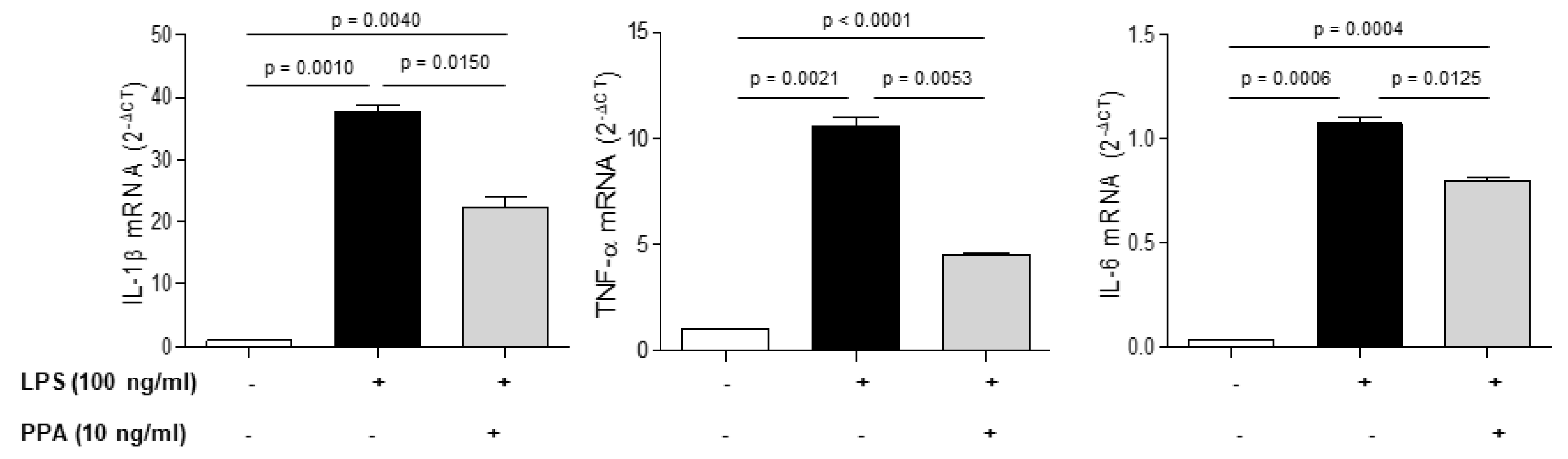
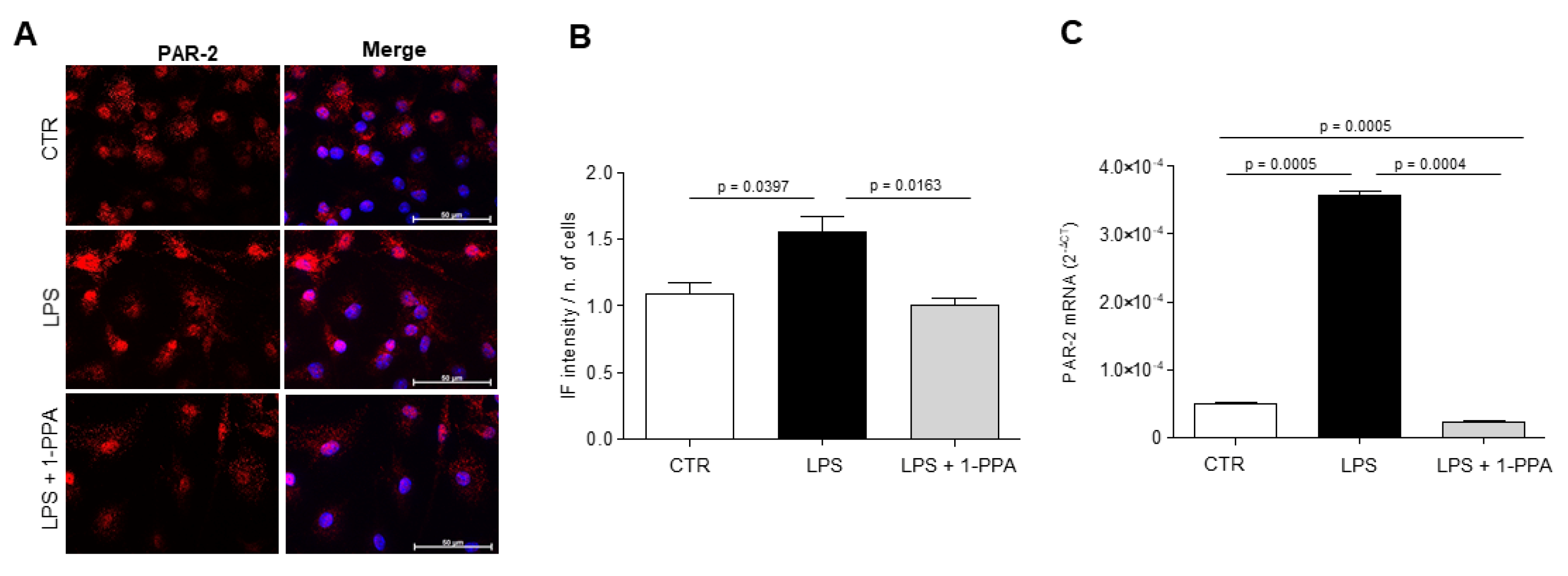
3.2. Primary mouse microglia
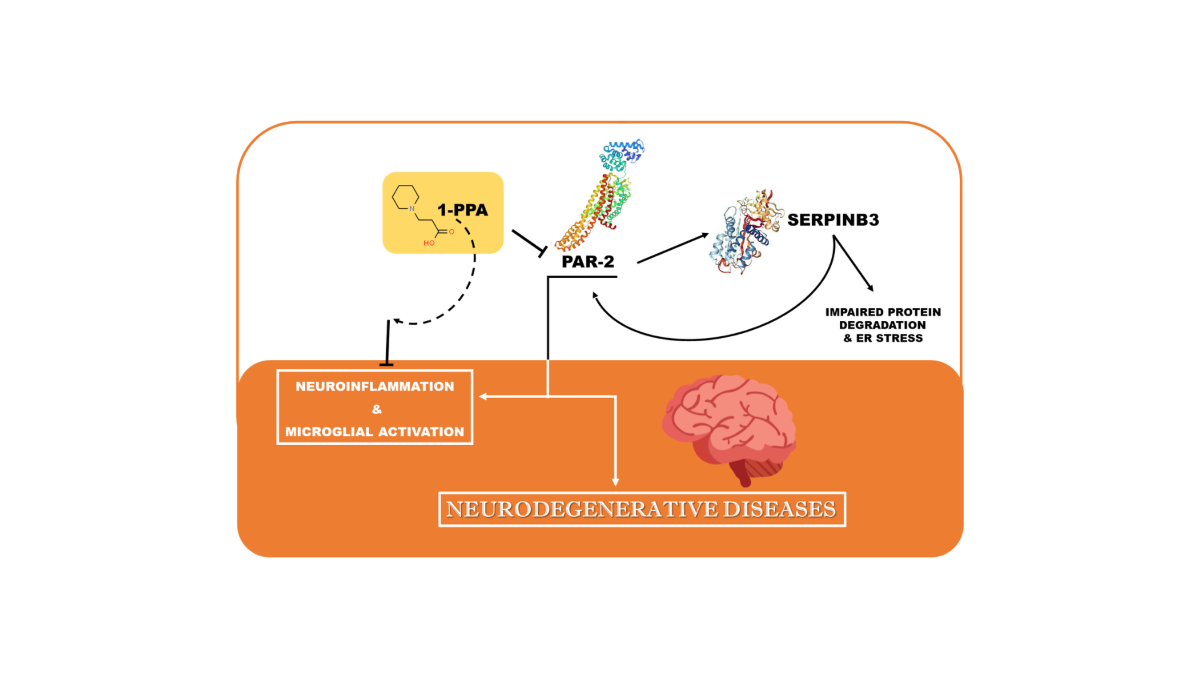
4. Discussion
5. Conclusion
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- S. L. Kowal, T. M. Dall, R. Chakrabarti, M. V. Storm, and A. Jain. The current and projected economic burden of Parkinson’s disease in the United States», Mov Disord, 2013, vol. 28 (3), pp. 311–318. [CrossRef]
- P. Liua, L. Sunb, X.L. Zhaoc, P. Zhangd, X-M Zhaoa, J. Zhang. PAR2-mediated epigenetic upregulation of α-synuclein contributes to the pathogenesis of Parkinson's disease. Brain Res 2014, vol. 1565, pp. 82-89. [CrossRef]
- Niraula, J. F. Sheridan, e J. P. Godbout. Microglia Priming with Aging and Stress. Neuropsychopharmacology, 2017, vol. 42 (1), pp. 318–333. [CrossRef]
- N. Zilka et al., Who fans the flames of Alzheimer’s disease brains? Misfolded tau on the crossroad of neurodegenerative and inflammatory pathways. J Neuroinflammation 2012, vol. 9 (1), p. 47. [CrossRef]
- H. Kettenmann, U.-K. Hanisch, M. Noda, and A. Verkhratsky, Physiology of Microglia. Physiol Rev, 2011, vol. 91 (2), pp. 461–553. [CrossRef]
- M.J. Hurley, P. F. Durrenberger, S.M. Gentleman, A. F. Walls, D. T. Dexter. Altered Expression of Brain Proteinase-Activated Receptor-2, Trypsin-2 and Serpin Proteinase Inhibitors in Parkinson's Disease. J Mol Neurosci. 2015, vol 57(1), pp48-62. [CrossRef]
- V. S. Ossovskaya e N. W. Bunnett. Protease-Activated Receptors: Contribution to Physiology and Disease, Physiol Rev 2004, vol. 84 (2), pp. 579–621. [CrossRef]
- S. R. Macfarlane, M. J. Seatter, T. Kanke, G. D. Hunter, e R. Plevin. Proteinase-activated receptors. Pharmacol Rev. 2001, vol. 53 (2), pp. 245–282.
- G. S. Cottrell, S. Amadesi, F. Schmidlin, N. Bunnett. Protease-activated receptor 2: activation, signalling and function. Biochem. Soc. Transact. 2003, vol. 31 (6), pp. 1191–1197. [CrossRef]
- M. Steinhoff, N. Vergnolle, S. H. Young, M. Tognetto, S. Amadesi, H. S. Ennes, M .Trevisani, M. D. Hollenberg, J. L. Wallace, et al. Agonists of proteinase-activated receptor 2 induce inflammation by a neurogenic mechanism. Nat Med 2000, vol. 6 (2), pp. 151–158. [CrossRef]
- N. Vergnolle, J. L. Wallace, N. W. Bunnett, e M. D. Hollenberg. Protease-activated receptors in inflammation, neuronal signaling and pain. Trends Pharmacol Sci. 2001, vol. 22 (3), pp. 146–152. [CrossRef]
- N. Vergnolle, M. Ferazzini, M. R. D’Andrea, J. Buddenkotte, M. Steinhoff. Proteinase-activated receptors: novel signals for peripheral nerves. Trends in Neurosci. 2003, vol. 26 (9), pp. 496–500. [CrossRef]
- Afkhami-Goli, F. Noorbakhsh, A.J. Keller, N. Vergnolle, D. Westaway, J. H. Jhamandas, P. Andrade-Gordon, M. D. Hollenberg, H.Arab, R.H. Dyck,et al. Proteinase-Activated Receptor-2 Exerts Protective and Pathogenic Cell Type-Specific Effects in Alzheimer’s Disease. J. Immunol. 2007, vol. 179 (8), pp. 5493–5503. [CrossRef]
- G. Jin, T. Hayashi, J.Kawagoe, T. Takizawa, T.Nagata, I. Nagano, M. Syoji, K. Abe et al. Deficiency of PAR-2 Gene Increases Acute Focal Ischemic Brain Injury. J Cereb Blood Flow Metab. 2005, vol. 25 (3), pp. 302–313. [CrossRef]
- F. Noorbakhsh, , S.Tsutsui, N. Vergnolle, L.A. Boven, N. Shariat, M. Vodjgani, K. G. Warren, P. Andrade-Gordon, M. D. Hollenberg, C.Power . Proteinase-activated receptor 2 modulates neuroinflammation in experimental autoimmune encephalomyelitis and multiple sclerosi. J Exp. Med. 2006, vol. 203 (2), pp. 425–435. [CrossRef]
- S.-C. Tang, J.D. Lathia, P. K. Selvaraj, D. Jo, M.R. Mughal, A. Cheng, D. A. Siler, W.R. Markesbery, T. V. Arumugam, M.P. Mattson. Toll-like receptor-4 mediates neuronal apoptosis induced by amyloid β-peptide and the membrane lipid peroxidation product 4-hydroxynonenal. Exp. Neurol., 2008, vol. 213 (1), pp. 114–121, set.. [CrossRef]
- M. Zeuner, K. Bieback, D. Widera. Controversial Role of Toll-like Receptor 4 in Adult Stem Cell., Stem Cell Rev and Rep. 2015, vol. 11 (4), pp. 621–634, ago.. [CrossRef]
- J.-P. Michaud, M. Hallé, A. Lampron, P.Thériault, P. Préfontaine, M. Filali, P. Tribout-Jover, A. Lanteigne, R. Jodoin, C.r Cluff et al. Toll-like receptor 4 stimulation with the detoxified ligand monophosphoryl lipid A improves Alzheimer’s disease-related pathology. Proc. Natl. Acad. Sci. U.S.A. 2013, vol. 110 (5), pp. 1941–1946. [CrossRef]
- Nimmerjahn, F. Kirchhoff, e F. Helmchen. Resting Microglial Cells Are Highly Dynamic Surveillants of Brain Parenchyma in Vivo. Science 2005, vol. 308 (5726) pp. 1314–1318. [CrossRef]
- Y. Wang, W. Luo, G. Reiser.Trypsin and trypsin-like proteases in the brain: Proteolysis and cellular functions. Cell. Mol. Life Sci. 2008, vol. 65 (2), pp. 237–252. [CrossRef]
- J. A. Huntington. Serpin structure, function and dysfunction. J Thromb. and Haemost. 2011, vol. 9, pp. 26–34. [CrossRef]
- M. H. Han, S.-I. Hwang, D. B. Roy, D. H. Lundgren, J. V. Price, S. S. Ousman, G. H. Fernald, B. Gerlitz, W. H. Robinson, et al. Proteomic analysis of active multiple sclerosis lesions reveals therapeutic targets. Nature 2008, vol. 451 (7182), pp. 1076–1081. [CrossRef]
- G. A. Hastings, T. A. Coleman, C. C. Haudenschild, S. Stefansson, E. P. Smith, R. Barthlow, S. Cherry, M. Sandkvist, D. A. Lawrence. Neuroserpin, a Brain-associated Inhibitor of Tissue Plasminogen Activator Is Localized Primarily in Neurons. J. Biol. Chem. 1997, vol. 272 (52), pp. 33062–33067. [CrossRef]
- S. Janciauskiene, H. T. Wright. Inflammation, antichymotrypsin, and lipid metabolism: autogenic etiology of Alzheimer’s disease. Bioessays, 1998, vol. 20 (12), pp. 1039–1046. [CrossRef]
- M.H. Seo, S.H. Kim, S. Yeo. Serping1 associated with alpha-synuclein increase in colonic smooth muscles of MPTP-induced Parkinson's disease mice. Sci Rep. 2024 vol. 14(1), p. 1140. [CrossRef]
- G. Villano, E.Novo, C. Turato, S. Quarta, M.Ruvoletto, A. Biasiolo, F. Protopapa, M. Chinellato, A. Martini, E. Trevellin . The protease activated receptor 2 - CCAAT/enhancer-binding protein beta - SerpinB3 axis inhibition as a novel strategy for the treatment of non-alcoholic steatohepatitis. Mol. Met. 2024vol. 81, p. 101889. [CrossRef]
- M. Chinellato, M. Gasparotto, S. Quarta, M. Ruvoletto, A. Biasiolo, F. Filippini, L.Spiezia, L. Cendron, P. Pontisso. 1-Piperidine Propionic Acid as an Allosteric Inhibitor of Protease Activated Receptor-2. Pharmaceuticals, 2023, vol. 16 (10), p. 1486,. [CrossRef]
- G. Auburger, M. Klinkenberg, J. Drost, K. Marcus, B. Morales-Gordo, W. S. Kunz, U. Brandt, V. Broccoli, H. Reichmann et al. Primary Skin Fibroblasts as a Model of Parkinson’s Disease. Mol. Neurobiol. 2012, vol. 46 (1), pp. 20–27. [CrossRef]
- Russo, G. Berti, N. Plotegher, G. Bernardo, R.Filograna, L. Bubacco, E. Greggio., Leucine-rich repeat kinase 2 positively regulates inflammation and down-regulates NF-κB p50 signaling in cultured microglia cells., J Neuroinflam. 2015, vol. 12 (1), p. 230. [CrossRef]
- H. Scheiblich, A. Schlütter, D. T. Golenbock, E. Latz, P. Martinez-Martinez, M. T. Heneka. Activation of the NLRP 3 inflammasome in microglia: the role of ceramide. J. Neurochem. 2017, vol. 143 (5), pp. 534–550, dic.. [CrossRef]
- K. J. Livak, T. D. Schmittgen. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, vol. 25 (4), pp. 402–408. [CrossRef]
- G. P. Connolly. Fibroblast models of neurological disorders: fluorescence measurement studies.Trends Pharmacol. Sci. 1998, vol. 19(5), pp. 171–177. [CrossRef]
- L. Gasparini, M. Racchi, G. Binetti, M. Trabucchi, S. B. Solerte, D. Alkon, R. Etcheberrigaray, G. Gibson, J. Blass, R. Paoletti, S. Govoni. Peripheral markers in testing pathophysiological hypotheses and diagnosing Alzheimer’s disease. FASEB J. 1998, vol. 12 (1), pp. 17–34. [CrossRef]
- L. Cui, W. Yang, J. Shuai, Y. Ma, Y. Yan. Lifestyle and Socioeconomic Transition and Health Consequences of Alzheimer's Disease and Other Dementias in Global, from 1990 to 2019. J Prev Alzheimers Dis. 2024, Vol 11(1), pp. 88-96. [CrossRef]
- J. Yang, W. Zhi, L. Wang. Role of Tau Protein in Neurodegenerative Diseases and Development of Its Targeted Drugs: A Literature Review. Molecules. Vol 29(12), 2024, p. 2812. [CrossRef]
- S. Cannito, B. Foglia, G. Villano, C. Turato, T. C. Delgado, E. Morello, F. Pin, E. Novo, L. Napione, S. Quarta, et al. SerpinB3 Differently Up-Regulates Hypoxia Inducible Factors -1α and -2α in Hepatocellular Carcinoma: Mechanisms Revealing Novel Potential Therapeutic Targets. Cancers 2019, vol 11, p. 1933. [CrossRef]
- Santiago-Balmaseda, A. Aguirre-Orozco, I.E. Valenzuela-Arzeta, M.M. Villegas-Rojas, I. Pérez-Segura, N. Jiménez-Barrios, E. Hurtado-Robles, L.D. Rodríguez-Hernández, E.R. Rivera-German, M. Guerra-Crespo, et al. Neurodegenerative Diseases: Unraveling the Heterogeneity of Astrocytes. Cells, 2024, vol. 13(11), p. 921. [CrossRef]
- D.A. Peterson. Blood–Brain Barrier. In: eLS. John Wiley & Sons, Ltd: Chichester, 2012. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



