Submitted:
26 June 2024
Posted:
28 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Result
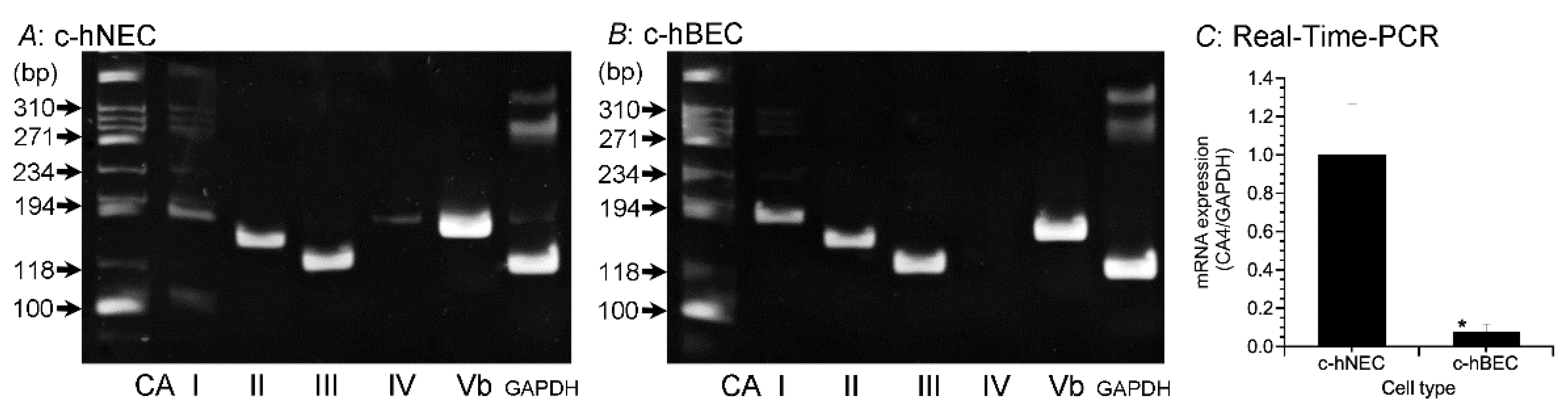
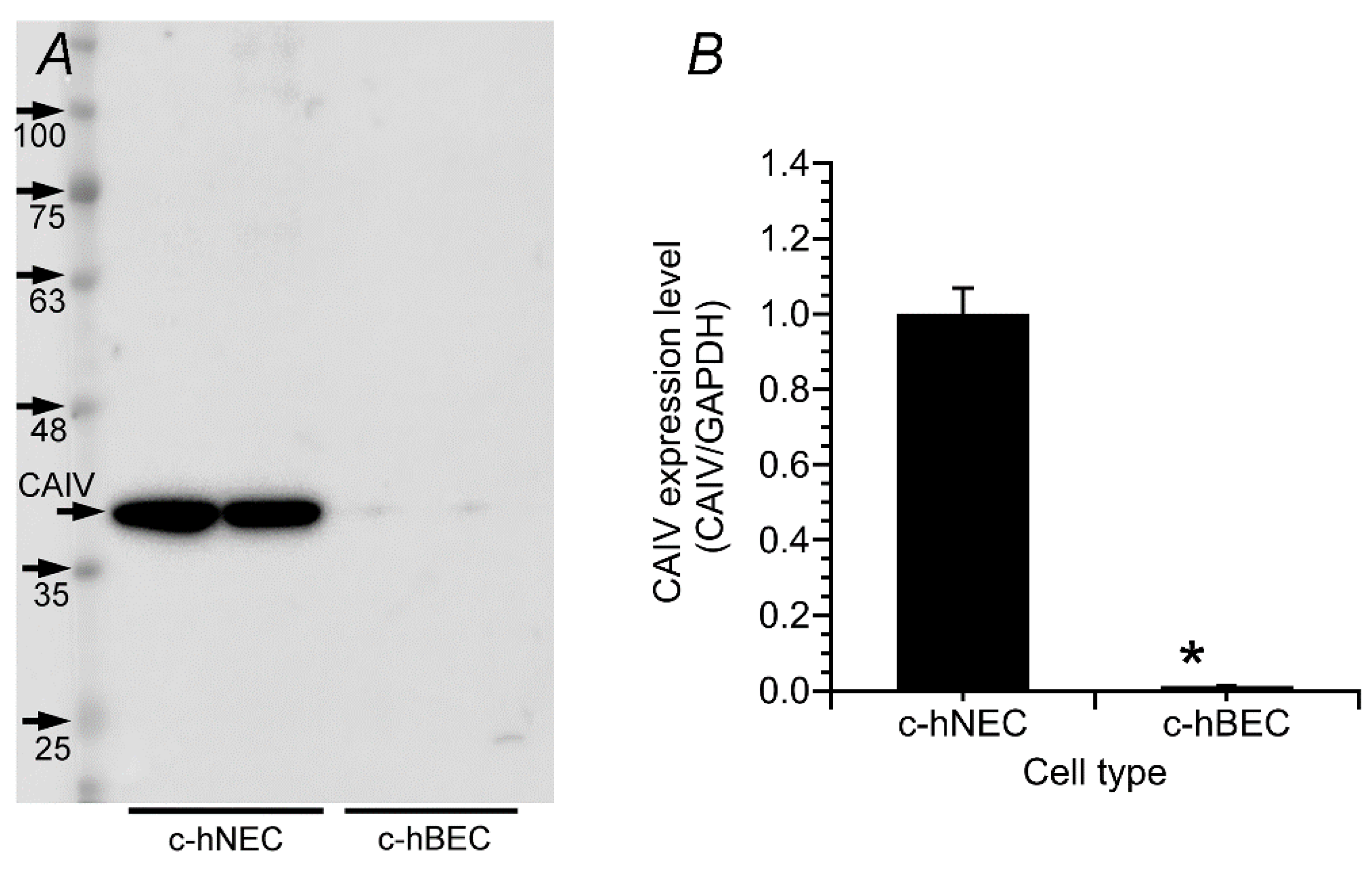
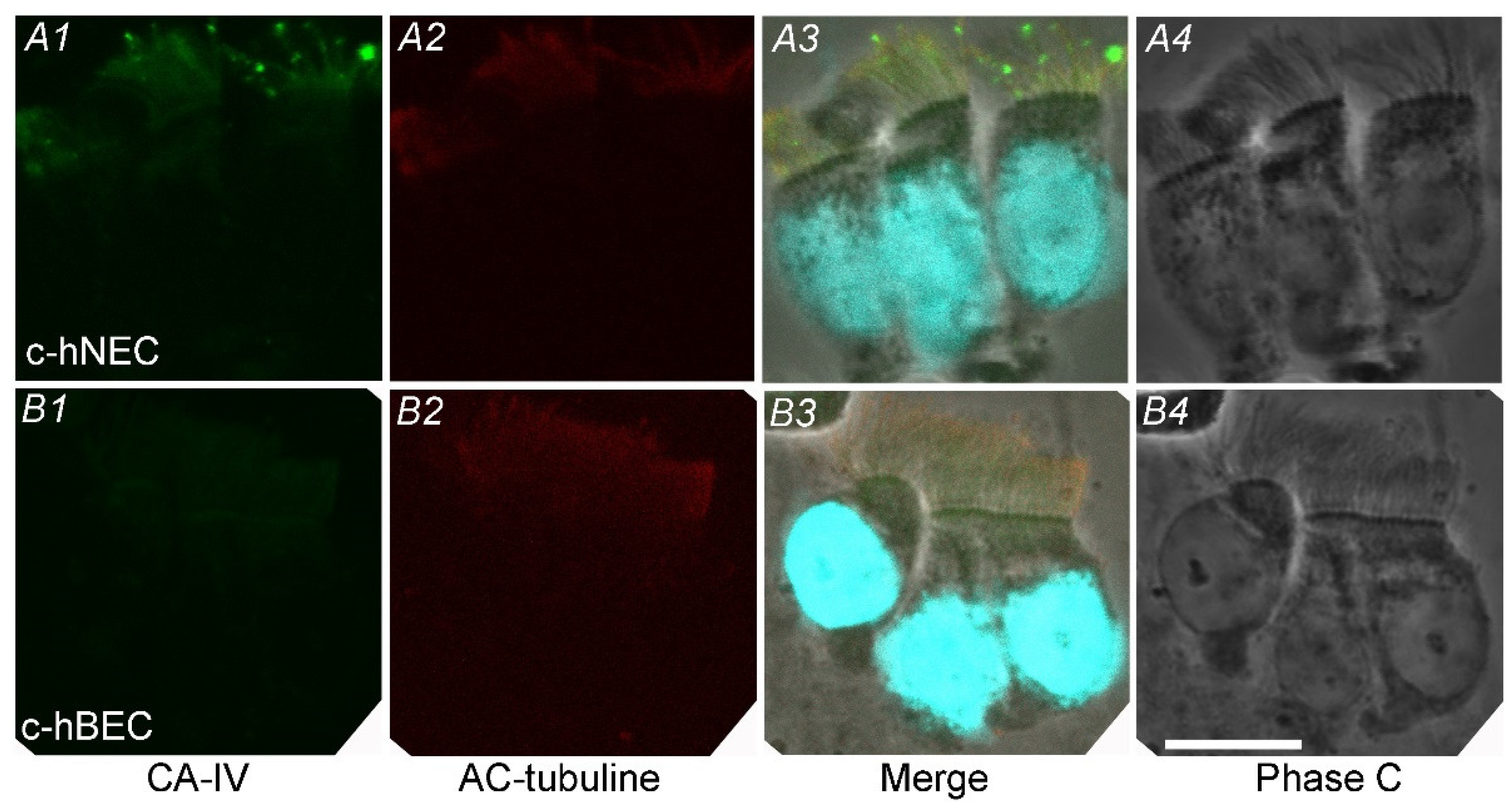
2.1. Expression of CA
2.1.1. PCR
2.1.2. Western Blotting
2.1.3. Immunofluorescence Examination for CAIV
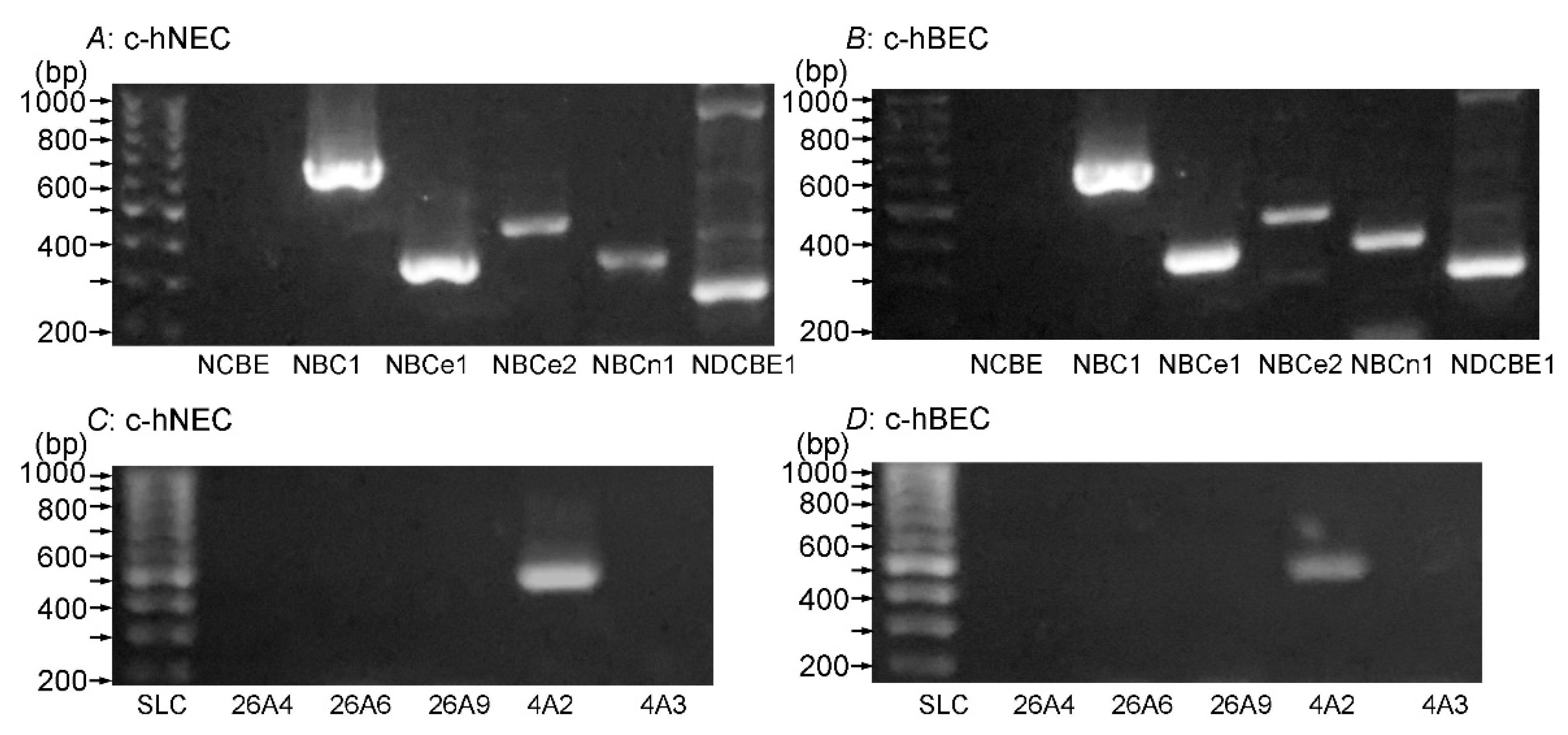
2.2. Expression of NBC and AE
2.3. CBF and CBD in c-hNECs and c-hBECs
2.3.1. Ciliated-hNECs
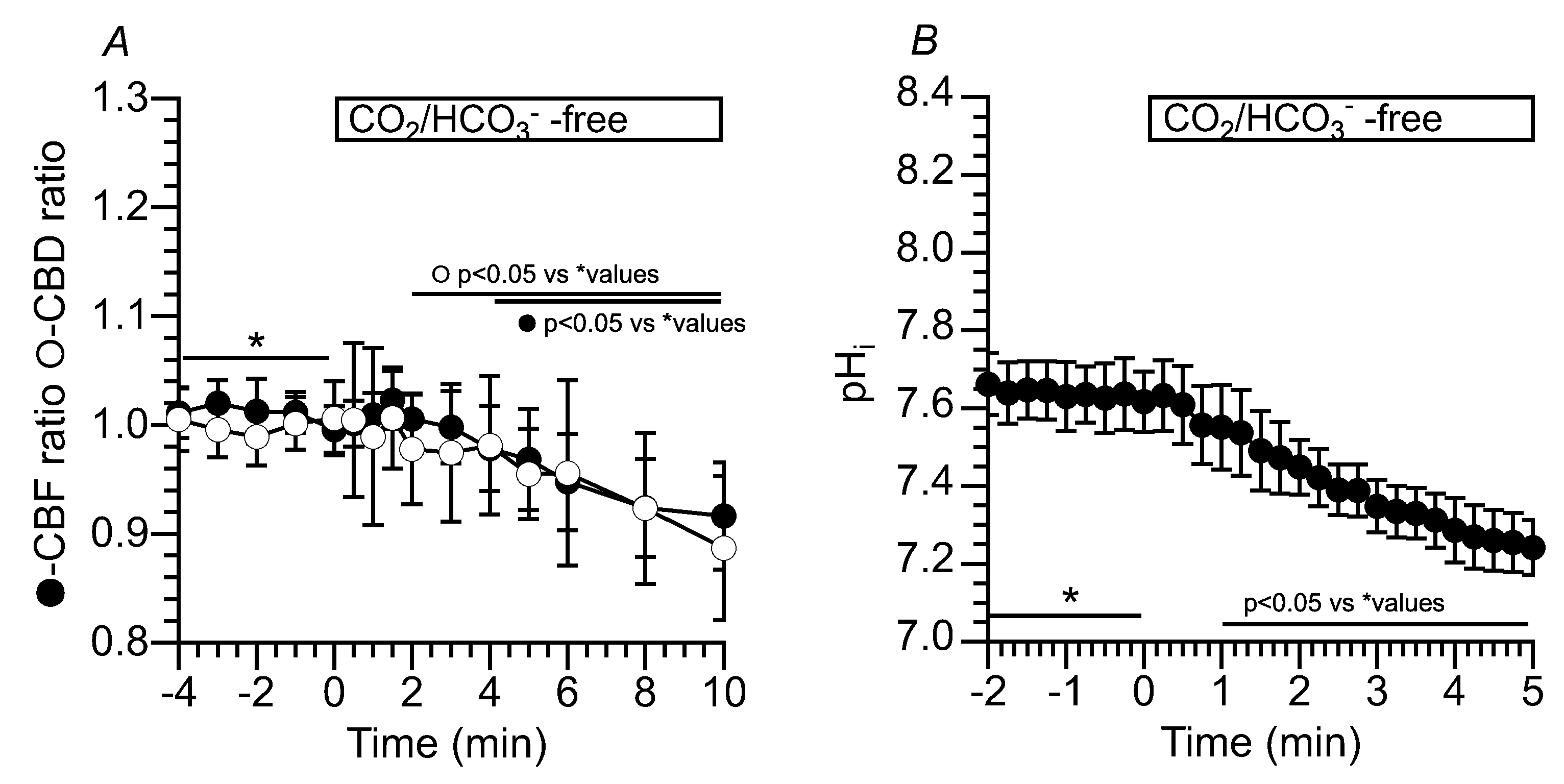
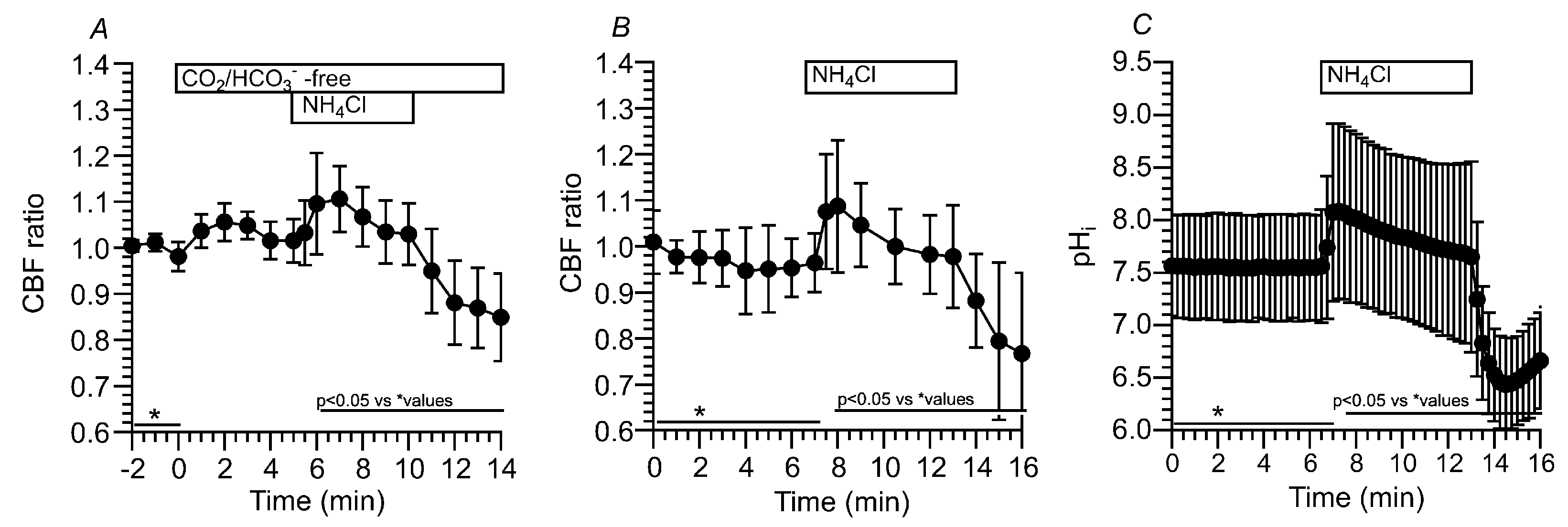
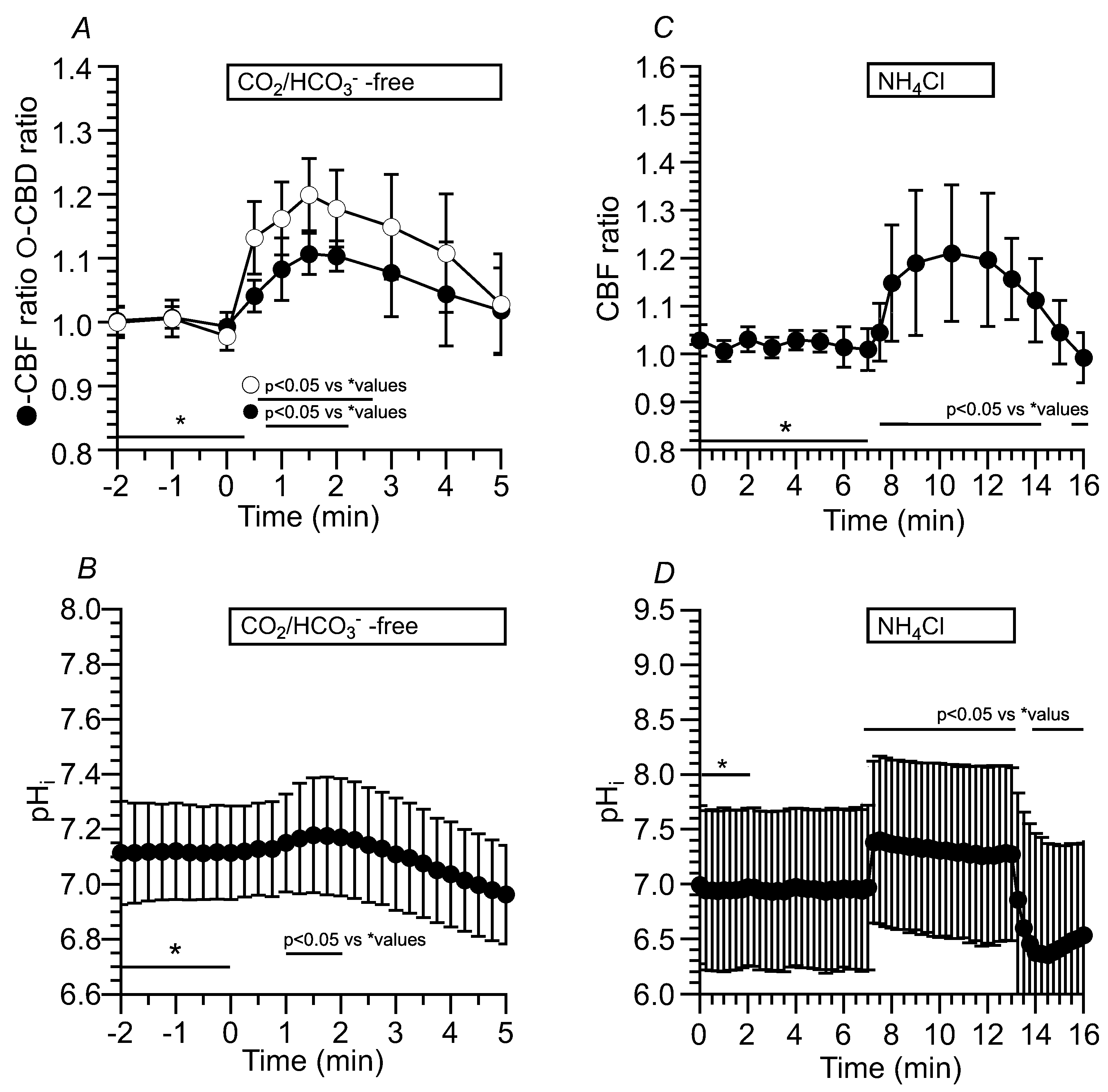
2.3.1.1. Effects of CO2/HCO3- -free solution (Zero-CO2) on CBF, CBD and pHi in c-hNECs
2.3.1.2. Effects of NH4+ Pulse on CBF, CBD and pHi in c-hNECs
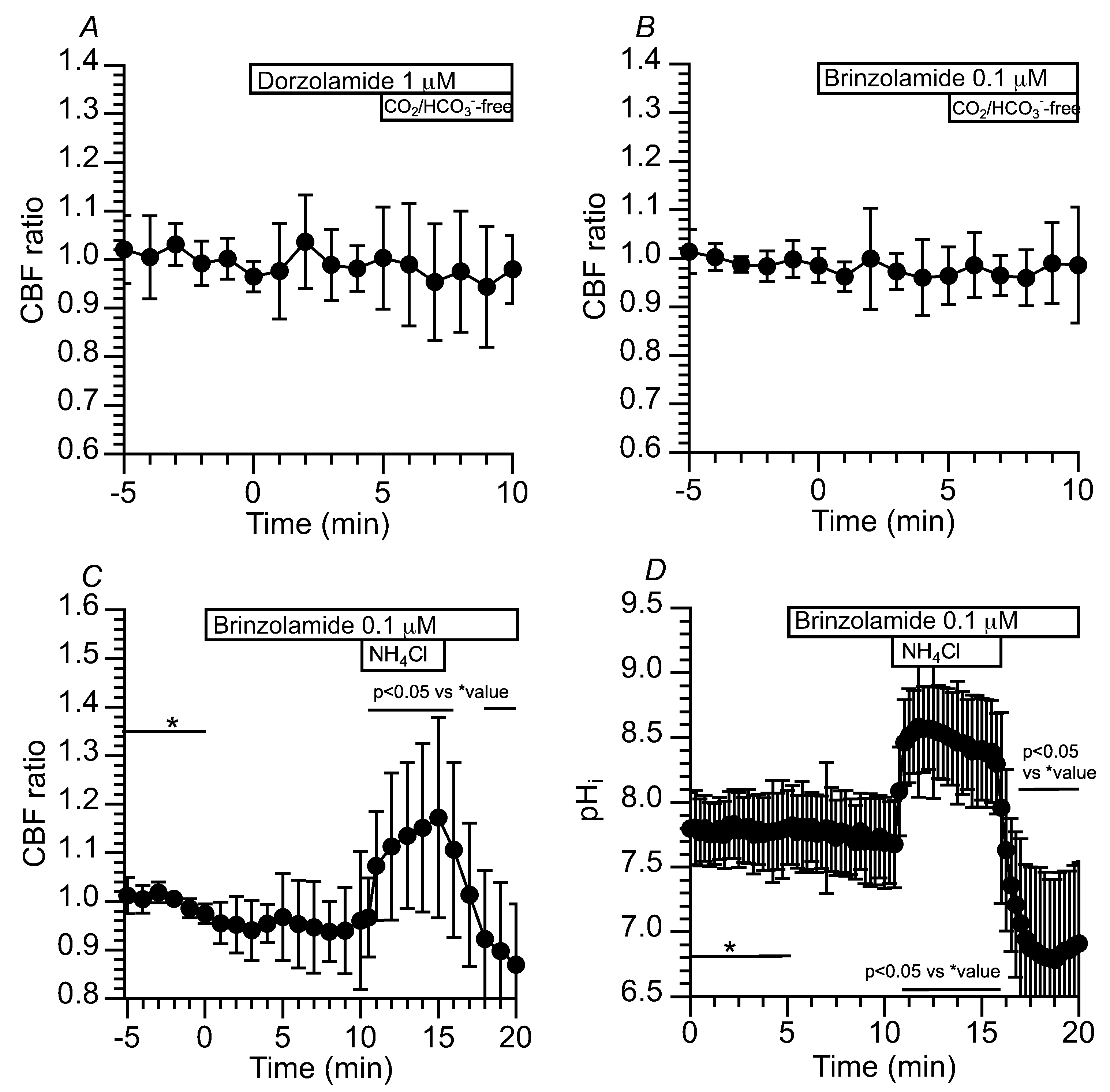
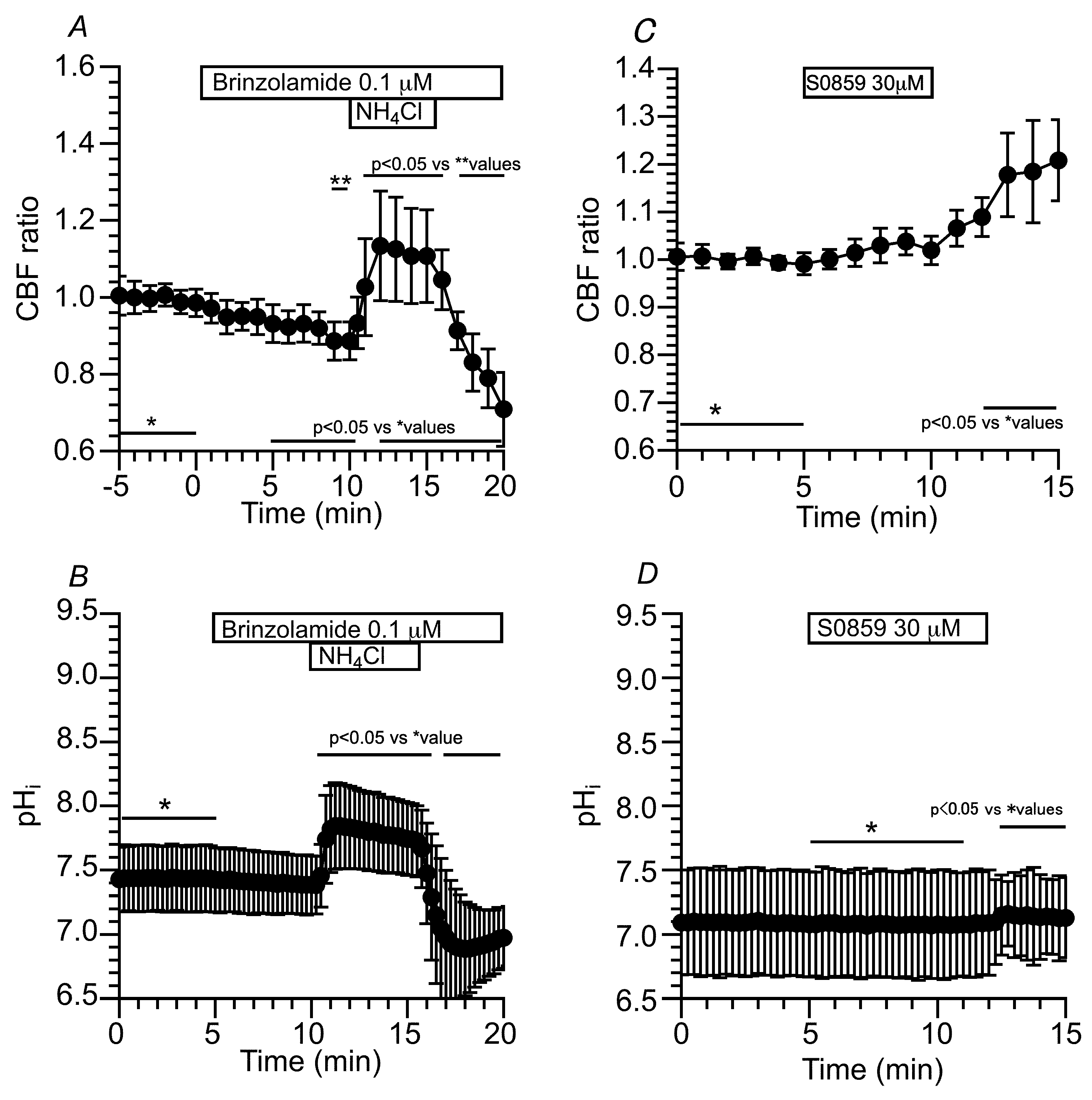
2.3.1.3. Effects of CA Inhibitors on CBF and pHi Changed by Applying the CO2/HCO3--Free Solution and NH4+ Pulse in c-hNECs
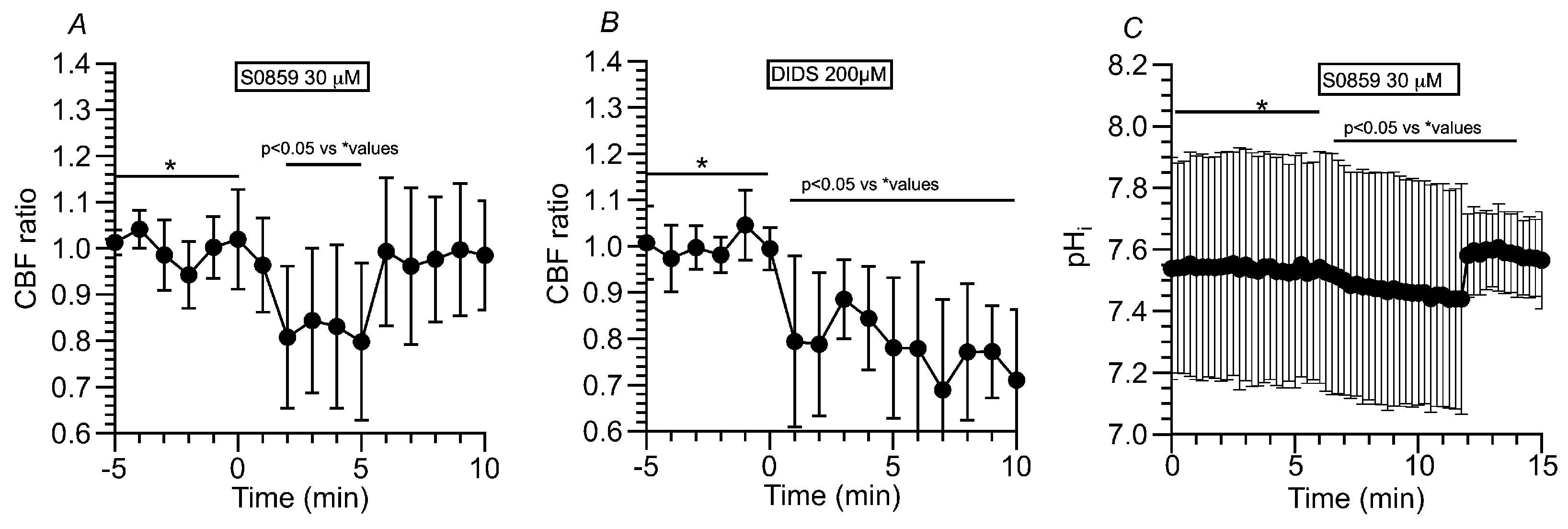
2.3.1.4. Effects of NBC Inhibitors (S0859 and DIDS) on CBF and pHi in c-hNECs
2.3.2. Ciliated-hBECs
2.3.2.1. Effects of Zero-CO2 and the NH4+ pulse on CBF and pHi in c-hBECs
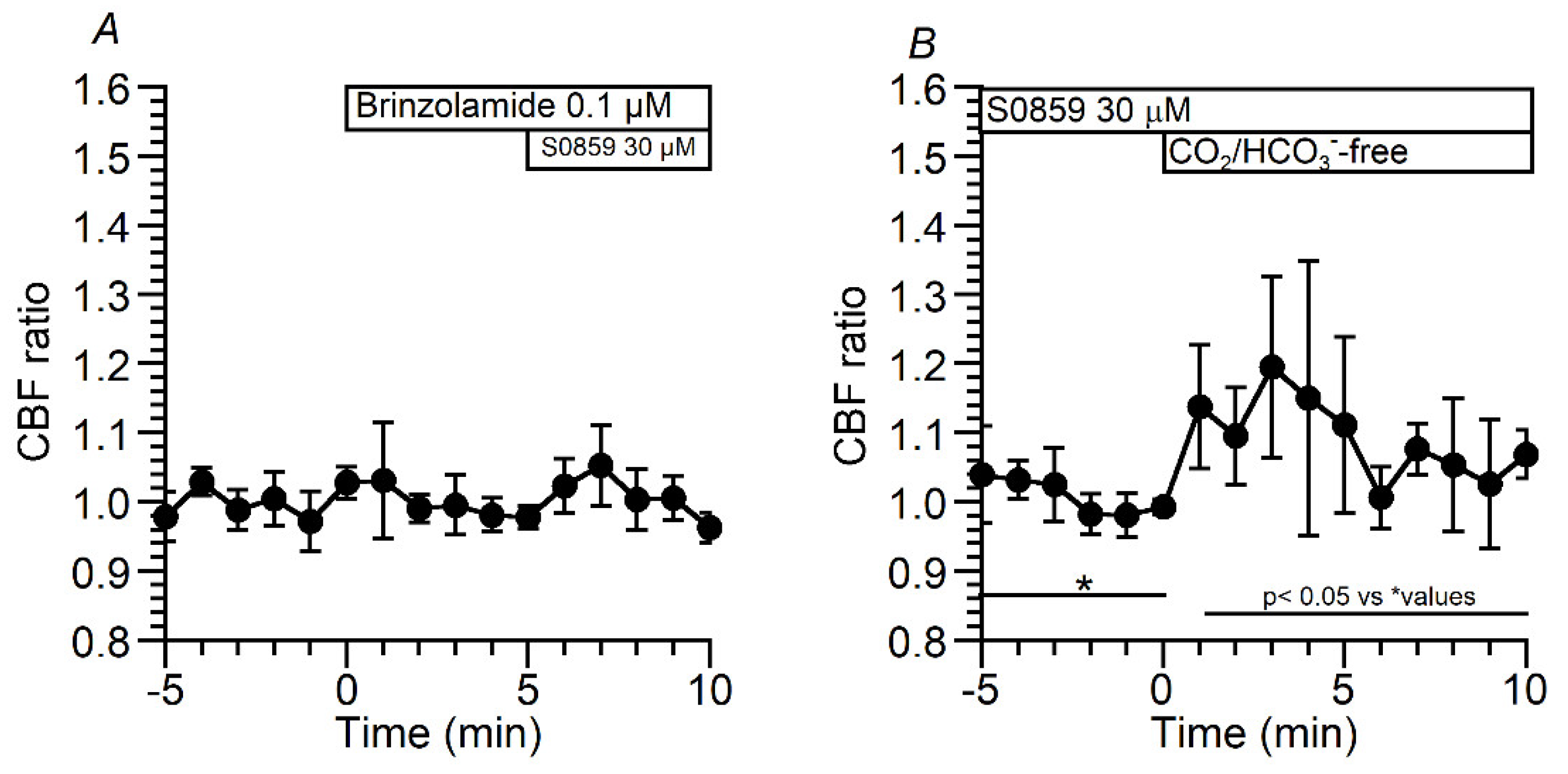
2.3.2.2. Effects of Brinzolamide and S0859 on CBF and pHi in c-hBECs
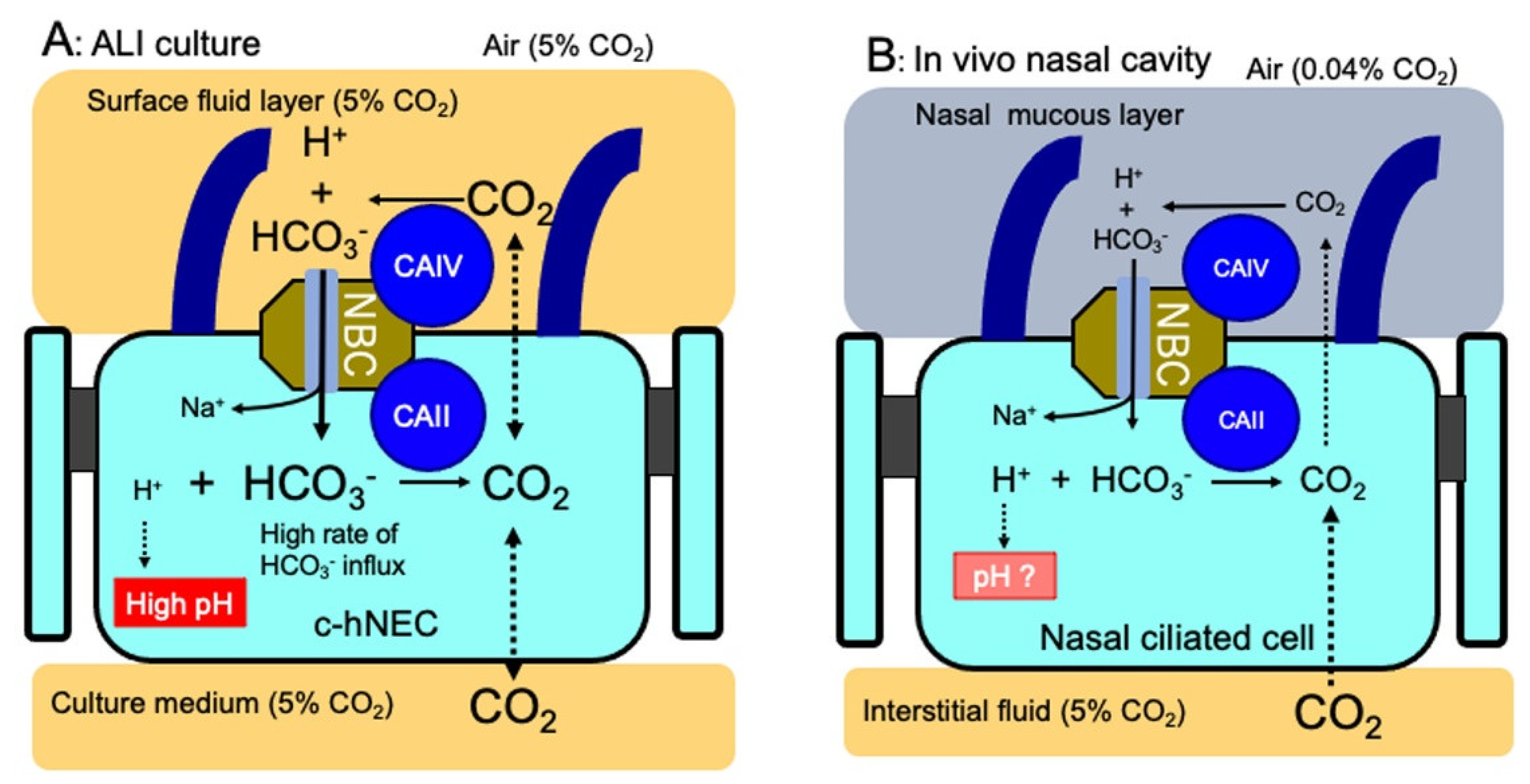
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Solution and Chemicals
4.3. Cell Culture Media
4.4. Antibodies
4.5. Cell preparation
4.6. Measurements of CBF and CBD
4.7. Measurement of pHi
4.8. RT-PCR
4.9. Western Blotting
4.10. Immunofluorescence Examination
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Afzelius, B.A. Cilia-related diseases. J Pathol 2004, 204, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Salathe, M. Regulation of mammalian ciliary beating. Annu Rev Physiol 2007, 69, 401–422. [Google Scholar] [CrossRef] [PubMed]
- Button, B.; Cai, L.H.; Ehre, C.; Kesimer, M.; Hill, D.B.; Sheehan, J.K.; Boucher, R.C.; Rubinstein, M. A periciliary brush promotes the lung health by separating the mucus layer from airway epithelia. Science 2012, 337, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Wanner, A.; Salathé, M.; O’Riordan, T.G. Mucociliary clearance in the airways. Am J Respir Crit Care Med 1996, 154, 1868–1902. [Google Scholar] [CrossRef] [PubMed]
- Delmotte, P.; Sanderson, M.J. Ciliary beat frequency is maintained at a maximal rate in the small airways of mouse lung slices. Am J Respir Cell Mol Biol 2006, 35, 110–117. [Google Scholar] [CrossRef]
- Inui, T.A.; Murakami, K.; Yasuda, M.; Hirano, S.; Ikeuchi, Y.; Kogiso, H.; Hosogi, S.; Inui, T.; Marunaka, Y.; Nakahari, T. Ciliary beating amplitude controlled by intracellular Cl(-) and a high rate of CO(2) production in ciliated human nasal epithelial cells. Pflugers Arch 2019, 471, 1127–1142. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, M.; Inui, T.A.; Hirano, S.; Asano, S.; Okazaki, T.; Inui, T.; Marunaka, Y.; Nakahari, T. Intracellular Cl(-) Regulation of Ciliary Beating in Ciliated Human Nasal Epithelial Cells: Frequency and Distance of Ciliary Beating Observed by High-Speed Video Microscopy. Int J Mol Sci 2020, 21, 4052. [Google Scholar] [CrossRef]
- Sutto, Z.; Conner, G.E.; Salathe, M. Regulation of human airway ciliary beat frequency by intracellular pH. J Physiol 2004, 560, 519–532. [Google Scholar] [CrossRef]
- Saito, D.; Suzuki, C.; Tanaka, S.; Hosogi, S.; Kawaguchi, K.; Asano, S.; Okamoto, S.; Yasuda, M.; Hirano, S.; Inui, T.; Marunaka, Y.; Nakahari, T. Ambroxol-enhanced ciliary beating via voltage-gated Ca(2+) channels in mouse airway ciliated cells. Eur J Pharmacol 2023, 941, 175496. [Google Scholar] [CrossRef]
- Nakahari, T.; Suzuki, C.; Kawaguchi, K.; Hosogi, S.; Tanaka, S.; Asano, S.; Inui, T.; Marunaka, Y. Ambroxol-Enhanced Frequency and Amplitude of Beating Cilia Controlled by a Voltage-Gated Ca(2+) Channel, Cav1.2, via pH(i) Increase and [Cl(-)](i) Decrease in the Lung Airway Epithelial Cells of Mice. Int J Mol Sci 2023, 24, 16976. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Koga, Y.; Wakasugi, T.; Kitamura, T.; Suzuki, H. TRPA1/M8 agonists upregulate ciliary beating through the pannexin-1 channel in the human nasal mucosa. Mol Biol Rep 2023, 50, 2085–2093. [Google Scholar] [CrossRef] [PubMed]
- Tarun, A.S.; Bryant, B.; Zhai, W.; Solomon, C.; Shusterman, D. Gene expression for carbonic anhydrase isoenzymes in human nasal mucosa. Chem Senses 2003, 28, 621–629. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, H.M.; Lee, S.H.; Kim, H.K.; Lee, J.H.; Oh, K.H.; Lee, S.H. Down-regulation of carbonic anhydrase isoenzymes in nasal polyps. Laryngoscope 2008, 118, 1856–1861. [Google Scholar] [CrossRef]
- Alvarez, B.V.; Loiselle, F.B.; Supuran, C.T.; Schwartz, G.J.; Casey, J.R. Direct extracellular interaction between carbonic anhydrase IV and the human NBC1 sodium/bicarbonate co-transporter. Biochemistry 2003, 42, 12321–12329. [Google Scholar] [CrossRef]
- Tsuruoka, S.; Swenson, E.R.; Petrovic, S.; Fujimura, A.; Schwartz, G.J. Role of basolateral carbonic anhydrase in proximal tubular fluid and bicarbonate absorption. Am J Physiol Renal Physiol 2001, 280, F146–F154. [Google Scholar] [CrossRef]
- Yang, Z.; Alvarez, B.V.; Chakarova, C.; Jiang, L.; Karan, G.; Frederick, J.M.; Zhao, Y.; Sauvé, Y.; Li, X.; Zrenner, E.; Wissinger, B.; Hollander, A.I.; Katz, B.; Baehr, W.; Cremers, F.P.; Casey, J.R.; Bhattacharya, S.S.; Zhang, K. Mutant carbonic anhydrase 4 impairs pH regulation and causes retinal photoreceptor degeneration. Hum Mol Genet 2005, 14, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Park, J.H.; Jung, H.H.; Lee, S.H.; Oh, J.W.; Lee, H.M.; Jun, H.S.; Cho, W.J.; Lee, J.Y. Expression and distribution of ion transport mRNAs in human nasal mucosa and nasal polyps. Acta Otolaryngol 2005, 125, 745–752. [Google Scholar] [PubMed]
- Rayner, R.E.; Makena, P.; Prasad, G.L.; Cormet-Boyaka, E. Optimization of Normal Human Bronchial Epithelial (NHBE) Cell 3D Cultures for in vitro Lung Model Studies. Sci Rep 2019, 9, 500. [Google Scholar] [CrossRef]
- Schwartz, G.J.; Kittelberger, A.M.; Barnhart, D.A.; Vijayakumar, S. Carbonic anhydrase IV is expressed in H(+)-secreting cells of rabbit kidney. Am J Physiol Renal Physiol 2000, 278, F894–F904. [Google Scholar] [CrossRef]
- Sterling, D.; Alvarez, B.V.; Casey, J.R. The extracellular component of a transport metabolon. Extracellular loop 4 of the human AE1 Cl-/HCO3- exchanger binds carbonic anhydrase IV. J Biol Chem 2002, 277, 25239–25246. [Google Scholar] [CrossRef]
- McMurtrie, H.L.; Cleary, H.J.; Alvarez, B.V.; Loiselle, F.B.; Sterling, D.; Morgan, P.E.; Johnson, D.E.; Casey, J.R. The bicarbonate transport metabolon. J Enzyme Inhib Med Chem 2004, 19, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, S.; Hisamitsu, T.; Pang, T.; Shigekawa, M. Kinetic dissection of two distinct proton binding sites in Na+/H+ exchangers by measurement of reverse mode reaction. J Biol Chem 2003, 278, 43580–43585. [Google Scholar] [CrossRef] [PubMed]
- Gross, E.; Pushkin, A.; Abuladze, N.; Fedotoff, O.; Kurtz, I. Regulation of the sodium bicarbonate cotransporter kNBC1 function: role of Asp(986), Asp(988) and kNBC1-carbonic anhydrase II binding. J Physiol 2002, 544, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, J.; Shen, B.; Vihinen, M.; Casini, A.; Scozzafava, A.; Supuran, C.T.; Parkkila, A.K.; Saarnio, J.; Kivelä, A.J.; Waheed, A.; Sly, W.S.; Parkkila, S. Characterization of CA XIII, a novel member of the carbonic anhydrase isozyme family. J Biol Chem 2004, 279, 2719–2727. [Google Scholar] [CrossRef] [PubMed]
- Supuran, C.T.; Scozzafava, A. Carbonic anhydrases as targets for medicinal chemistry. Bioorg Med Chem 2007, 15, 4336–4350. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, Y.; Oishi, M.; Amasaki, T.; Soeta, S.; Ichihara, N.; Nishita, T.; Murakami, M.; Amasaki, H.; Asari, M. Immunohistochemical localization and gene expression of carbonic anhydrase isoenzymes CA-II and CA-VI in canine lower airways and lung. J Vet Med Sci 2009, 71, 1525–1528. [Google Scholar] [CrossRef] [PubMed]
- Lesburg, C.A.; Huang, C.; Christianson, D.W.; Fierke, C.A. Histidine --> carboxamide ligand substitutions in the zinc binding site of carbonic anhydrase II alter metal coordination geometry but retain catalytic activity. Biochemistry 1997, 36, 15780–15791. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K.; Okazaki, K.; Nishi, T.; Uose, S.; Nakase, H.; Ohana, M.; Matsushima, Y.; Omori, K.; Chiba, T. Experimental immune-mediated pancreatitis in neonatally thymectomized mice immunized with carbonic anhydrase II and lactoferrin. Lab Invest 2002, 82, 411–424. [Google Scholar] [CrossRef]
- Liu, L.; Yamamoto, A.; Yamaguchi, M.; Taniguchi, I.; Nomura, N.; Nakakuki, M.; Kozawa, Y.; Fukuyasu, T.; Higuchi, M.; Niwa, E.; Tamada, T.; Ishiguro, H. Bicarbonate transport of airway surface epithelia in luminally perfused mice bronchioles. J Physiol Sci 2022, 72, 4. [Google Scholar] [CrossRef]
- Thornell, I.M.; Li, X.; Tang, X.X.; Brommel, C.M.; Karp, P.H.; Welsh, M.J.; Zabner, J. Nominal carbonic anhydrase activity minimizes airway-surface liquid pH changes during breathing. Physiol Rep 2018, 6, e13569. [Google Scholar] [CrossRef]
- Zajac, M.; Dreano, E.; Edwards, A.; Planelles, G.; Sermet-Gaudelus, I. Airway Surface Liquid pH Regulation in Airway Epithelium Current Understandings and Gaps in Knowledge. Int J Mol Sci 2021, 22, 3384. [Google Scholar] [CrossRef] [PubMed]
| Transcript | Direction | Sequence | Size (bp) |
| CA1 | Sense | AGCTGCCTCAAAGGCTGATG | 181 |
| Antisense | GGTCCAGAAATCCAGGGATGAA | ||
| CA2 | Sense | TTACTGGACCTACCCAGGCTCAC | 167 |
| Antisense | GCCAGTTGTCCACCATCAGTTC | ||
| CA3 | Sense | CATGAGAATGGCGACTTCCAGA | 141 |
| Antisense | GAATGAGCCCTGGTAGGTCCAGTA | ||
| CA4 | Sense | TCCCTAGAAACCTAGGGTCATTTCA | 156 |
| Antisense | TGGAGCTAGATCACGTTTCACAA | ||
| CA5b | Sense | TGTTCTGAAGTGAAAGTCTGGTCTG | 172 |
| Antisense | CCAAACTAGAGTGCCCTGGATG |
| NBC | |||
| Transcript | Direction | Sequence | Size (bp) |
| NCBE (SLC4A10) |
Sense | GCAGGTCAGGTTGTTTCTCCTC | 498 |
| Antisense | TCTTCCTCTTCTCCTGGGAAGG | ||
| NBC1 (SLC4A11) |
Sense | GGCCTGTGGAACAGTTTCTTCC | 690 |
| Antisense | TGCCCTTCACCAGCCTGTTCTC | ||
| NBCe1 (SLC4A4) |
Sense | GGTGTGCAGTTCATGGATCGTC | 336 |
| Antisense | GTCACTGTCCAGACTTCCCTTC | ||
| NBCe2 (SLC4A5) |
Sense | ATCTTCATGGACCAGCAGATCAC | 468 |
| Antisense | TGCTTGGCTGGCATCAGGAGG | ||
| NBCn1 (SLC4A7) |
Sense | CAGATGCAAGCAGCCTTGTGTG | 328 |
| Antisense | GGTCCATGATGACCACAAGCTG | ||
| NDCBE1 (SLC4A8) |
Sense | GCTCAAGAAAGGCTGTGGCTAC | 243 |
| Antisense | CATGAAGACTGAGCAGCCCATC | ||
| AE | |||
| AE2 | Sense | GAAGATTCCTGAGAATGCCT | 181 |
| Antisense | GTCCATGTTGGCAGTAGTCG | ||
| AE3 | Sense | ATCTGAGGCAGAACCTGTGG | 418 |
| Antisense | TTTCACTAAGTGTCGCCGC | ||
| SLC26A4 | Sense | GTTTACTAGCTGGCCTTATATTTGGACTGT | 484 |
| Antisense | AGGCTATGGATTGGCACTTTGGGAACG | ||
| SLC26A6 | Sense | TAGGGGAGGTTGGGCCAGGGATGC | 456 |
| Antisense | TGCCGGGAAGTGCCAAACAGGAAGAAGTAGAT | ||
| SLC26A9 | Sense | TCCAGGTCTTCAACAATGCCAC | 400 |
| Antisense | CGAGTCTTGTGCATGTAGCGAG | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




