Submitted:
21 June 2024
Posted:
21 June 2024
You are already at the latest version
Abstract
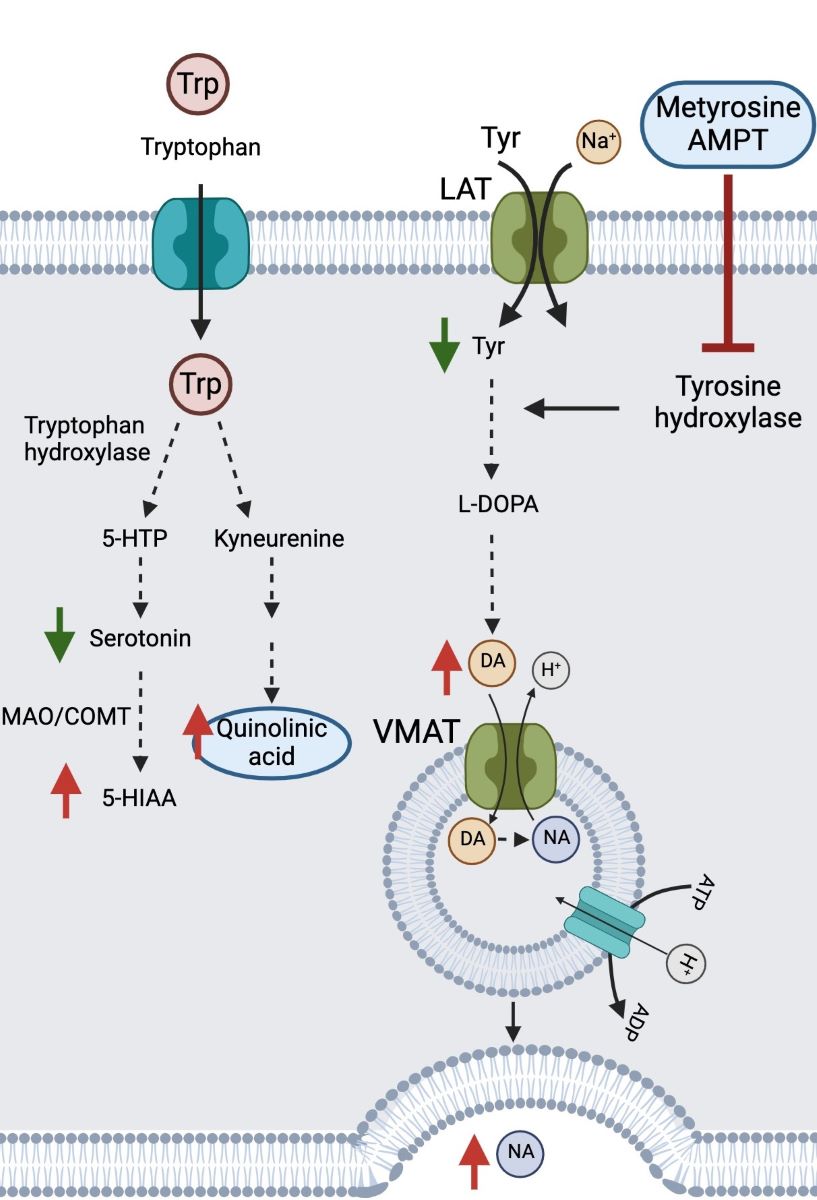
Keywords:
1. Introduction
2. Case Description
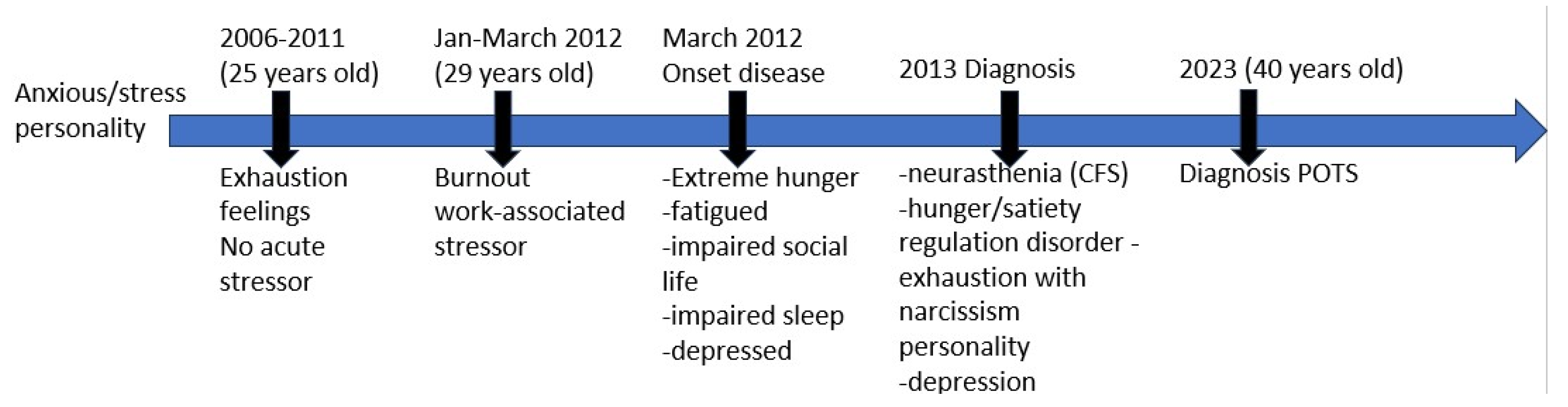
2.1. Diagnostic Timeline
2.2. Biochemical and Genetic Analyses
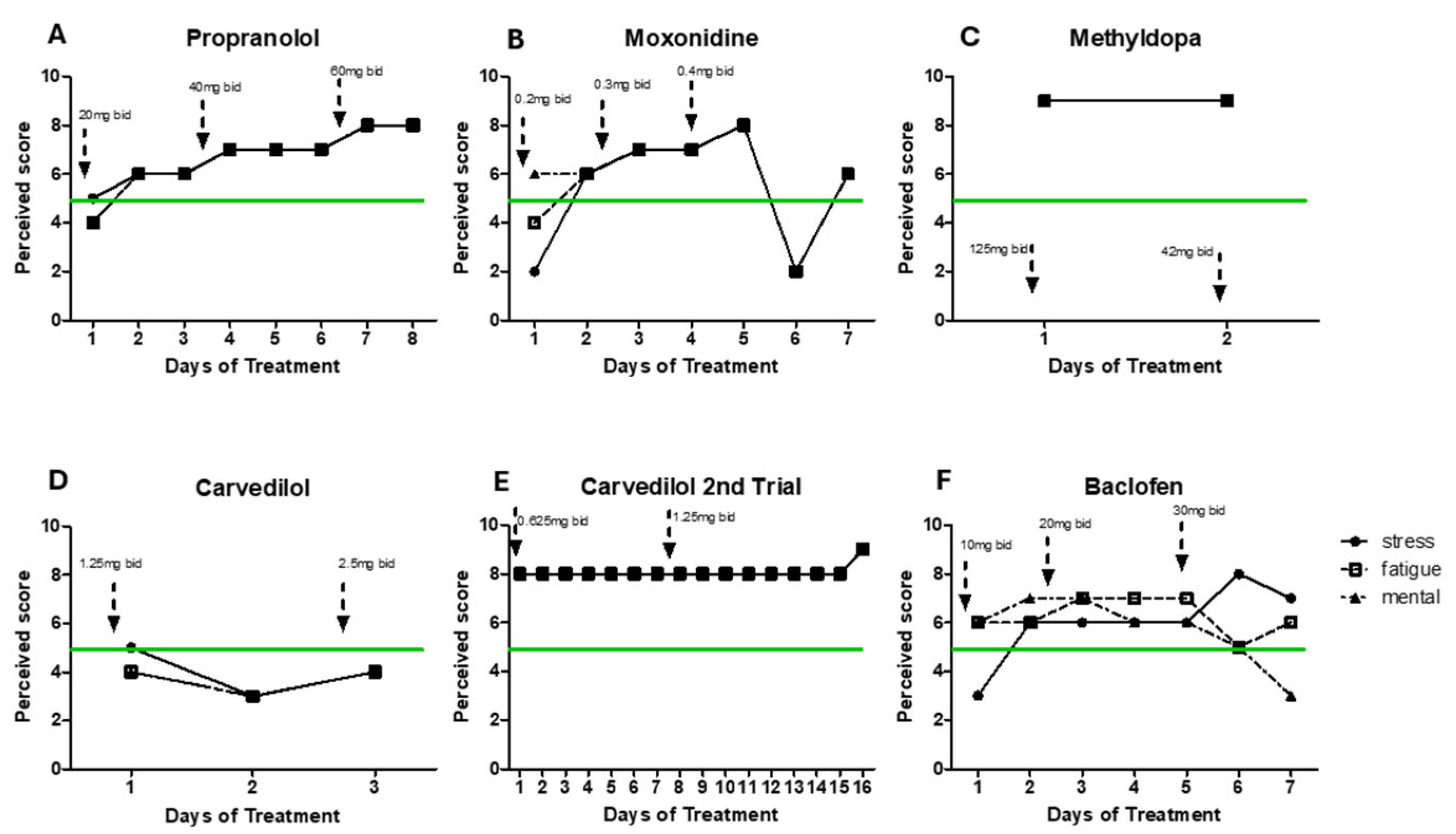
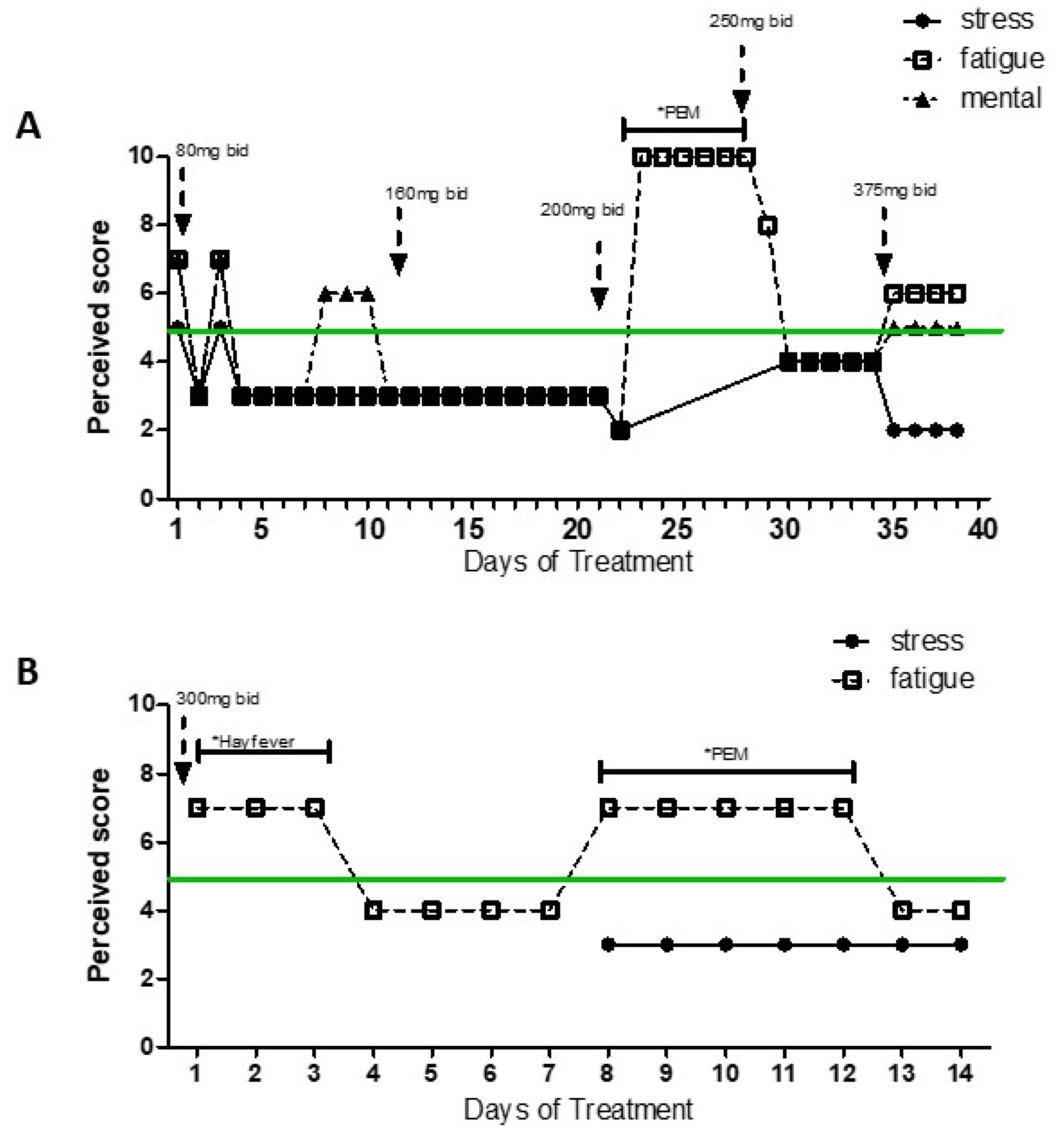
3. Treatment and Course of Symptoms
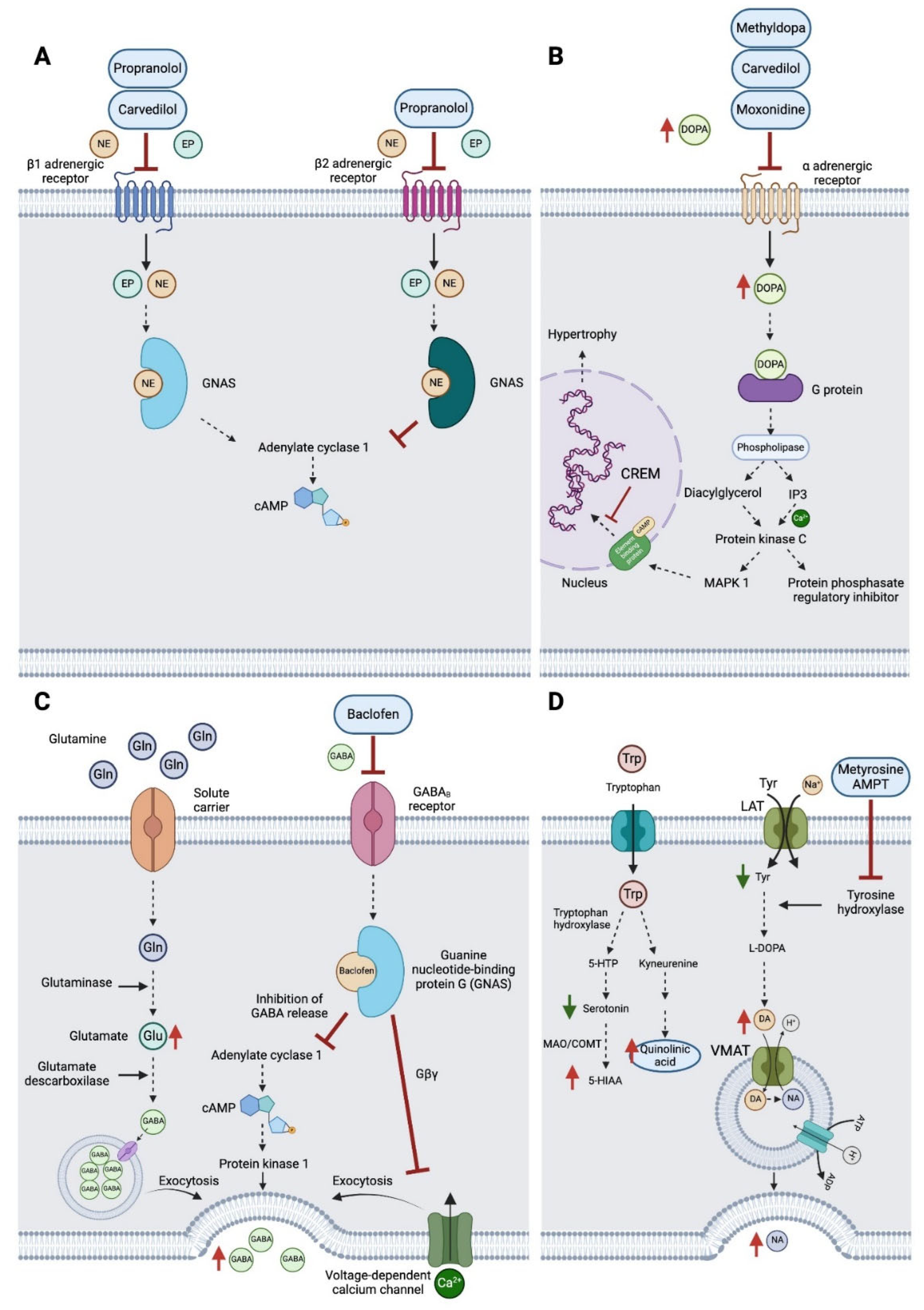
4. Materials and Methods
5. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harrison, J.E.; Weber, S.; Jakob, R.; Chute, C.G. ICD-11: an international classification of diseases for the twenty-first century. BMC Med Informatics Decis. Mak. 2021, 21, 1–10. [Google Scholar] [CrossRef]
- Carruthers, B.M.; Van De Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef]
- Sapra, A., & Bhandari, P. (2023). Chronic fatigue syndrome. In StatPearls [Internet]. StatPearls Publishing. Available from https://www.ncbi.nlm.nih.gov/books/NBK557676/.
- Abdulla, J. , & Torpy, B. D. J. (2000). Chronic fatigue syndrome. In Feingold, K. R., Anawalt, B., Blackman, M. R., Boyce, A., Chrousos, G., Corpas, E.,... & Wilson, D. P. (Eds.), *Endotext*. MDText.com, Inc.
- de Vega, W.C.; Herrera, S.; Vernon, S.D.; McGowan, P.O. Epigenetic modifications and glucocorticoid sensitivity in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). BMC Med Genom. 2017, 10, 1–14. [Google Scholar] [CrossRef]
- Allen, C. D. (2014). Use of low-dose beta-blockers to treat symptoms of chronic fatigue syndrome (Master's thesis, The University of Utah).
- Montoya, J.G.; Holmes, T.H.; Anderson, J.N.; Maecker, H.T.; Rosenberg-Hasson, Y.; Valencia, I.J.; Chu, L.; Younger, J.W.; Tato, C.M.; Davis, M.M. Cytokine signature associated with disease severity in chronic fatigue syndrome patients. Proc. Natl. Acad. Sci. 2017, 114, E7150–E7158. [Google Scholar] [CrossRef]
- Brenu, E.W.; Hardcastle, S.L.; Atkinson, G.M.; van Driel, M.L.; Kreijkamp-Kaspers, S.; Ashton, K.J.; Staines, D.R.; Marshall-Gradisnik, S.M. Natural killer cells in patients with severe chronic fatigue syndrome. Autoimmun. Highlights 2013, 4, 69–80. [Google Scholar] [CrossRef]
- Janal, M.N.; Ciccone, D.S.; Natelson, B.H. Sub-typing CFS patients on the basis of ‘minor’ symptoms. Biol. Psychol. 2006, 73, 124–131. [Google Scholar] [CrossRef]
- Pi, I.G.; Sein-Echaluce, M.L.G.; Aubach, L.R.; Puig-Gros, J.T.; Solà, J.F. Stressful Events in the Onset of Chronic Fatigue Syndrome. . 2016, 90, e1–7. [Google Scholar]
- Borsini, A.; Hepgul, N.; Mondelli, V.; Chalder, T.; Pariante, C.M. Childhood stressors in the development of fatigue syndromes: a review of the past 20 years of research. Psychol. Med. 2013, 44, 1809–1823. [Google Scholar] [CrossRef]
- Hall, D.L.; Lattie, E.G.; Milrad, S.F.; Czaja, S.; Fletcher, M.A.; Klimas, N.; Perdomo, D.; Antoni, M.H. Telephone-administered versus live group cognitive behavioral stress management for adults with CFS. J. Psychosom. Res. 2016, 93, 41–47. [Google Scholar] [CrossRef]
- Hall, D.L.; Lattie, E.G.; Antoni, M.H.; Fletcher, M.A.; Czaja, S.; Perdomo, D.; Klimas, N.G. Stress management skills, cortisol awakening response, and post-exertional malaise in Chronic Fatigue Syndrome. Psychoneuroendocrinology 2014, 49, 26–31. [Google Scholar] [CrossRef]
- Powell, D.J.; Liossi, C.; Moss-Morris, R.; Schlotz, W. Unstimulated cortisol secretory activity in everyday life and its relationship with fatigue and chronic fatigue syndrome: A systematic review and subset meta-analysis. Psychoneuroendocrinology 2013, 38, 2405–2422. [Google Scholar] [CrossRef]
- Rajeevan, M.S.; Smith, A.K.; Dimulescu, I.; Unger, E.R.; Vernon, S.D.; Heim, C.; Reeves, W.C. Glucocorticoid receptor polymorphisms and haplotypes associated with chronic fatigue syndrome. Genes, Brain Behav. 2006, 6, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Wyller, V.B.; Eriksen, H.R.; Malterud, K. Can sustained arousal explain the Chronic Fatigue Syndrome? Behav. Brain Funct. 2009, 5, 10–10. [Google Scholar] [CrossRef]
- Mar, P.L.; Raj, S.R. Postural Orthostatic Tachycardia Syndrome: Mechanisms and New Therapies. Annu. Rev. Med. 2020, 71, 235–248. [Google Scholar] [CrossRef]
- Clinician Coalition ME/CFS Treatment Recommendations US ME/CFS Version 1. (2021). *Bateman Horne Center*. Retrieved from https://batemanhornecenter.org/wp-content/uploads/filebase/Treatment-Recs-MECFS-Clinician-Coalition-V1-Feb.-2021.pdf.
- Sommerfeldt, L.; Portilla, H.; Jacobsen, L.; Gjerstad, J.; Wyller, V.B. Polymorphisms of adrenergic cardiovascular control genes are associated with adolescent chronic fatigue syndrome. Acta Paediatr. 2011, 100, 293–298. [Google Scholar] [CrossRef]
- Hall, K.T.; Kossowsky, J.; Oberlander, T.F.; Kaptchuk, T.J.; Saul, J.P.; Wyller, V.B.; Fagermoen, E.; Sulheim, D.; Gjerstad, J.; Winger, A.; et al. Genetic variation in catechol-O-methyltransferase modifies effects of clonidine treatment in chronic fatigue syndrome. Pharmacogenomics J. 2016, 16, 454–60. [Google Scholar] [CrossRef]
- Polli, A.; Hendrix, J.; Ickmans, K.; Bakusic, J.; Ghosh, M.; Monteyne, D.; Velkeniers, B.; Bekaert, B.; Nijs, J.; Godderis, L. Genetic and epigenetic regulation of Catechol-O-methyltransferase in relation to inflammation in chronic fatigue syndrome and Fibromyalgia. J. Transl. Med. 2022, 20, 1–11. [Google Scholar] [CrossRef]
- Riley, D.S.; Barber, M.S.; Kienle, G.S.; Aronson, J.K.; von Schoen-Angerer, T.; Tugwell, P.; Kiene, H.; Helfand, M.; Altman, D.G.; Sox, H.; et al. CARE guidelines for case reports: explanation and elaboration document. J. Clin. Epidemiol. 2017, 89, 218–235. [Google Scholar] [CrossRef]
- Pan, Y.-W.; Zhang, Y.-N.; Wang, Y.-C.; Lu, Y.-B.; Huang, X.-L.; Lao, Y.-F.; Zhang, L.; Yang, J.; Shi, M.; Ma, H.-L. Myeloperoxidase: a new target for the treatment of stroke? Neural Regen. Res. 2022, 17, 1711–1716. [Google Scholar] [CrossRef]
- Palma-Gudiel, H.; Córdova-Palomera, A.; Leza, J.C.; Fañanás, L. Glucocorticoid receptor gene (NR3C1) methylation processes as mediators of early adversity in stress-related disorders causality: A critical review. Neurosci. Biobehav. Rev. 2015, 55, 520–535. [Google Scholar] [CrossRef]
- Jiménez, K.M.; Pereira-Morales, A.J.; Forero, D.A. MTHFR gene methylation is associated with perceived stress in healthy young adults. Psychiatr. Genet. 2018, 28, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Yin, J.; Miller, A.H.; Xiao, C. A systematic review of the association between fatigue and genetic polymorphisms. Brain, Behav. Immun. 2017, 62, 230–244. [Google Scholar] [CrossRef]
- Prichard, B.N.; Owens, C.W.; Graham, B.R. Pharmacology and clinical use of moxonidine, a new centrally acting sympatholytic antihypertensive agent. 1997, S29–45. [Google Scholar]
- Boot, E.; Booij, J.; Hasler, G.; Zinkstok, J.R.; de Haan, L.; Linszen, D.H.; van Amelsvoort, T.A. AMPT-induced monoamine depletion in humans: evaluation of two alternative [123I]IBZM SPECT procedures. Eur. J. Nucl. Med. 2008, 35, 1350–1356. [Google Scholar] [CrossRef]
- Ballester-Servera, C.; Cañes, L.; Alonso, J.; Puertas, L.; Taurón, M.; Rodríguez, C.; Martínez-González, J. El receptor nuclear NOR-1 (Neuron-derived Orphan Receptor-1) en el remodelado vascular patológico. Clin. E Investig. En Arter. 2022, 34, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Graf, W.D.; Unis, A.S.; Yates, C.M.; Sulzbacher, S.; Dinulos, M.B.; Jack, R.M.; Dugaw, K.A.; Paddock, M.N.; Parson, W.W. Catecholamines in patients with 22q11.2 deletion syndrome and the low-activity COMT polymorphism. Neurology 2001, 57, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Longhurst, J.G.; Carpenter, L.L.; Epperson, C.; Price, L.H.; McDougle, C.J. Effects of catecholamine depletion with AMPT (alpha-methyl-para-tyrosine) in obsessive-compulsive disorder. Biol. Psychiatry 1999, 46, 573–576. [Google Scholar] [CrossRef]
- Voruganti, L.; Msc; Slomka, P.; Msc, P.Z.; Bsc, G.C.; Bsc, A.S.; Mattar, A.; Awad, A.G. Subjective Effects of AMPT-induced Dopamine Depletion in Schizophrenia Correlation between Dysphoric Responses and Striatal D2 Binding Ratios on SPECT Imaging. Neuropsychopharmacology 2001, 25, 642–650. [Google Scholar] [CrossRef]
- Alves, F.d.S.; Bakker, G.; Schmitz, N.; Abeling, N.; Hasler, G.; van der Meer, J.; Nederveen, A.; de Haan, L.; Linszen, D.; van Amelsvoort, T. Dopaminergic modulation of the reward system in schizophrenia: A placebo-controlled dopamine depletion fMRI study. Eur. Neuropsychopharmacol. 2013, 23, 1577–1586. [Google Scholar] [CrossRef]
- Miller, H.L.; Delgado, P.L.; Salomon, R.M.; Heninger, G.R.; Charney, D.S. Effects of α-methyl-para-tyrosine (AMPT) in drug-free depressed patients. Neuropsychopharmacology 1996, 14, 151–157. [Google Scholar] [CrossRef]
| Biochemical target | Normal range | Unit | Level | Value | Date of measurement |
|---|---|---|---|---|---|
| 5-HIAA (5-Hydroxyindoleacetic Acid) | <2.40 | mg/g | High | 3.05* | Nov 2021 |
| Adrenaline | <20 | μg | Normal | 11 | Aug 2012 |
| Normal | 3.5 | July 2022 | |||
| Normal | 10.22 | March 2023 | |||
| B1 adrenergic receptor autoantibody | <15 | U/ml | High | 17.9* | July 2021 |
| B2 adrenergic receptor autoantibody | <8 | U/ml | High | 13.5* | July 2021 |
| Dopamine (plasma) | <85 | ng/l | Normal | 81 | March 2023 |
| GABA urine/creatinine | 3-13.6 | μmol/g | High | 15.4* | July 2022 |
| Glutamate urine/creatinine | 8- 25 | μmol/gCrea | High | 42.4* | July 2022 |
| M3-muscarinic autoantibodies AChR | <6 | U/ml | High | 8.4* | July 2021 |
| Metanephrine | <350 | μg | Normal | 78 | Aug 2012 |
| High | 427* | March 2023 | |||
| Noradrenaline | <90 | μg | Normal | 77 | Aug 2012 |
| Normal | 19.5 | July 2022 | |||
| Noradrenaline | 70-750 (lying) | ng/l | Normal | 167 | March 2023 |
| Noradrenaline | 200-1700 (standing) | ng/l | Normal | 518 | March 2023 |
| Noradrenaline urine/creatinine | 32-58 | μg/gCrea | High | 64.9* | July 2022 |
| Noradrenaline/Adrenaline | 3-6 | quotient | Normal | 5.6 | July 2022 |
| Normetanephrine | <600 | μg | Normal | 270 | Aug 2012 |
| Quinolinic acid | <5.50 | mg/g | High | 6.11* | Nov 2021 |
| Serotonin | 50-20 | μg/l | Normal | 39.7 | July 2022 |
| Serotonin urine/creatinine | 148-230 | μg/gCrea | Low | 131.9* | July 2022 |
| Taurine | 5.4-31.3 | mg/l | Low | 4.8* | July 2022 |
| Tryptophan | 7.30-12.50 | mg/g | Normal | 9.86 | Nov 2021 |
| Normal | 10.5 | July 2022 | |||
| Tyrosine | 38.5-84.3 | μmol/l | Low | 35.18* | Nov 2021 |
| Low | 36.43* | July 2022 | |||
| VMA (Vanillyl Mandelic Acid) | <3 | mg/g | High | 3.14* | Nov 2021 |
| Gene target | Locus (GRCh38) | rs ID | Genotype | Gene region |
|---|---|---|---|---|
| COMT | 22q11.21 | rs4680 | A/A homozygote | coding |
| MAOA | Xp11.3 | VNTR30bp | High copy number | promoter |
| MAOB | Xp11.3 | rs1799836 | T/T homozygote | intron |
| FKBP5 | 6p21.31 | rs1360780 | C/T heterozygote | intron |
| FKBP5 | 6p21.31 | rs9470080 | C/T heterozygote | intron |
| FKBP5 | 6p21.31 | rs4713916 | G/A heterozygote | intron |
| FKBP5 | 6p21.31 | rs9296158 | G/A heterozygote | intron |
| NR3C1 | 5q31.3 | rs6198 | G/A heterozygote | 3´UTR |
| MTHFR | 1p36.22 | rs1801133 | C/T heterozygote | coding |
| SOD2 | 6q25.3 | rs4880 | C/T heterozygote | coding |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




