Submitted:
18 June 2024
Posted:
18 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sampling Location and Sample Collection
2.2. Physiochemical Properties
2.3. DNA Isolation
2.4. Whole Genome Library Preparation and Sequencing
2.5. Diversity Analysis and Taxonomy Classification
2.6. CAZy Annotation
2.7. Statistical Analysis
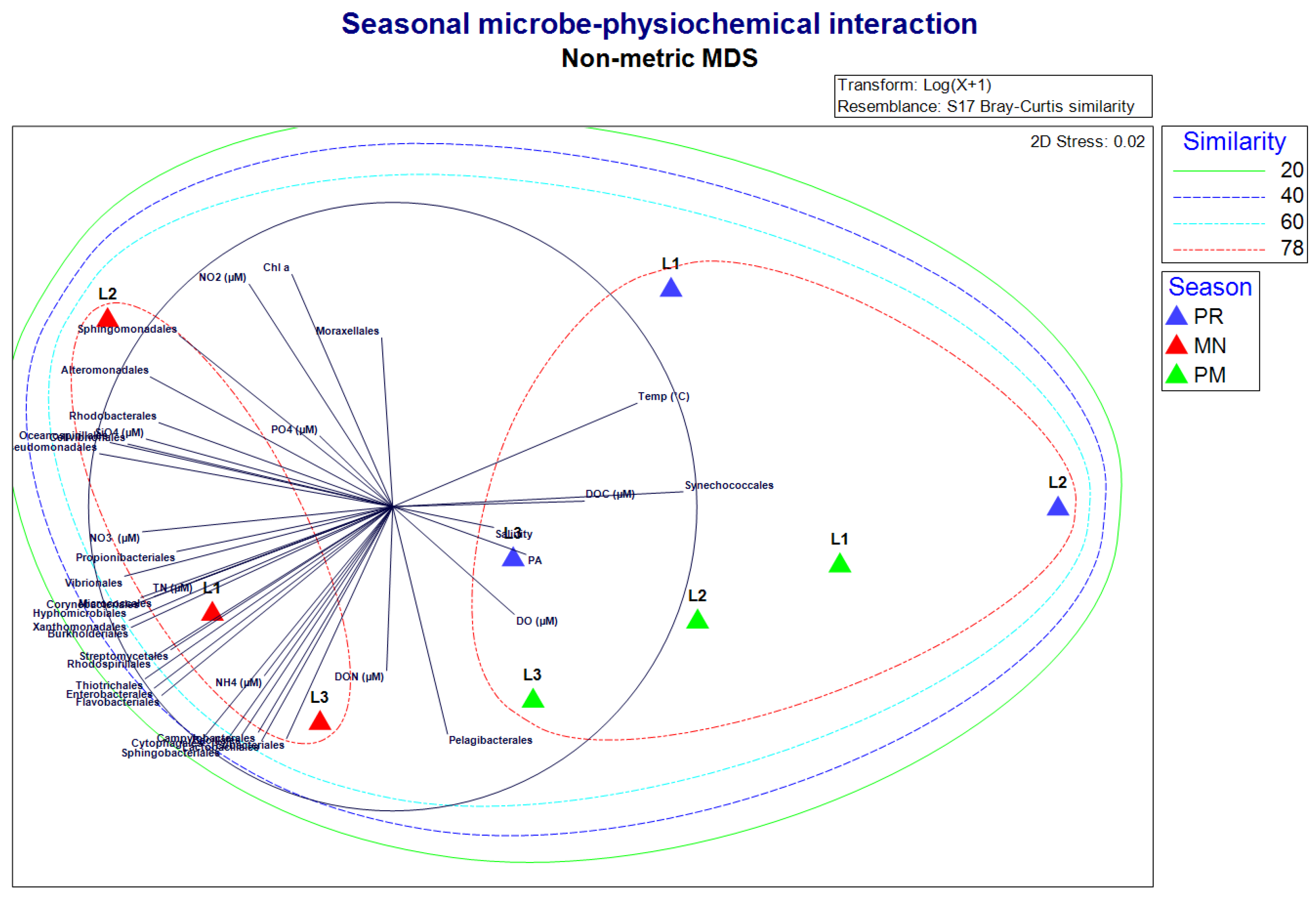
3. Results
3.1. Estimation of Physiochemical Parameters
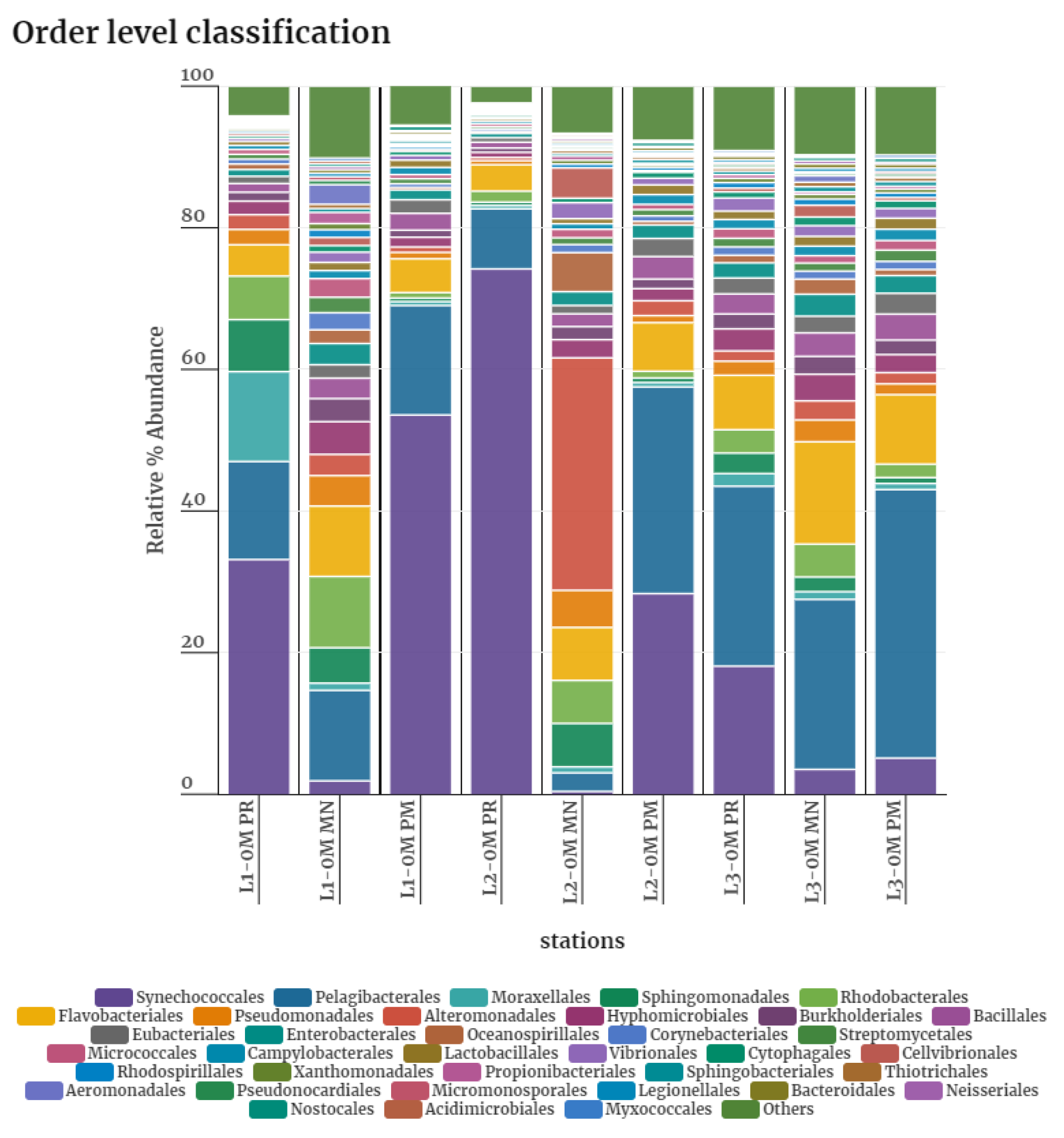
3.2. Bacterial Diversity
3.3. Taxonomic Diversity at the Phylum Level
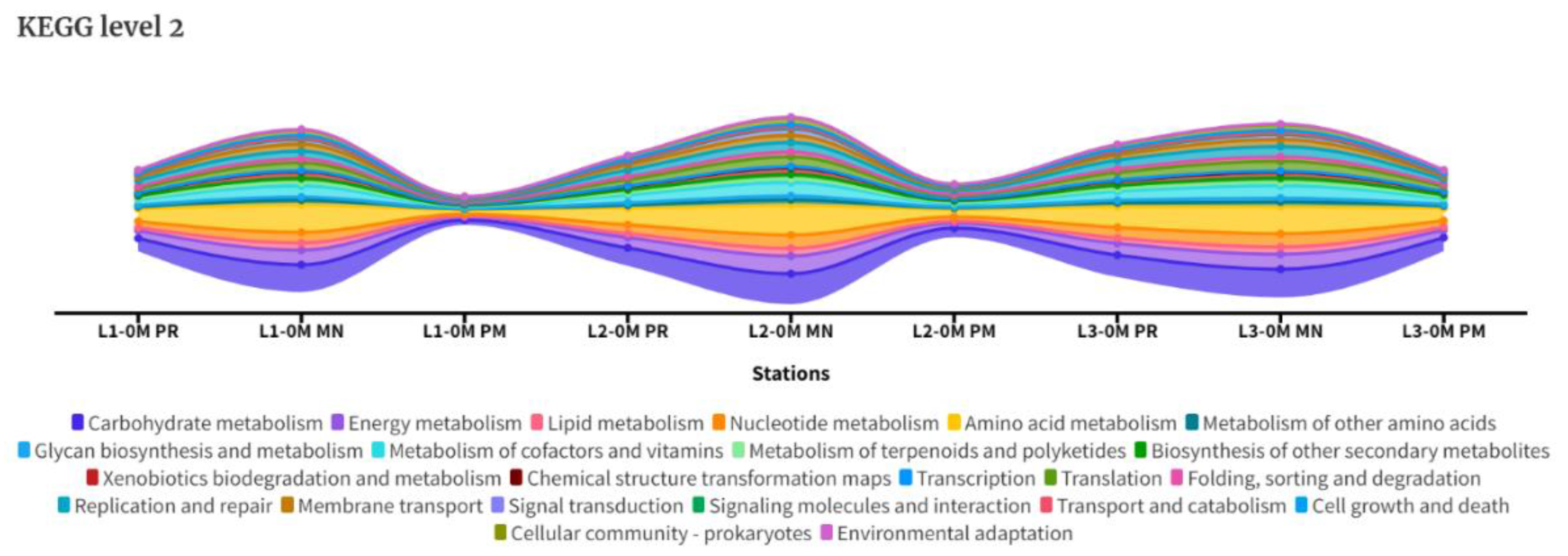
3.4. KEGG Analysis
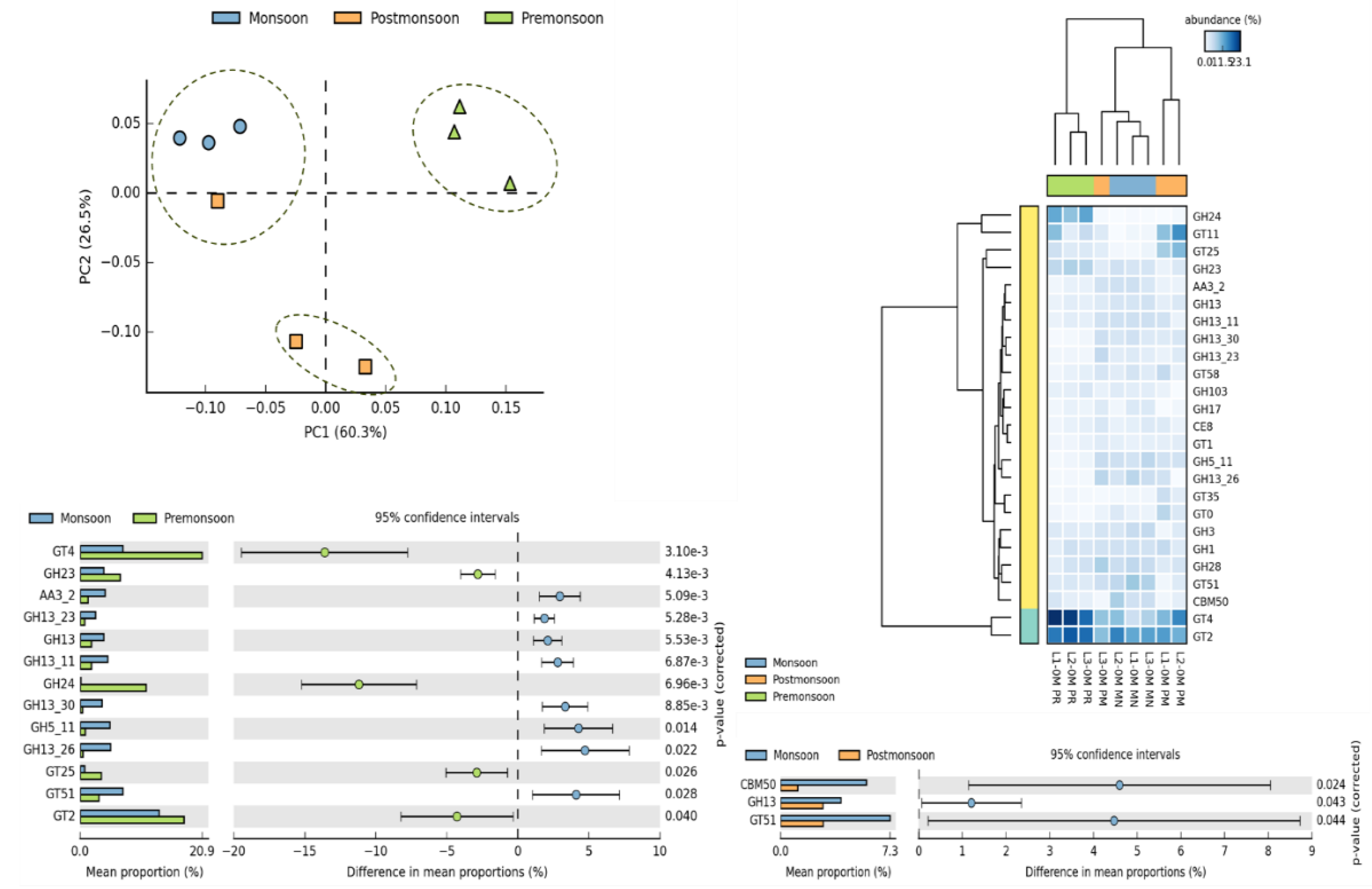
3.5. CAZymes
3.6. Statistical Analysis
4. Discussion
4.1. Physicochemical Settings during the Study
4.2. Taxonomic Distribution during PR
4.3. Taxonomic Distribution during PM
4.4. Taxonomic Distribution during MN
4.5. Functional Analysis
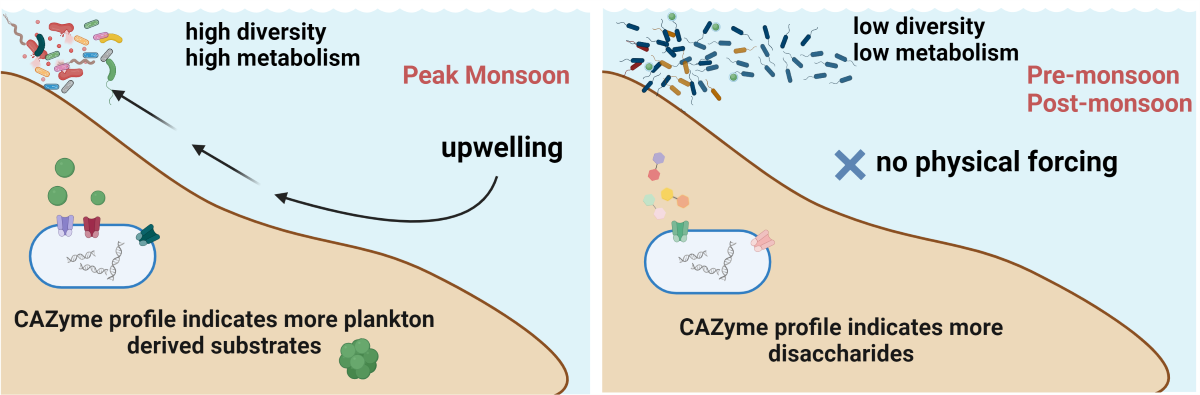
- Increased nutrient availability: During the spring intermonsoon period (PR), surface layers of the Arabian Sea are nutrient-depleted, but during summer monsoon, phytoplankton growth is fueled by upwelling events. This upwelling brought on nutrient-rich cold subsurface water into the euphotic zone, with reported three-fold increases in nitrate compared to surrounding areas (Figure S3). Consequently, high productivity occurs in the Arabian Sea during this season, providing increased organic material for bacterial metabolism [76]. Earlier research conducted in the South China Sea [63], Western Subtropical Pacific [77], and Arabian Sea [78]corroborated these findings.
- Riverine Influxes with Anthropogenic Inputs: August correspond24s to one of the peak riverine runoff months [79]. Our study corroborates previous findings [80,81] as it demonstrates a higher nutrient concentrations during the MN period and these could also be anthropogenic driven. Studies shows a 4-6 fold increase in anthropogenic nutrient flux in the past 50 years [82,83] (Figure S3). Research indicates that riverine ecosystems exhibit higher bacterial abundance compared to intertidal and ocean systems [84]. Thus the riverine run off during monsoon could positively contribute to the bacterial diversity.
- Chlorophyll a (Chl a) as a Proxy for Phytoplankton Biomass: High Chl a levels indicate elevated productivity and organic matter availability, supporting increased bacterial growth [85]. Enhanced levels of nitrates, nitrites, and silicates directly promote phytoplankton growth [86], which contributes significantly during the MN. Such enhanced levels of nutrients were observed during this study. Moreover, Upwelling positively influence the phytoplankton blooms [87] and researchers have investigated for decades, the relationship between bacteria and algae involving the assimilation and remineralization of phytoplankton-derived organic matter by bacteria [88,89]. All these attributes to the conclusion that phytoplankton blooms due to upwelled nutrients could be a reason for higher bacterial diversity during MN in the study region.
- Deeper Water Communities: Bacterial communities adapted to deeper waters ascend to the surface during the MN, sustained by optimal conditions created during upwelling—lower temperatures, higher nutrients, and salinity [86]. These communities revert to their native compositions when surface water conditions return to normal physicochemical levels. Recent studies in ocean systems indicate that deeper waters harbour more diverse bacterial community than surface waters [90]. During upwelling events, these diverse communities ascend to the surface and possibly attempt to adapt to the environment.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- S. P. Kumar, R. P. Roshin, J. Narvekar, P. D. Kumar, and E. Vivekanandan, ‘What drives the increased phytoplankton biomass in the Arabian Sea?’, Current Science, pp. 101–106, 2010.
- J. Retnamma, K. Chinnadurai, J. Loganathan, A. Nagarathinam, P. Singaram, and A. K. Jose, ‘Ecological responses of autotrophic microplankton to the eutrophication of the coastal upwelling along the Southwest coast of India’, Environmental Science and Pollution Research, vol. 28, pp. 11401–11414, 2021.
- S. Parab, A. S. Jagtap, R. M. Meena, and C. S. Manohar, ‘Bacterial dynamics along the west coast of India during the non-monsoon and monsoon season’, Continental Shelf Research, vol. 251, p. 104876, Dec. 2022. [CrossRef]
- S. Prakash and R. Ramesh, ‘Is the Arabian Sea getting more productive?’, CURRENT SCIENCE, vol. 92, no. 5, p. 5, 2007.
- V. Narayanan Nampoothiri S, C. V. Ramu, K. Rasheed, Y. V. B. Sarma, and G. V. M. Gupta, ‘Observational evidence on the coastal upwelling along the northwest coast of India during summer monsoon’, Environ Monit Assess, vol. 194, no. 1, p. 5, Dec. 2021. [CrossRef]
- K. J. Albin et al., ‘Distinctive phytoplankton size responses to the nutrient enrichment of coastal upwelling and winter convection in the eastern Arabian Sea’, Progress in Oceanography, vol. 203, p. 102779, Apr. 2022. [CrossRef]
- C. K. Sherin et al., ‘Nutriclines and nutrient stoichiometry in the eastern Arabian Sea: Intra-annual variations and controlling mechanisms’, Progress in Oceanography, vol. 215, p. 103048, Jul. 2023. [CrossRef]
- R. T. Barber et al., ‘Primary productivity and its regulation in the Arabian Sea during 1995’, Deep Sea Research Part II: Topical Studies in Oceanography, vol. 48, no. 6–7, pp. 1127–1172, 2001.
- J. I. Goes, P. G. Thoppil, H. do R. Gomes, and J. T. Fasullo, ‘Warming of the Eurasian landmass is making the Arabian Sea more productive’, Science, vol. 308, no. 5721, pp. 545–547, 2005.
- S. Smith, ‘The Arabian Sea of the 1990s: New biogeochemical understanding’, Progress in Oceanography, vol. 2, no. 65, pp. 113–115, 2005.
- C. Bird, J. Martinez Martinez, A. G. O’Donnell, and M. Wyman, ‘Spatial distribution and transcriptional activity of an uncultured clade of planktonic diazotrophic γ-proteobacteria in the Arabian Sea’, Applied and environmental microbiology, vol. 71, no. 4, pp. 2079–2085, 2005.
- J. Barcelos e Ramos et al., ‘Nutrient-specific responses of a phytoplankton community: a case study of the North Atlantic Gyre, Azores’, Journal of Plankton Research, vol. 39, no. 4, pp. 744–761, 2017.
- M. P. Sison-Mangus, S. Jiang, R. M. Kudela, and S. Mehic, ‘Phytoplankton-Associated Bacterial Community Composition and Succession during Toxic Diatom Bloom and Non-Bloom Events’, Frontiers in Microbiology, vol. 7, 2016, Accessed: Feb. 18, 2023. [Online]. Available: https://www.frontiersin.org/articles/10.3389/fmicb.2016.01433.
- N. Arandia-Gorostidi et al., ‘Novel Interactions Between Phytoplankton and Bacteria Shape Microbial Seasonal Dynamics in Coastal Ocean Waters’, Frontiers in Marine Science, vol. 9, 2022, Accessed: Feb. 18, 2023. [Online]. Available: https://www.frontiersin.org/articles/10.3389/fmars.2022.901201.
- V. Fernandes and K. Bogati, ‘Analysis of Bacteria–Phytoplankton relationships at three discrete locations in the Eastern Arabian Sea during winter’, Continental Shelf Research, vol. 243, p. 104751, 2022.
- B. S. Halpern et al., ‘A global map of human impact on marine ecosystems’, science, vol. 319, no. 5865, pp. 948–952, 2008.
- B. Nogales, M. P. Lanfranconi, J. M. Piña-Villalonga, and R. Bosch, ‘Anthropogenic perturbations in marine microbial communities’, FEMS Microbiology reviews, vol. 35, no. 2, pp. 275–298, 2011.
- V. Sachithanandam et al., ‘Microbial diversity from the continental shelf regions of the Eastern Arabian Sea: A metagenomic approach’, Saudi Journal of Biological Sciences, vol. 27, no. 8, pp. 2065–2075, Aug. 2020. [CrossRef]
- F. Muller-Karger et al., ‘Annual cycle of primary production in the Cariaco Basin: Response to upwelling and implications for vertical export’, Journal of Geophysical Research: Oceans, vol. 106, no. C3, pp. 4527–4542, 2001.
- K. Grasshoff, ‘Determination of nutrients’, Methods of seawater analysis, pp. 125–187, 1983.
- E. García-Robledo, A. Corzo, and S. Papaspyrou, ‘A fast and direct spectrophotometric method for the sequential determination of nitrate and nitrite at low concentrations in small volumes’, Marine Chemistry, vol. 162, pp. 30–36, 2014.
- E. Yakushev, E. Vinogradova, A. Dubinin, A. Kostyleva, N. Men’shikova, and S. V. Pakhomova, ‘On determination of low oxygen concentrations with Winkler technique’, Oceanology, vol. 52, Feb. 2012. [CrossRef]
- M. Kanehisa and S. Goto, ‘KEGG: kyoto encyclopedia of genes and genomes’, Nucleic Acids Res, vol. 28, no. 1, pp. 27–30, Jan. 2000. [CrossRef]
- E. Drula, M.-L. Garron, S. Dogan, V. Lombard, B. Henrissat, and N. Terrapon, ‘The carbohydrate-active enzyme database: functions and literature’, Nucleic Acids Research, vol. 50, no. D1, pp. D571–D577, Jan. 2022. [CrossRef]
- M. Anderson, ‘PERMANOVA+ for PRIMER: guide to software and statistical methods.’, Primer-E Limited., 2008.
- D. H. Parks, G. W. Tyson, P. Hugenholtz, and R. G. Beiko, ‘STAMP: statistical analysis of taxonomic and functional profiles’, Bioinformatics, vol. 30, no. 21, pp. 3123–3124, 2014.
- J. Marra and R. T. Barber, ‘Primary productivity in the Arabian Sea: A synthesis of JGOFS data’, Progress in Oceanography, vol. 65, no. 2, pp. 159–175, May 2005. [CrossRef]
- S. Bauer, G. L. Hitchcock, and D. B. Olson, ‘Influence of monsoonally-forced Ekman dynamics upon surface layer depth and plankton biomass distribution in the Arabian Sea’, Deep Sea Research Part A. Oceanographic Research Papers, vol. 38, no. 5, pp. 531–553, 1991.
- C. Jayaram, N. Chacko, K. A. Joseph, and A. Balchand, ‘Interannual variability of upwelling indices in the southeastern Arabian Sea: a satellite based study’, Ocean Science Journal, vol. 45, pp. 27–40, 2010.
- R. Jyothibabu et al., ‘Impact of freshwater influx on microzooplankton mediated food web in a tropical estuary (Cochin backwaters – India)’, Estuarine, Coastal and Shelf Science, vol. 69, no. 3, pp. 505–518, Sep. 2006. [CrossRef]
- M. Madhupratap et al., ‘Arabian Sea oceanography and fisheries of the west coast of India’, Current Science, vol. 81, no. 4, pp. 355–361, 2001.
- P. Muraleedharan and S. Prasannakumar, ‘Arabian Sea upwelling-A comparison between coastal and open ocean regions’, 1996.
- B. Smitha, V. Sanjeevan, K. Vimalkumar, and C. Revichandran, ‘On the upwelling off the southern tip and along the west coast of India’, Journal of Coastal Research, no. 24, pp. 95–102, 2008.
- H. L. Roman-Stork, B. Subrahmanyam, and V. S. N. Murty, ‘The Role of Salinity in the Southeastern Arabian Sea in Determining Monsoon Onset and Strength’, Journal of Geophysical Research: Oceans, vol. 125, no. 1, p. e2019JC015592, 2020. [CrossRef]
- G. Gupta et al., ‘The world’s largest coastal deoxygenation zone is not anthropogenically driven’, Environmental Research Letters, vol. 16, no. 5, p. 054009, 2021.
- R. J. Newton et al., ‘Genome characteristics of a generalist marine bacterial lineage’, The ISME journal, vol. 4, no. 6, pp. 784–798, 2010.
- T. O. Delmont et al., ‘Nitrogen-fixing populations of Planctomycetes and Proteobacteria are abundant in surface ocean metagenomes’, Nature microbiology, vol. 3, no. 7, pp. 804–813, 2018.
- A. L. Hauptmann et al., ‘Bacterial diversity in snow on North Pole ice floes’, Extremophiles, vol. 18, pp. 945–951, 2014.
- J. A. Huber et al., ‘Microbial population structures in the deep marine biosphere’, science, vol. 318, no. 5847, pp. 97–100, 2007.
- Z. Zhou, P. Q. Tran, K. Kieft, and K. Anantharaman, ‘Genome diversification in globally distributed novel marine Proteobacteria is linked to environmental adaptation’, ISME J, vol. 14, no. 8, pp. 2060–2077, Aug. 2020. [CrossRef]
- C. Molina-Pardines, J. M. Haro-Moreno, and M. López-Pérez, ‘Phosphate-related genomic islands as drivers of environmental adaptation in the streamlined marine alphaproteobacterial HIMB59’, MSystems, vol. 8, no. 6, pp. e00898-23, 2023.
- H. P. Nair, R. M. Puthusseri, H. Vincent, and S. G. Bhat, ‘16S rDNA-based bacterial diversity analysis of Arabian Sea sediments: A metagenomic approach’, Ecological Genetics and Genomics, vol. 3–5, pp. 47–51, Nov. 2017. [CrossRef]
- X. Lai, X. Zeng, S. Fang, Y. Huang, L. Cao, and S. Zhou, ‘Denaturing gradient gel electrophoresis (DGGE) analysis of bacterial community composition in deep-sea sediments of the South China Sea’, World Journal of Microbiology and Biotechnology, vol. 22, pp. 1337–1345, 2006.
- M. A. Moran et al., ‘Ecological genomics of marine Roseobacters’, Appl Environ Microbiol, vol. 73, no. 14, pp. 4559–4569, Jul. 2007. [CrossRef]
- R. M. Morris et al., ‘SAR11 clade dominates ocean surface bacterioplankton communities’, Nature, vol. 420, no. 6917, pp. 806–810, Dec. 2002. [CrossRef]
- J. C. Thrash et al., ‘Phylogenomic evidence for a common ancestor of mitochondria and the SAR11 clade’, Sci Rep, vol. 1, p. 13, Jun. 2011. [CrossRef]
- M. S. Rappé, S. A. Connon, K. L. Vergin, and S. J. Giovannoni, ‘Cultivation of the ubiquitous SAR11 marine bacterioplankton clade’, Nature, vol. 418, no. 6898, pp. 630–633, Aug. 2002. [CrossRef]
- J. Vijayan, P. Ammini, and V. K. Nathan, ‘Diversity pattern of marine culturable heterotrophic bacteria in a region with coexisting upwelling and mud banks in the southeastern Arabian Sea’, Environ Sci Pollut Res, vol. 29, no. 3, pp. 3967–3982, Jan. 2022. [CrossRef]
- M. T. Cottrell and D. L. Kirchman, ‘Natural assemblages of marine proteobacteria and members of the Cytophaga-Flavobacter cluster consuming low-and high-molecular-weight dissolved organic matter’, Applied and environmental microbiology, vol. 66, no. 4, pp. 1692–1697, 2000.
- V. Sharma et al., ‘Phylogenomics of the Phylum Proteobacteria: Resolving the Complex Relationships’, Curr Microbiol, vol. 79, no. 8, p. 224, Jun. 2022. [CrossRef]
- H. W. Paerl and T. G. Otten, ‘Harmful cyanobacterial blooms: causes, consequences, and controls’, Microbial ecology, vol. 65, pp. 995–1010, 2013.
- Z. Chen, X. Wang, Y. Song, Q. Zeng, Y. Zhang, and H. Luo, ‘Prochlorococcus have low global mutation rate and small effective population size’, Nat Ecol Evol, vol. 6, no. 2, pp. 183–194, Feb. 2022. [CrossRef]
- F. Partensky, J. Blanchot, and D. Vaulot, ‘Differential distribution and ecology of Prochlorococcus and Synechococcus in oceanic waters: a review’, Bulletin de l’Institut Océanographique - Special issue: Marine cyanobacteria, vol. 19, pp. 457–476, Jul. 1999.
- H. Cheng et al., ‘Influence of phosphorus fertilization patterns on the bacterial community in upland farmland’, Industrial Crops and Products, vol. 155, p. 112761, Nov. 2020. [CrossRef]
- P. R. Gómez-Pereira et al., ‘Genomic content of uncultured Bacteroidetes from contrasting oceanic provinces in the North Atlantic Ocean’, Environmental Microbiology, vol. 14, no. 1, pp. 52–66, 2012.
- D. L. Kirchman, ‘The ecology of Cytophaga–Flavobacteria in aquatic environments’, FEMS microbiology ecology, vol. 39, no. 2, pp. 91–100, 2002.
- P. G. Thoppil, A. J. Wallcraft, and T. G. Jensen, ‘Winter convective mixing in the northern Arabian Sea during contrasting monsoons’, Journal of Physical Oceanography, vol. 52, no. 3, pp. 313–327, 2022.
- L. Alonso-Sáez et al., ‘Transcriptional Mechanisms of Thermal Acclimation in Prochlorococcus’, mBio, vol. 14, no. 3, pp. e03425-22, Apr. 2023. [CrossRef]
- C. Wu et al., ‘Diversity, structure, and distribution of bacterioplankton and diazotroph communities in the Bay of Bengal during the winter monsoon’, Front. Microbiol., vol. 13, Nov. 2022. [CrossRef]
- A. Vuillemin, ‘Nitrogen cycling activities during decreased stratification in the coastal oxygen minimum zone off Namibia’, Front Microbiol, vol. 14, p. 1101902, Feb. 2023. [CrossRef]
- J. Blanchot, M. Rodier, and A. Le Bouteiller, ‘Effect of El Niño Southern Oscillation events on the distribution and abundance of phytoplankton in the Western Pacific Tropical Ocean along 165°E’, Journal of Plankton Research, vol. 14, no. 1, pp. 137–156, Jan. 1992. [CrossRef]
- Y. Liu et al., ‘Cell surface acid-base properties of the cyanobacterium Synechococcus: Influences of nitrogen source, growth phase and N:P ratios’, Geochimica et Cosmochimica Acta, vol. 187, pp. 179–194, Aug. 2016. [CrossRef]
- X. Liu, N. Xie, J. Li, M. Bai, B. Sen, and G. Wang, ‘Potential Contribution of Coastal Upwelling to Carbon Sink through Interaction between Cyanobacteria and Microbial Eukaryotes’, Water, vol. 14, no. 19, Art. no. 19, Jan. 2022. [CrossRef]
- C. Nikolova and T. Gutierrez, ‘Chapter 6 - Novel approaches in the use of biosurfactants in the oil industry and environmental remediation’, in Biosurfactants, G. Soberón-Chávez, Ed., in Foundations and Frontiers in Enzymology., Academic Press, 2023, pp. 107–128. [CrossRef]
- F. Bian et al., ‘Genome sequences of six Pseudoalteromonas strains isolated from Arctic sea ice’, J Bacteriol, vol. 194, no. 4, pp. 908–909, Feb. 2012. [CrossRef]
- D. Li, J. O. Sharp, and J. E. Drewes, ‘Microbial genetic potential for xenobiotic metabolism increases with depth during biofiltration’, Environ. Sci.: Processes Impacts, vol. 22, no. 10, pp. 2058–2069, Oct. 2020. [CrossRef]
- E. Cardenas, J. Kranabetter, G. Hope, K. R. Maas, S. Hallam, and W. W. Mohn, ‘Forest harvesting reduces the soil metagenomic potential for biomass decomposition’, The ISME Journal, vol. 9, no. 11, pp. 2465–2476, 2015.
- L. Manoharan, S. K. Kushwaha, K. Hedlund, and D. Ahrén, ‘Captured metagenomics: large-scale targeting of genes based on ‘sequence capture’reveals functional diversity in soils’, DNA Research, vol. 22, no. 6, pp. 451–460, 2015.
- X. Xia et al., ‘Distinct metabolic strategies of the dominant heterotrophic bacterial groups associated with marine Synechococcus’, Science of The Total Environment, vol. 798, p. 149208, Dec. 2021. [CrossRef]
- L. Jiang et al., ‘Identification and characterization of a novel trehalose synthase gene derived from saline-alkali soil metagenomes’, PLoS One, vol. 8, no. 10, p. e77437, 2013.
- M. Walmagh, R. Zhao, and T. Desmet, ‘Trehalose analogues: latest insights in properties and biocatalytic production’, International Journal of Molecular Sciences, vol. 16, no. 6, pp. 13729–13745, 2015.
- T. Collins, C. Gerday, and G. Feller, ‘Xylanases, xylanase families and extremophilic xylanases’, FEMS microbiology reviews, vol. 29, no. 1, pp. 3–23, 2005.
- M. T. Cottrell, L. Yu, and D. L. Kirchman, ‘Sequence and expression analyses of Cytophaga-like hydrolases in a Western arctic metagenomic library and the Sargasso Sea’, Applied and environmental microbiology, vol. 71, no. 12, pp. 8506–8513, 2005.
- L. E. Taylor, B. Henrissat, P. M. Coutinho, N. A. Ekborg, S. W. Hutcheson, and R. M. Weiner, ‘Complete cellulase system in the marine bacterium Saccharophagus degradans strain 2-40T’, Journal of bacteriology, vol. 188, no. 11, pp. 3849–3861, 2006.
- S. Violot, R. Haser, G. Sonan, D. Georlette, G. Feller, and N. Aghajari, ‘Expression, purification, crystallization and preliminary X-ray crystallographic studies of a psychrophilic cellulase from Pseudoalteromonas haloplanktis’, Acta Crystallographica Section D: Biological Crystallography, vol. 59, no. 7, pp. 1256–1258, 2003.
- B. Kumar and V. Sarma, ‘Variations in concentrations and sources of bioavailable organic compounds in the Indian estuaries and their fluxes to coastal waters’, Continental Shelf Research, vol. 166, pp. 22–33, 2018.
- A.-Y. Tsai, G.-C. Gong, R. W. Sanders, C.-F. Chao, and K.-P. Chiang, ‘Microbial dynamics in an oligotrophic bay of the western subtropical Pacific: Impact of short-term heavy freshwater runoff and upwelling’, Journal of oceanography, vol. 66, pp. 873–883, 2010.
- A. Malik et al., ‘Interactions between trophic levels in upwelling and non-upwelling regions during summer monsoon’, Journal of sea research, vol. 95, pp. 56–69, 2015.
- A. Shivaprasad, J. Vinita, C. Revichandran, N. T. Manoj, K. V. Jayalakshmy, and K. R. Muraleedharan, ‘Ambiguities in the classification of Cochin Estuary, West Coast of India’, Hydrology and Earth System Sciences Discussions, vol. 10, no. 3, pp. 3595–3628, Mar. 2013. [CrossRef]
- W. Shi and M. Wang, ‘Phytoplankton biomass dynamics in the Arabian Sea from VIIRS observations’, Journal of Marine Systems, vol. 227, p. 103670, 2022.
- P. N. M. Vinayachandran et al., ‘Reviews and syntheses: Physical and biogeochemical processes associated with upwelling in the Indian Ocean’, Biogeosciences, vol. 18, no. 22, pp. 5967–6029, Nov. 2021. [CrossRef]
- M. Krishna, M. Prasad, D. Rao, R. Viswanadham, V. Sarma, and N. Reddy, ‘Export of dissolved inorganic nutrients to the northern Indian Ocean from the Indian monsoonal rivers during discharge period’, Geochimica et Cosmochimica Acta, vol. 172, pp. 430–443, 2016.
- G. Martin et al., ‘Eutrophication induced changes in benthic community structure of a flow-restricted tropical estuary (Cochin backwaters), India’, Environmental monitoring and assessment, vol. 176, pp. 427–438, 2011.
- Y. Wang et al., ‘Comparison of the Levels of Bacterial Diversity in Freshwater, Intertidal Wetland, and Marine Sediments by Using Millions of Illumina Tags’, Appl Environ Microbiol, vol. 78, no. 23, pp. 8264–8271, Dec. 2012. [CrossRef]
- A. Debbab, A. H. Aly, W. H. Lin, and P. Proksch, ‘Bioactive compounds from marine bacteria and fungi’, Microbial biotechnology, vol. 3, no. 5, pp. 544–563, 2010.
- P. S. Kumar et al., ‘Influence of nutrient fluxes on phytoplankton community and harmful algal blooms along the coastal waters of southeastern Arabian Sea’, Continental Shelf Research, vol. 161, pp. 20–28, Jun. 2018. [CrossRef]
- A. Ferreira, V. Brotas, C. Palma, C. Borges, and A. C. Brito, ‘Assessing phytoplankton bloom phenology in upwelling-influenced regions using ocean color remote sensing’, Remote Sensing, vol. 13, no. 4, p. 675, 2021.
- A. Buchan, G. R. LeCleir, C. A. Gulvik, and J. M. González, ‘Master recyclers: features and functions of bacteria associated with phytoplankton blooms’, Nature Reviews Microbiology, vol. 12, no. 10, pp. 686–698, 2014.
- C. Costas-Selas et al., ‘Linking the impact of bacteria on phytoplankton growth with microbial community composition and co-occurrence patterns’, Marine Environmental Research, vol. 193, p. 106262, Jan. 2024. [CrossRef]
- M. Bandekar, N. Ramaiah, A. Jain, and R. M. Meena, ‘Seasonal and depth-wise variations in bacterial and archaeal groups in the Arabian Sea oxygen minimum zone’, Deep Sea Research Part II: Topical Studies in Oceanography, vol. 156, pp. 4–18, 2018.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



