Submitted:
17 June 2024
Posted:
18 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Rat Models
2.2. Patients
2.3. Infrared Thermal Imaging
2.4. Infrared Thermal Image Analysis
2.5. Statistical Analysis
3. Results
3.1. Rat Models
3.2. Patients
3.2.1. Patient Demographics & Flap Characteristics
3.2.2. Infrared Thermal Imaging Analysis
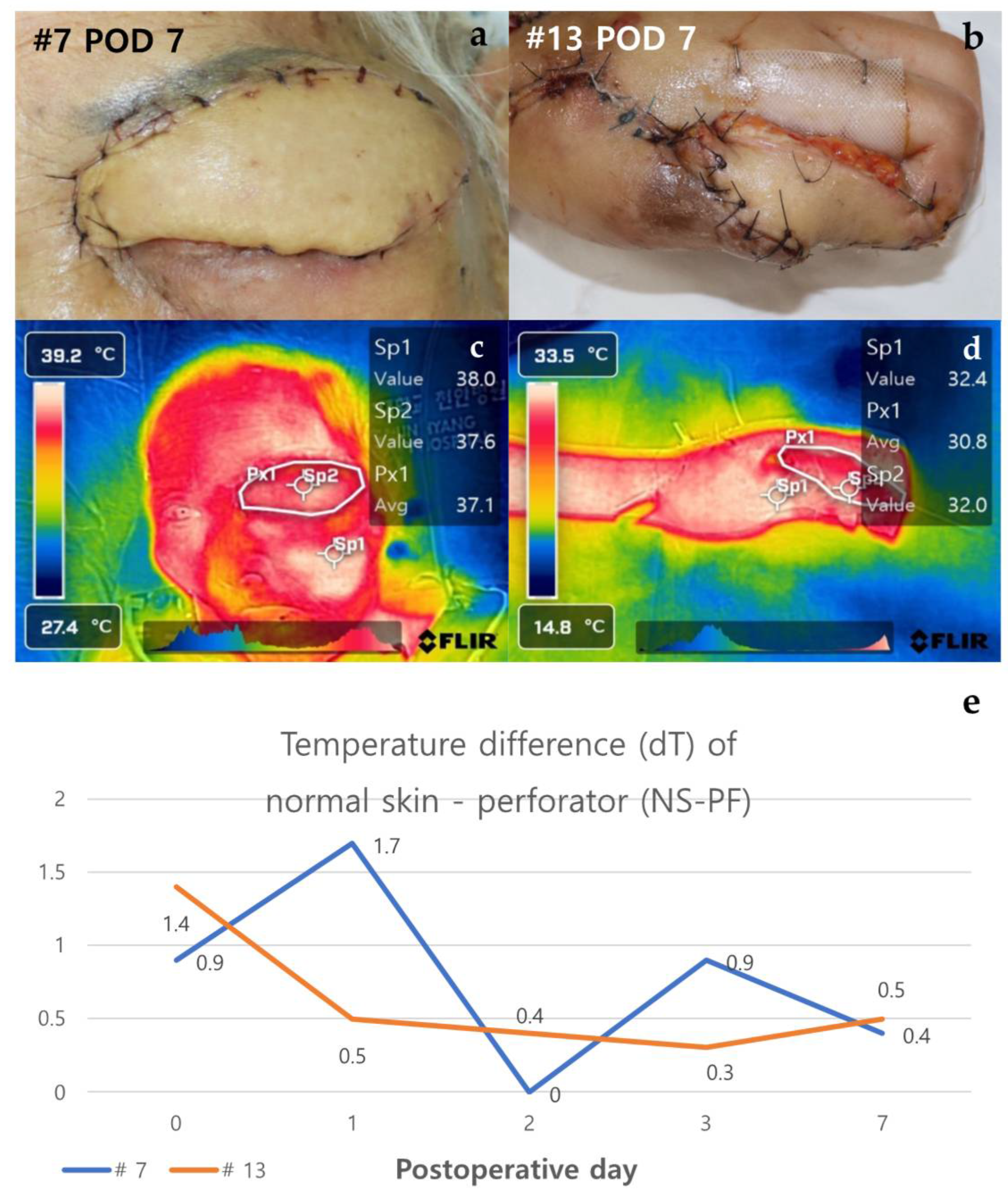
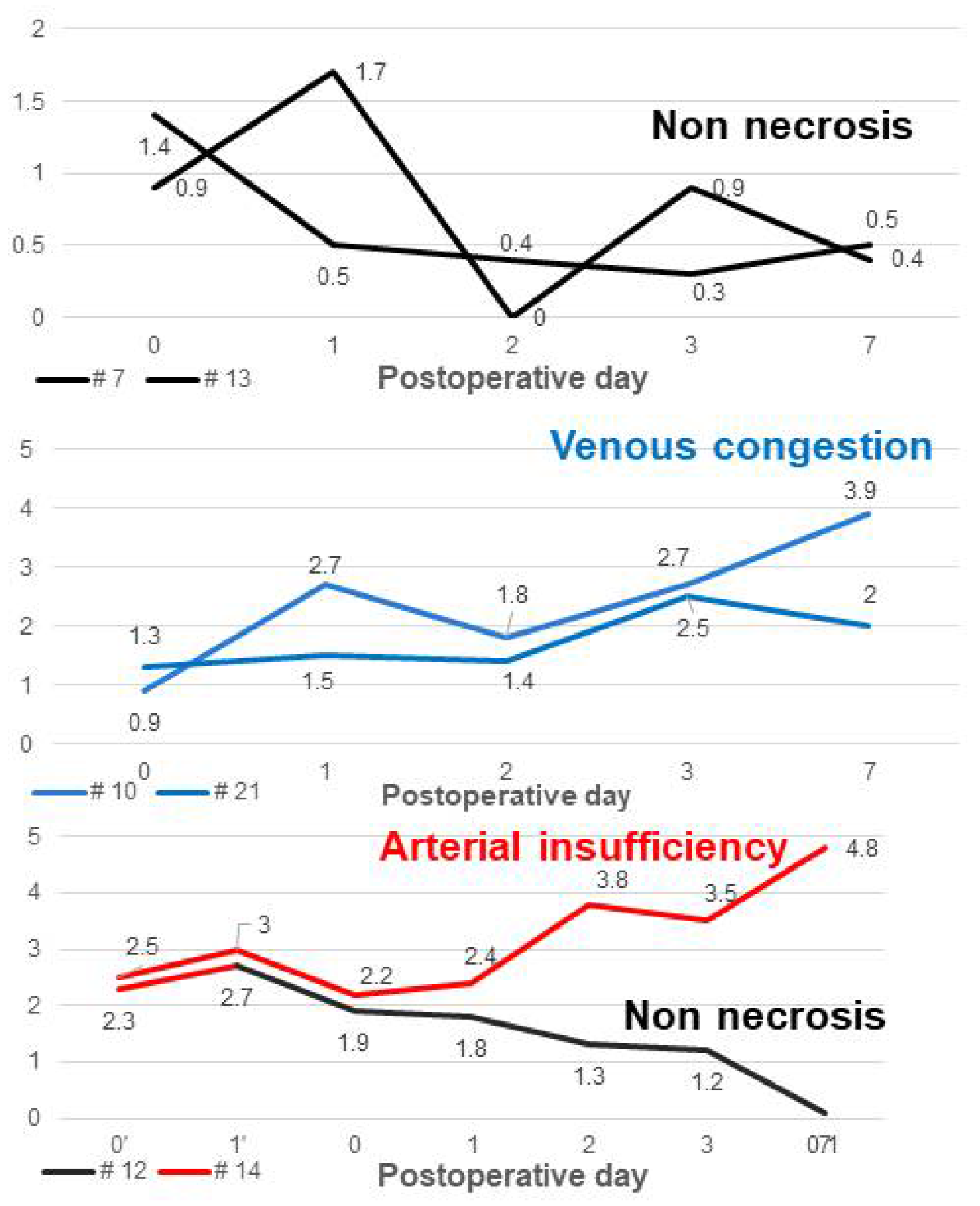
3.2.3. Case of Non-Necrosis Group (Case #7 & Case #13)
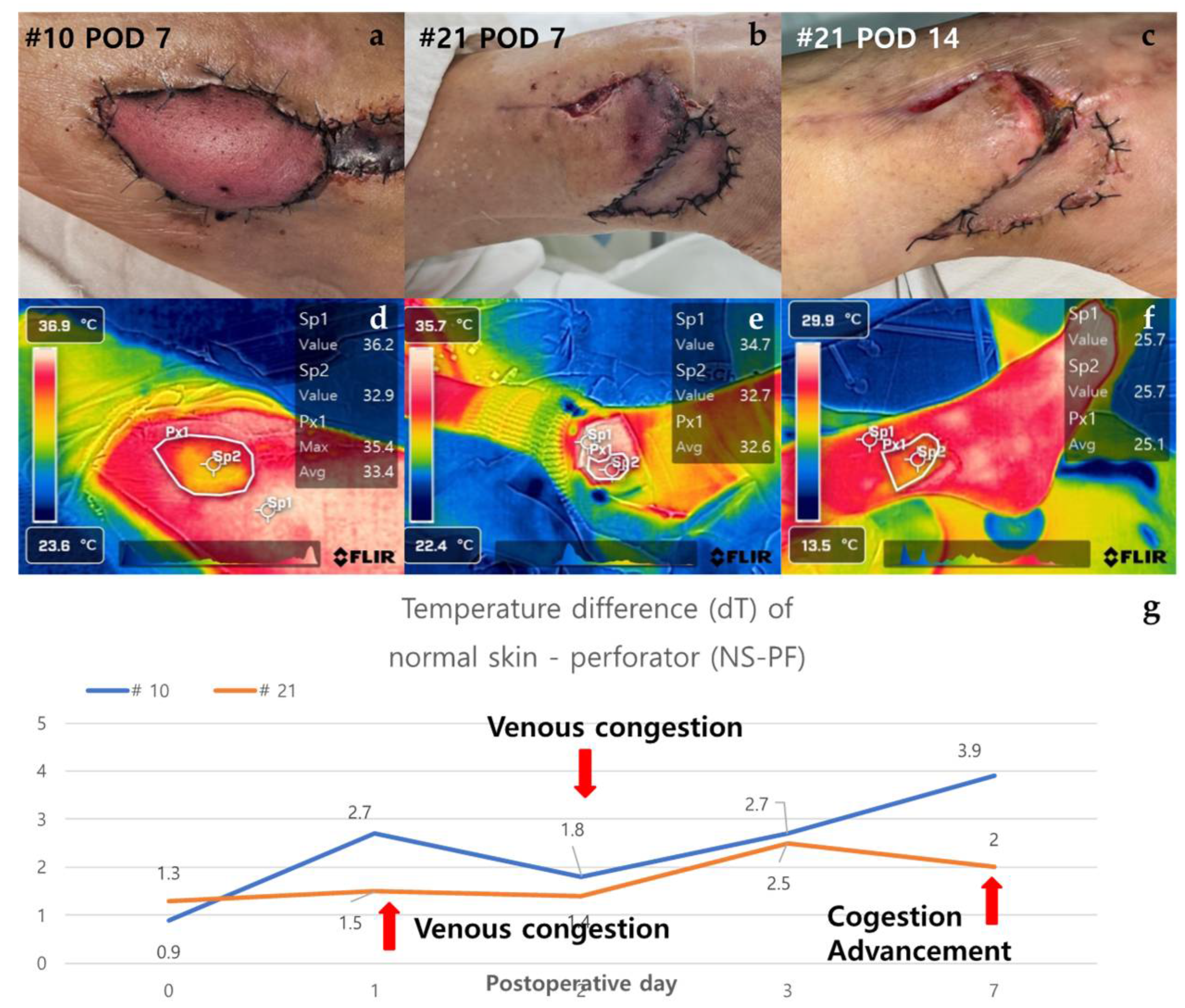
3.2.4. Case of Venous Congestion in Necrosis Group (Case #10 & Case #21)
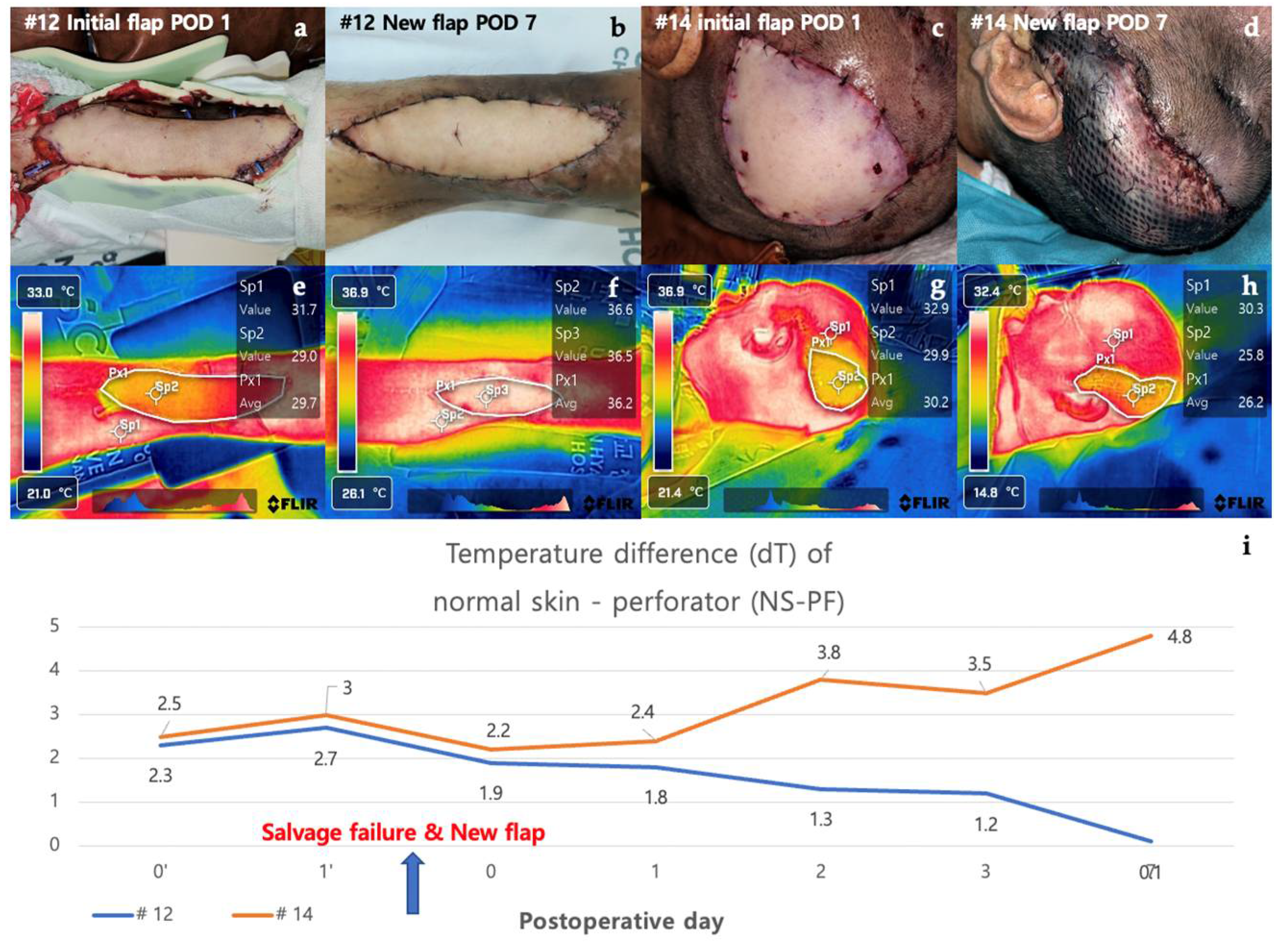
3.2.5. Case in Which a New Flap Was Applied after Salvage Procedure for Arterial Insufficiency (Case #12 & Case #14)
3.2.6. The Difference between the Temperature of Perforator Area and the Average Temperature of Total Flap (dT of PF-AFP).
4. Discussion
5. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gottlieb, L.J.; Krieger, L.M. From the reconstructive ladder to the reconstructive elevator. Plastic and Reconstructive Surgery 1994, 93, 1503–1504. [Google Scholar] [CrossRef] [PubMed]
- Shen, A.Y.; Lonie, S.; Lim, K.; Farthing, H.; Hunter-Smith, D.J.; Rozen, W.M. Free flap monitoring, salvage, and failure timing: A systematic review. Journal of Reconstructive Microsurgery 2021, 37, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Knoedler, S.; Hoch, C.C.; Huelsboemer, L.; Knoedler, L.; Stögner, V.A.; Pomahac, B.; Kauke-Navarro, M.; Colen, D. Postoperative free flap monitoring in reconstructive surgery-man or machine? Frontiers in Surgery 2023, 10, 1130566. [Google Scholar] [CrossRef] [PubMed]
- Hosein, R.C.; Cornejo, A.; Wang, H.T. Postoperative monitoring of free flap reconstruction: A comparison of external Doppler ultrasonography and the implantable Doppler probe. Plastic Surgery (Oakville) 2016, 24, 11–19. [Google Scholar] [CrossRef]
- Rabbani, M.J.; Bhatti, A.Z.; Shahzad, A. Flap monitoring using thermal imaging camera: A contactless method. Journal of the College of Physicians and Surgeons Pakistan 2021, 30, 703–706. [Google Scholar]
- John, H.E.; Niumsawatt, V.; Rozen, W.M.; Whitaker, I.S. Clinical applications of dynamic infrared thermography in plastic surgery: A systematic review. Gland Surgery 2016, 5, 122–132. [Google Scholar]
- Hennessy, O.; Potter, S.M. Use of infrared thermography for the assessment of free flap perforators in autologous breast reconstruction: A systematic review. JPRAS Open 2019, 23, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Sittitrai, P.; Ruenmarkkaew, D.; Klibngern, H. Pedicled flaps versus free flaps for oral cavity cancer reconstruction: A comparison of complications, hospital costs, and functional outcomes. International Archives of Otorhinolaryngology 2022, 27, e32–e42. [Google Scholar] [CrossRef]
- Neusner, A.D.; Pribaz, J.J.; Guo, L. Free your mind, not your flap. Plastic and Reconstructive Surgery - Global Open 2022, 10, e4384. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.S.; Kontos, M. Autologous tissue immediate breast reconstruction: Desired but oncologically safe? International Journal of Clinical Practice 2009, 63, 1642–1646. [Google Scholar] [CrossRef]
- Suh, J.M.; Chung, C.H.; Chang, Y.J. Head and neck reconstruction using free flaps: A 30-year medical record review. Archives of Craniofacial Surgery 2021, 22, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Khouri, R.K.; Shaw, W.W. Monitoring of free flaps with surface-temperature recordings: Is it reliable? Plastic and Reconstructive Surgery 1992, 89, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, G.K.; Hwang, B.H.; Zhang, Y.; Monahan, J.F.; Davis, G.B.; Lee, Y.S.; Ragina, N.P.; Wang, C.; Zhou, Z.Y.; Hong, Y.K.; Spivak, R.M.; Wong, A.K. Novel biomarkers of arterial and venous ischemia in microvascular flaps. PLoS One 2013, 8, e71628. [Google Scholar] [CrossRef] [PubMed]
- Amon, M.; Menger, M.D.; Vollmar, B. Heme oxygenase and nitric oxide synthase mediate cooling-associated protection against TNF-alpha-induced microcirculatory dysfunction and apoptotic cell death. FASEB Journal 2003, 17, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Menger, M.D.; Rücker, M.; Vollmar, B. Capillary dysfunction in striated muscle ischemia/reperfusion: On the mechanisms of capillary “no-reflow”. Shock 1997, 8, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Akita, S. Advances and challenges in perforator flap surgery. Global Health & Medicine 2020, 2, 114–121. [Google Scholar] [CrossRef]
- Shen, A.Y.; Lonie, S.; Lim, K.; Farthing, H.; Hunter-Smith, D.J.; Rozen, W.M. Free flap monitoring, salvage, and failure timing: A systematic review. Journal of Reconstructive Microsurgery 2021, 37, 300–308. [Google Scholar] [CrossRef]
- Chao, A.H.; Meyerson, J.; Povoski, S.P.; Kocak, E. A review of devices used in the monitoring of microvascular free tissue transfers. Expert Review of Medical Devices 2013, 10, 649–660. [Google Scholar] [CrossRef]
- Nguyen, G.K.; Hwang, B.H.; Zhang, Y.; Monahan, J.F.; Davis, G.B.; Lee, Y.S.; Ragina, N.P.; Wang, C.; Zhou, Z.Y.; Hong, Y.K.; et al. Novel biomarkers of arterial and venous ischemia in microvascular flaps. PLoS One 2013, 8, e71628. [Google Scholar] [CrossRef]
- Lohman, R.F.; Langevin, C.J.; Bozkurt, M.; Kundu, N.; Djohan, R. A prospective analysis of free flap monitoring techniques: Physical examination, external Doppler, implantable Doppler, and tissue oximetry. Journal of Reconstructive Microsurgery 2013, 29, 51–56. [Google Scholar] [CrossRef]
- Chao, A.H.; Meyerson, J.; Povoski, S.P.; Kocak, E. A review of devices used in the monitoring of microvascular free tissue transfers. Expert Review of Medical Devices 2013, 10, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Shen, Z.; Shao, Z.; Yu, P. Free flap monitoring using near-infrared spectroscopy: A systemic review. Annals of Plastic Surgery 2016, 76, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Ricci, J.A.; Vargas, C.R.; Lin, S.J.; Tobias, A.M.; Taghinia, A.H.; Lee, B.T. A novel free flap monitoring system using tissue oximetry with text message alerts. Journal of Reconstructive Microsurgery 2016, 32, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jiao, L.; Chen, S.; Li, Z.; Xiao, Y.; Du, F.; Huang, J.; Long, X. Flap perfusion assessment with indocyanine green angiography in deep inferior epigastric perforator flap breast reconstruction: A systematic review and meta-analysis. Microsurgery 2023, 43, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.W.; Hwang, Y.S.; Byeon, J.Y.; Kim, J.H.; Choi, H.J. Does the advantage of transcutaneous oximetry measurements in diabetic foot ulcer apply equally to free flap reconstruction? World Journal of Clinical Cases 2023, 11, 7570–7582. [Google Scholar] [CrossRef] [PubMed]
- Nam, H.J.; Wee, S.Y.; Kim, S.Y.; Jeong, H.G.; Lee, D.W.; Byeon, J.Y.; Park, S.H.; Choi, H.J. The correlation between transcutaneous oxygen pressure (TcPO2) and forward-looking infrared (FLIR) thermography in the evaluation of lower extremity perfusion according to angiosome. International Wound Journal. [CrossRef]
- Asif 2023, A.; Poyiatzis, C.; Raheman, F.J. The use of infrared thermography (IRT) in burns depth assessment: A diagnostic accuracy meta-analysis. European Burn Journal 2022, 3, 432–446. [Google Scholar] [CrossRef]
- Berner, J.E.; Pereira, N.; Troisi, L.; Will, P.; Nanchahal, J.; Jain, A. Accuracy of infrared thermography for perforator mapping: A systematic review and meta-analysis of diagnostic studies. Journal of Plastic 2021, Reconstructive & Aesthetic Surgery, 74, 1173-1179.
- Park, C. Applications of the reverse McFarlane flap in hand surgery. Journal of Hand Surgery 2021, 46, 432–439. [Google Scholar]
- Lee, Y.S.; Kim, S.H. Reconstructive techniques using reverse McFarlane flaps. Plastic and Reconstructive Surgery 2022, 49, 210–215. [Google Scholar]
- Patel, R.; Tan, E. Vascular considerations in reverse McFarlane flap procedures. International Journal of Reconstructive Surgery 2020, 36, 95–102. [Google Scholar]
- Nguyen, A.; Patel, V. Retrograde flap hemodynamics and clinical applications. Journal of Microsurgery 2022, 39, 315–320. [Google Scholar]
- Sowa, M.G.; Friesen, J.R.; Levasseur, M.; Schattka, B.; Sigurdson, L.; Hayakawa, T. The utility of near infrared imaging in intra-operative prediction of flap outcome: A reverse McFarlane skin flap model study. Journal of Near Infrared Spectroscopy 2012, 20, 601–615. [Google Scholar] [CrossRef]
- Ibne Mahbub, M.S.; Kim, Y.J.; Choi, H.; Lee, B.T. Papaverine loaded injectable and thermosensitive hydrogel system for improving survival of rat dorsal skin flaps. Journal of Materials Science: Materials in Medicine 2023, 34, 28. [Google Scholar] [CrossRef] [PubMed]
- Czapla, N.; Lokaj, M.; Falkowski, A.; Prowans, P. The use of thermography to design tissue flaps - experimental studies on animals. Videosurgery and Other Miniinvasive Techniques 2014, 9, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, R.; Lorenzen, J.; Knobloch, K.; Papst, S.; Kabbani, M.; Koennecker, S.; Vogt, P.M. Free flap microcirculatory monitoring correlates to free flap temperature assessment. Journal of Plastic 2011, Reconstructive & Aesthetic Surgery, 64, 1353-1358.
- Hummelink, S.; Kruit, A.S.; van Vlaenderen, A.R.W.; et al. Post-operative monitoring of free flaps using a low-cost thermal camera: A pilot study. European Journal of Plastic Surgery 2020, 43, 589–596. [Google Scholar] [CrossRef]
- Papillion, P.; Wong, L.; Waldrop, J.; Sargent, L.; Brzezienski, M.; Kennedy, W.; Rehm, J. Infrared surface temperature monitoring in the postoperative management of free tissue transfers. Canadian Journal of Plastic Surgery 2009, 17, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Shen, A.Y.; Lonie, S.; Lim, K.; Farthing, H.; Hunter-Smith, D.J.; Rozen, W.M. Free flap monitoring, salvage, and failure timing: A systematic review. Journal of Reconstructive Microsurgery 2021, 37, 300–308. [Google Scholar] [CrossRef]
- Whitaker, I.S.; Lie, K.H.; Rozen, W.M.; et al. Dynamic infrared thermography for the preoperative planning of microsurgical breast reconstruction: A comparison with CTA. Journal of Plastic 2012, Reconstructive & Aesthetic Surgery, 65, 130-132.
- Perng, C.K.; Ma, H.; Chiu, Y.J.; Lin, P.H.; Tsai, C.H. Detection of free flap pedicle thrombosis by infrared surface temperature imaging. Journal of Surgical Research 2018, 229, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Moran-Romero, M.A.; Lopez-Mendoza, F.J. Postoperative monitoring of free flaps using smartphone thermal imaging may lead to ambiguous results: Three case reports. International Microsurgery 2022, 6, 4. [Google Scholar] [CrossRef]
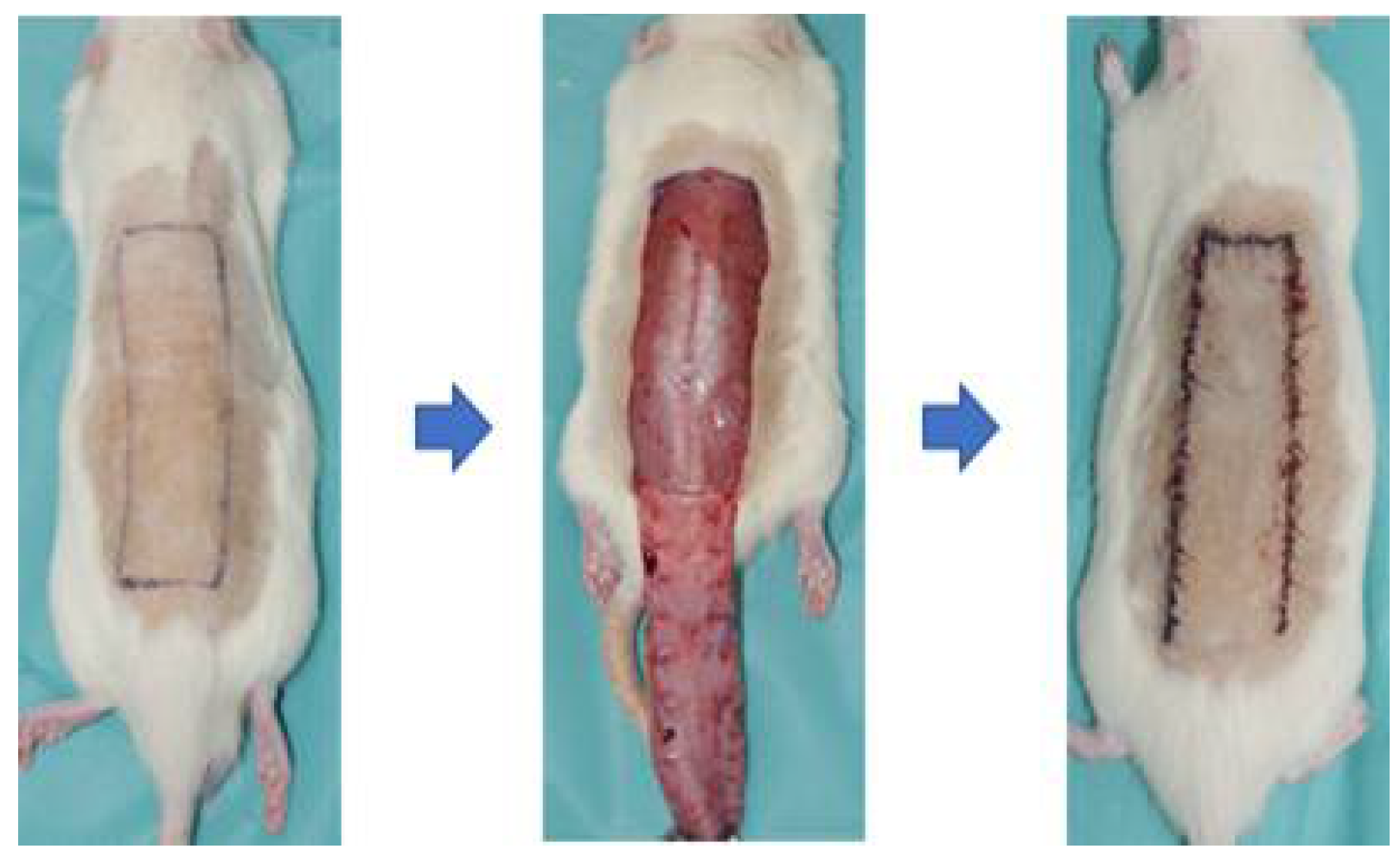

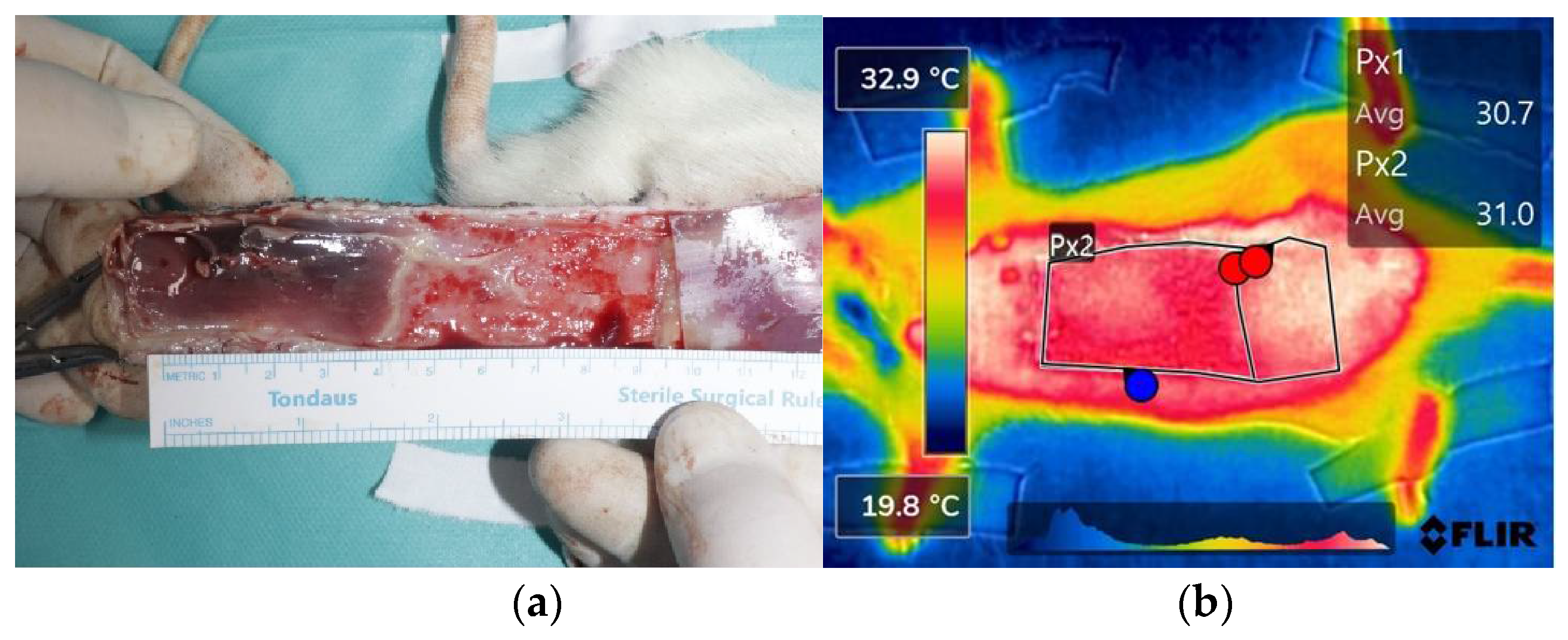

| Flap models (n=28) | POD 3 | POD 7 |
|---|---|---|
| Necrotic area/Total flap ratio (%) | 24.4 | 27.5 |
| Temperature difference between total flap and necrotic area | 0.800 | 0.792 |
| No. of patient | Sex | Age | Location | Flap | Flap type | Flap necrosis |
|---|---|---|---|---|---|---|
| 1 | M | 61 | Lt. ankle | ALT free flap | Free flap | No necrosis |
| 2 | F | 27 | Scalp | ALT free flap | Free flap | No necrosis |
| 3 | M | 16 | Lt. ankle | ALT free flap | Free flap | No necrosis |
| 4 | M | 55 | Lt. lower leg | ALT free flap | Free flap | No necrosis |
| 5 | M | 72 | Philtrum | ALT free flap | Free flap | No necrosis |
| 6 | F | 66 | Lt. ankle | ALT free flap | Free flap | No necrosis |
| 7 | F | 100 | Lt. upper eyelid | ALT free flap | Free flap | No necrosis |
| 8 | M | 60 | Lt. foot | ALT free flap | Free flap | Total necrosis |
| 9 | M | 63 | Lt. ankle | ALT free flap | Free flap | No necrosis |
| 10 | M | 51 | Lt. foot | ALT free flap | Free flap | Total necrosis |
| 11 | F | 70 | Rt. foot | ALT free flap | Free flap | No necrosis |
| 12 | M | 48 | Lt. ankle | TDAP free flap → ALT free flap | Free flap | No necrosis |
| 13 | F | 23 | Lt. hand | SCIP free flap | Free flap | No necrosis |
| 14 | M | 75 | Scalp | ALT free flap → Vastus lateralis muscle free flap | Free flap | Total necrosis |
| 15 | M | 81 | Nose | Nasolabial fold flap | Pedicled flap | No necrosis |
| 16 | M | 56 | Nose | Nasolabial fold flap | Pedicled flap | No necrosis |
| 17 | M | 52 | Nose | Nasolabial fold flap | Pedicled flap | No necrosis |
| 18 | M | 91 | Nose | Paramedian forehead flap | Pedicled flap | No necrosis |
| 19 | M | 57 | Nose | Paramedian forehead flap | Pedicled flap | No necrosis |
| 20 | M | 57 | Rt. lower leg | ALT pedicled flap | Pedicled flap | No necrosis |
| 21 | F | 49 | Rt. ankle | Peroneal artery perforator based FC rotation flap | Pedicled flap | Partial necrosis |
| Necrosis goupe (n=4) | |
|---|---|
| Necrosis type | |
| Total necrosis | 3 |
| Partial necrosis | 1 |
| Vascular compromise | |
| Arterial insufficiency | 2 |
| Venous congestion | 2 |
| Non necrosis (n=18) | Necrosis (n=4) | |||
| Total necrosis (n=3) | Partial necrosis (n=1) | |||
| Age (yrs) | 57.167 | 58.75 | ||
| Sex | ||||
| Male | 13 | 3 | ||
| Female | 5 | 1 | ||
| Temperature difference (dT) of Normal skin – Perforator (NS-PF) (℃) |
||||
| POD 0 | 1.817 | 1.55 (0.484) | 1.633 (0.740) | 1.3 (0.526) |
| POD 1 | 1.533 | 1.875 (0.434) | 2 (0.262) | 1.5 (0.842) |
| POD 2 | 1.528 | 2.2 (0.434) | 2.467 (0.262) | 1.4 (0.842) |
| POD 3 | 0.989 | 2.525 (0.001*) | 2.533 (0.006*) | 2.5 (0.105) |
| POD 7 | 0.706 | 3.5 (<0.001*) | 4 (0.002*) | 2 (0.105) |
| Temperature difference (dT) of Perforator – Average of flap (PF-AFP) (℃) |
||||
| POD 0 | 0.539 | 0.175 (0.118) | 0.2 | 0.1 |
| POD 1 | 0.35 | -0.175 (0.002*) | -0.2 | -0.1 |
| POD 2 | 0.333 | -0.225 (0.003*) | -0.2 | -0.3 |
| POD 3 | 0.617 | -0.375 (<0.001*) | -0.233 | -0.8 |
| POD 7 | 0.489 | -0.225 (<0.001*) | -0.333 | -0.1 |
| Monitoring Method | Advantages | Limitations | References |
|---|---|---|---|
|
Clinical Examination |
Non-invasive Widely available Low cost |
Limited applicability in buried flaps Risk of poor interrater agreement due to inconsistent flap (failure) appearances |
[20] |
|
Acoustic Doppler Sonography |
Non-invasive High sensitivity & specificity Real-time monitoring |
Limited applicability in buried flaps Operator-dependent Limited ability to detect venous thrombosis |
[20,21] |
|
Implantable Doppler |
Continuous monitoring High sensitivity & specificity Real-time monitoring |
Invasive Requires surgical implantation Risk of infection Limited applicability in buried flaps |
[20,21] |
|
Indocyanine Green Fluorescence Angiography |
Non-invasive High sensitivity & specificity Real-time monitoring Ability to detect venous thrombosis |
Limited applicability in buried flaps Requires specialized equipment Limited ability to detect arterial thrombosis |
[20,23] |
| Near-Infrared Spectroscopy | Non-invasive Real-time monitoring Ability to detect arterial thrombosis |
Limited applicability in buried flaps Requires specialized equipment Limited ability to detect venous thrombosis |
[22] |
| Tissue Oximetry | Non-invasive Real-time monitoring Ability to detect arterial thrombosis |
Limited applicability in buried flaps Requires specialized equipment Limited ability to detect venous thrombosis |
[24] |
|
Transcutaneous Oximetry measurement |
Non-invasive Quantifying measurement Potential for thermal injury |
Limited applicability in buried flaps Time required for measurement Low sensitivity |
[25,26] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




