Submitted:
11 June 2024
Posted:
12 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Laboratory Measurements
2.3. Clinical Data
2.3. Statistical Methods
3. Results
3.1. Prevalence of ME/CFS-, POTS-, SFN- and MCAS-Associated Symptoms in PACVS
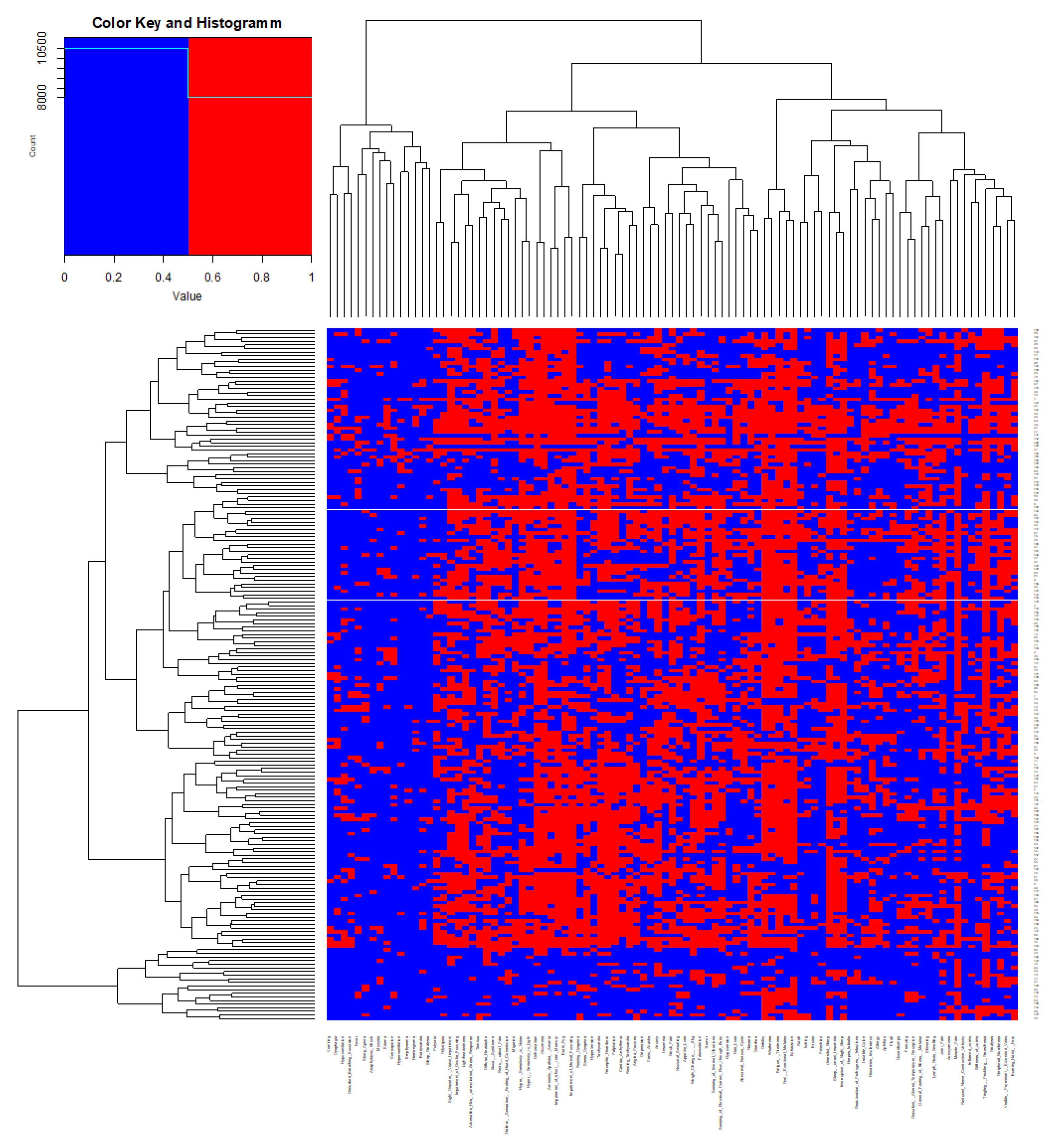
3.2. Unbiased Clustering of PACVS-Associated Symptoms
3.3. Prevalence of Major PACVS-Associated Symptoms
3.4. Abnormalities of Blood Markers in PACVS
4. Discussion
4.1. The Clinical Phenotype of PACVS
4.2. PACVS-Associated Alterations of Diagnostic Markers
4.2.1. Increased eGFR
4.2.2. Low fT3
4.2.3. Disturbance of Iron Storage
4.2.4. Increase in Soluble Neurofilament Light Chains (sNFL)
4.2.5. IgG-Subclass Imbalances
4.2.6. Systemic Inflammation
4.2.7. Lipids
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
 |
References
- Scholkmann, F.; May, C.-A. COVID-19, post-acute COVID-19 syndrome (PACS, “long COVID”) and post-COVID-19 vaccination syndrome (PCVS, “post-COVIDvacsyndrome”):Similarities and differences. Pathology - Research and Practice 2023. [CrossRef]
- Semmler, A.; Mundorf, A.K.; Kuechler, A.S.; Schulze-Bosse, K.; Heidecke, H.; Schulze-Forster, K.; Schott, M.; Uhrberg, M.; Weinhold, S.; Lackner, K.J.; et al. Chronic Fatigue and Dysautonomia following COVID-19 Vaccination Is Distinguished from Normal Vaccination Response by Altered Blood Markers. Vaccines (Basel) 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.J.; Son, C.G. Review of case definitions for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). J Transl Med 2020, 18, 289. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, B.M.; van de Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J Intern Med 2011, 270, 327–338. [Google Scholar] [CrossRef]
- Fedorowski, A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med 2019, 285, 352–366. [Google Scholar] [CrossRef]
- Bair, M.J.; Krebs, E.E. Fibromyalgia. Ann Intern Med 2020, 172, ITC33–ITC48. [Google Scholar] [CrossRef] [PubMed]
- Tavee, J.; Zhou, L. Small fiber neuropathy: A burning problem. Cleve Clin J Med 2009, 76, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Valent, P.; Hartmann, K.; Bonadonna, P.; Niedoszytko, M.; Triggiani, M.; Arock, M.; Brockow, K. Mast Cell Activation Syndromes: Collegium Internationale Allergologicum Update 2022. Int Arch Allergy Immunol 2022, 183, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Kuechler, A.S.; Weinhold, S.; Boege, F.; Adams, O.; Muller, L.; Babor, F.; Bennstein, S.B.; Pham, T.U.; Hejazi, M.; Reusing, S.B.; et al. A Diagnostic Strategy for Gauging Individual Humoral Ex Vivo Immune Responsiveness Following COVID-19 Vaccination. Vaccines (Basel) 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Benkert, P.; Meier, S.; Schaedelin, S.; Manouchehrinia, A.; Yaldizli, O.; Maceski, A.; Oechtering, J.; Achtnichts, L.; Conen, D.; Derfuss, T.; et al. Serum neurofilament light chain for individual prognostication of disease activity in people with multiple sclerosis: a retrospective modelling and validation study. Lancet Neurol 2022, 21, 246–257. [Google Scholar] [CrossRef]
- Reinsberg, J.; Dembinski, J.; Dorn, C.; Behrendt, D.; Bartmann, P.; van Der Ven, H. Determination of total interleukin-8 in whole blood after cell lysis. Clin Chem 2000, 46, 1387–1394. [Google Scholar] [CrossRef]
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine 2021, 38, 101019. [Google Scholar] [CrossRef]
- Cancarevic, I.; Nassar, M.; Medina, L.; Sanchez, A.; Parikh, A.; Hosna, A.; Devanabanda, B.; Vest, M.; Ayotunde, F.; Ghallab, M.; et al. Nephrotic Syndrome in Adult Patients With COVID-19 Infection or Post COVID-19 Vaccine: A Systematic Review. Cureus 2022, 14, e29613. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xu, Z.; Wang, P.; Li, X.M.; Shuai, Z.W.; Ye, D.Q.; Pan, H.F. New-onset autoimmune phenomena post-COVID-19 vaccination. Immunology 2022, 165, 386–401. [Google Scholar] [CrossRef] [PubMed]
- Finterer, J.; Scorza, F.A. A retrospective analysis of clinically confirmed long post-COVID vaccination syndrome. J Clin Transl Res 2022, 8, 506–508. [Google Scholar] [PubMed]
- Finsterer, J.; Scorza, F.A.; Scorza, C.A. Post SARS-CoV-2 vaccination Guillain-Barre syndrome in 19 patients. Clinics (Sao Paulo) 2021, 76, e3286. [Google Scholar] [CrossRef] [PubMed]
- Abolmaali, M.; Rezania, F.; Behnagh, A.K.; Hamidabad, N.M.; Gorji, A.; Mirzaasgari, Z. Guillain-Barré syndrome in association with COVID-19 vaccination: a systematic review. Immunologic Research 2022, 70, 752–764. [Google Scholar] [CrossRef]
- Finsterer, J. Exacerbating Guillain-Barre Syndrome Eight Days after Vector-Based COVID-19 Vaccination. Case Rep Infect Dis 2021, 2021, 3619131. [Google Scholar] [CrossRef]
- Murvelashvili, N.; Tessnow, A. A Case of Hypophysitis Following Immunization With the mRNA-1273 SARS-CoV-2 Vaccine. J Investig Med High Impact Case Rep 2021, 9, 23247096211043386. [Google Scholar] [CrossRef] [PubMed]
- Ankireddypalli, A.R.; Chow, L.S.; Radulescu, A.; Kawakami, Y.; Araki, T. A Case of Hypophysitis Associated With SARS-CoV-2 Vaccination. AACE Clin Case Rep 2022, 8, 204–209. [Google Scholar] [CrossRef]
- Bouca, B.; Roldao, M.; Bogalho, P.; Cerqueira, L.; Silva-Nunes, J. Central Diabetes Insipidus Following Immunization With BNT162b2 mRNA COVID-19 Vaccine: A Case Report. Front Endocrinol (Lausanne) 2022, 13, 889074. [Google Scholar] [CrossRef]
- Moura Neto, A.; Zantut-Wittmann, D.E. Abnormalities of Thyroid Hormone Metabolism during Systemic Illness: The Low T3 Syndrome in Different Clinical Settings. International Journal of Endocrinology 2016, 2016, 2157583. [Google Scholar] [CrossRef]
- Suriawinata, E.; Mehta, K.J. Iron and iron-related proteins in COVID-19. Clin Exp Med 2022, 1–23. [Google Scholar] [CrossRef]
- Legler, F.; Meyer-Arndt, L.; Mödl, L.; Kedor, C.; Freitag, H.; Stein, E.; Hoppmann, U.; Rust, R.; Wittke, K.; Siebert, N.; et al. Long-term symptom severity and clinical biomarkers in post-COVID-19/chronic fatigue syndrome: results from a prospective observational cohort. eClinicalMedicine 2023, 63. [Google Scholar] [CrossRef]
- Lutz, L.; Rohrhofer, J.; Zehetmayer, S.; Stingl, M.; Untersmayr, E. Evaluation of Immune Dysregulation in an Austrian Patient Cohort Suffering from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Biomolecules 2021, 11. [Google Scholar] [CrossRef]
- Gordon, C.L.; Johnson, P.D.; Permezel, M.; Holmes, N.E.; Gutteridge, G.; McDonald, C.F.; Eisen, D.P.; Stewardson, A.J.; Edington, J.; Charles, P.G.; et al. Association between severe pandemic 2009 influenza A (H1N1) virus infection and immunoglobulin G(2) subclass deficiency. Clin Infect Dis 2010, 50, 672–678. [Google Scholar] [CrossRef]
- Yates, J.L.; Ehrbar, D.J.; Hunt, D.T.; Girardin, R.C.; Dupuis, A.P., 2nd; Payne, A.F.; Sowizral, M.; Varney, S.; Kulas, K.E.; Demarest, V.L.; et al. Serological analysis reveals an imbalanced IgG subclass composition associated with COVID-19 disease severity. Cell Rep Med 2021, 2, 100329. [Google Scholar] [CrossRef]
- Schultheiss, C.; Willscher, E.; Paschold, L.; Gottschick, C.; Klee, B.; Bosurgi, L.; Dutzmann, J.; Sedding, D.; Frese, T.; Girndt, M.; et al. Liquid biomarkers of macrophage dysregulation and circulating spike protein illustrate the biological heterogeneity in patients with post-acute sequelae of COVID-19. J Med Virol 2023, 95, e28364. [Google Scholar] [CrossRef]
- Kedor, C.; Freitag, H.; Meyer-Arndt, L.; Wittke, K.; Hanitsch, L.G.; Zoller, T.; Steinbeis, F.; Haffke, M.; Rudolf, G.; Heidecker, B.; et al. A prospective observational study of post-COVID-19 chronic fatigue syndrome following the first pandemic wave in Germany and biomarkers associated with symptom severity. Nat Commun 2022, 13, 5104. [Google Scholar] [CrossRef]
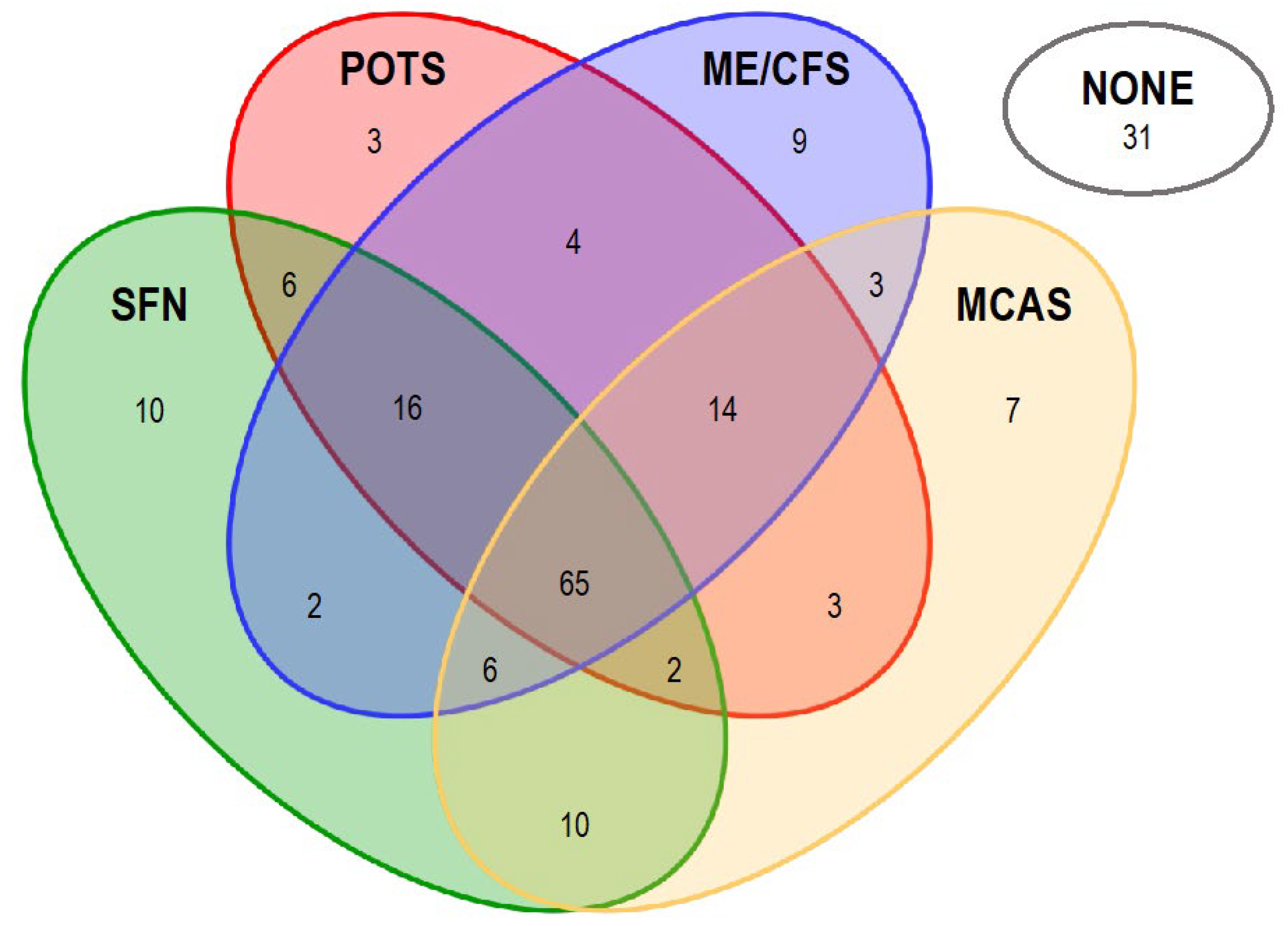

| Symptom/Diagnosis1 | Prevalence (%)Syndrome Assoc.2,3 | |
|---|---|---|
| Exhaustion Debility Muscle Pain Unrestful Sleep Dizziness Tingling/ Prickling/ Paresthesia Impairment of Mental Focusing Fatigue/ Tiredness Orthostatism Brain Fog |
84.82 83.77 80.63 80.63 80.10 79.58 78.53 76.96 76.44 76.44 |
ME/CFS, POTS ME/CFS, POTS ME/CFS, POTS, SFN ME/CFS ME/CFS, POTS, SFN SFN ME/CFS, POTS ME/CFS, POTS ME/CFS, POTS, SFN ME/CFS, POTS |
| Interruption of Night Sleep Weakness Perceptible Heartbeat Post-Exertional Malaise Fasciculation Anxiety Injury from Immunization Tachycardia Impairment of Short-Term Memory Hyper-Sensitivity to Noise Sleep-onset Insomnia Neck Pain Diffuse Headache Peripheral Numbness Amnestic Aphasia/ Anomia Joint Pain Sight Disorder/ Vision Impairment Stress Dyspnea Palpitation Sensing of internal Vibrations Lightheadedness Resting Tachycardia Feel. Electr. Current Flow through Body Impairment of Ocular Focusing Hyper-Sensitivity to Light Weight Change ≥/≤ 5kg Nausea |
74.87 74.35 72.77 70.68 70.68 69.11 67.54 65.97 65.45 64.92 64.40 64.40 63.35 62.83 60.73 60.73 60.21 59.69 58.64 58.12 57.59 57.07 56.02 56.02 55.50 53.93 52.36 |
ME/CFS, POTS ME/CFS, POTS ME/CFS, POTS, SFN ME/CFS ME/CFS, POTS, SFN ME/CFS, POTS ME/CFS, POST, SFN ME/CFS, POTS ME/CFS, POTS ME/CFS, POTS ME/CFS, POTS, MCAS SFN ME/CFS, POTS ME/CFS ME/CFS ME/CFS, MCAS ME/CFS, POTS, SFN SFN ME/CFS, POTS ME/CFS, POTS, SFN SFN ME/CFS ME/CFS, POTS ME/CFS, POTS ME/CFS, POTS, MCAS |
| Sicca-Syndrome Tinnitus Post-Vaccine-Syndrome Nocturnal Sweating Angina Pectoris Burning Hand/ Feet Histamine Intolerance Retro-orbital Pain Appetite Loss Panic Attacks Cardiac Arrhythmia Abnormal Estrous Cycle Disturbed/ Altered Temp Perception Freezing Diarrhea General Feeling of Illness/ Malaise Resting Dyspnea Adrenal. Hits/ Unmotiv. Stress-Response Helmet-Sensation/ Head Constriction Lymph Node Swelling Visible/ Prominent/ Extended Veins Polyuria Hair Loss/Allopezia Insomnia Long Covid without Covid-Infection Hypertension Tremor Depression Shivering Polydipsia ME/CFS Paralysis Migraine Allergy Stiffness of Joints Itching Irritable Colon Spont. Bruising (unrelated to trauma) Reactivation of Pathogens/ Infections Hypotension Rash |
49.74 48.17 47.64 47.12 46.40 46.07 44.50 43.98 43.46 43.46 42.93 42.93 40.84 40.31 40.31 39.27 39.27 39.27 39.27 38.74 37.70 37.17 37.17 36.13 36.13 34.55 34.55 32.98 32.46 31.94 31.94 31.41 30.37 29.84 29.32 29.32 28.27 27.75 26.70 26.18 26.18 |
ME/CFS, SFN SFN ME/CFS ME/CFS, POTS ME/CFS, POTS, SFN SFN POTS, MCAS ME/CFS MCAS ME/CFS, POTS, MCAS ME/CFS ME/CFS, POTS, SFN ME/CFS, POTS SFN ME/CFS SFN ME/CFS, POTS ME/CFS MCAS ME/CFS ME/CFS, SFN, MCAS POTS |
| Aphthae Goosebumps Vaccine-Induced Inflamm. Response Petechiae Hypoventilation Hyper-Inflammation Herpes labialis POTS Inflamed Joints Flush Fever Acrocyanosis Reduced Nerve Conduction Velocity Dysphagia MCAS Myocarditis/ Pericarditis Fibromyalgia SFN Constipation Edema Bradycardia Disturbed Breathing Excursion Mycosis Vomiting Sleep Apnea Sleep Paralysis Hyperventilation Hemangioma Thrombosis Anaphylactic Shock Shingles Guillain-Barré Syndrome Emphysema Facial Nerve Paresis |
24.61 24.08 24.08 23.04 21.99 21.99 21.47 20.94 20.42 20.42 19.90 19.90 19.90 19.90 19.37 18.85 17.80 17.80 17.28 14.14 13.61 13.61 12.57 12.57 12.04 7.85 7.85 7.33 6.81 5.76 5.76 4.71 3.66 3.14 |
POTS MCAS POTS POTS, MCAS POTS MCAS POTS, SFN MCAS POTS MCAS |
Cluster 1: General Unwellness
|
| normal range (n.r.)1 [dimension] |
below n.r.2 N (%) |
above n.r.2 N (%) |
Remarks | |
|---|---|---|---|---|
|
Kidney eGFR (CAPA) eGFR (CKD-EPI) Urea |
60-120 [ml/min/1.73 m2] 60-120 [ml/min/1.73 m2] ♀ 21-43, ♂ 18-55 [mg/dl] |
14 (7) 1 (1) 70 (37) |
33 (17) 30 (16) 1 (1) |
GFR >120 always associated with urea <18 |
|
Thyroid TSH fT3 fT4 anti-TPO anti-TSH-R |
0,27-4,2 [µIU/ml] ♀ 2,6-5, ♂ 2-4,4 [ng/ml] ♀ 9.8-16.3, ♂ 9.3-17.1 [pg/ml] ≤ 34 [IU/ml) ≤ 1.7 [IU/L] |
4 (2) 65 (34) 14 (7) - - |
4 (2) - 7 (4) 18 (9) 1 (1) |
All other fT3-values in lower half of n.r. |
|
Cardiac markers ProBNP hsTroponin T |
≤ 125 [pg/ml] ♀ ≤ 14, ♂ ≤ 11 [ng/L] |
|
- 1 (1) |
|
|
Liver AST ALT ALP gammaGT Bilirubin |
♀ ≤ 3, ♂ ≤ 35 [U/L] ♀ ≤ 35, ♂ ≤ 45 [U/L] ♀ ≤ 104, ♂ ≤ 129 [U/L] ♀ ≤ 38, ♂ ≤ 55 [U/L] ≤ 1.2 [mg/dL] |
|
20 (10) 11 (6) 1 (1) 7 (4) 8 (4) |
AST/ALT ≤ 1 in all cases no value > 2 |
|
Lipids Cholesterol Trigliceride LDL HDL LDL/HDL |
≤ 200 [mg/dL] ≤ 150 [mg/dL] ≤ 160 [mg|dL] ≥ 40 [mg/dL] 2.5 - 3.5 |
32 (17) 117 (62) |
61 (32) 39 (20) 10 (5) 4 (2) |
fasting state not assured |
|
Inflammation Il-6 Il-8 CRP |
≤ 7 [pg/mL] ≤ 62 pg/mL] ≤ 0.5 [mg/dL] |
- - - |
115 (60) 172 (90) 13 (7) |
IL-6 up to 50,000 IL-8 up to 2,000,000 |
|
IgG-Subclasses total IgG1 IgG2 IgG3 IgG4 |
700-1600 [mg/dL] 280-800 [mg/dL] 115-570 [mg/dL] 24-125 [mg/dL] 5.2-125 [mg/dL] |
19 (10) 1 (1) 2 (1) 88 (46) 12 (6) |
3 (2) 18 (9) 5 (3) 17 (9) 21 (11) |
|
|
Iron-metabolism Fe Ferritin Transferrin Transf. Saturation Sol Transf. Recept. Ferritin Index |
♀ 37-165, ♂ 40-155 [µg/dL] ♀ 13-150, ♂ 30-400 [µg/L] 200-360 [mg/dL] 16-45 [%] 0.81-1.75 [mg/L] 0.63-2.2 |
9 (5) 11 (6) 34 (18) 27 (14) 36 (19) 117 (61) |
20 (10) 3 (2) 3 (2) 20 (10) 4 (2) 2 (1) |
|
|
Neurodegeneration sNFL sNFL |
< 1.5 (zScore) 10-90 (Percentile) |
6 (3) |
39 (20) 52 (27) |
adjusted for age, BMI, GFR according to ref. [10] |
| BMI | 18.5-30 | 19 (10) | 16 (8) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




