Submitted:
05 June 2024
Posted:
07 June 2024
You are already at the latest version
Abstract
Keywords:
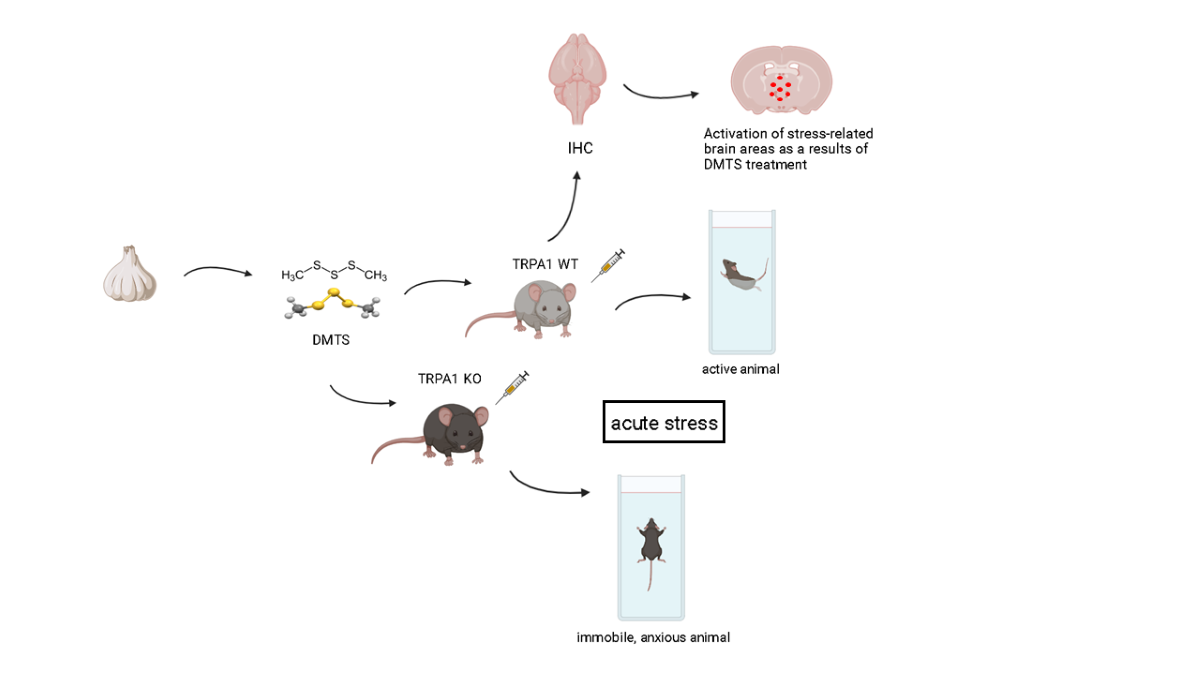
1. Introduction
2. Results
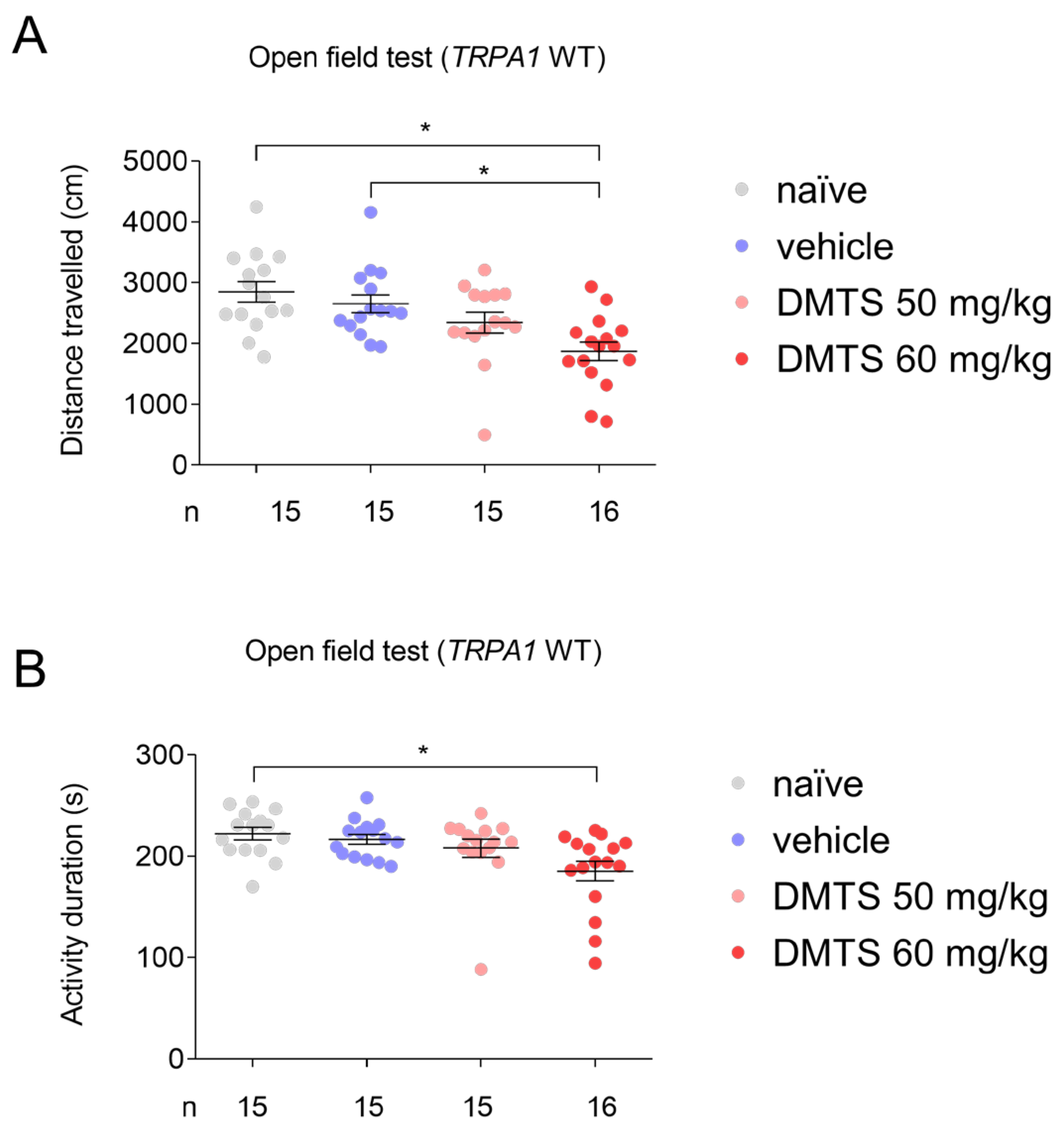
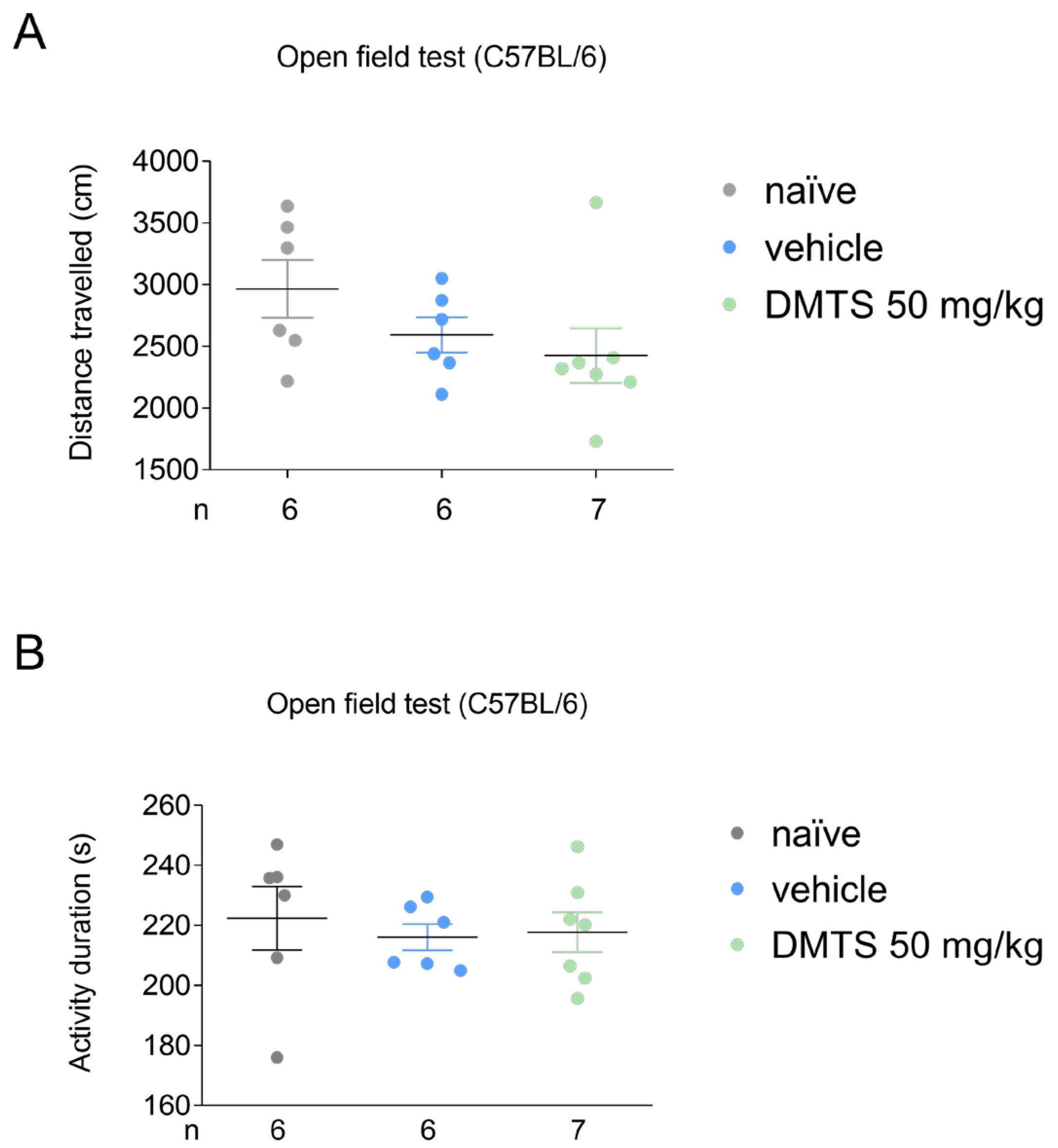
2.1. Finding the suitable dose of DMTS via open field test
2.2. Behavioural tests
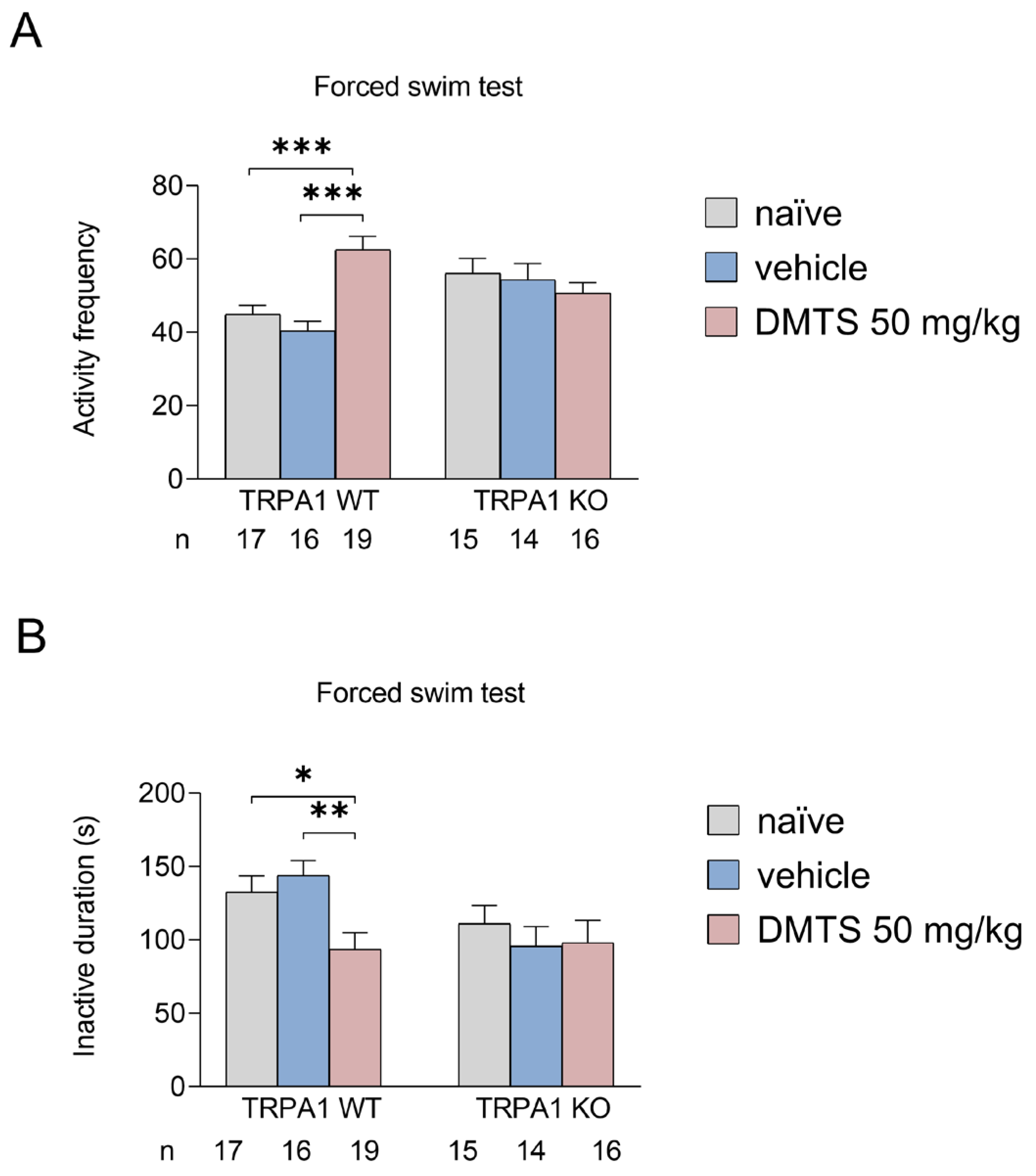
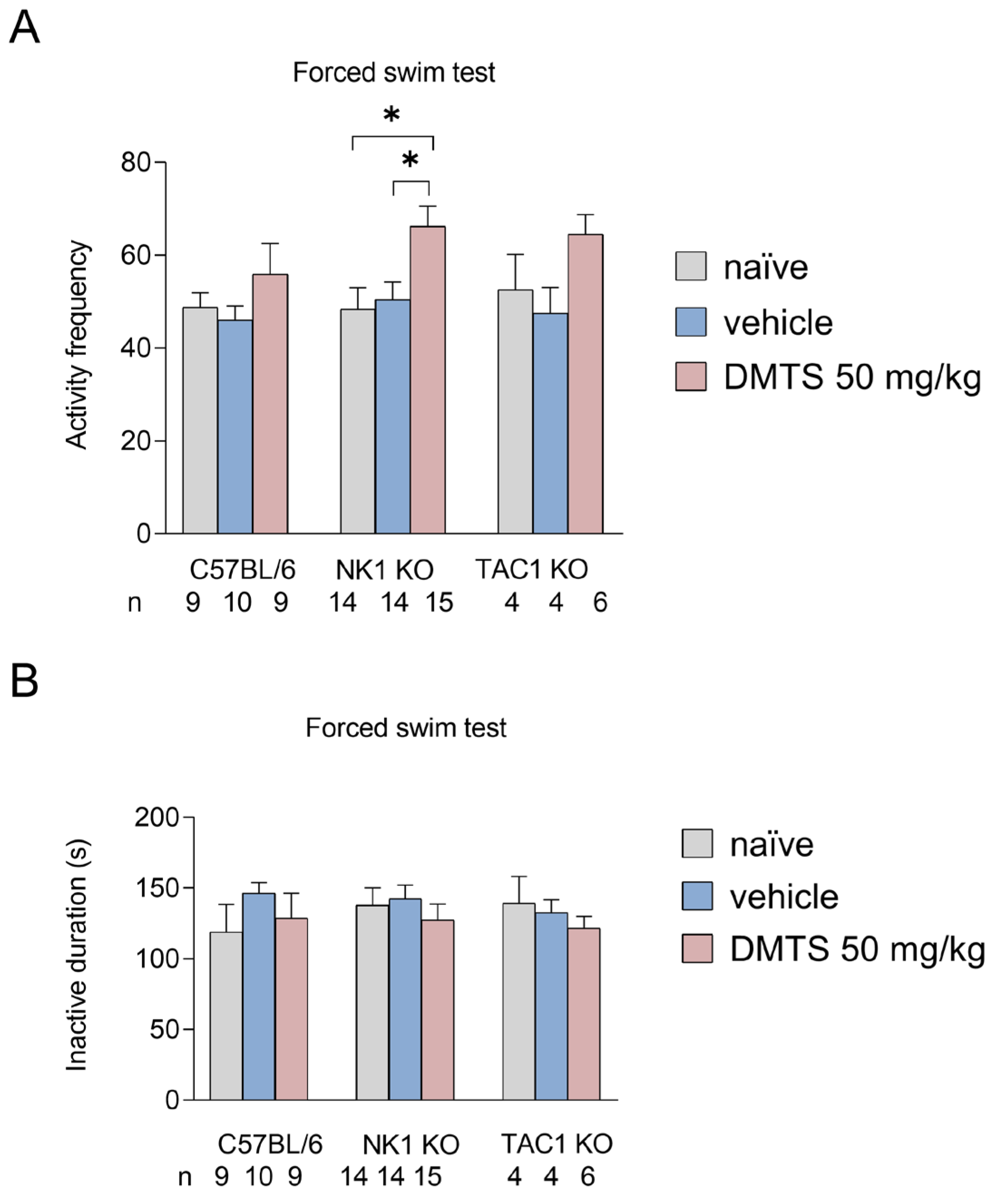
2.2.1. Effect of DMTS on TRPA1 WT and KO animals in the forced swim test
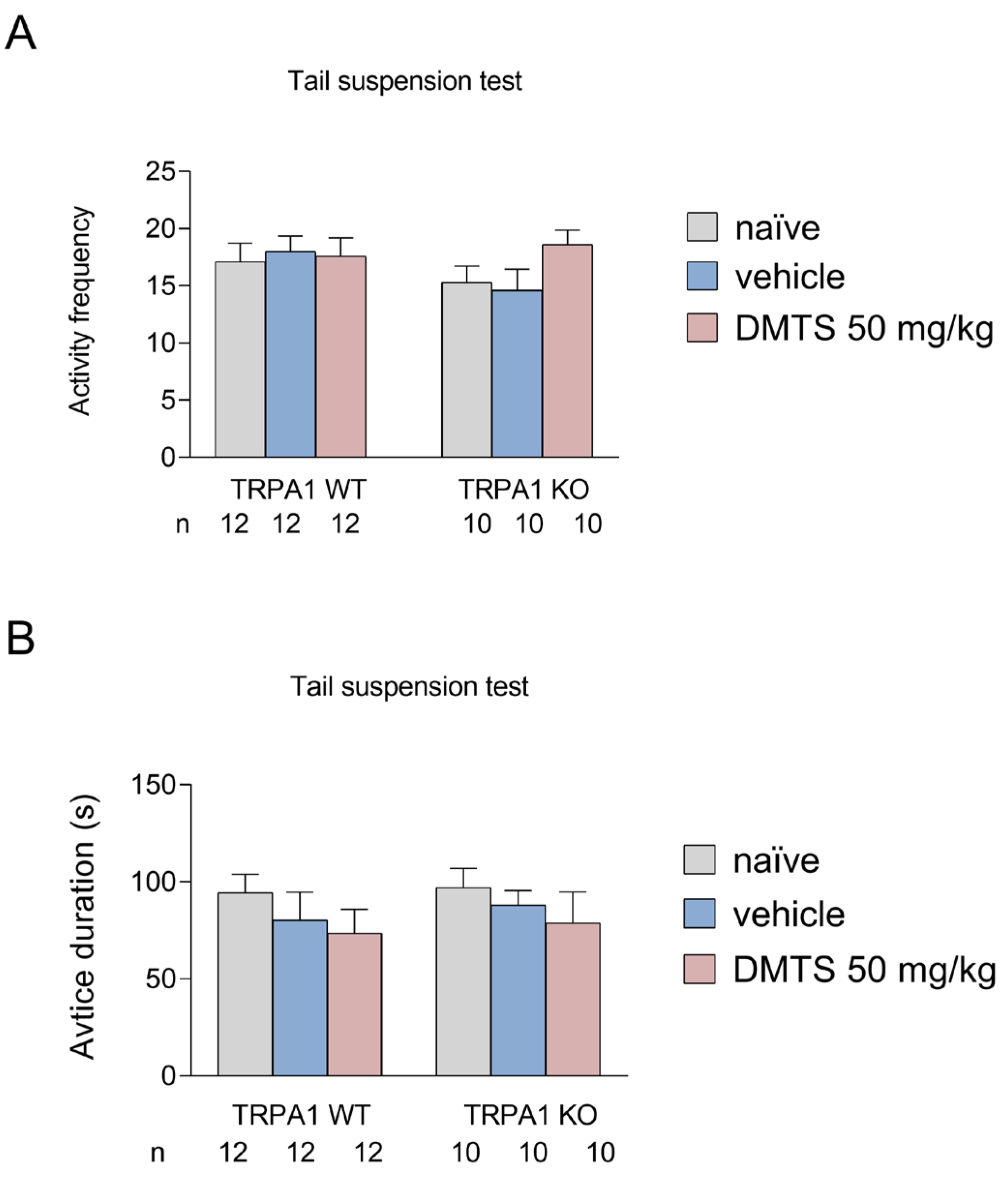
2.2.2. Effect of DMTS on TRPA1 WT and KO animals in the tail suspension test
2.2.3. Exploring the involvement of substance P in mediating the effect of DMTS
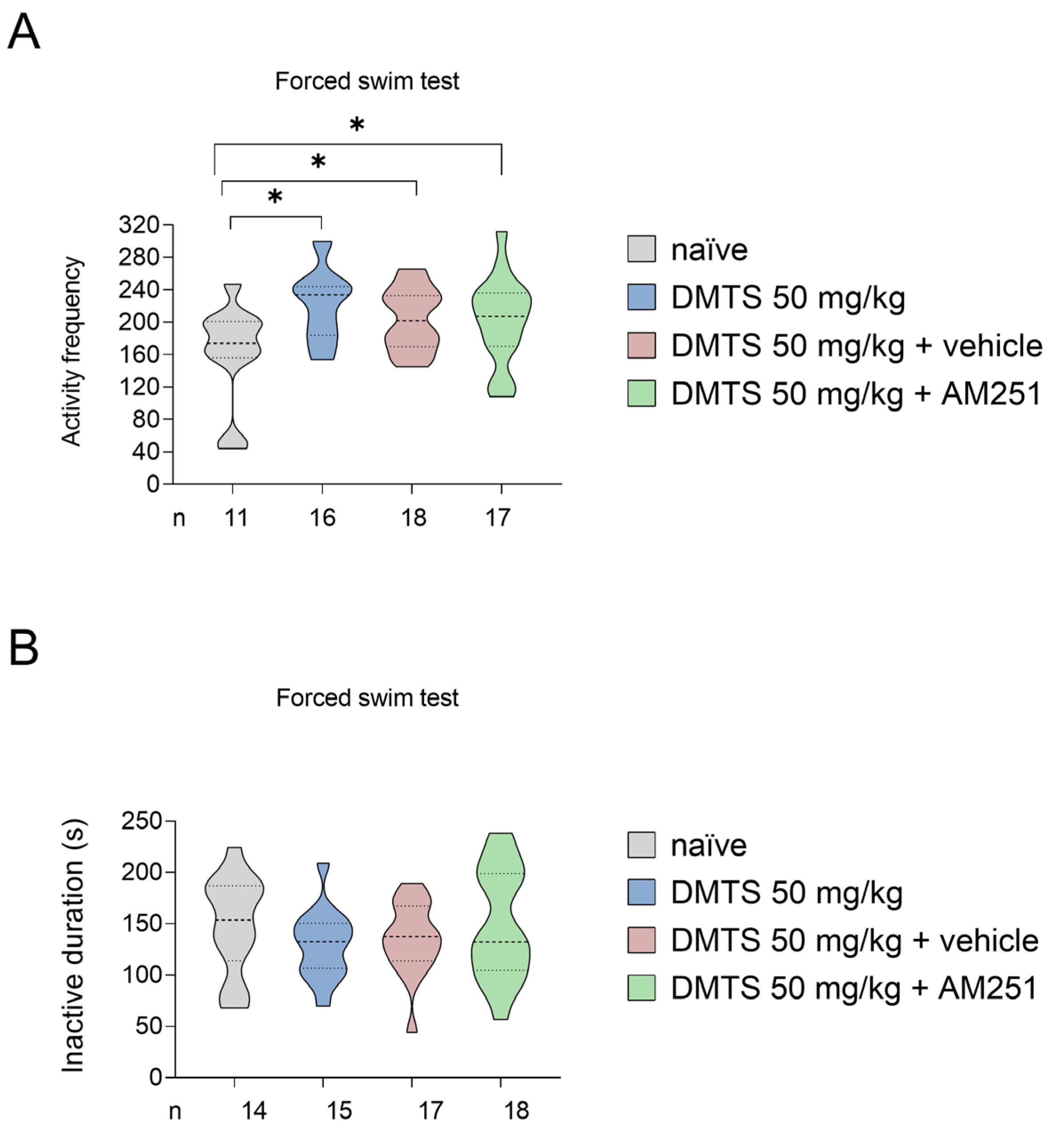
2.2.4. Investigation of the contribution of the endocannabinoid system in mediating the effects of DMTS
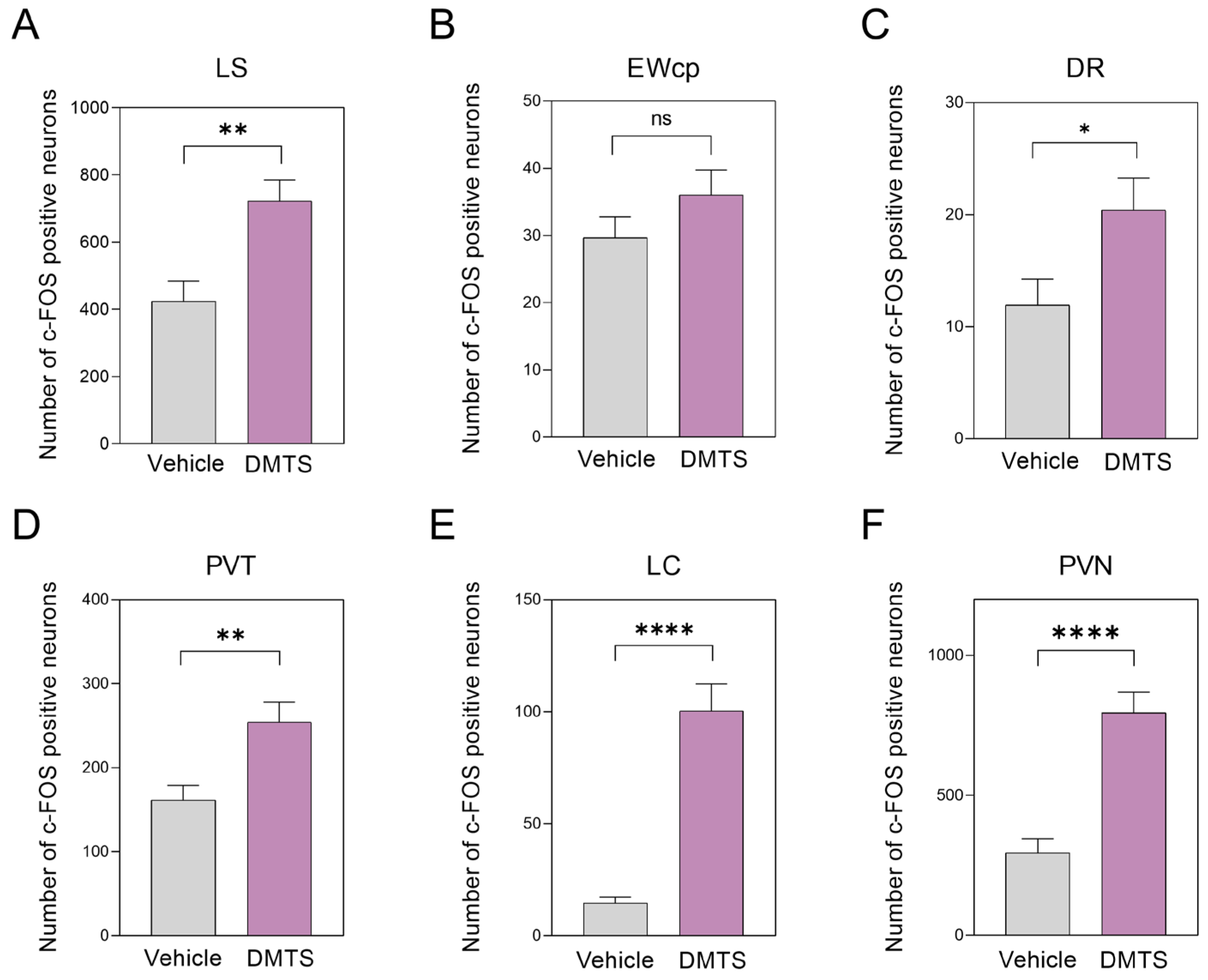
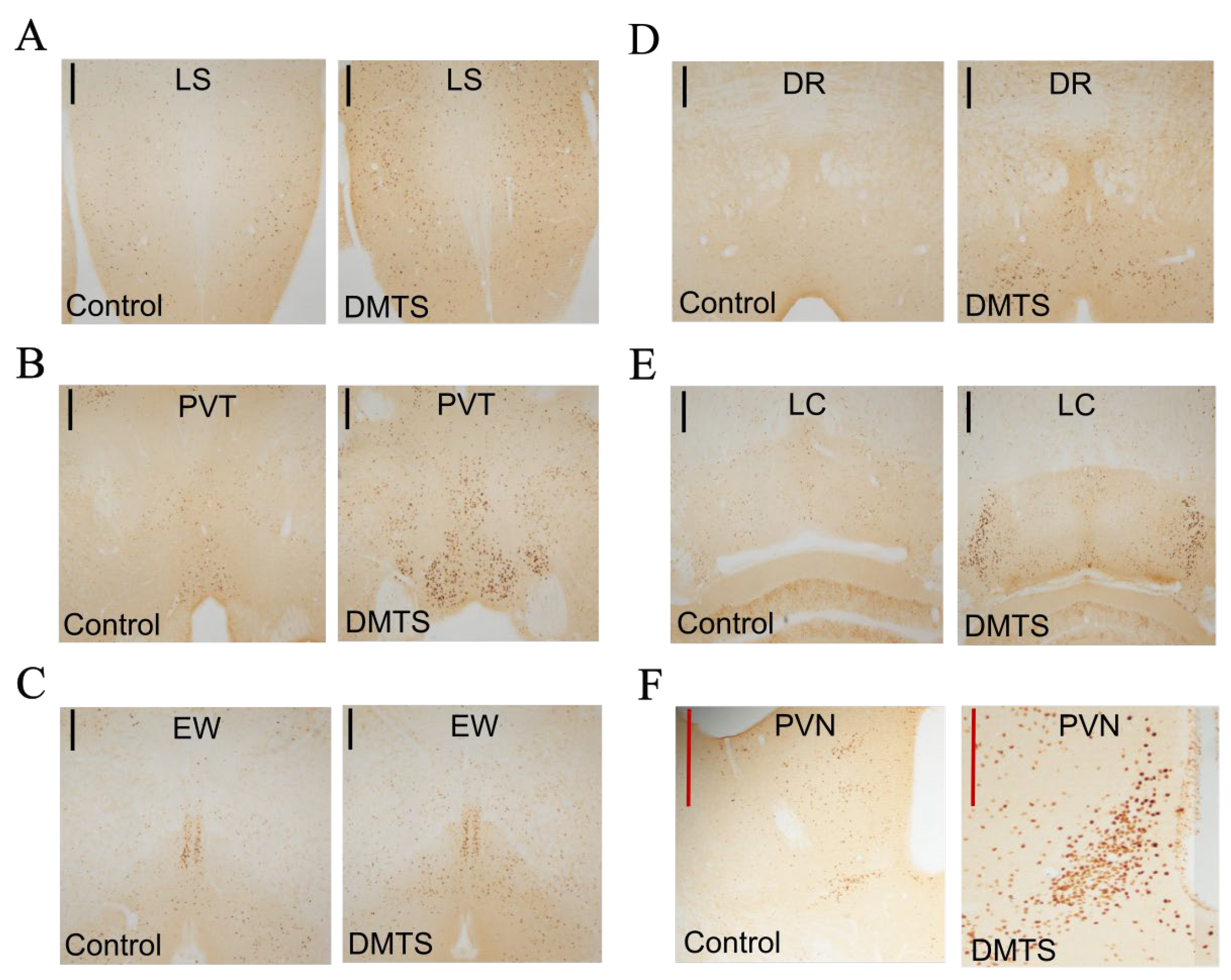
2.3. Immunohistochemistry
3. Discussion
4. Materials and Methods
4.1. Animals and experimental design
4.2. Behavioural tests
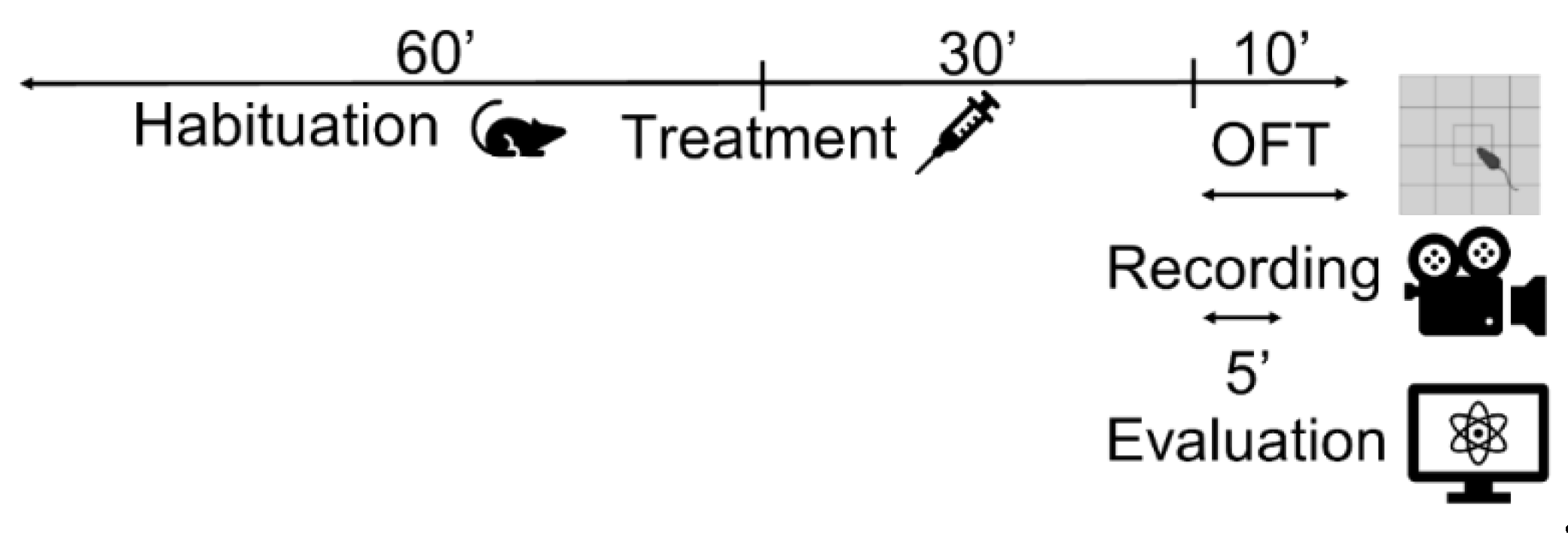
4.2.1. Open field test
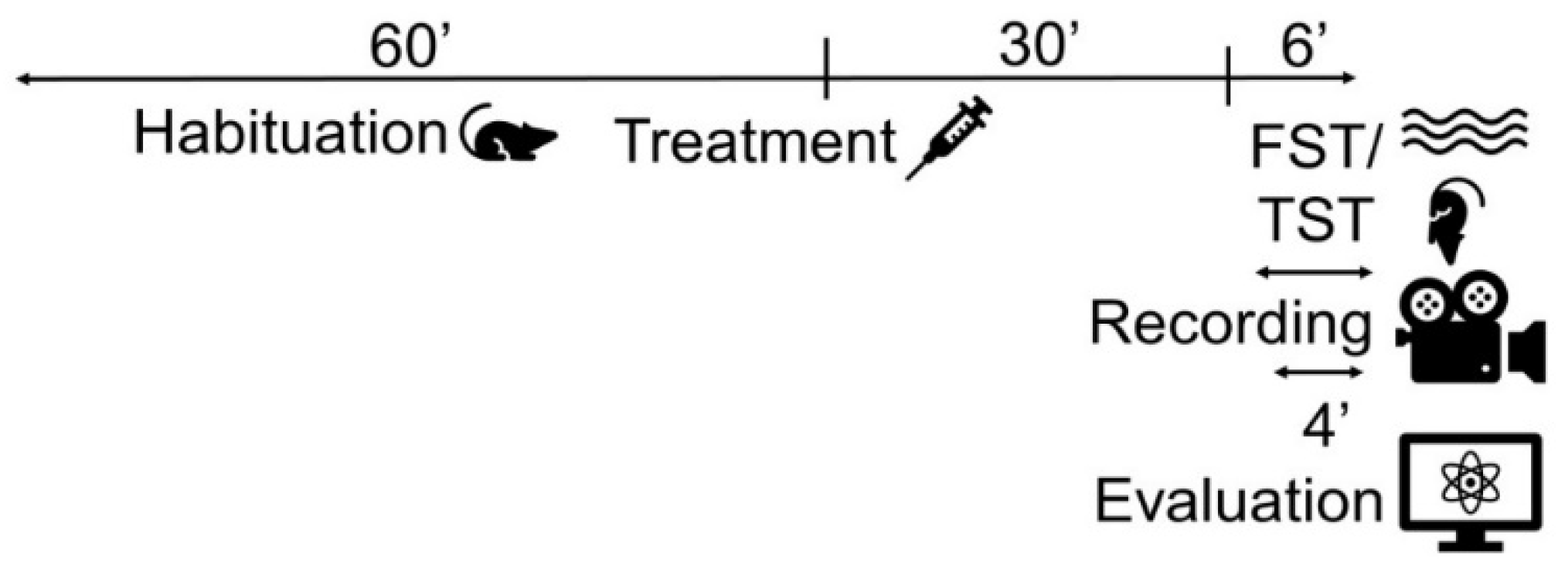
4.2.2. Forced swim test and tail suspension test
2.3. Perfusion and tissue collection
2.3.1. c-FOS immunohistochemistry
2.4. Evaluation methods
2.4.1. Noldus EthoVision XT 15
2.4.2. ImageJ
2.5. Statistics
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
7. Abbreviations
References
- McEwen, B.S. The Neurobiology of Stress: From Serendipity to Clinical Relevance. Brain Research 2000.
- Wong, M.-L.; Licinio, J. Research and Treatment Approaches to Depression. Nat Rev Neurosci 2001, 2, 343–351. [Google Scholar] [CrossRef]
- Sun, H.-J.; Wu, Z.-Y.; Nie, X.-W.; Bian, J.-S. Role of Hydrogen Sulfide and Polysulfides in Neurological Diseases: Focus on Protein S-Persulfidation. CN 2021, 19, 868–884. [Google Scholar] [CrossRef] [PubMed]
- Pozsgai, G.; Payrits, M.; Sághy, É.; Sebestyén-Bátai, R.; Steen, E.; Szőke, É.; Sándor, Z.; Solymár, M.; Garami, A.; Orvos, P.; et al. Analgesic Effect of Dimethyl Trisulfide in Mice Is Mediated by TRPA1 and Sst 4 Receptors. Nitric Oxide 2017, 65, 10–21. [Google Scholar] [CrossRef]
- Dombi, Á.; Sánta, C.; Bátai, I.Z.; Kormos, V.; Kecskés, A.; Tékus, V.; Pohóczky, K.; Bölcskei, K.; Pintér, E.; Pozsgai, G. Dimethyl Trisulfide Diminishes Traumatic Neuropathic Pain Acting on TRPA1 Receptors in Mice. IJMS 2021, 22, 3363. [Google Scholar] [CrossRef]
- Kimura, H. Signalling by Hydrogen Sulfide and Polysulfides via Protein S-sulfuration. British Pharmacological Society, 2020; 177, 720–733. [Google Scholar] [CrossRef]
- Cortese-Krott, M.M.; Kuhnle, G.G.C.; Dyson, A.; Fernandez, B.O.; Grman, M.; DuMond, J.F.; Barrow, M.P.; McLeod, G.; Nakagawa, H.; Ondrias, K.; et al. Key Bioactive Reaction Products of the NO/H 2 S Interaction Are S/N-Hybrid Species, Polysulfides, and Nitroxyl. Proc. Natl. Acad. Sci. U.S.A. 2015, 112. [Google Scholar] [CrossRef] [PubMed]
- Koike, S.; Kawamura, K.; Kimura, Y.; Shibuya, N.; Kimura, H.; Ogasawara, Y. Analysis of Endogenous H2S and H2Sn in Mouse Brain by High-Performance Liquid Chromatography with Fluorescence and Tandem Mass Spectrometric Detection. Free Radical Biology and Medicine 2017, 113, 355–362. [Google Scholar] [CrossRef]
- Chen, W.-L.; Xie, B.; Zhang, C.; Xu, K.-L.; Niu, Y.-Y.; Tang, X.-Q.; Zhang, P.; Zou, W.; Hu, B.; Tian, Y. Antidepressant-like and Anxiolytic-like Effects of Hydrogen Sulfide in Behavioral Models of Depression and Anxiety. Behavioural Pharmacology 2013, 24, 590–597. [Google Scholar] [CrossRef]
- Nagy, P.; Pálinkás, Z.; Nagy, A.; Budai, B.; Tóth, I.; Vasas, A. Chemical Aspects of Hydrogen Sulfide Measurements in Physiological Samples. Biochimica et Biophysica Acta (BBA) - General Subjects 2014, 1840, 876–891. [Google Scholar] [CrossRef]
- De Silva, D.; Lee, S.; Duke, A.; Angalakurthi, S.; Chou, C.-E.; Ebrahimpour, A.; Thompson, D.E.; Petrikovics, I. Intravascular Residence Time Determination for the Cyanide Antidote Dimethyl Trisulfide in Rat by Using Liquid-Liquid Extraction Coupled with High Performance Liquid Chromatography. Journal of Analytical Methods in Chemistry 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Bátai, I.Z.; Sár, C.P.; Horváth, Á.; Borbély, É.; Bölcskei, K.; Kemény, Á.; Sándor, Z.; Nemes, B.; Helyes, Z.; Perkecz, A.; et al. TRPA1 Ion Channel Determines Beneficial and Detrimental Effects of GYY4137 in Murine Serum-Transfer Arthritis. Front. Pharmacol. 2019, 10, 964. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Mikami, Y.; Osumi, K.; Tsugane, M.; Oka, J.; Kimura, H. Polysulfides Are Possible H 2 S-derived Signaling Molecules in Rat Brain. FASEB j. 2013, 27, 2451–2457. [Google Scholar] [CrossRef] [PubMed]
- Kiss, L.; Holmes, S.; Chou, C.-E.; Dong, X.; Ross, J.; Brown, D.; Mendenhall, B.; Coronado, V.; De Silva, D.; Rockwood, G.A.; et al. Method Development for Detecting the Novel Cyanide Antidote Dimethyl Trisulfide from Blood and Brain, and Its Interaction with Blood. Journal of Chromatography B, 2017; 1044–1045. [Google Scholar] [CrossRef]
- Kiss, L.; Bocsik, A.; Walter, F.R.; Ross, J.; Brown, D.; Mendenhall, B.A.; Crews, S.R.; Lowry, J.; Coronado, V.; Thompson, D.E.; et al. From the Cover: In Vitro and In Vivo Blood-Brain Barrier Penetration Studies with the Novel Cyanide Antidote Candidate Dimethyl Trisulfide in Mice. Toxicological Sciences 2017, 160, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y. ; The 173rd Clinical Department of PLA 421rd Hospital, Guangdong, China; Huang, C.; The 173rd Clinical Department of PLA 421rd Hospital, Guangdong, China; Deng, H.; Department of Anesthesiology, Guangzhou General Hospital of Guangzhou Military Command, Guangdong, China; Jia, J.; Department of Anesthesiology, Guangzhou General Hospital of Guangzhou Military Command, Guangdong, China; Wu, Y.; Department of Anesthesiology, Guangzhou General Hospital of Guangzhou Military Command, Guangdong, China; et al. TRPA1 and Substance P Mediate Stress Induced Duodenal Lesions in Water Immersion Restraint Stress Rat Model. Turk J Gastroenterol, 2018; 692–700. [Google Scholar] [CrossRef]
- Hökfelt, T.; Kellerth, J.O.; Nilsson, G.; Pernow, B. Substance P: Localization in the Central Nervous System and in Some Primary Sensory Neurons. Science 1975, 190, 889–890. [Google Scholar] [CrossRef] [PubMed]
- Bilkei-Gorzo, A.; Racz, I.; Michel, K.; Zimmer, A. Diminished Anxiety- and Depression-Related Behaviors in Mice with Selective Deletion of the Tac1 Gene. J. Neurosci. 2002, 22, 10046–10052. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, M. H2S and Substance P in Inflammation. In Methods in Enzymology; Elsevier, 2015; Vol. 555, pp. 195–205 ISBN 978-0-12-801511-7.
- Egertova, M.; Giang, D.K.; Cravatt, B.F.; Elphick, M.R. A New Perspective on Cannabinoid Signalling: Complementary Localization of Fatty Acid Amide Hydrolase and the CB1 Receptor in Rat Brain. 1998.
- Huang, W.-J.; Chen, W.-W.; Zhang, X. Endocannabinoid System: Role in Depression, Reward and Pain Control (Review). Molecular Medicine Reports 2016, 14, 2899–2903. [Google Scholar] [CrossRef] [PubMed]
- Kapanda, C.N.; Muccioli, G.G.; Labar, G.; Poupaert, J.H.; Lambert, D.M. Bis(Dialkylaminethiocarbonyl)Disulfides as Potent and Selective Monoglyceride Lipase Inhibitors. J. Med. Chem. 2009, 52, 7310–7314. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Hillard, C.J. Role of Endocannabinoid Signaling in Anxiety and Depression. In Behavioral Neurobiology of the Endocannabinoid System; Kendall, D., Alexander, S., Eds.; Current Topics in Behavioral Neurosciences; Springer Berlin Heidelberg: Berlin, Heidelberg, 2009; Volume 1, pp. 347–347. ISBN 978-3-540-88954-0. [Google Scholar]
- Saito, V.M.; Wotjak, C.T.; Moreira, F.A. Exploração Farmacológica Do Sistema Endocanabinoide: Novas Perspectivas Para o Tratamento de Transtornos de Ansiedade e Depressão? Rev. Bras. Psiquiatr. 2010, 32, 57–514. [Google Scholar] [CrossRef]
- Viveros, M.; Marco, E.; File, S. Endocannabinoid System and Stress and Anxiety Responses. Pharmacology Biochemistry and Behavior 2005, 81, 331–342. [Google Scholar] [CrossRef]
- Mechoulam, R.; Parker, L.A. The Endocannabinoid System and the Brain. Annu. Rev. Psychol. 2013, 64, 21–47. [Google Scholar] [CrossRef]
- Herkenham, M.; Lynn, A.B.; Little, M.D.; Johnson, M.R.; Melvin, L.S.; De Costa, B.R.; Rice, K.C. Cannabinoid Receptor Localization in Brain. Proc. Natl. Acad. Sci. U.S.A. 1990, 87, 1932–1936. [Google Scholar] [CrossRef]
- Hill, M.N.; Hillard, C.J.; Bambico, F.R.; Patel, S.; Gorzalka, B.B.; Gobbi, G. The Therapeutic Potential of the Endocannabinoid System for the Development of a Novel Class of Antidepressants. Trends in Pharmacological Sciences 2009, 30, 484–493. [Google Scholar] [CrossRef]
- Tejeda-Martínez, A.R.; Viveros-Paredes, J.M.; Hidalgo-Franco, G.V.; Pardo-González, E.; Chaparro-Huerta, V.; González-Castañeda, R.E.; Flores-Soto, M.E. Chronic Inhibition of FAAH Reduces Depressive-Like Behavior and Improves Dentate Gyrus Proliferation after Chronic Unpredictable Stress Exposure. Behavioural Neurology 2021, 2021, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Ware, T.B.; Lee, H.-C.; Hsu, K.-L. Lipid-Metabolizing Serine Hydrolases in the Mammalian Central Nervous System: Endocannabinoids and Beyond. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 2019, 1864, 907–921. [Google Scholar] [CrossRef] [PubMed]
- Gomes-de-Souza, L.; A Oliveira, L.; Benini, R.; Rodella, P.; Costa-Ferreira, W.; Crestani, C.C. Involvement of Endocannabinoid Neurotransmission in the Bed Nucleus of Stria Terminalis in Cardiovascular Responses to Acute Restraint Stress in Rats. British Journal of Pharmacology 2016, 2016, 173. [Google Scholar] [CrossRef]
- Seibenhener, M.L.; Wooten, M.C. Use of the Open Field Maze to Measure Locomotor and Anxiety-like Behavior in Mice. JoVE 2015, 52434. [Google Scholar] [CrossRef]
- Cohen, I.L.; Gardner, J.M.; Karmel, B.Z.; Kim, S.-Y. Rating Scale Measures Are Associated with Noldus EthoVision-XT Video Tracking of Behaviors of Children on the Autism Spectrum. Mol Autism 2014, 5, 15. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An Analysis of Variance Test for Normality (Complete Samples). 2024.
- Kimura, H. Hydrogen Sulfide (H2S) and Polysulfide (H2Sn) Signaling: The First 25 Years. Biomolecules 2021, 11, 896. [Google Scholar] [CrossRef] [PubMed]
- Kormos, V.; Kecskés, A.; Farkas, J.; Gaszner, T.; Csernus, V.; Alomari, A.; Hegedüs, D.; Renner, É.; Palkovits, M.; Zelena, D.; et al. Peptidergic Neurons of the Edinger–Westphal Nucleus Express TRPA1 Ion Channel That Is Downregulated Both upon Chronic Variable Mild Stress in Male Mice and in Humans Who Died by Suicide. jpn 2022, 47, E162–E175. [Google Scholar] [CrossRef]
- Al-Omari, A.; Kecskés, M.; Gaszner, B.; Biró-Sütő, T.; Fazekas, B.; Berta, G.; Kuzma, M.; Pintér, E.; Kormos, V. Functionally Active TRPA1 Ion Channel Is Downregulated in Peptidergic Neurons of the Edinger-Westphal Nucleus upon Acute Alcohol Exposure. Front. Cell Dev. Biol. 2023, 10, 1046559. [Google Scholar] [CrossRef]
- Konkoly, J.; Kormos, V.; Gaszner, B.; Correia, P.; Berta, G.; Biró-Sütő, T.; Zelena, D.; Pintér, E. Transient Receptor Potential Ankyrin 1 Ion Channel Expressed by the Edinger-Westphal Nucleus Contributes to Stress Adaptation in Murine Model of Posttraumatic Stress Disorder. Front. Cell Dev. Biol. 2022, 10, 1059073. [Google Scholar] [CrossRef]
- Renard, C.E.; Dailly, E.; David, D.J.P.; Hascoet, M.; Bourin, M. Monoamine Metabolism Changes Following the Mouse Forced Swimming Test but Not the Tail Suspension Test. Fundamemntal Clinical Pharma 2003, 17, 449–455. [Google Scholar] [CrossRef]
- Bhatia, M. H2S and Substance P in Inflammation. In Methods in Enzymology; Elsevier, 2015; Vol. 555, pp. 195–205 ISBN 978-0-12-801511-7.
- Milicic, M.; Gaszner, B.; Berta, G.; Pintér, E.; Kormos, V. The Lack of TRPA1 Ion Channel Does Not Affect the Chronic Stress-Induced Activation of the Locus Ceruleus. IJMS 2024, 25, 1765. [Google Scholar] [CrossRef] [PubMed]
- Kozicz, T.; Tilburg-Ouwens, D.; Faludi, G.; Palkovits, M.; Roubos, E. Gender-Related Urocortin 1 and Brain-Derived Neurotrophic Factor Expression in the Adult Human Midbrain of Suicide Victims with Major Depression. Neuroscience 2008, 152, 1015–1023. [Google Scholar] [CrossRef]
- Bouwknecht, J.A.; Spiga, F.; Staub, D.R.; Hale, M.W.; Shekhar, A.; Lowry, C.A. Differential Effects of Exposure to Low-Light or High-Light Open-Field on Anxiety-Related Behaviors: Relationship to c-Fos Expression in Serotonergic and Non-Serotonergic Neurons in the Dorsal Raphe Nucleus. Brain Research Bulletin 2007, 72, 32–43. [Google Scholar] [CrossRef]
- Neufeld-Cohen, A.; Tsoory, M.M.; Evans, A.K.; Getselter, D.; Gil, S.; Lowry, C.A.; Vale, W.W.; Chen, A. A Triple Urocortin Knockout Mouse Model Reveals an Essential Role for Urocortins in Stress Recovery. Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 19020–19025. [Google Scholar] [CrossRef]
- Kozicz, T. The Missing Link; the Significance of Urocortin 1/Urocortin 2 in the Modulation of the Dorsal Raphe Serotoninergic System. Mol Psychiatry 2010, 15, 340–341. [Google Scholar] [CrossRef]
- Kovács, K.J. Measurement of Immediate-Early Gene Activation- C-fos and Beyond. J Neuroendocrinology 2008, 20, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Bittencourt, J.C.; Vaughan, J.; Arias, C.; Rissman, R.A.; Vale, W.W.; Sawchenko, P.E. Urocortin Expression in Rat Brain: Evidence Against a Pervasive Relationship of Urocortin-Containing Projections With Targets Bearing Type 2 CRF Receptors. J. Comp. Neurol. 1999, 415, 285–312. [Google Scholar] [CrossRef]
- Kozicz, T. On the Role of Urocortin 1 in the Non-Preganglionic Edinger–Westphal Nucleus in Stress Adaptation. General and Comparative Endocrinology 2007, 153, 235–240. [Google Scholar] [CrossRef]
- Derks, N.M.; Roubos, E.W.; Kozicz, T. Presence of Estrogen Receptor β in Urocortin 1-Neurons in the Mouse Non-Preganglionic Edinger–Westphal Nucleus. General and Comparative Endocrinology 2007, 153, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Isgor, C.; Cecchi, M.; Kabbaj, M.; Akil, H.; Watson, S.J. Estrogen Receptor β in the Paraventricular Nucleus of Hypothalamus Regulates the Neuroendocrine Response to Stress and Is Regulated by Corticosterone. Neuroscience 2003, 121, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Silva, I.A.; Helena, C.V.V.; Franci, C.R.; Lucion, A.B.; Anselmo-Franci, J.A. Modulatory Role of Locus Coeruleus and Estradiol on the Stress Response of Female Rats. Endocr 2009, 35, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.; Kawano, J.; Yanai, A.; Fujinaga, R.; Tanaka, M.; Watanabe, Y.; Shinoda, K. Expression of Estrogen Receptors (α, β) and Androgen Receptor in Serotonin Neurons of the Rat and Mouse Dorsal Raphe Nuclei; Sex and Species Differences. Neuroscience Research 2004, 49, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Hasunuma, K.; Murakawa, T.; Takenawa, S.; Mitsui, K.; Hatsukano, T.; Sano, K.; Nakata, M.; Ogawa, S. Estrogen Receptor β in the Lateral Septum Mediates Estrogen Regulation of Social Anxiety-like Behavior in Male Mice. Neuroscience 2024, 537, 126–140. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



