Submitted:
31 May 2024
Posted:
03 June 2024
You are already at the latest version
Abstract
Keywords:
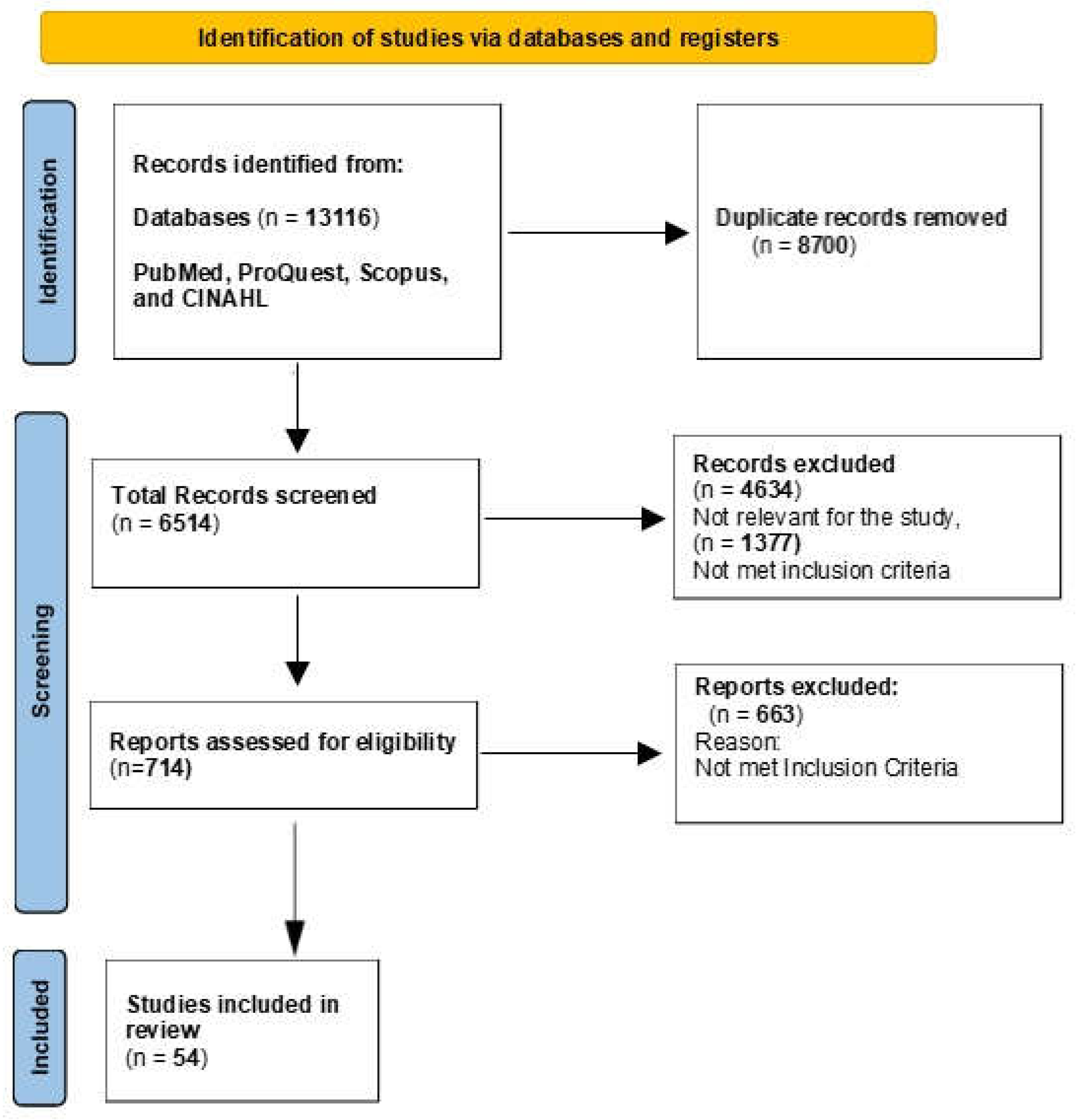
Introduction
Materials and Methods
Results
Discussion
Treatment Modalities
1. Conventional Therapy
1.1. Transfusion Therapy with Iron Chelation Therapy (ICT)
1.2. Splenectomy
1.3. Hemoglobin F Inducers
2. Novel Modalities
2.1. Gene Therapy
- Extraction of hematopoietic stem and progenitor cells (HSPCs): HSPCs, which can undergo self-renewal and multilineage differentiation, are found in the bone marrow. These cells express CD34 and are responsible for producing blood cells throughout a person's lifetime. Collection of the cells can be done either from the bone marrow or the peripheral blood [30]. The conventional method of collecting autologous bone marrow is invasive and yields insufficient CD34+ HSPCs. Patients with B-thalassemia have an increased accumulation of erythroid precursors in their bone marrow, which can hinder the harvesting of an adequate dose of stem cells. Therefore, it is necessary to mobilize the stem cells [31].
- Mobilization and apheresis: Mobilization of cells from bone marrow to peripheral blood is carried out using the recommended combination of granulocyte-colony stimulating factor (G-CSF) and plerixafor before collection by apheresis [32]. A recent study shows that the combined use of G-CSF and plerixafor provides an adequate yield of stem cells. [33]
- Myeloablation: Myeloablation of the bone marrow is the next step and is performed to create enough space for the gene modified HSPCs to be reinserted, ensuring efficient engraftment. The recommended myeloablative agent is Busulfan since the dose can be tailored and modified for each patient depending on the response to the drug [34].
- Modified HSPCs infusion: The final step is the infusion of the cryopreserved genetically modified HSPCs, using 5% dimethylsulfoxide (DMSO) solution, intravenously after it meets all the release criteria (sterility, viability, purity, and vector copy number [VCN] for gene-insertions), and minimum cell dose criteria (>2-3 × 106 CD34+/kg) [34]. Gene therapy for TDT can be divided into two parts based on the method used, gene insertion and gene editing.
2.1.1. Gene Insertion
2.1.2. Gene Editing
2.1.3. Zynteglo
Conclusion
Acknowledgments
Declaration of Interest
References
- Genetic Counseling and Testing for Hereditary Hemochromatosis. In Hereditary Hemochromatosis; National Center for Biotechnology Information (US), 2018; Available online: https://www.ncbi.nlm.nih.gov/books/NBK545151/.
- Rund, D.; Rachmilewitz, E. Beta-Thalassemia. N Engl J Med 2005, 353, 1135–1146. [Google Scholar] [CrossRef] [PubMed]
- Cappellini, M.D.; Cohen, A.; Porter, J.; Taher, A.; Viprakasit, V. Guidelines for the management of transfusion dependent thalassaemia (TDT). Thalassemia International Federation 2014. [Google Scholar] [CrossRef]
- Fucharoen, S.; Weatherall, D.J. The Hemoglobin E Thalassemias. Cold Spring Harb Perspect Med 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- Taher, A.; Musallam, K.; Inati, A.; Weatherall, D.J. The Overlooked Genetic Blood Disorder: Beta Thalassemia Intermedia. JAMA 2021, 326, 980–990. [Google Scholar] [CrossRef]
- Ben Salah, N.; Bou-Fakhredin, R.; Mellouli, F.; Taher, A.T. Revisiting beta thalassemia intermedia: past, present, and future prospects. Hematology 2017, 22, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Kattamis, A.; Kwiatkowski, J.L.; Aydinok, Y. Thalassaemia. Lancet 2022, 399, 2310–2324. [Google Scholar] [CrossRef] [PubMed]
- Adly, A.A.; Ismail, E.A. Management of Children With β-Thalassemia Intermedia: Overview, Recent Advances, and Treatment Challenges. J Pediatr Hematol Oncol 2018, 40, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.T.; Sayani, F.; Trompeter, S.; Drasar, E.; Piga, A. Challenges of blood transfusions in β-thalassemia. Blood Rev 2019, 37, 100588. [Google Scholar] [CrossRef]
- Cappellini, M.D.; Cohen, A.; Porter, J.; Taher, A.; Viprakasit, V. (Eds.) Guidelines for the Management of Transfusion Dependent Thalassaemia (TDT), 3rd ed. Thalassaemia International Federation: Nicosia, Cyprus, 2014. [Google Scholar] [CrossRef]
- Vichinsky, E.; Levine, L.; Bhatia, S.; et al. Standards of Care Guidelines for Thalassemia. Children's Hospital and Research Center: Oakland, CA, 2012. [Google Scholar]
- Davis, B.A.; Porter, J.B. Results of long term iron chelation treatment with deferoxamine. In Iron Chelation Therapy; Hershko, C., Konijn, A.M., Link, G., Eds.; Kluwer Academic Publishers: Boston, MA, 2002; pp. 91–125. [Google Scholar] [CrossRef]
- Gabutti, V.; Piga, A. Results of long-term iron-chelating therapy. Acta Haematol. 1996, 95, 26–36. [Google Scholar] [CrossRef]
- Yardumian, A.; Telfer, P.; Constantinou, G.; et al. Standards for the Clinical Care of Children and Adults with Thalassaemia in the UK. United Kingdom Thalassaemia Society: London, UK, 2005. [Google Scholar]
- Ladis, V.; Chouliaras, G.; Berdoukas, V.; et al. Relation of chelation regimes to cardiac mortality and morbidity in patients with thalassaemia major: an observational study from a large Greek Unit. Eur J Haematol 2010, 85, 335–344. [Google Scholar] [CrossRef]
- Borgna-Pignatti, C.; Rugolotto, S.; De Stefano, P.; et al. Survival and complications in patients with thalassemia major treated with transfusion and deferoxamine. Haematologica 2004, 89, 1187–1193. [Google Scholar]
- Ali, S.; Mumtaz, S.; Shakir, H.A.; et al. Current status of beta-thalassemia and its treatment strategies. Mol Genet Genomic Med 2021, 9. [Google Scholar] [CrossRef]
- Pecorari, L.; Savelli, A.; Cuna, C.D.; Fracchia, S.; Borgna-Pignatti, C. The role of splenectomy in thalassemia major. An update. Acta Pediatr Mediterr 2008, 24, 57–60. [Google Scholar]
- Ikeda, M.; Sekimoto, M.; Takiguchi, S.; et al. High incidence of thrombosis of the portal venous system after laparoscopic splenectomy: a prospective study with contrast-enhanced CT scan. Ann Surg. 2005, 241, 208–214. [Google Scholar] [CrossRef]
- Merchant, R.H.; Shah, A.R.; Ahmad, J.; Karnik, A.; Rai, N. Post splenectomy outcome in β-thalassemia. Indian J Pediatr. 2015, 82, 1097–1100. [Google Scholar] [CrossRef]
- Hassan, M.N.; Tahereb, G.M.; Ahmad, T.; et al. Correlation of splenectomy with portal vein thrombosis in beta-thalassemia major. J Pak Med Assoc. 2011, 61, 760–762. [Google Scholar]
- Borgna-Pignatti, C.; Carnelli, V.; Caruso, V.; et al. Thromboembolic events in beta thalassemia major: an Italian multicenter study. Acta Haematol. 1998, 99, 76–79. [Google Scholar] [CrossRef]
- Musallam, K.M.; Taher, A.T.; Cappellini, M.D.; Sankaran, V.G. Clinical experience with fetal hemoglobin induction therapy in patients with β-thalassemia. Blood. 2013, 121, 2199–2205. [Google Scholar] [CrossRef]
- Finotti, A.; Breda, L.; Lederer, C.W.; et al. Recent trends in the gene therapy of β-thalassemia. J Blood Med. 2015, 6, 69–85. [Google Scholar] [CrossRef]
- Pace, B.S.; Liu, L.; Li, B.; Makala, L.H. Cell signaling pathways involved in drug-mediated fetal hemoglobin induction: Strategies to treat sickle cell disease. Exp Biol Med (Maywood) 2015, 240, 1050–1064. [Google Scholar] [CrossRef]
- Cappellini, M.D.; Porter, J.B.; Viprakasit, V.; Taher, A.T. A paradigm shift on beta-thalassaemia treatment: How will we manage this old disease with new therapies? Blood Rev. 2018, 32, 300–311. [Google Scholar] [CrossRef]
- Foong, W.C.; Ho, J.J.; Loh, C.K.; Viprakasit, V. Hydroxyurea for reducing blood transfusion in non-transfusion dependent beta thalassaemias. Cochrane Database Syst Rev 2016. [Google Scholar] [CrossRef]
- Taher, A.; Vichinsky, E.; Musallam, K.; Cappellini, M.D.; Viprakasit, V. Guidelines for the Management of Non Transfusion Dependent Thalassaemia (NTDT). Thalassaemia International Federation: Nicosia, Cyprus, 2013. [Google Scholar]
- Soni, S. Gene Therapies for Transfusion-Dependent β-Thalassemia. Indian Pediatr. 2021, 58, 667–674. [Google Scholar] [CrossRef]
- Bhushan, B.; Khanna, V.K.; Dubey, A.; et al. Emerging trends in gene therapy for β-thalassemia: A systematic review. Mol Ther Nucleic Acids 2020, 21, 1028–1038. [Google Scholar] [CrossRef]
- Origa, R.; Galanello, R.; Ganz, T.; et al. Liver iron concentrations and urinary hepcidin in β-thalassemia. Haematologica 2009, 94, 503–507. [Google Scholar] [CrossRef]
- Origa, R.; Galanello, R.; Ganz, T.; et al. Liver iron concentrations and urinary hepcidin in β-thalassemia. Haematologica 2009, 94, 503–507. [Google Scholar] [CrossRef]
- Davis, B.A.; O'Sullivan, C.; Jarritt, P.H.; Porter, J.B. Value of sequential monitoring of left ventricular ejection fraction in the management of thalassemia major. Blood Cells Mol Dis 2004, 33, 1–5. [Google Scholar] [CrossRef]
- Schulz, C.; Von Andrian, U.H.; Massberg, S. Hematopoietic stem and progenitor cells: their mobilization and homing to bone marrow and peripheral tissue. Immunol Res. 2009, 44, 160–168. [Google Scholar] [CrossRef]
- Soni, S. Gene therapies for transfusion dependent β-thalassemia: Current status and critical criteria for success. Am J Hematol. 2020, 95, 1099–1112. [Google Scholar] [CrossRef]
- Hargrove, P.W.; Kepes, S.; Hanawa, H.; et al. Globin Lentiviral Vector Insertions Can Perturb the Expression of Endogenous Genes in β-thalassemic Hematopoietic Cells. Mol Ther. 2008, 16, 525–533. [Google Scholar] [CrossRef]
- Thompson, A.A.; Walters, M.C.; Kwiatkowski, J.; et al. Gene Therapy in Patients with Transfusion-Dependent β-Thalassemia. N Engl J Med. 2018, 378, 1479–1493. [Google Scholar] [CrossRef]
- Marktel, S.; Scaramuzza, S.; Cicalese, M.P.; et al. Intrabone hematopoietic stem cell gene therapy for adult and pediatric patients affected by transfusion-dependent β-thalassemia. Nat Med. 2019, 25, 234–241. [Google Scholar] [CrossRef]
- Lal, A.; Locatelli, F.; Kwiatkowski, J.L.; et al. Northstar-3: interim results from a phase 3 study evaluating Lentiglobin gene therapy in patients with transfusion-dependent β-thalassemia and either a β0 or IVS-I-110 mutation at both alleles of the HBB gene. Blood 2019, 134 (Suppl. 1), 815. [Google Scholar] [CrossRef]
- Hoban, M.D.; Bauer, D.E. A genome editing primer for the hematologist. Blood. 2016, 127, 2525–2535. [Google Scholar] [CrossRef]
- Zhang, X.; Tee, L.Y.; Wang, X.; Huang, Q.; Yang, S. Off-target Effects in CRISPR/Cas9-mediated Genome Engineering. Mol Ther Nucleic Acids. 2015, 4. [Google Scholar] [CrossRef]
- Shariati, L.; Rohani, F.; Heidari Hafshejani, N.; et al. Disruption of SOX6 gene using CRISPR/Cas9 technology for gamma-globin reactivation: An approach towards gene therapy of β-thalassemia. J Cell Biochem. 2018, 119, 9357–9363. [Google Scholar] [CrossRef]
- Paschoudi, K.; Yannaki, E.; Psatha, N. Precision Editing as a Therapeutic Approach for β-Hemoglobinopathies. Int J Mol Sci. 2023, 24, 9527. [Google Scholar] [CrossRef]
- Gene therapy for transfusion-dependent β-thalassemia and sickle cell disease. ClinicalTrials.gov. Identifier: NCT03655678.
- Lentiglobin gene therapy for β-thalassemia. ClinicalTrials.gov. Identifier: NCT03745287.
- Frangoul, H.; Altshuler, D.; Cappellini, M.D.; et al. CRISPR-Cas9 Gene Editing for Sickle Cell Disease and β-Thalassemia. N Engl J Med. 2021, 384, 252–260. [Google Scholar] [CrossRef]
- Amorosi, D. Gene therapy a potential 'functional cure' for sickle cell disease, beta thalassemia. Hematol Oncol Today. 2023, 24, 6. [Google Scholar]
- Zynteglo (betibeglogene autotemcel) package insert, U.S. Food and Drug Administration website. Available online: https://www.fda.gov/vaccines-blood-biologics/zynteglo.
- Thompson, A.A.; Walters, M.C.; Kwiatkowski, J.; et al. Gene Therapy in Patients with Transfusion-Dependent β-Thalassemia. N Engl J Med. 2018, 378, 1479–1493. [Google Scholar] [CrossRef]
- Gonsalves, S.; Salpeter, S.R.; Kaplan, R.M.; et al. The impact of gene therapy for transfusion-dependent β-thalassemia: A systematic review and meta-analysis. Am J Hematol. 2021, 96, 768–776. [Google Scholar] [CrossRef]
- Cappellini, M.D.; Cohen, A.R.; Porter, J.B.; et al. Improvement in Health-Related Quality of Life and Well-being in Patients with Transfusion-Dependent β-Thalassemia (TDT) Treated with Betibeglogene Autotemcel (LentiGlobin for β-Thalassemia, beti-cel): Northstar-2 and Northstar-3 Studies. Blood. 2021, 138 (Suppl. 1), 3085. [Google Scholar] [CrossRef]
- Zynteglo (betibeglogene autotemcel) package information, U.S. Food and Drug Administration website. Available online: https://www.fda.gov/media/160991/download.
- Arya, Y.A.; Sahi, P.K. Cell-Based Gene Therapy for β-Thalassemia. Indian Pediatr. 2023, 60, 313–315. [Google Scholar] [CrossRef] [PubMed]
- Bluebird Bio announces U.S. commercial infrastructure to enable Zynteglo. Bluebird Bio website. Available online: https://investor.bluebirdbio.com/news-releases/news-release-details/bluebird-bio-announces-us-commercial-infrastructure-enable.
- Zynteglo. European Medicines Agency website. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/zynteglo.
- Locatelli, F.; Thompson, A.A.; Kwiatkowski, J.L.; et al. Betibeglogene Autotemcel Gene Therapy for Non–β0/β0 Genotype β-Thalassemia. N Engl J Med. 2021, 384, 547–559. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).