Submitted:
29 May 2024
Posted:
30 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
COVID-19 During the 2023-2024 Season
Pediatric Population
Adults 18-49 Years
Adults 50-64 Years
Older Adults Age ≥ 65 Years
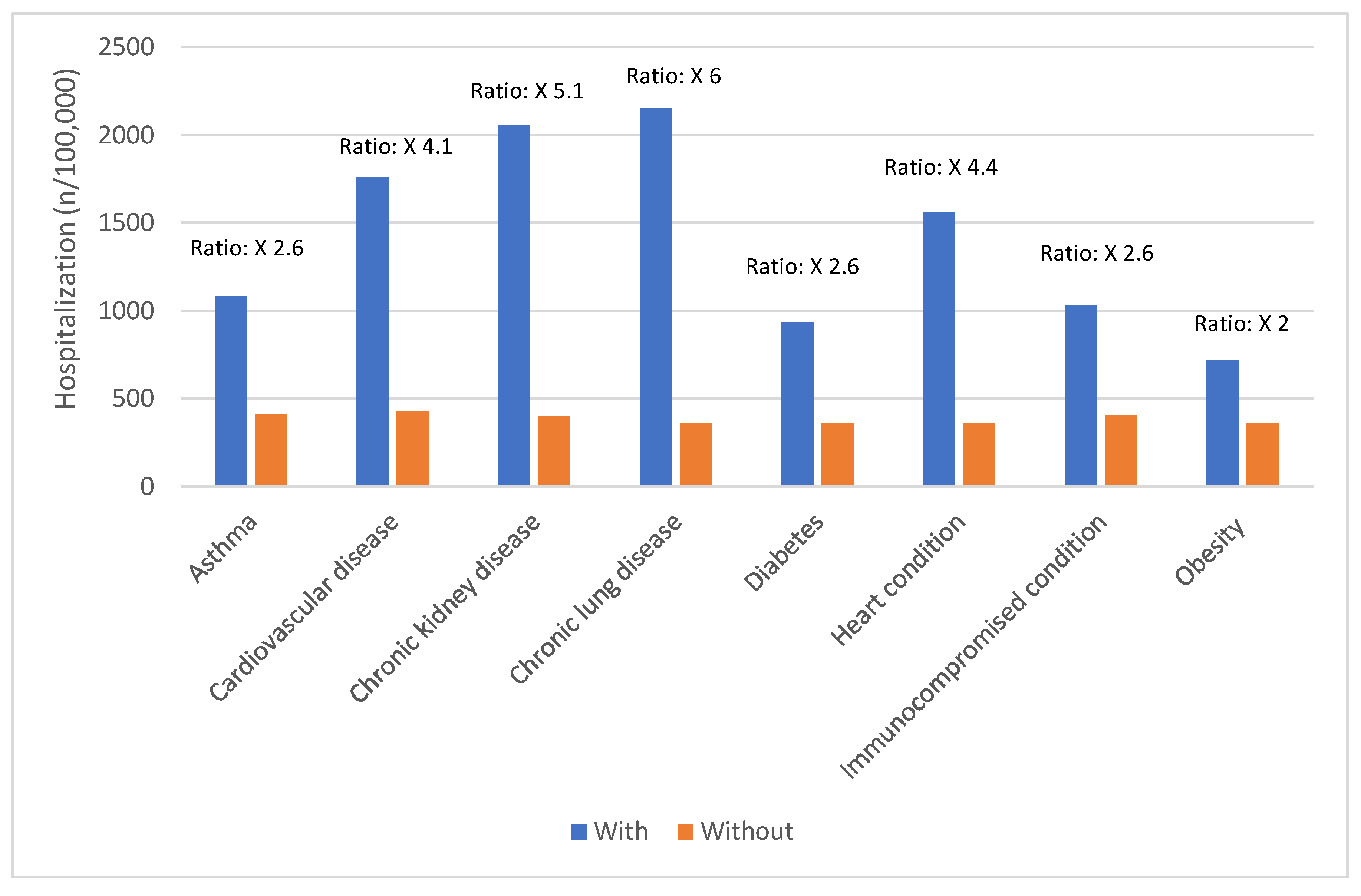
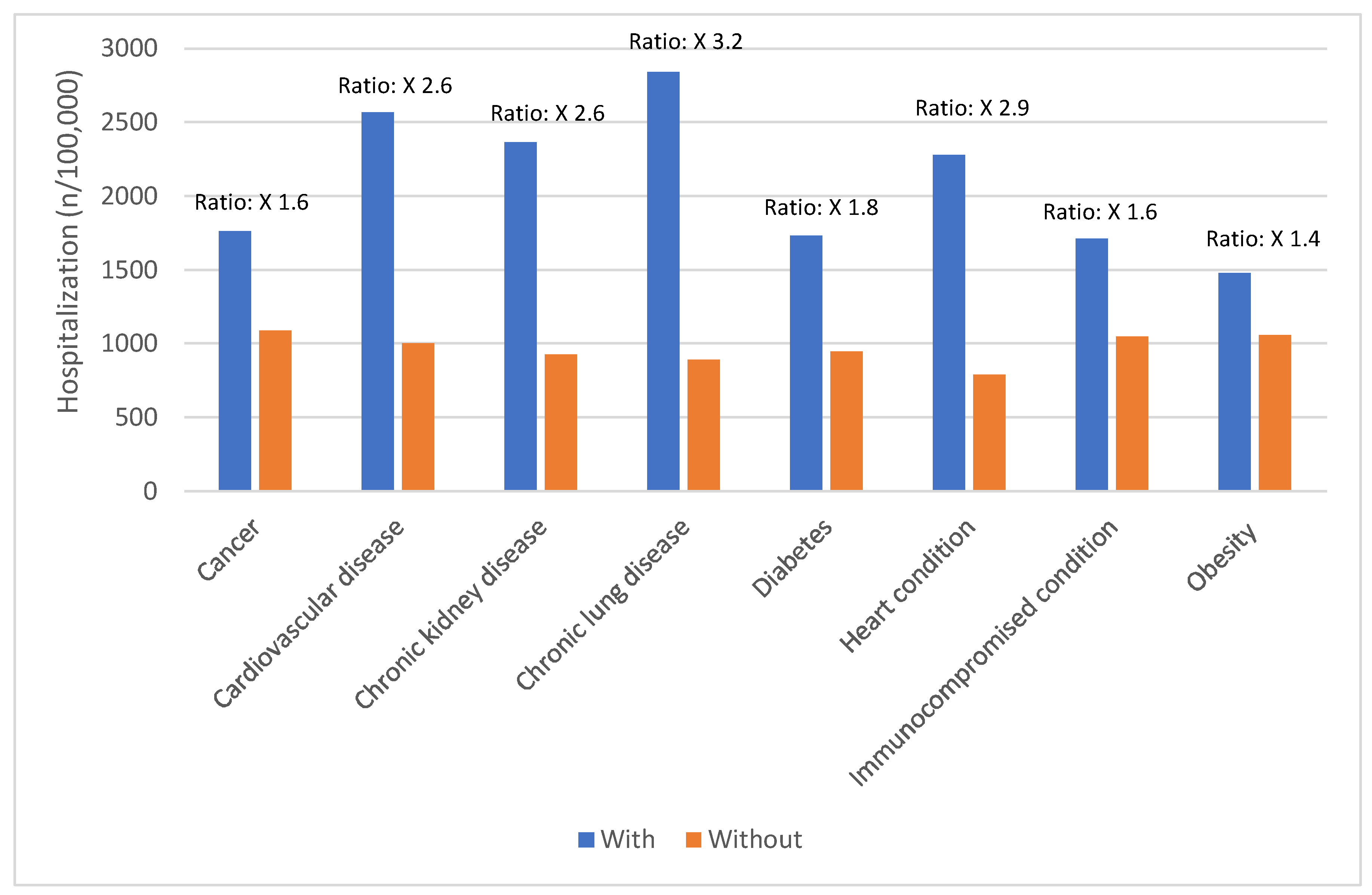
Risk Factors for Severe COVID-19 Outcomes
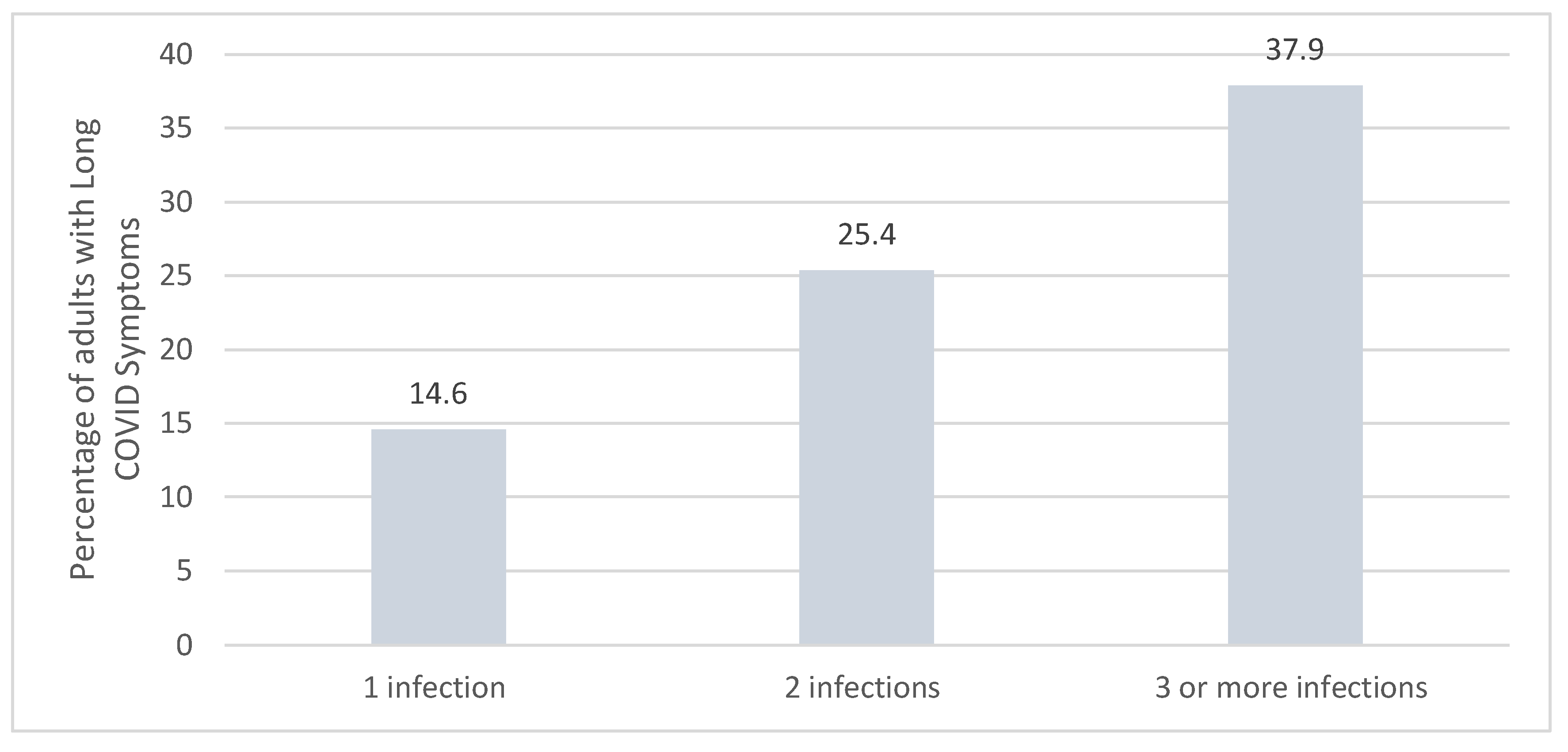
The Compounding Effect of Long COVID
COVID-19 Burden vs. Influenza
The Continued Evolution of COVID-19
COVID-19 2023/2024 Vaccine
FDA Approval and ACIP Recommendation
Safety, Immunogenicity, and Effectiveness of the Vaccine
2023/2024. Vaccine Uptake in Different Populations
- Perceived risk of getting ill or severity of COVID-19 versus safety/tolerability of the vaccine
- Effectiveness of the COVID-19 vaccine
- Safety of the COVID-19 vaccine.
Adapting Strategies for Future COVID-19 Seasons
- a)
- Changes in the COVID-19 vaccine approval process to align with influenza to allow for co-administration
- b)
- Increased integration of COVID-19 vaccine into routine delivery of care
- c)
- Communication and engagement of healthcare providers for optimal patient education, and overcoming the challenges of misinformation and distrust, along with vaccine delivery.
COVID-19 Vaccine Approval Process
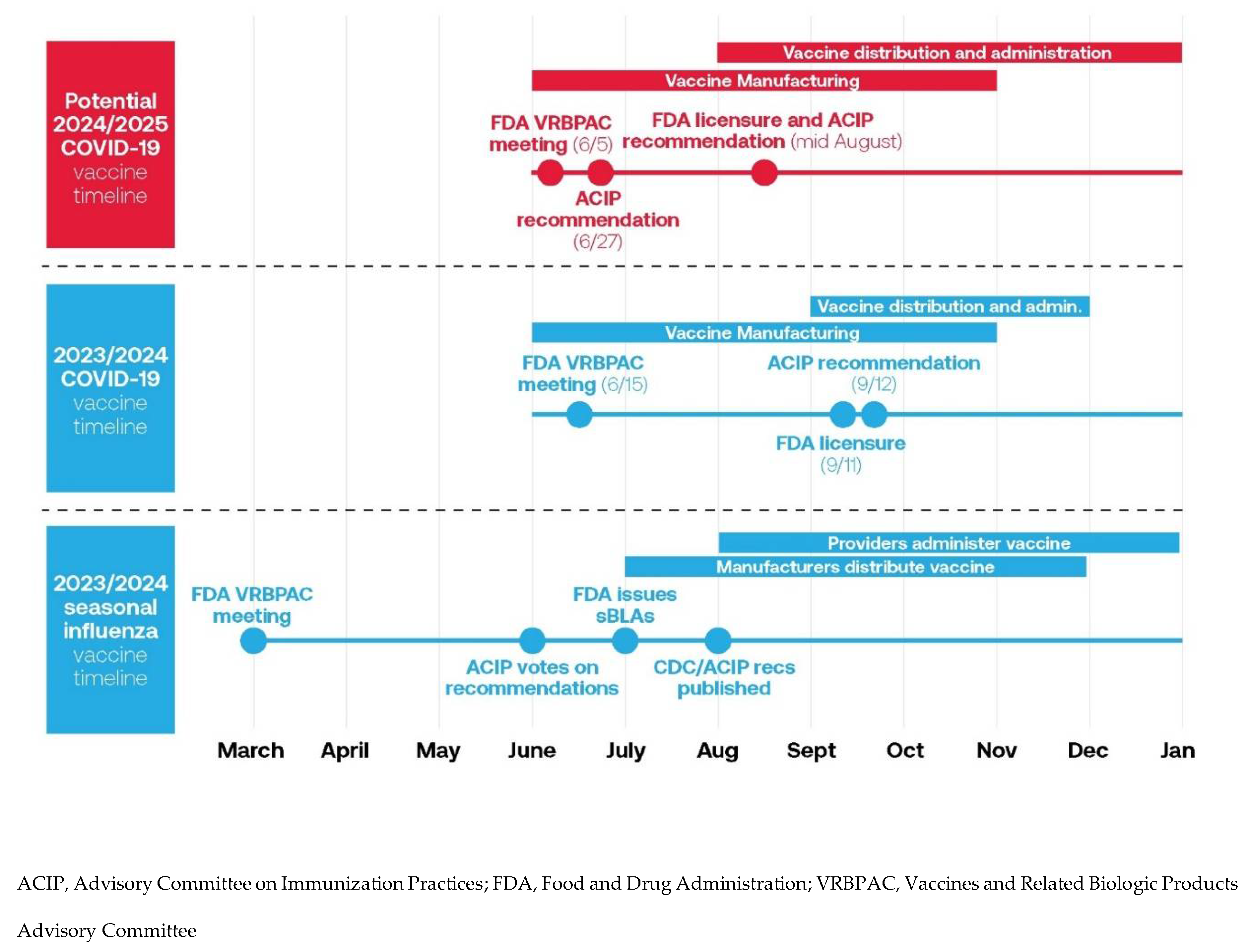
Increased Integration of COVID-19 Vaccine into Routine Delivery of Care
Engaging Providers for Fall 2024 Delivery
Addressing Key Barriers to COVID-19 Vaccine Uptake
Improving Vaccine Access
Simplifying Vaccine Messaging and the Delivery of COVID-19 Vaccines
- The perceived risk of getting ill or severity of COVID-19 versus the efficacy and safety of the vaccine.
- Effectiveness of the vaccine.
- Safety of the vaccine.
Combating Misinformation and Disinformation
Key Takeaways and Recommendations
Acknowledgments
Conflicts of Interest
References
- The Commonwealth Fund. Two Years of U.S. COVID-19 Vaccines Have Prevented Millions of Hospitalizations and Deaths. [CrossRef]
- CDC. COVID Data Tracker. Centers for Disease Control and Prevention. Accessed April 12, 2024. https://covid.cdc.gov/covid-data-tracker.
- CDC. Trends in United States COVID-19 Hospitalizations, Deaths, Emergency Department (ED) Visits, and Test Positivity by Geographic Area. Centers for Disease Control and Prevention. Accessed April 15, 2024. https://covid.cdc.gov/covid-data-tracker.
- CDC. Adult Coverage and Intent. Published April 9, 2024. Accessed April 15, 2024. https://www.cdc.gov/vaccines/imz-managers/coverage/covidvaxview/interactive/adult-coverage-vaccination.html.
- CDC. Child Coverage and Parental Intent for Vaccination. Published March 6, 2024. Accessed April 15, 2024. https://www.cdc.gov/vaccines/imz-managers/coverage/covidvaxview/interactive/children-coverage-vaccination.html.
- CDC. COVID-19 Vaccinations in the United States. Centers for Disease Control and Prevention. Accessed April 15, 2024. https://covid.cdc.gov/covid-data-tracker.
- Regan JJ. Use of Updated COVID-19 Vaccines 2023–2024 Formula for Persons Aged ≥6 Months: Recommendations of the Advisory Committee on Immunization Practices — United States, September 2023. MMWR Morb Mortal Wkly Rep. 2023;72. [CrossRef]
- CDC. COVID-NET - COVID-19-Associated Hospitalization Surveillance Network: A Respiratory Virus Hospitalization Surveillance Network (RESP-NET) Platform. Centers for Disease Control and Prevention. Published February 11, 2020. Accessed May 6, 2024. https://www.cdc.gov/coronavirus/2019-ncov/covidnetdashboard/de/powerbi/dashboard.html.
- CDC. Laboratory-Confirmed Influenza Hospitalizations. Accessed May 6, 2024. https://gis.cdc.gov/GRASP/Fluview/FluHospRates.html.
- Zambrano LD, Newhams MM, Simeone RM, et al. Characteristics and Clinical Outcomes of Vaccine-Eligible US Children Under-5 Years Hospitalized for Acute COVID-19 in a National Network. The Pediatric Infectious Disease Journal. 2024;43(3):242. [CrossRef]
- Link-Gelles R. Estimates of Bivalent mRNA Vaccine Durability in Preventing COVID-19–Associated Hospitalization and Critical Illness Among Adults with and Without Immunocompromising Conditions — VISION Network, September 2022–April 2023. MMWR Morb Mortal Wkly Rep. 2023;72. [CrossRef]
- Havers, FP. COVID-19–Associated Hospitalizations among Infants, Children and Adults — COVID-NET, January–August 2023. Accessed May 6, 2024. https://stacks.cdc.gov/view/cdc/132883.
- CDC. Health Policy Data Requests - Percent of U.S. Adults 55 and Over with Chronic Conditions. Published February 7, 2019. Accessed May 6, 2024. https://www.cdc.gov/nchs/health_policy/adult_chronic_conditions.htm.
- CDC. Underlying Medical Conditions Associated with Higher Risk for Severe COVID-19: Information for Healthcare Professionals. Centers for Disease Control and Prevention. Published , 2024. Accessed May 6, 2024. https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-care/underlyingconditions. 12 April.
- Taylor, C.A. COVID-19–Associated Hospitalizations Among U.S. Adults Aged ≥65 Years — COVID-NET, 13 States, January–August 2023. <italic>MMWR Morb Mortal Wkly Rep</italic>. 2023;72. [CrossRef]
- CDC. Preliminary Flu Burden Estimates, 2022-23 Season. Centers for Disease Control and Prevention. Published November 22, 2023. Accessed May 6, 2024. https://www.cdc.gov/flu/about/burden/2022-2023.htm.
- CDC. The Changing Threat of COVID-19. Published March 1, 2024. Accessed April 15, 2024. https://www.cdc.gov/ncird/whats-new/changing-threat-covid-19.html.
- Adams ML, Katz DL, Grandpre J. Population-Based Estimates of Chronic Conditions Affecting Risk for Complications from Coronavirus Disease, United States - Volume 26, Number 8—August 2020 - Emerging Infectious Diseases journal - CDC. [CrossRef]
- Kompaniyets L. Underlying Medical Conditions and Severe Illness Among 540,667 Adults Hospitalized With COVID-19, March 2020–March 2021. <italic>Prev Chronic Dis</italic>. 2021;18. [CrossRef]
- Bench2Practice. Hospitalizations* by Underlying Medical Condition. Accessed May 7, 2024. https://atlas.modernatx.com/en-US/bench2practice/Interactive-dashboard.
- Kopel H, Bogdanov A, Winer-Jones JP, et al. Comparison of COVID-19 and Influenza-Related Outcomes in the United States during Fall–Winter 2022–2023: A Cross-Sectional Retrospective Study. <italic>Diseases</italic>. 2024;12(1):16. [CrossRef]
- CDC. Long COVID - Household Pulse Survey - COVID-19. Published , 2024. Accessed May 6, 2024. https://www.cdc.gov/nchs/covid19/pulse/long-covid.htm.
- United States Census Bureau. International Database. Accessed April 28, 2024. https://www.census.gov/data-tools/demo/idb/#/pop?COUNTRY_YR_ANIM=2021&menu=popViz&COUNTRY_YEAR=2021&TREND_RANGE=1950,2100&TREND_STEP=10&TREND_ADD_YRS=&POP_YEARS=2024&CCODE=US&CCODE_SINGLE=US&popPages=BYAGE&ageGroup=5Y.
- Zheng YB, Zeng N, Yuan K, et al. Prevalence and risk factor for long COVID in children and adolescents: A meta-analysis and systematic review. <italic>J Infect Public Health</italic>. 2023;16(5):660-672. [CrossRef]
- Statistics Canada Government of Canada. Experiences of Canadians with long-term symptoms following COVID-19. Published December 8, 2023. Accessed May 7, 2024. https://www150.statcan.gc.ca/n1/pub/75-006-x/2023001/article/00015-eng.htm.
- 26. Koumpias AM, Schwartzman D, Fleming O. Long-haul COVID: healthcare utilization and medical expenditures 6 months post-diagnosis. BMC Health Services Research, 1010. [CrossRef]
- Michael B, Wood G, Sargent B, et al. Post-COVID cognitive deficits at one year are global and associated with elevated brain injury markers and grey matter volume reduction: national prospective study. Published online January 5, 2024. [CrossRef]
- CDC. RESP-NET Interactive Dashboard. Published October 6, 2023. Accessed April 15, 2024. https://www.cdc.gov/surveillance/resp-net/dashboard.html.
- Epic Research. Respiratory Illnesses. Epic Research. Accessed April 15, 2024. https://epicresearch.org/data-tracker/respiratory-illnesses.
- Kopel H, Bogdanov A, Winer-Jones JP, et al. Comparison of COVID-19 and Influenza-Related Outcomes in the United States during Fall–Winter 2022–2023: A Cross-Sectional Retrospective Study. <italic>Diseases</italic>. 2024;12(1):16. [CrossRef]
- CDC. Influenza Vaccination Coverage, Adults: COVIDVaxView. Published April 11, 2024. Accessed April 15, 2024. https://www.cdc.gov/flu/fluvaxview/dashboard/vaccination-adult-coverage.html.
- CDC. Monitoring Variant Proportions. Centers for Disease Control and Prevention. Published March 28, 2020. Accessed April 15, 2024. https://covid.cdc.gov/covid-data-tracker.
- Wang Q, Guo Y, Bowen A, et al. XBB.1.5 monovalent mRNA vaccine booster elicits robust neutralizing antibodies against emerging SARS-CoV-2 variants. Published online December 6, 2023:2023.11.26.568730. [CrossRef]
- Gayed J, Diya O, Lowry FS, et al. Safety and Immunogenicity of the Monovalent Omicron XBB.1.5-Adapted BNT162b2 COVID-19 Vaccine in Individuals ≥12 Years Old: A Phase 2/3 Trial. <italic>Vaccines</italic>. 2024;12(2):118. [CrossRef]
- Chalkias S, McGhee N, Whatley JL, et al. Safety and Immunogenicity of XBB.1.5-Containing mRNA Vaccines. Published online September 7, 2023:2023.08.22.23293434. [CrossRef]
- Andersson NW, Thiesson EM, Hviid A. Adverse Events After XBB.1.5-Containing COVID-19 mRNA Vaccines. <italic>JAMA</italic>. 2024;331(12):1057-1059. [CrossRef]
- DeCuir J. Interim Effectiveness of Updated 2023–2024 (Monovalent XBB.1.5) COVID-19 Vaccines Against COVID-19–Associated Emergency Department and Urgent Care Encounters and Hospitalization Among Immunocompetent Adults Aged ≥18 Years — VISION and IVY Networks, September 2023–January 2024. <italic>MMWR Morb Mortal Wkly Rep</italic>. 2024;73. [CrossRef]
- Kopel H, Araujo AB, Bogdanov A, et al. Effectiveness of the 2023-2024 Omicron XBB.1.5-containing mRNA COVID-19 vaccine (mRNA-1273.815) in preventing COVID-19-related hospitalizations and medical encounters among adults in the United States: An interim analysis. Published online April 12, 2024. [CrossRef]
- Wallace M. Evidence to Recommendations Framework: Accessed May 6, 2024. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2023-09-12/11-COVID-Wallace-508.pdf.
- Harris E. Amid Health Misinformation, Most Trust Physicians for Truth. <italic>JAMA</italic>. 2023;330(12):1128. [CrossRef]
- Lakshmi Panagiotakopouluos. Next Steps for the COVID-19 Vaccine Program. Presented at: ACIP Meeting; 2024/02/28. Accessed April 15, 2024. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2024-02-28-29/07-COVID-Panagiotakopoulos-508.pdf.
- CDC. 2023-2024 CDC Flu Vaccination Recommendations Adopted. Centers for Disease Control and Prevention. Published June 29, 2023. Accessed April 28, 2024. https://www.cdc.gov/flu/spotlights/2022-2023/flu-vaccination-recommendations-adopted.htm.
- Chatham-Stephens K. An update on COVID-19 vaccination coverage. Accessed May 6, 2024. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2024-02-28-29/03-COVID-Chatham-Stevens-508.pdf.
- Shen AK, Browne S, Srivastava T, Kornides ML, Tan ASL. Trusted messengers and trusted messages: The role for community-based organizations in promoting COVID-19 and routine immunizations. <italic>Vaccine</italic>. 2023;41(12):1994-2002. [CrossRef]
- CDC. Adult Immunization Schedule by Age. Published March 11, 2024. Accessed April 16, 2024. https://www.cdc.gov/vaccines/schedules/hcp/imz/adult.html.
- CDC. Adult Vaccinations Administered. Published April 9, 2024. Accessed April 16, 2024. https://www.cdc.gov/vaccines/imz-managers/coverage/covidvaxview/interactive/adult-vaccinations-administered.html.
- CDC. Influenza Vaccinations Administered in Pharmacies and Physician Medical Offices, Adults, United States | FluVaxView | Seasonal Influenza (Flu). Published April 11, 2024. Accessed April 16, 2024. https://www.cdc.gov/flu/fluvaxview/dashboard/vaccination-administered.html.
- CDC. Interim Clinical Considerations for Use of COVID-19 Vaccines. Published March 1, 2024. Accessed April 16, 2024. https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html.
- Shanahan EA, DeLeo RA, Albright EA, et al. Visual policy narrative messaging improves COVID-19 vaccine uptake. <italic>PNAS Nexus</italic>. 2023;2(4):pgad080. [CrossRef]
- Nyhan B, Reifler J, Richey S, Freed GL. Effective Messages in Vaccine Promotion: A Randomized Trial. <italic>Pediatrics</italic>. Published online March 3, 2014:peds.2013-2365. [CrossRef]
- Brewer NT, Chapman GB, Rothman AJ, Leask J, Kempe A. Increasing Vaccination: Putting Psychological Science Into Action. <italic>Psychol Sci Public Interest</italic>. 2017;18(3):149-207. [CrossRef]
- Brewer NT, Hall ME, Malo TL, Gilkey MB, Quinn B, Lathren C. Announcements Versus Conversations to Improve HPV Vaccination Coverage: A Randomized Trial. <italic>Pediatrics</italic>. 2016;139:e20161764. [CrossRef]
- Opel DJ, Heritage J, Taylor JA, et al. The Architecture of Provider-Parent Vaccine Discussions at Health Supervision Visits. <italic>Pediatrics</italic>. 2013;132(6):1037-1046. [CrossRef]
- Fogarty CT, Crues L. How to Talk to Reluctant Patients About the Flu Shot. <italic>fpm</italic>. 2017;24(5):6-8.
- Infodemiology. Infodemiology training for public health. Accessed May 28, 2024. https://training.infodemiology.com/publichealth.
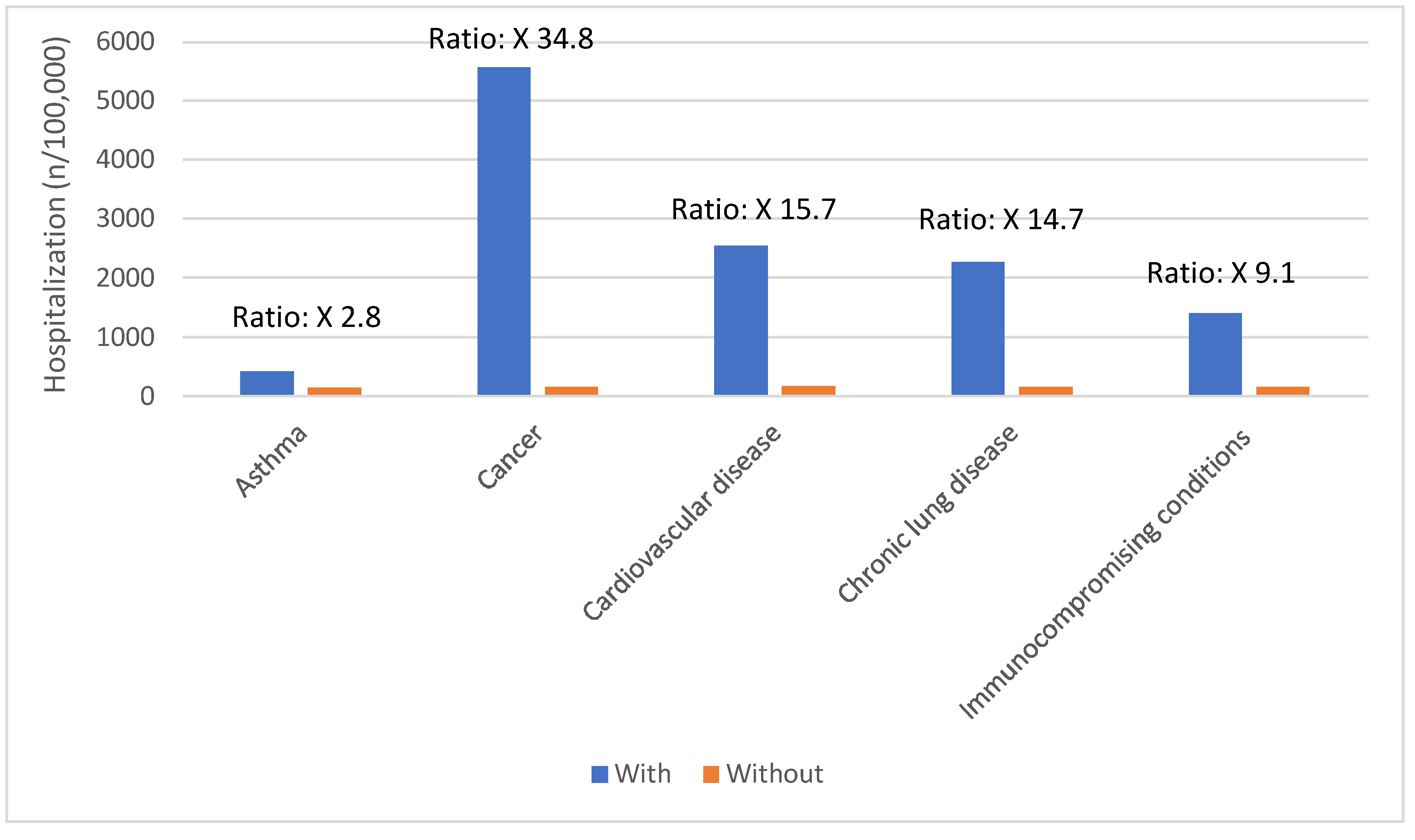
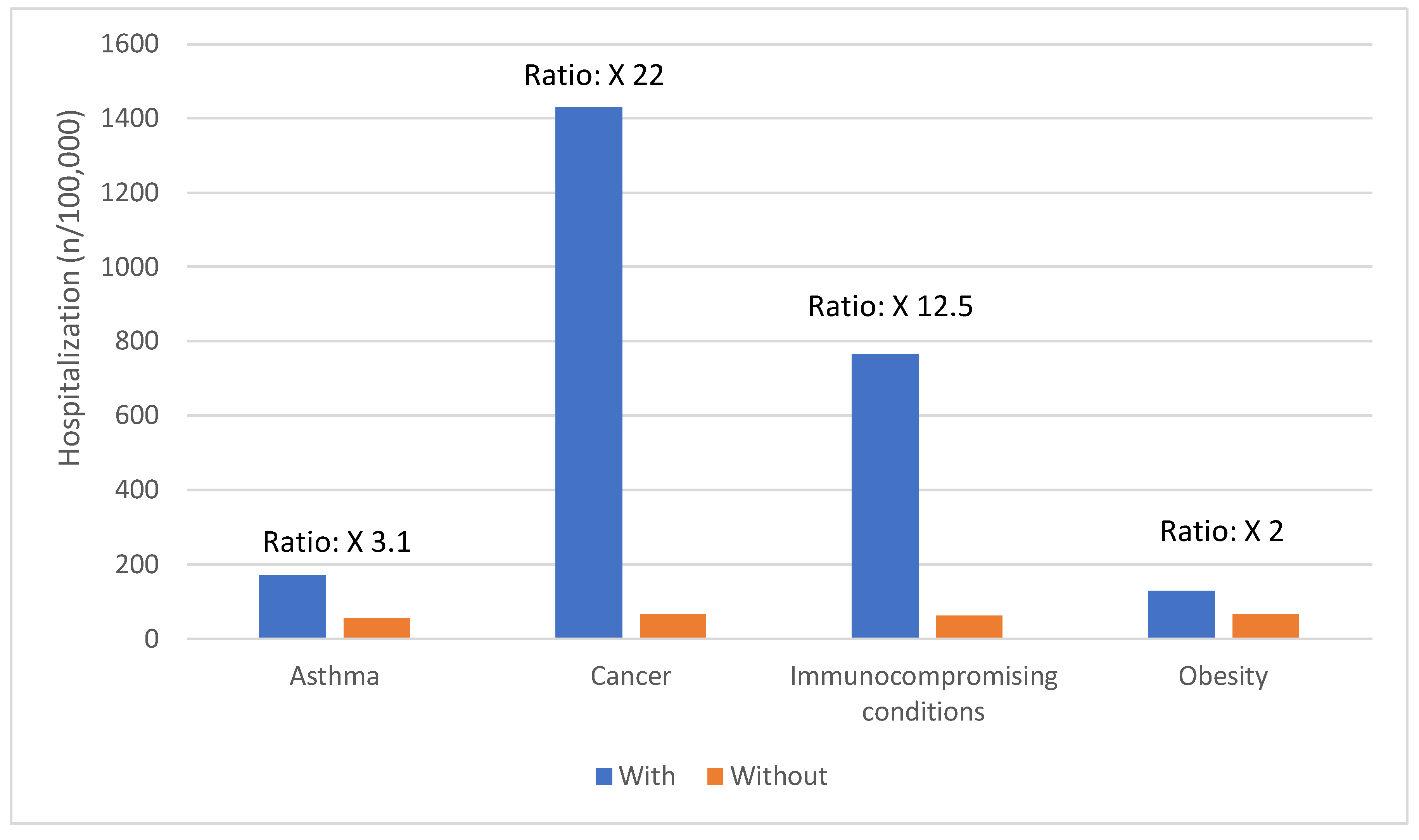
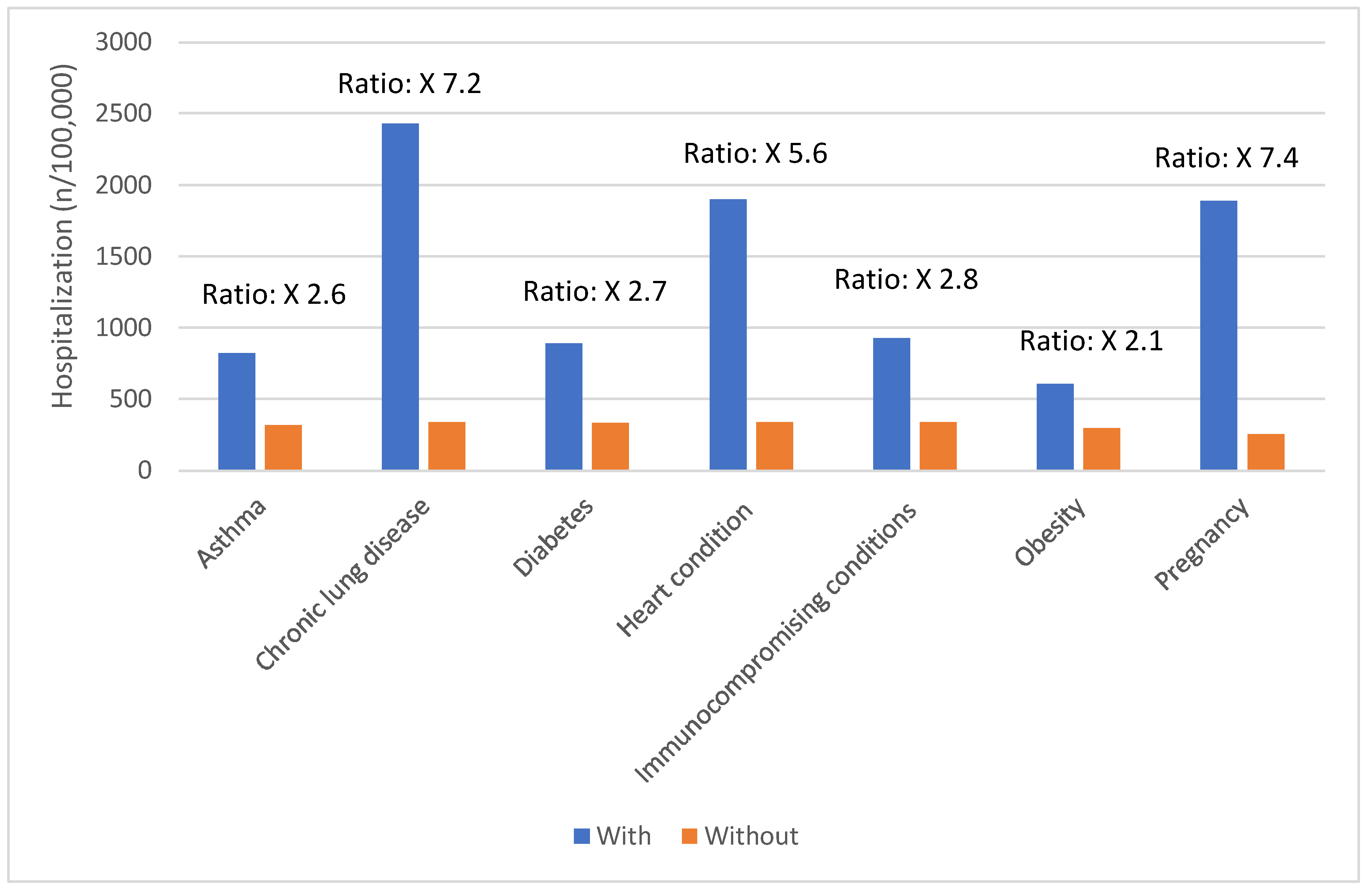
| Age group | COVID-19 hospitalizations per 100,000 population |
|---|---|
| 0-4 years | 72.8 |
| •6-< 12 months | 162.1 |
| •1-4 years | 32.1 |
| 5-17 years | 10.3 |
| •5-11 years | 8.9 |
| •12-17 years | 11.9 |
| 18-49 years | 37.4 |
| •18-29 years | 26.1 |
| •30-39 years | 39.6 |
| •40-49 years | 49.1 |
| 50-64 years | 109.2 |
| ≥ 65 years | 537.6 |
| •65-74 years | 277.6 |
| •≥ 75 years | 917.3 |
| Age group | COVID-19 N=93,888 |
Influenza N=20,561 |
Count Ratio of COVID-19 versus influenza |
|---|---|---|---|
| 0-5 years | 706* | 564 | 1.3 |
| 6-17 years | 1529* | 1260 | 1.2 |
| 18-49 years | 26,242* | 4693 | 5.6 |
| 50-64 years | 22,947* | 5529 | 4.2 |
| ≥ 65 years | 42,464* | 8515 | 5 |
| 6m-4 y | 5-11 y | 12-17 y | 18-49 y | 50-64 y | ≥ 65 y | |
|---|---|---|---|---|---|---|
| Influenza vaccine uptake | 59.8% | 52.3% | 47.0% | 36.8% | 51.2% | 73.7% |
| COVID-19 Vaccine uptake | 5.6% | 12.6% | 17.8% | 13.6% | 25.3% | 42.5% |
| American Indian | Pacific Islander | Asian | Black | Hispanic | Multiple or other races | White, non-Hispanic |
|---|---|---|---|---|---|---|
| 15.6% | 17.9% | 21.8% | 19.6% | 15.6% | 17.0% | 25.5% |
|
|
1.COVID-19 is the leading cause of respiratory infectious disease leading to hospitalizations and deaths regardless of age and comorbidities, both during and between seasonal peaks. Healthcare providers need to inform and educate their patients or patient’s parents/caregivers on the disease burden and their risk for COVID-19-related morbidity and mortality. 2.The SARS-CoV-2 virus mutates rapidly, measured over weeks and months rather than years. This explains the need for the COVID-19 vaccine to be updated regularly to ensure it covers the predominant circulating variants. Healthcare providers should inform their patients or patient’s parents/caregivers of the need for remaining up to date with the most recent COVID-19 vaccine to maintain ongoing protection. 3.COVID-19 mRNA vaccines have demonstrated consistent clinical protection, significantly reducing the risk for hospitalization and death. Healthcare providers should presumptively recommend COVID-19 vaccination to all their patients or patient’s parents/caregivers at the point-of-care (in all clinical settings and in retail pharmacies). 4.For the upcoming 2024 fall season, COVID-19 vaccines will be licensed, recommended, and available at the point-of-care by the start of the respiratory vaccination campaign. HCPs should be co-administering COVID-19 vaccines with Influenza vaccines at the same visit towards improving vaccination acceptance. 5.Patients recognize their healthcare providers as the most trusted source of healthcare information. Delivering a strong, clear presumptive COVID-19 vaccine recommendation at the point-of-care (clinic setting or the pharmacy) has consistently shown to increase vaccination acceptance whilst reducing the time needed with patient. Providers should presumptively recommend the COVID-19 vaccine to every person 6 months of age and older at every opportunity. 6.The misinformation that drives online conversations day after day is the misinformation that healthcare providers will hear from their patients when delivering a vaccination recommendation. Awareness of these trending topics and themes is critical for healthcare providers to optimally inform and educate their patients. Providers should regularly consult an infodemiology dashboard for awareness of the trending issues and how to categorize them within distinct themes of either perceived risk, safety, or vaccine effectiveness to be able to effectively provide evidence-based reassurance to their patients. 7.Healthcare Providersfrom their schooling through their residencies and into their professional careers need to be trained in Communication skills to be able to better make clear recommendations and learn how to better communicate with patients/families with questions and/or concerns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




