Submitted:
23 May 2024
Posted:
24 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Overview of Cosmetic and Dermatology Sciences
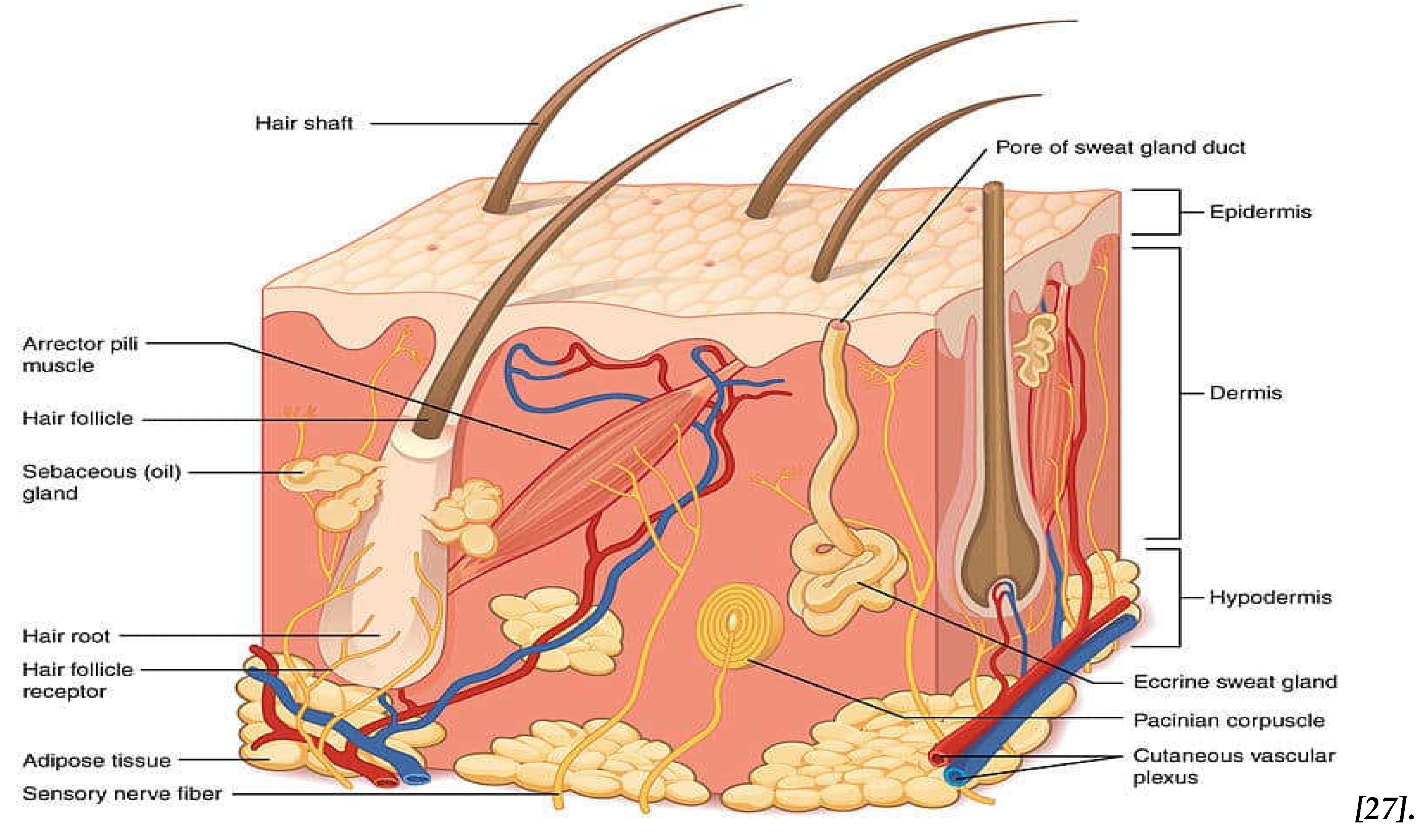
1.2. The Skin and Skin Anatomy
2. Overview of Hydrogels
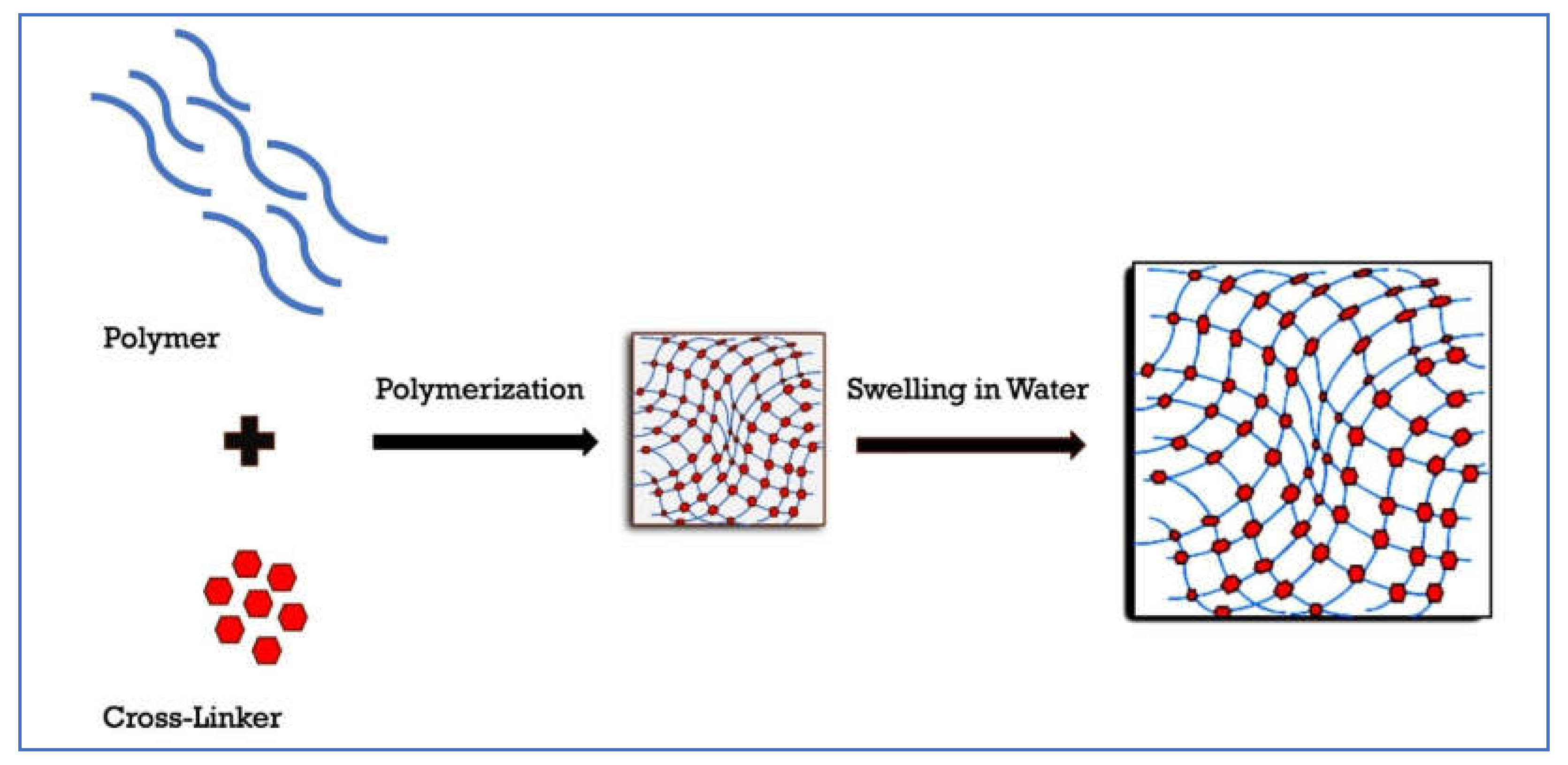
2.1. Introduction to Hydrogels
2.2. Types of Hydrogels
2.3. Rheological Properties of Hydrogels
2.4. Characterization of the Physical and Chemical Properties of the Hydrogels
3. Hyaluronic Acid
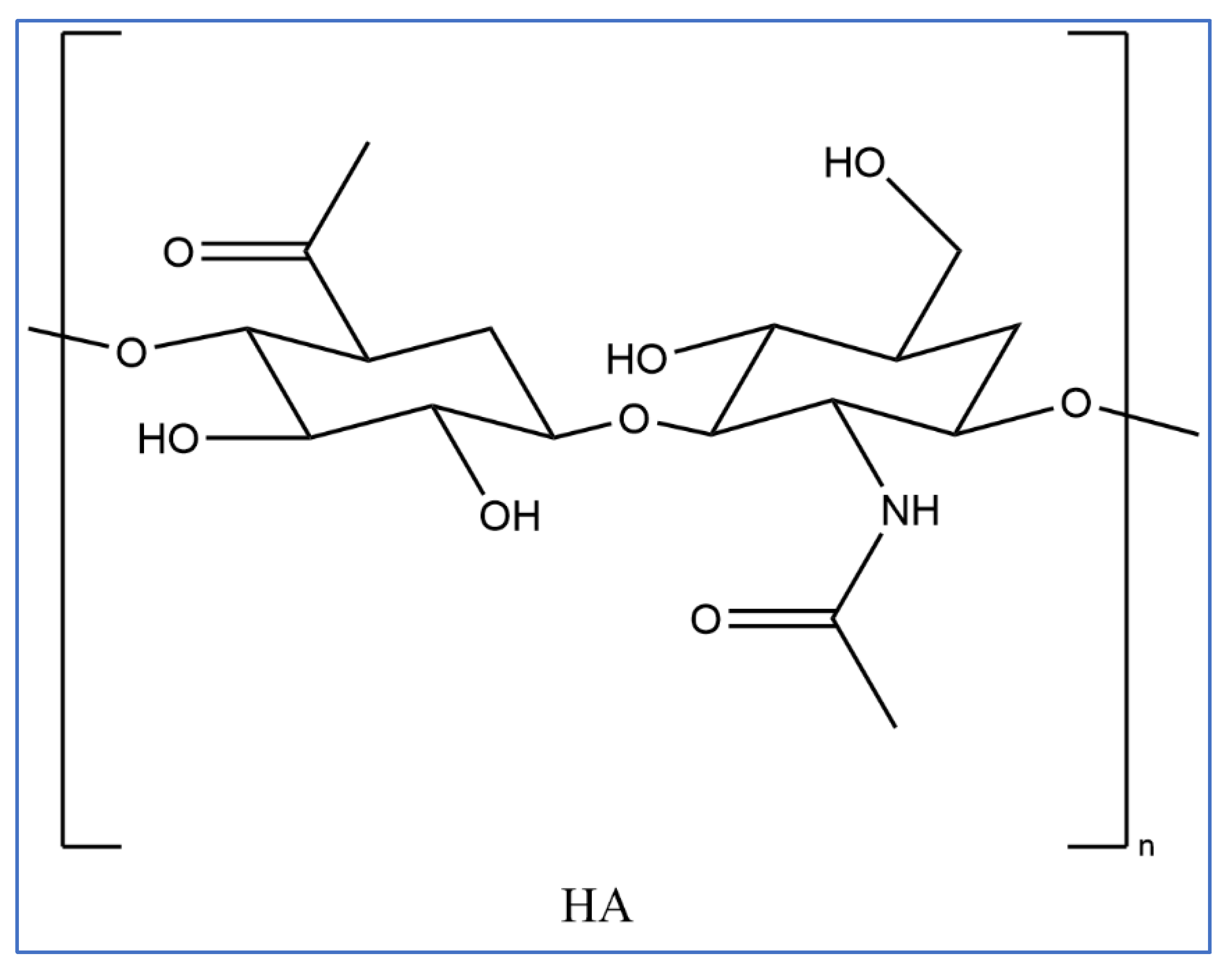
3.1. Introduction on Hyaluronic Acid
3.2. Classification of Hyaluronic Acid
3.3. Physical and Chemical Properties of Hyaluronic Acid
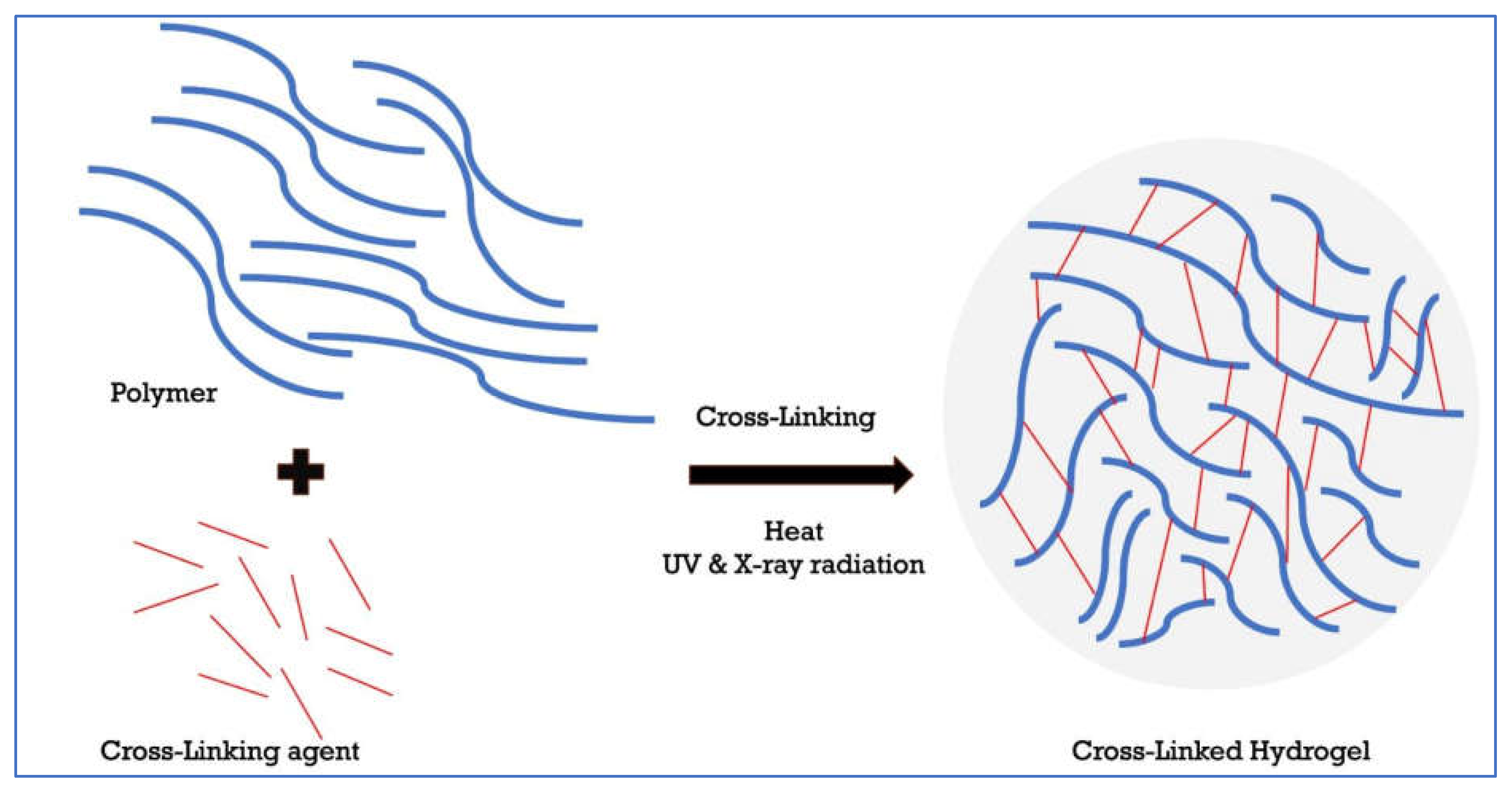
3.4. Types of Gelling Agents Used for Preparation of Hyaluronic Acid Hydrogels
3.4.1. Specific Types of Cross-Linking Agents
3.4.2. Temperature Dependence of Gelation Mechanism to Prepare HA Hydrogels.
3.4.3. pH Dependence of Gelation Mechanism to Prepare HA Hydrogels
3.5. Preparation of Hyaluronic Acid Hydrogels Using Cross-Linking Agents
3.6. Methods of Preparation for Hyaluronic Acid Hydrogels in Different
3.6.1. Hyaluronic Acid Molecular Weight
3.6.2. pH
3.6.3. Temperature
3.7. Preparation of High Molecular Weight Hydrogel Platform Loaded with Low Molecular Weight Hyaluronic Acid
3.8. Pharmaceutical and Cosmetic Uses of Hyaluronic Acid
3.8.1. Uses of Hyaluronic Acid in Pharmaceutical Applications and its Advantages
3.8.2. Uses of Hyaluronic Acid in the Cosmetic Field and its Advantages
3.9. Hydrogel Based Hyaluronic Acid Phase Transition by Changing the pH for Full or Completed Absorption via Skin
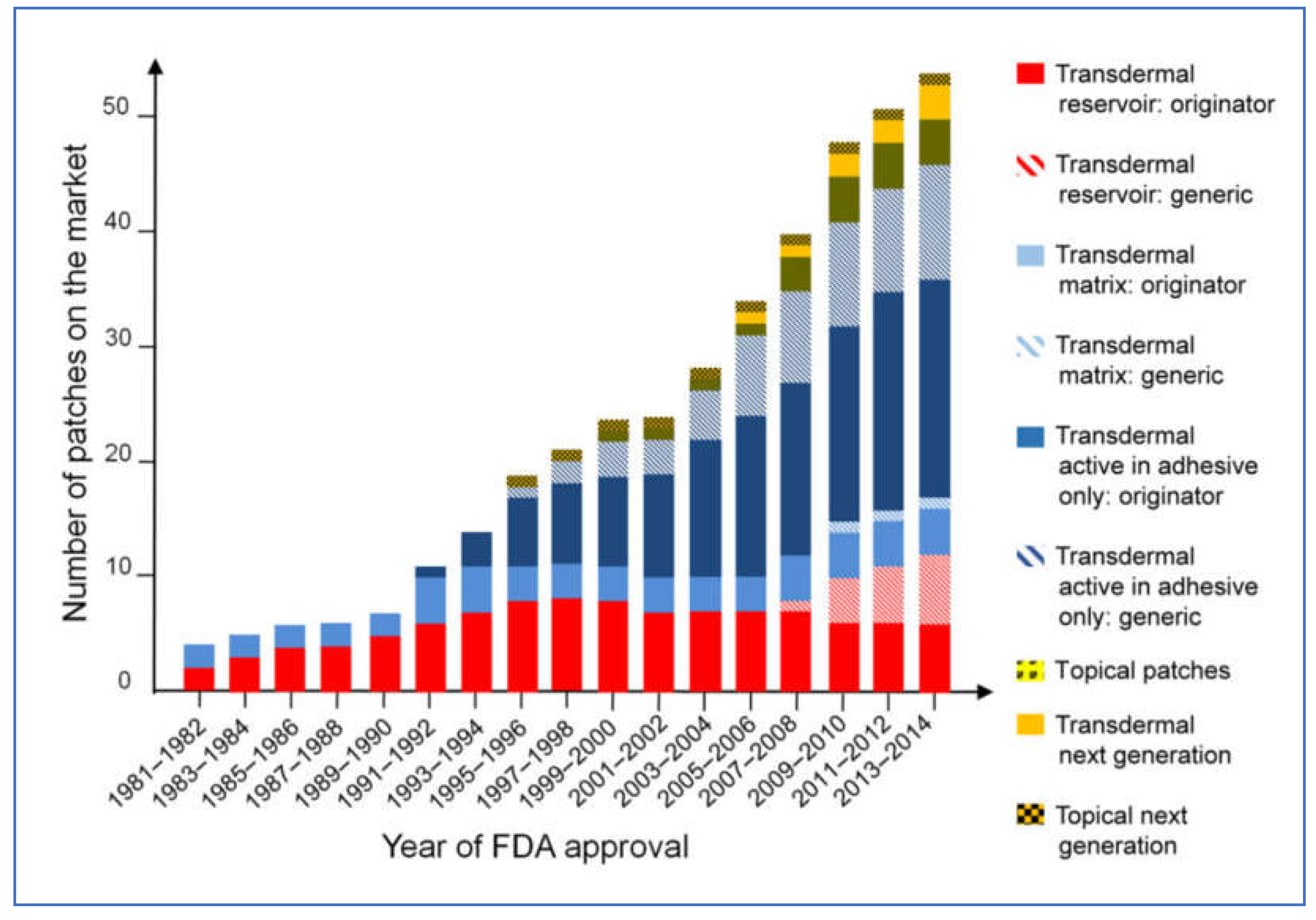
4. Overview of the Development of Transdermal Delivery Systems
Drug Delivery via Skin Patches
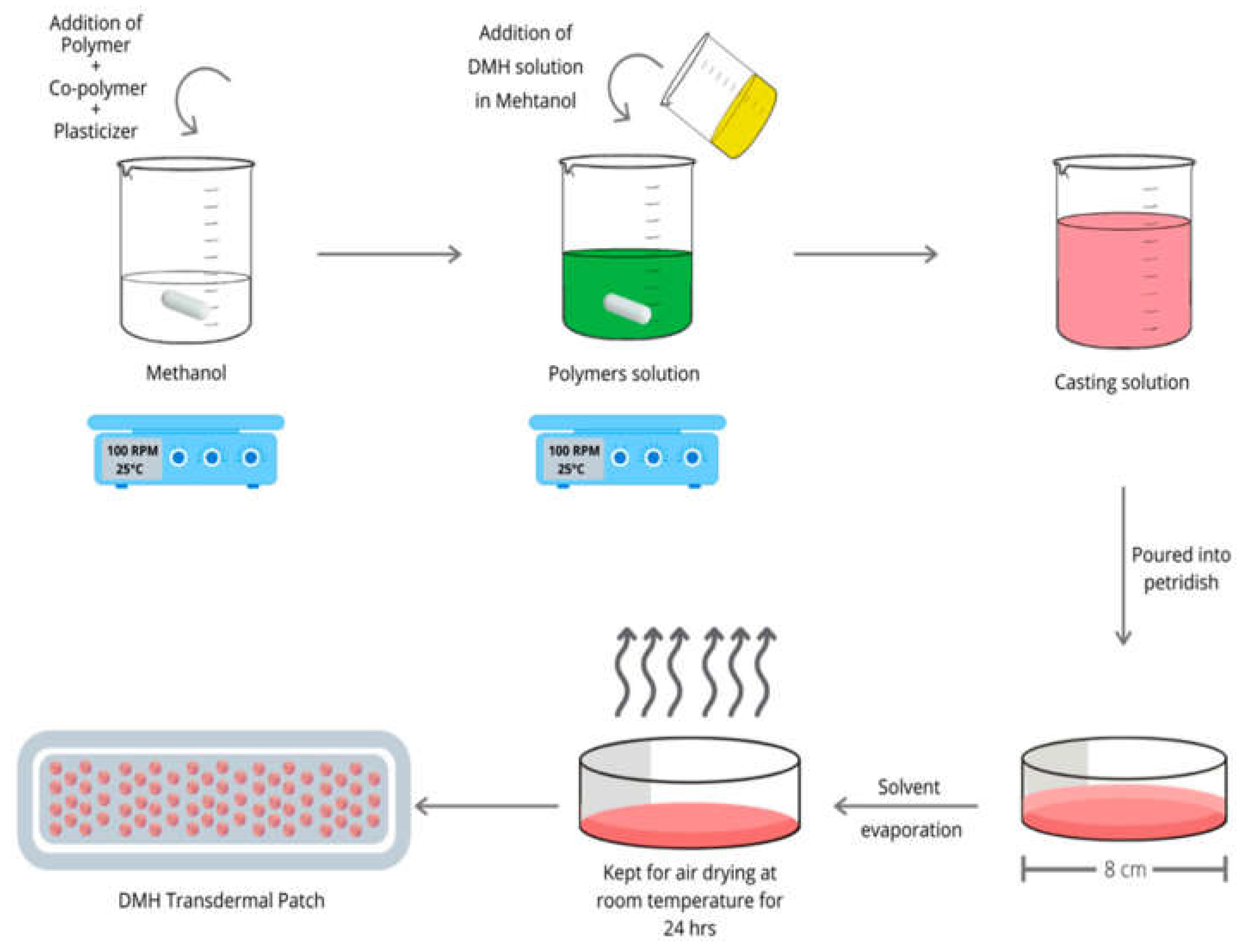
5. Drug and Actives Loading in Hydrogel Films
5.1. Post-Loading (Osmosis Dependent Loading) Method in the Hydrogel
5.2. In-Situ Loading Method of the Actives and Drugs in the Hydrogel
6. Drugs and Actives Release from the Hydrogel Film
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Surya, M. and S. Gunasekaran, A Review on Recent Scenario of Cosmetics. International Journal of Pharmaceutical Sciences Review and Research, 2021.
- Barel, A.O. , Handbook of Cosmetic Science and Technology. 2009, CRC: Press EBooks.
- Cosmeceuticals and Active Cosmetics, Drugs vs. Cosmetics. 2005.
- Raja, S. , et al., Cosmeceuticals and Active Cosmetics. 2015.
- Sharma, A.; Kuhad, A.; Bhandari, R. Novel nanotechnological approaches for treatment of skin-aging. J. Tissue Viability 2022, 31, 374–386. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, D.H.; Neudecker, B.A.; Dinardo, J.C.; Lewis, J.A., II; Maibach, H.I. Clinical efficacy assessment in photodamaged skin of 0.5% and 1.0% idebenone. J. Cosmet. Dermatol. 2005, 4, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Milani, M.; Colombo, F. Skin Anti-Aging Effect of Oral Vitamin A Supplementation in Combination with Topical Retinoic Acid Treatment in Comparison with Topical Treatment Alone: A Randomized, Prospective, Assessor-Blinded, Parallel Trial. Cosmetics 2023, 10, 144. [Google Scholar] [CrossRef]
- Handbook of Cosmetic Science and Technology. 2014.
- Iwata, H. and K. Shimada, Formulas, Ingredients and Production of Cosmetics: Technology of Skin- and Hair-Care Products in Japan. Formulas, Ingredients and Production of Cosmetics, 2012.
- McMullen, R.L.; Dell’acqua, G. History of Natural Ingredients in Cosmetics. Cosmetics 2023, 10, 71. [Google Scholar] [CrossRef]
- Polymers for Personal Care Products and Cosmetics, ed. X.J. Loh. 2016: The Royal Society of Chemistry.
- Patil, A.A. and M. S. Ferritto. Polymers for personal care and cosmetics. 2013.
- Wanjari, N. and J.S. Waghmare, A Review on Latest Trend of Cosmetics-Cosmeceuticals. International Journal of Pharma Research & Review, 2015.
- Rashid, F.; Childs, S.; Dodou, K. Comparison of Analytical Methods for the Detection of Residual Crosslinker in Hyaluronic Acid Hydrogel Films. Cosmetics 2023, 10, 70. [Google Scholar] [CrossRef]
- Gupta, V.; Mohapatra, S.; Mishra, H.; Farooq, U.; Kumar, K.; Ansari, M.J.; Aldawsari, M.F.; Alalaiwe, A.S.; Mirza, M.A.; Iqbal, Z. Nanotechnology in Cosmetics and Cosmeceuticals—A Review of Latest Advancements. Gels 2022, 8, 173. [Google Scholar] [CrossRef] [PubMed]
- Manful, M.E.; Ahmed, L.; Barry-Ryan, C. Cosmetic Formulations from Natural Sources: Safety Considerations and Legislative Frameworks in the European Union. Cosmetics 2024, 11, 72. [Google Scholar] [CrossRef]
- Sarkic, A.; Stappen, I. Essential Oils and Their Single Compounds in Cosmetics—A Critical Review. Cosmetics 2018, 5, 11. [Google Scholar] [CrossRef]
- Hay, R.J.; Johns, N.E.; Williams, H.C.; Bolliger, I.; Dellavalle, R.P.; Margolis, D.J.; Marks, R.; Naldi, L.; Weinstock, M.A.; Wulf, S.K.; et al. The Global Burden of Skin Disease in 2010: An Analysis of the Prevalence and Impact of Skin Conditions. J. Investig. Dermatol. 2014, 134, 1527–1534. [Google Scholar] [CrossRef]
- Pacifico, M.D.; Pearl, R.; Grover, R. The UK Government Two-Week Rule and its Impact on Melanoma Prognosis: An Evidence-Based Study. Ind. Mark. Manag. 2007, 89, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Conforti, C.; Zalaudek, I. Epidemiology and Risk Factors of Melanoma: A Review. Dermatol. Pr. Concept. 2021, 11, 2021161S–2021161S. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.-W.; Jee, S.-H. Strategies to Develop a Suitable Formulation for Inflammatory Skin Disease Treatment. Int. J. Mol. Sci. 2021, 22, 6078. [Google Scholar] [CrossRef] [PubMed]
- Textbook of Cosmetic Dermatology. 2017.
- Moser, K.; Kriwet, K.; Naik, A.; Kalia, Y.N.; Guy, R.H. Passive skin penetration enhancement and its quantification in vitro. Eur. J. Pharm. Biopharm. 2001, 52, 103–112. [Google Scholar] [CrossRef]
- C, M.B.Z., S. Grushchak, and J. Newman, Skin Anatomy and Analysis. Facial Plast Surg Clin North Am, 2023. 31(4): p. 433-442.
- Draelos, Z.D. Cosmetics and Dermatologic Problems and Solutions; Taylor & Francis Ltd: London, United Kingdom, 2011. [Google Scholar]
- Alkilani, A.Z.; McCrudden, M.T.C.; Donnelly, R.F. Transdermal Drug Delivery: Innovative Pharmaceutical Developments Based on Disruption of the Barrier Properties of the Stratum Corneum. Pharmaceutics 2015, 7, 438–470. [Google Scholar] [CrossRef] [PubMed]
- Skin Ultrastructure Epidermis - Dermis [cited 2023 27/Nov./2023]; - TeachMeAnatomy]. Available from: https://teachmeanatomy.info/the-basics/ultrastructure/skin/.
- Rostkowska, E.; Poleszak, E.; Wojciechowska, K.; Szewczyk, K.D.S. Dermatological Management of Aged Skin. Cosmetics 2023, 10, 55. [Google Scholar] [CrossRef]
- Rahrovan, S.; Fanian, F.; Mehryan, P.; Humbert, P.; Firooz, A. Male versus female skin: What dermatologists and cosmeticians should know. Int. J. Women's Dermatol. 2018, 4, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Abdo, J.M.; Sopko, N.A.; Milner, S.M. The applied anatomy of human skin: A model for regeneration. Wound Med. 2020, 28, 100179. [Google Scholar] [CrossRef]
- Lai-Cheong, J.E. and J.A. McGrath, Structure and function of skin, hair and nails. Medicine, 2013. 41(6): p. 317-320.
- Ahmed, E.M. , Hydrogel: Preparation, characterization, and applications: A review. Journal of Advanced Research, 2015. 6(2): p. 105-121.
- Akhtar, M.F., M. Hanif, and N.M. Ranjha, Methods of synthesis of hydrogels … A review. Saudi Pharmaceutical Journal, 2016. 24(5): p. 554-559.
- Vigata, M.; Meinert, C.; Hutmacher, D.W.; Bock, N. Hydrogels as Drug Delivery Systems: A Review of Current Characterization and Evaluation Techniques. Pharmaceutics 2020, 12, 1188. [Google Scholar] [CrossRef]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Morteza, B., M. Naimeh, and M. Mehdi, An Introduction to Hydrogels and Some Recent Applications, in Emerging Concepts in Analysis and Applications of Hydrogels, M. Sutapa Biswas, Editor. 2016, IntechOpen: Rijeka. p. Ch. 2.
- Singh, S.K., A. Dhyani, and D.S. Juyal, Hydrogel: Preparation, Characterization and Applications. The Pharma Innovation Journal, 2017. 6: p. 25-32.
- Tang, G.; Du, B.; Stadler, F.J. A novel approach to analyze the rheological properties of hydrogels with network structure simulation. J. Polym. Res. 2017, 25, 1–10. [Google Scholar] [CrossRef]
- Di Giuseppe, E.; Corbi, F.; Funiciello, F.; Massmeyer, A.; Santimano, T.; Rosenau, M.; Davaille, A. Characterization of Carbopol® hydrogel rheology for experimental tectonics and geodynamics. Tectonophysics 2015, 642, 29–45. [Google Scholar] [CrossRef]
- Liao, Y.-H.; Jones, S.A.; Forbes, B.; Martin, G.P.; Brown, M.B. Hyaluronan: Pharmaceutical Characterization and Drug Delivery. Drug Deliv. 2005, 12, 327–342. [Google Scholar] [CrossRef] [PubMed]
- Parhi, R. Cross-Linked Hydrogel for Pharmaceutical Applications: A Review. Adv. Pharm. Bull. 2017, 7, 515–530. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, A.K.; Shukla, S.K.; Bhanu, S.; Kankane, S. Responsive polymers in controlled drug delivery. Prog. Polym. Sci. 2008, 33, 1088–1118. Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for Biomedical Applications: Their Characteristics and the Mechanisms behind Them. Gels 2017, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermato-Endocrinol. 2012, 4, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Iaconisi, G.N.; Lunetti, P.; Gallo, N.; Cappello, A.R.; Fiermonte, G.; Dolce, V.; Capobianco, L. Hyaluronic Acid: A Powerful Biomolecule with Wide-Ranging Applications—A Comprehensive Review. Int. J. Mol. Sci. 2023, 24, 10296. [Google Scholar] [CrossRef] [PubMed]
- Necas, J.; Bartosikova, L.; Brauner, P.; Kolar, J. Hyaluronic acid (hyaluronan): A review. Vet. Med. 2008, 53, 397–411. [Google Scholar] [CrossRef]
- Maneiro, E.; De Andres, M.C.; Fernández-Sueiro, J.L.; Galdo, F.; Blanco, F.J. The biological action of hyaluronan on human osteoartritic articular chondrocytes: the importance of molecular weight. . 2004, 22, 307–12. [Google Scholar]
- Chen, L.H.; Xue, J.F.; Zheng, Z.Y.; Shuhaidi, M.; Thu, H.E.; Hussain, Z. Hyaluronic acid, an efficient biomacromolecule for treatment of inflammatory skin and joint diseases: A review of recent developments and critical appraisal of preclinical and clinical investigations. Int. J. Biol. Macromol. 2018, 116, 572–584. [Google Scholar] [CrossRef]
- Rashid, F.; Carter, P.; Childs, S. Novel Injectable Hydrogel Formulations and Gas Chromatography Analysis of the Residual Crosslinker in Formulations Intended for Pharmaceutical and Cosmetic Applications. Gels 2024, 10, 280. [Google Scholar] [CrossRef] [PubMed]
- Maharjan, A.S.; Pilling, D.; Gomer, R.H. High and Low Molecular Weight Hyaluronic Acid Differentially Regulate Human Fibrocyte Differentiation. PLOS ONE 2011, 6, e26078. [Google Scholar] [CrossRef] [PubMed]
- Albano, G.D. , et al., A 3D “In Vitro” Model to Study Hyaluronan Effect in Nasal Epithelial Cell Line Exposed to Double-Stranded RNA Poly(I:C). Biomol Ther (Seoul), 2020. 28(3): p. 272-281.
- Lee, Y. , et al., Thermo-sensitive, injectable, and tissue adhesive sol–gel transition hyaluronic acid/pluronic composite hydrogels prepared from bio-inspired catechol-thiol reaction. Soft Matter, 2010. 6(5): p. 977-983.
- Cowman, M.K.; Lee, H.-G.; Schwertfeger, K.L.; McCarthy, J.B.; Turley, E.A. The Content and Size of Hyaluronan in Biological Fluids and Tissues. Front. Immunol. 2015, 6, 261. [Google Scholar] [CrossRef] [PubMed]
- Jabbari, F.; Babaeipour, V.; Saharkhiz, S. Comprehensive review on biosynthesis of hyaluronic acid with different molecular weights and its biomedical applications. Int. J. Biol. Macromol. 2023, 240, 124484. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Wang, Y.-M.; Yang, J.; Luo, X.-S. Hyaluronic acid: a versatile biomaterial in tissue engineering. Plast. Aesthetic Res. 2017, 4, 219. [Google Scholar] [CrossRef]
- Rashid, F.; Albayati, M.; Dodou, K. Studies on Novel Methods for Formulating Novel Cross-Linked Hydrogel Films of Hyaluronic Acid. Cosmetics 2019, 6, 59. [Google Scholar] [CrossRef]
- Ström, A.; Larsson, A.; Okay, O. Preparation and physical properties of hyaluronic acid-based cryogels. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Gulrez, S., S. Al-Assaf, and G. Phillips, Hydrogels: Methods of Preparation, Characterisation and Applications in Molecular and Environmental Bioengineering. 2011.
- Ahmad, Z.; Salman, S.; Khan, S.A.; Amin, A.; Rahman, Z.U.; Al-Ghamdi, Y.O.; Akhtar, K.; Bakhsh, E.M.; Khan, S.B. Versatility of Hydrogels: From Synthetic Strategies, Classification, and Properties to Biomedical Applications. Gels 2022, 8, 167. [Google Scholar] [CrossRef]
- Fujiwara, J.; Takahashi, M.; Hatakeyama, T.; Hatakeyama, H. Gelation of hyaluronic acid through annealing. Polym. Int. 2000, 49, 1604–1608. [Google Scholar] [CrossRef]
- Gatej, I.; Popa, M.; Rinaudo, M. Role of the pH on Hyaluronan Behavior in Aqueous Solution. Biomacromolecules 2004, 6, 61–67. [Google Scholar] [CrossRef]
- Maleki, A.; Kjøniksen, A.; Nyström, B. Effect of pH on the Behavior of Hyaluronic Acid in Dilute and Semidilute Aqueous Solutions. Macromol. Symp. 2008, 274, 131–140. [Google Scholar] [CrossRef]
- Caicco, M.J.; Zahir, T.; Mothe, A.J.; Ballios, B.G.; Kihm, A.J.; Tator, C.H.; Shoichet, M.S. Characterization of hyaluronan–methylcellulose hydrogels for cell delivery to the injured spinal cord. J. Biomed. Mater. Res. Part A 2012, 101A, 1472–1477. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, P.; Bisht, A.; Alexander, A.; Dave, V.; Sharma, S. Biomedical applications of hydrogels in drug delivery system: An update. J. Drug Deliv. Sci. Technol. 2021, 66, 102914. [Google Scholar] [CrossRef]
- Lv, Z.; Chang, L.; Long, X.; Liu, J.; Xiang, Y.; Liu, J.; Liu, J.; Deng, H.; Deng, L.; Dong, A. Thermosensitive in situ hydrogel based on the hybrid of hyaluronic acid and modified PCL/PEG triblock copolymer. Carbohydr. Polym. 2014, 108, 26–33. [Google Scholar] [CrossRef]
- Snetkov, P.; Zakharova, K.; Morozkina, S.; Olekhnovich, R.; Uspenskaya, M. Hyaluronic Acid: The Influence of Molecular Weight on Structural, Physical, Physico-Chemical, and Degradable Properties of Biopolymer. Polymers 2020, 12, 1800. [Google Scholar] [CrossRef] [PubMed]
- Chun, C.; Lee, D.Y.; Kim, J.-T.; Kwon, M.-K.; Kim, Y.-Z.; Kim, S.-S. Effect of molecular weight of hyaluronic acid (HA) on viscoelasticity and particle texturing feel of HA dermal biphasic fillers. Biomater. Res. 2016, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Larraneta, E.; Henry, M.; Irwin, N.J.; Trotter, J.; Perminova, A.A.; Donnelly, R.F. Synthesis and characterization of hyaluronic acid hydrogels crosslinked using a solvent-free process for potential biomedical applications. Carbohydr. Polym. 2018, 181, 1194–1205. [Google Scholar] [CrossRef] [PubMed]
- Tavsanli, B.; Okay, O. Preparation and fracture process of high strength hyaluronic acid hydrogels cross-linked by ethylene glycol diglycidyl ether. React. Funct. Polym. 2016, 109, 42–51. [Google Scholar] [CrossRef]
- Khunmanee, S.; Jeong, Y.; Park, H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J. Tissue Eng. 2017, 8. [Google Scholar] [CrossRef]
- Xu, L.; Zhong, S.; Gao, Y.; Cui, X. Thermo-responsive poly(N-isopropylacrylamide)-hyaluronic acid nano-hydrogel and its multiple applications. Int. J. Biol. Macromol. 2022, 194, 811–818. [Google Scholar] [CrossRef]
- Ha, D.I.; Lee, S.B.; Chong, M.S.; Lee, Y.M.; Kim, S.Y.; Park, Y.H. Preparation of thermo-responsive and injectable hydrogels based on hyaluronic acid and poly(N-isopropylacrylamide) and their drug release behaviors. Macromol. Res. 2006, 14, 87–93. [Google Scholar] [CrossRef]
- Farwick, M., P. Lersch, and G. Strutz, Low molecular weight hyaluronic acid: Its effects on epidermal gene expression & skin ageing. SÖFW J., 2008. 11: p. 134-137.
- Huang, G.; Huang, H. Application of hyaluronic acid as carriers in drug delivery. Drug Deliv. 2018, 25, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Tripodo, G.; Trapani, A.; Torre, M.L.; Giammona, G.; Trapani, G.; Mandracchia, D. Hyaluronic acid and its derivatives in drug delivery and imaging: Recent advances and challenges. Eur. J. Pharm. Biopharm. 2015, 97, 400–416. [Google Scholar] [CrossRef]
- Neuman, M.G.; Nanau, R.M.; Oruña-Sanchez, L.; Coto, G. Hyaluronic Acid and Wound Healing. J. Pharm. Pharm. Sci. 2015, 18, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Galvez-Martin, P.; Soto-Fernandez, C.; Romero-Rueda, J.; Cabañas, J.; Torrent, A.; Castells, G.; Martinez-Puig, D. A Novel Hyaluronic Acid Matrix Ingredient with Regenerative, Anti-Aging and Antioxidant Capacity. Int. J. Mol. Sci. 2023, 24, 4774. [Google Scholar] [CrossRef] [PubMed]
- Gold, M. Use of hyaluronic acid fillers for the treatment of the aging face. Clin. Interv. Aging 2007, ume 2, 369–376. [Google Scholar] [CrossRef]
- Kondiah, P.J. , et al., A Review of Injectable Polymeric Hydrogel Systems for Application in Bone Tissue Engineering. Molecules, 2016. 21(11).
- Maitra, J. and V. Shukla, Cross-linking in hydrogels - a review. Am J Polym Sci, 2014. 4: p. 25-31.
- Qiu, Y.; Park, K. Environment-sensitive hydrogels for drug delivery. Adv. Drug Deliv. Rev. 2001, 53, 321–339. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, A.C.; Mitragotri, S. An overview of clinical and commercial impact of drug delivery systems. J. Control. Release 2014, 190, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Das, D.B. Potential of combined ultrasound and microneedles for enhanced transdermal drug permeation: A review. Eur. J. Pharm. Biopharm. 2015, 89, 312–328. [Google Scholar] [CrossRef]
- Brambilla, D.; Luciani, P.; Leroux, J.-C. Breakthrough discoveries in drug delivery technologies: The next 30 years. J. Control. Release 2014, 190, 9–14. [Google Scholar] [CrossRef]
- Ita, K. Transdermal drug delivery: progress and challenges. J. Drug Deliv. Sci. Technol. 2014, 24, 245–250. [Google Scholar] [CrossRef]
- Schoellhammer, C.M.; Blankschtein, D.; Langer, R. Skin permeabilization for transdermal drug delivery: recent advances and future prospects. Expert Opin. Drug Deliv. 2014, 11, 393–407. [Google Scholar] [CrossRef]
- McCrudden, M.T.; Singh, T.R.R.; Migalska, K.; Donnelly, R.F. Strategies for Enhanced Peptide and Protein Delivery. Ther. Deliv. 2013, 4, 593–614. [Google Scholar] [CrossRef] [PubMed]
- Kermode, M. Unsafe injections in low-income country health settings: need for injection safety promotion to prevent the spread of blood-borne viruses. Heal. Promot. Int. 2004, 19, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; Singh, T.R.R.; Morrow, D.I.J.; Woolfson, A.D. Microneedle-Mediated Transdermal and Intradermal Drug Delivery; Wiley, 2012;
- Kretsos, K.; Kasting, G.B. A geometrical model of dermal capillary clearance. Math. Biosci. 2007, 208, 430–453. [Google Scholar] [CrossRef] [PubMed]
- Pastore, M.N.; Kalia, Y.N.; Horstmann, M.; Roberts, M.S. Transdermal patches: history, development and pharmacology. Br. J. Pharmacol. 2015, 172, 2179–2209. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M. Solute-Vehicle-Skin Interactions in Percutaneous Absorption: The Principles and the People. Ski. Pharmacol. Physiol. 2013, 26, 356–370. [Google Scholar] [CrossRef] [PubMed]
- Benson, H.A.E. and A.C. Watkinson. Topical and Transdermal Drug Delivery: Principles and Practice. 2011.
- Kasting, G. Lipid Solubility and Molecular Weight: Whose Idea Was That. Ski. Pharmacol. Physiol. 2013, 26, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Walters, K.A. Dermatological And Transdermal Formulations. 2002.
- Magnusson, B.M.; Anissimov, Y.G.; Cross, S.E.; Roberts, M.S. Molecular Size as the Main Determinant of Solute Maximum Flux Across the Skin. J. Investig. Dermatol. 2004, 122, 993–999. [Google Scholar] [CrossRef]
- Wiedersberg, S. and R.H. Guy, Transdermal drug delivery: 30+ years of war and still fighting! Journal of Controlled Release, 2014. 190: p. 150-156.
- Penn, M.J.; Hennessy, M.G. Optimal loading of hydrogel-based drug-delivery systems. Appl. Math. Model. 2022, 112, 649–668. [Google Scholar] [CrossRef]
- Singh, B.; Chauhan, G.; Kumar, S.; Chauhan, N. Synthesis, characterization and swelling responses of pH sensitive psyllium and polyacrylamide based hydrogels for the use in drug delivery (I). Carbohydr. Polym. 2007, 67, 190–200. [Google Scholar] [CrossRef]
- Dong, K. , et al., Assessment of the safety, targeting, and distribution characteristics of a novel pH-sensitive hydrogel. Colloids Surf B Biointerfaces, 2014. 123: p. 965-73.
- Chaturvedi, K.; Ganguly, K.; Nadagouda, M.N.; Aminabhavi, T.M. Polymeric hydrogels for oral insulin delivery. J. Control. Release 2013, 165, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Cavalieri, F.; Chiessi, E.; Villa, R.; Viganò, L.; Zaffaroni, N.; Telling, M.F.; Paradossi, G. Novel PVA-Based Hydrogel Microparticles for Doxorubicin Delivery. Biomacromolecules 2008, 9, 1967–1973. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Ding, J.; Fei, L.; He, M.; Cui, F.; Tang, C.; Yin, C. Beneficial properties for insulin absorption using superporous hydrogel containing interpenetrating polymer network as oral delivery vehicles. Int. J. Pharm. 2008, 350, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Rashid, F.; Albayati, M.; Dodou, K. Novel Crosslinked HA Hydrogel Films for the Immediate Release of Active Ingredients. Cosmetics 2022, 10, 6. [Google Scholar] [CrossRef]
- Cursaru, B.; Teodorescu, M.; Boscornea, C.; Stanescu, P.O.; Stoleriu, S. Drug absorption and release properties of crosslinked hydrogels based on diepoxy-terminated poly(ethylene glycol)s and aliphatic polyamines — a study on the effect of the gel molecular structure. Mater. Sci. Eng. C 2013, 33, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J. , et al., Swelling Kinetics of Interpenetrating Polymer Hydrogels Composed of Poly(Vinyl Alcohol)/Chitosan. Journal of Macromolecular Science, Part A, 2003. 40(5): p. 501-510.
- Kim, S.W., Y. H. Bae, and T. Okano, Hydrogels: swelling, drug loading, and release. Pharm Res, 1992. 9(3): p. 283-90.
- Rasool, B.K.A.; Mohammed, A.A.; Salem, Y.Y. The Optimization of a Dimenhydrinate Transdermal Patch Formulation Based on the Quantitative Analysis of In Vitro Release Data by DDSolver through Skin Penetration Studies. Sci. Pharm. 2021, 89, 33. [Google Scholar] [CrossRef]
- Lei, L.; Bai, Y.; Qin, X.; Liu, J.; Huang, W.; Lv, Q. Current Understanding of Hydrogel for Drug Release and Tissue Engineering. Gels 2022, 8, 301. [Google Scholar] [CrossRef] [PubMed]
- Serra, L.; Doménech, J.; Peppas, N.A. Drug transport mechanisms and release kinetics from molecularly designed poly(acrylic acid-g-ethylene glycol) hydrogels. Biomaterials 2006, 27, 5440–5451. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Liu, W.-Q.; Chen, W.-X.; Sun, L.; Zhang, G.-B. Investigation of swelling and controlled-release behaviors of hydrophobically modified poly(methacrylic acid) hydrogels. Polymer 2007, 48, 2665–2671. [Google Scholar] [CrossRef]
- Reddy, O.S.; Subha, M.C.S.; Jithendra, T.; Madhavi, C.; Rao, K.C. Fabrication and characterization of smart karaya gum/sodium alginate semi-IPN microbeads for controlled release of D-penicillamine drug. Polym. Polym. Compos. 2021, 29, 163–175. [Google Scholar] [CrossRef]
- Chen, J.; Liu, M.; Liu, H.; Ma, L. Synthesis, swelling and drug release behavior of poly(N,N-diethylacrylamide-co-N-hydroxymethyl acrylamide) hydrogel. Mater. Sci. Eng. C 2009, 29, 2116–2123. [Google Scholar] [CrossRef]
- Dadsetan, M.; Liu, Z.; Pumberger, M.; Giraldo, C.V.; Ruesink, T.; Lu, L.; Yaszemski, M.J. A stimuli-responsive hydrogel for doxorubicin delivery. Biomaterials 2010, 31, 8051–8062. [Google Scholar] [CrossRef] [PubMed]
- Sullad, A.G.; Manjeshwar, L.S.; Aminabhavi, T.M. Blend microspheres of chitosan and polyurethane for controlled release of water-soluble antihypertensitive drugs. Polym. Bull. 2014, 72, 265–280. [Google Scholar] [CrossRef]
- Kim, I.S. and I.J. Oh, Drug release from the enzyme-degradable and pH-sensitive hydrogel composed of glycidyl methacrylate dextran and poly(acrylic acid). Arch Pharm Res, 2005. 28(8): p. 983-7.
- Adepu, S.; Ramakrishna, S. Controlled Drug Delivery Systems: Current Status and Future Directions. Molecules 2021, 26, 5905. [Google Scholar] [CrossRef]
- Fu, Y.; Kao, W.J. Drug release kinetics and transport mechanisms of non-degradable and degradable polymeric delivery systems. Expert Opin. Drug Deliv. 2010, 7, 429–444. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



