Submitted:
18 May 2024
Posted:
21 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Overview
2.2. Exclusion and Inclusion Criteria
2.3. Records Retrieval
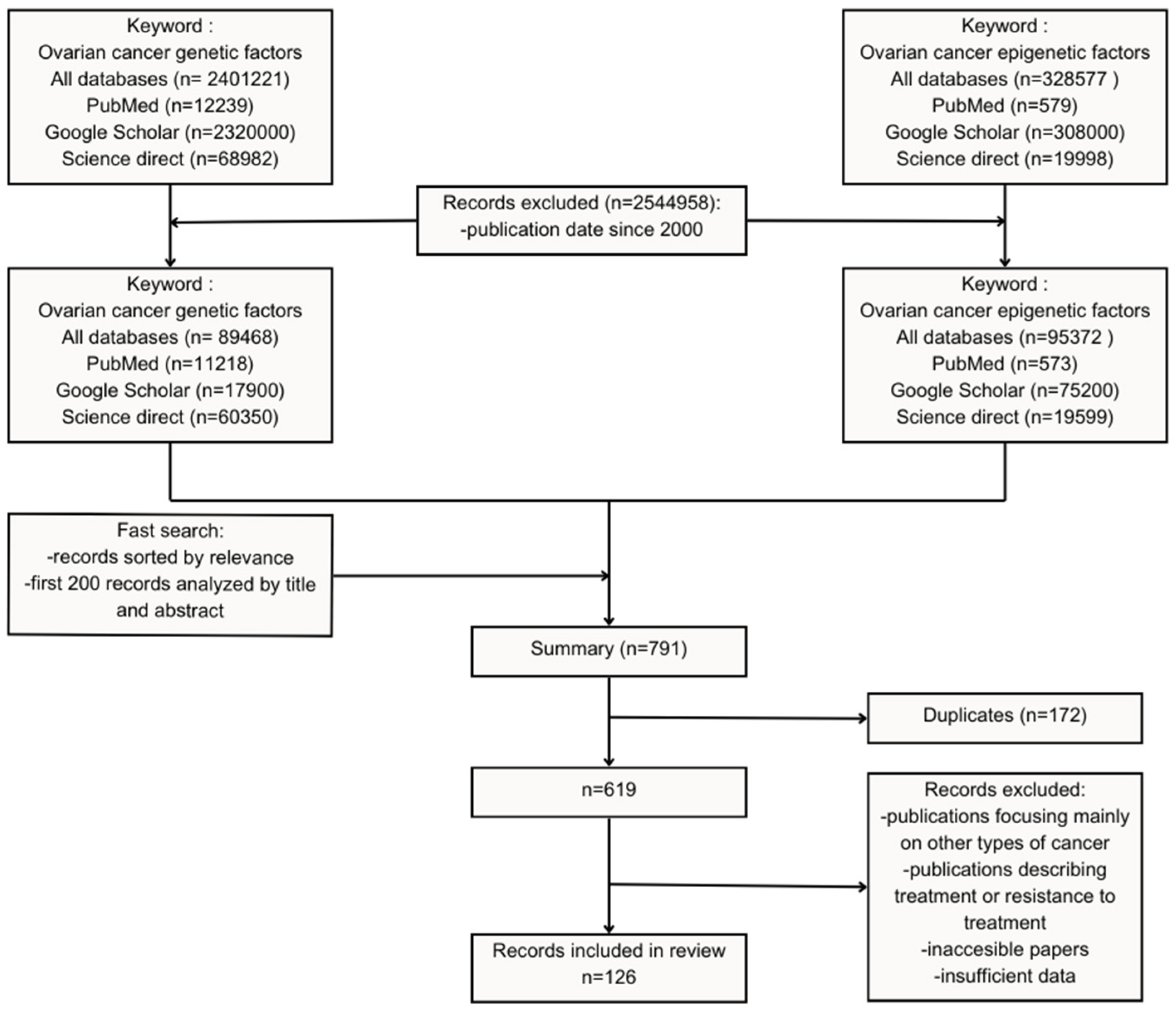
2.4. Data Analysis
2.5. Data Compilation
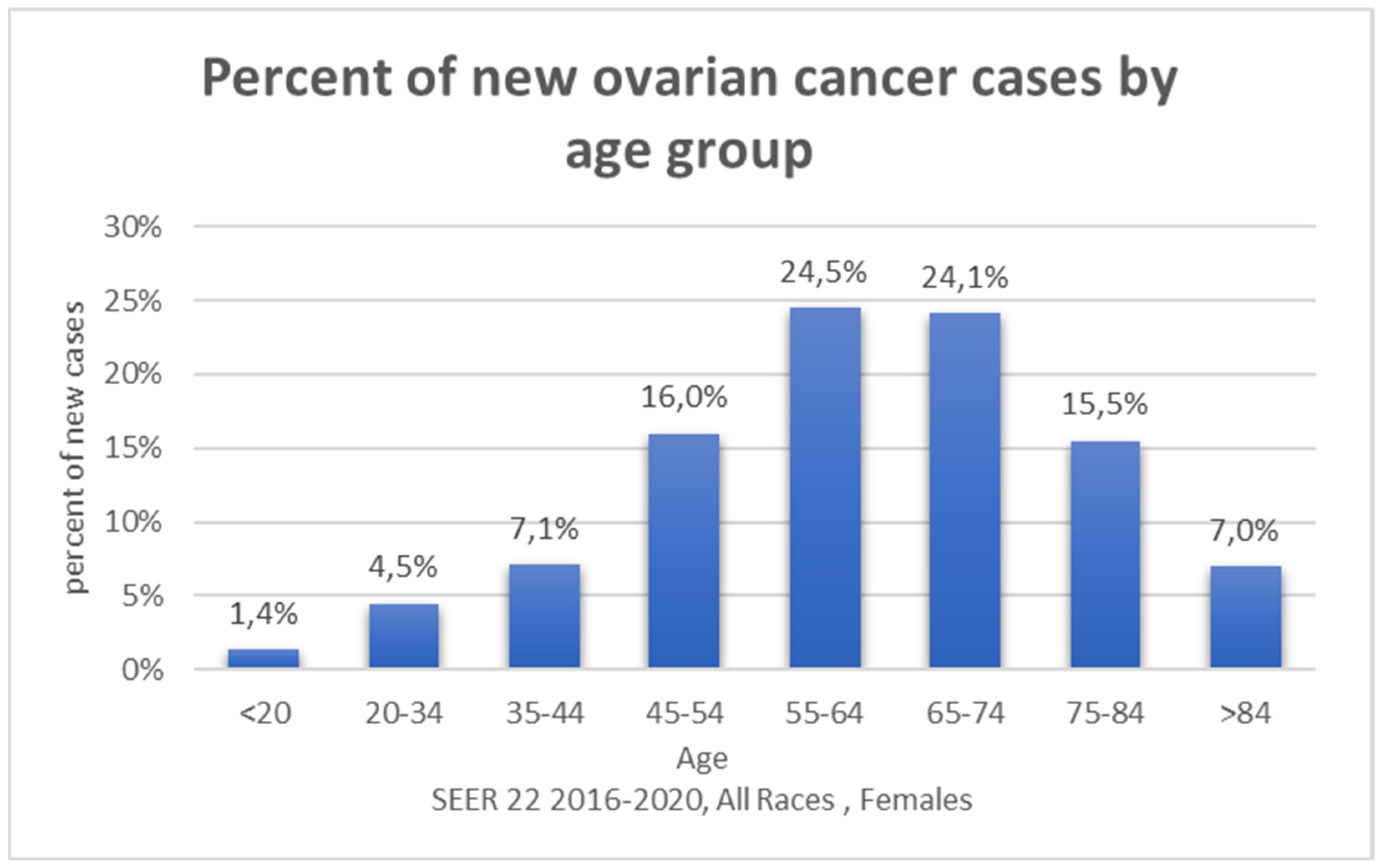
3. Results
3.1. Selected Genetic FACTORS in Ovarian Carcinogenesis
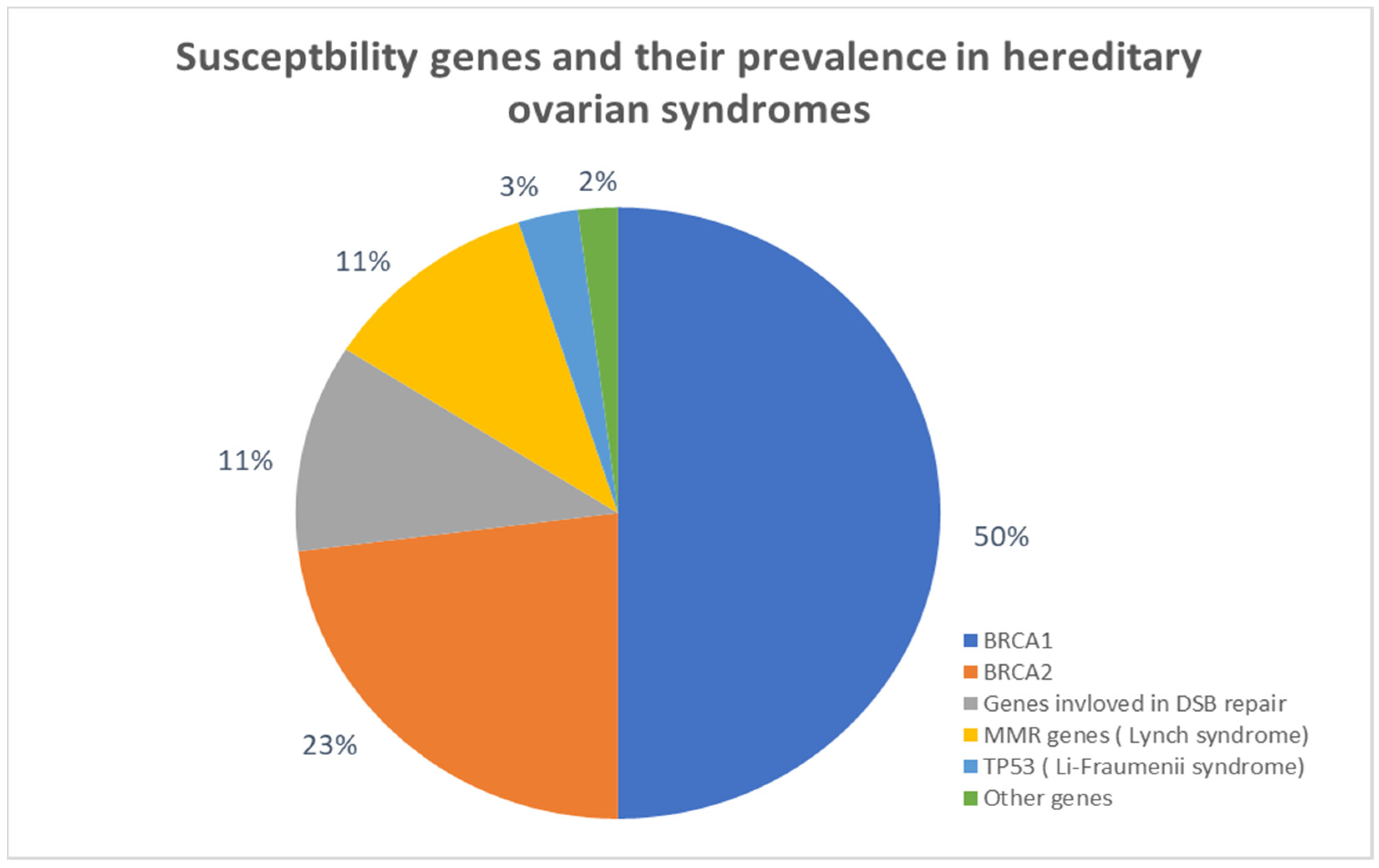
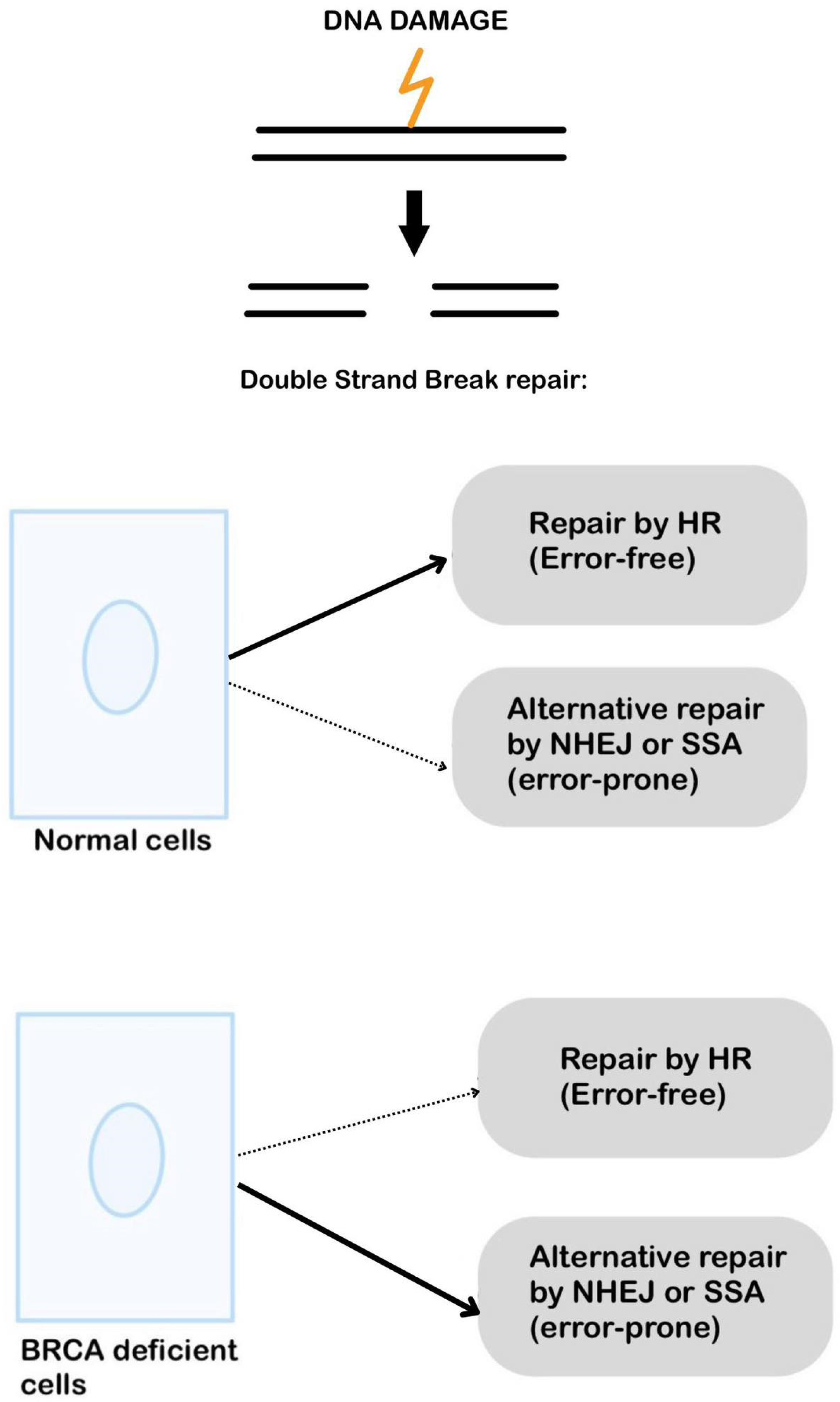
3.1.1. BRCA1/2 and Double Strand Break Repair
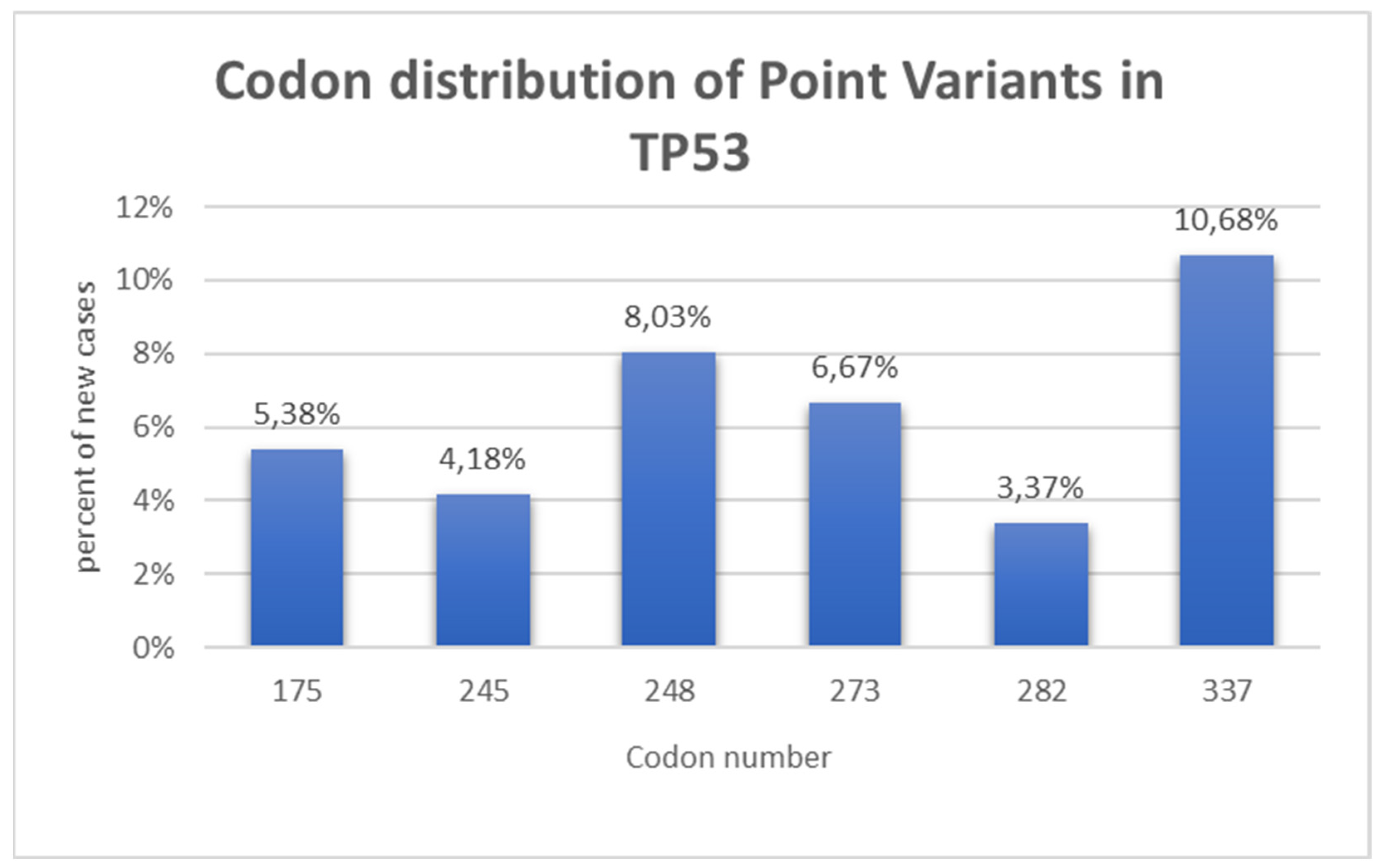
3.1.2. Li-Fraumeni Syndrome
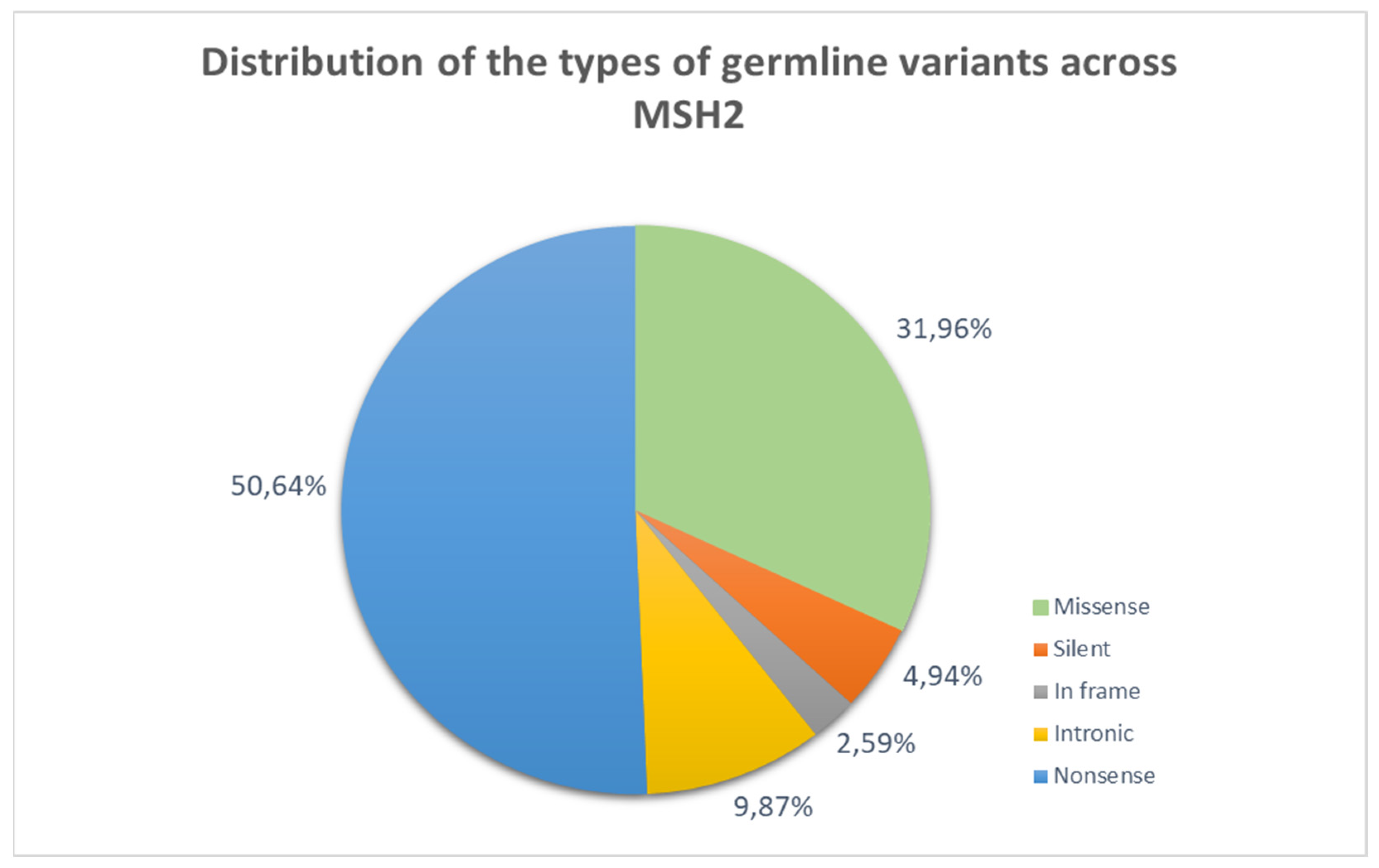
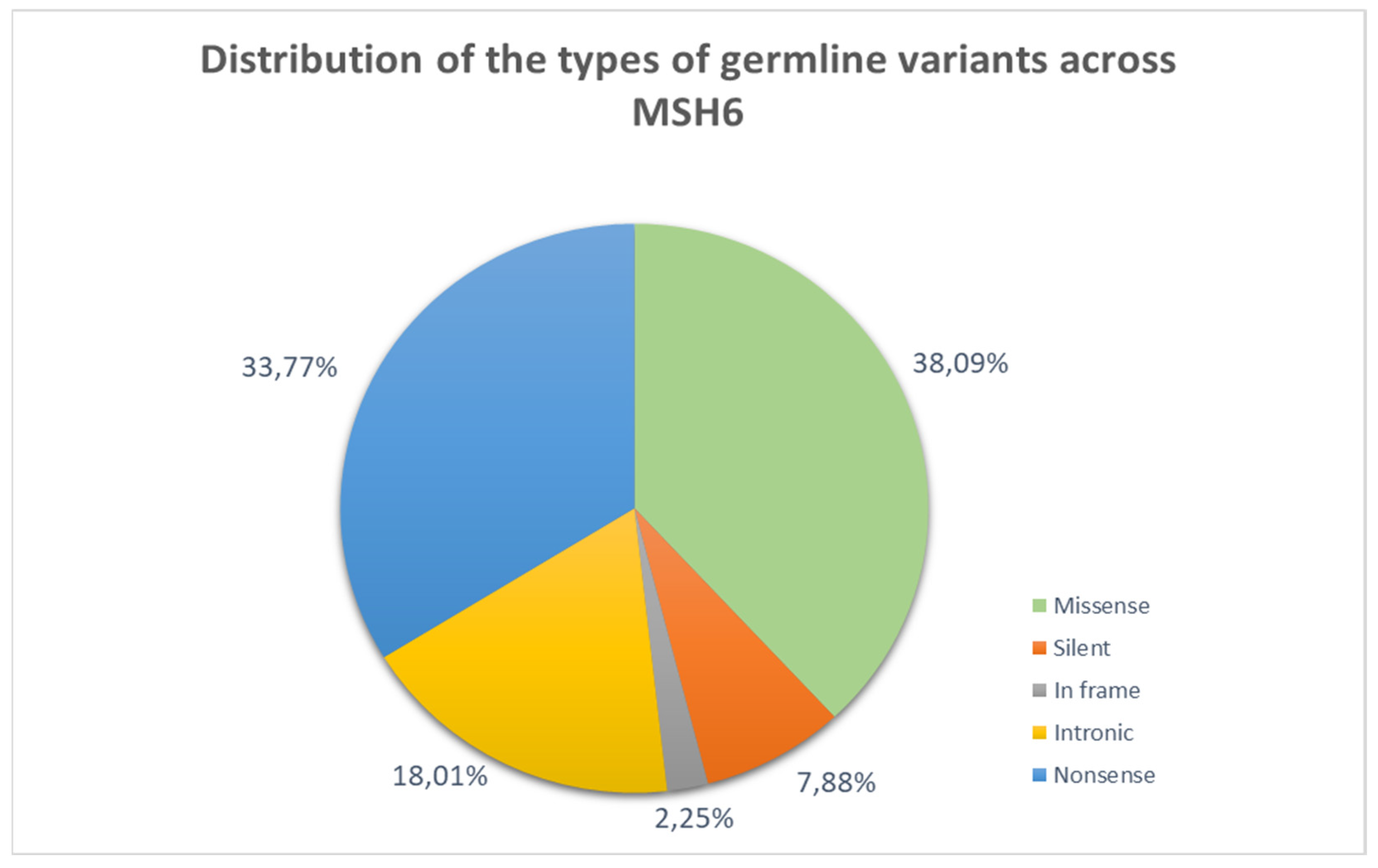
3.1.3. Lynch Syndrome
3.1.4. Phosphatase and Tensin Homolog Protein Expression
3.2. Hormonal Factors
3.2.1. Estrogens
3.2.2. Androgens
3.2.3. Progesterone
3.2.4. Progesterone Receptor
3.3. Environmental Factors
3.3.1. PCOS and Age of Menopause
3.3.2. Pregnancy and Breastfeeding
3.3.3. Combined Oral Contraceptive Pills
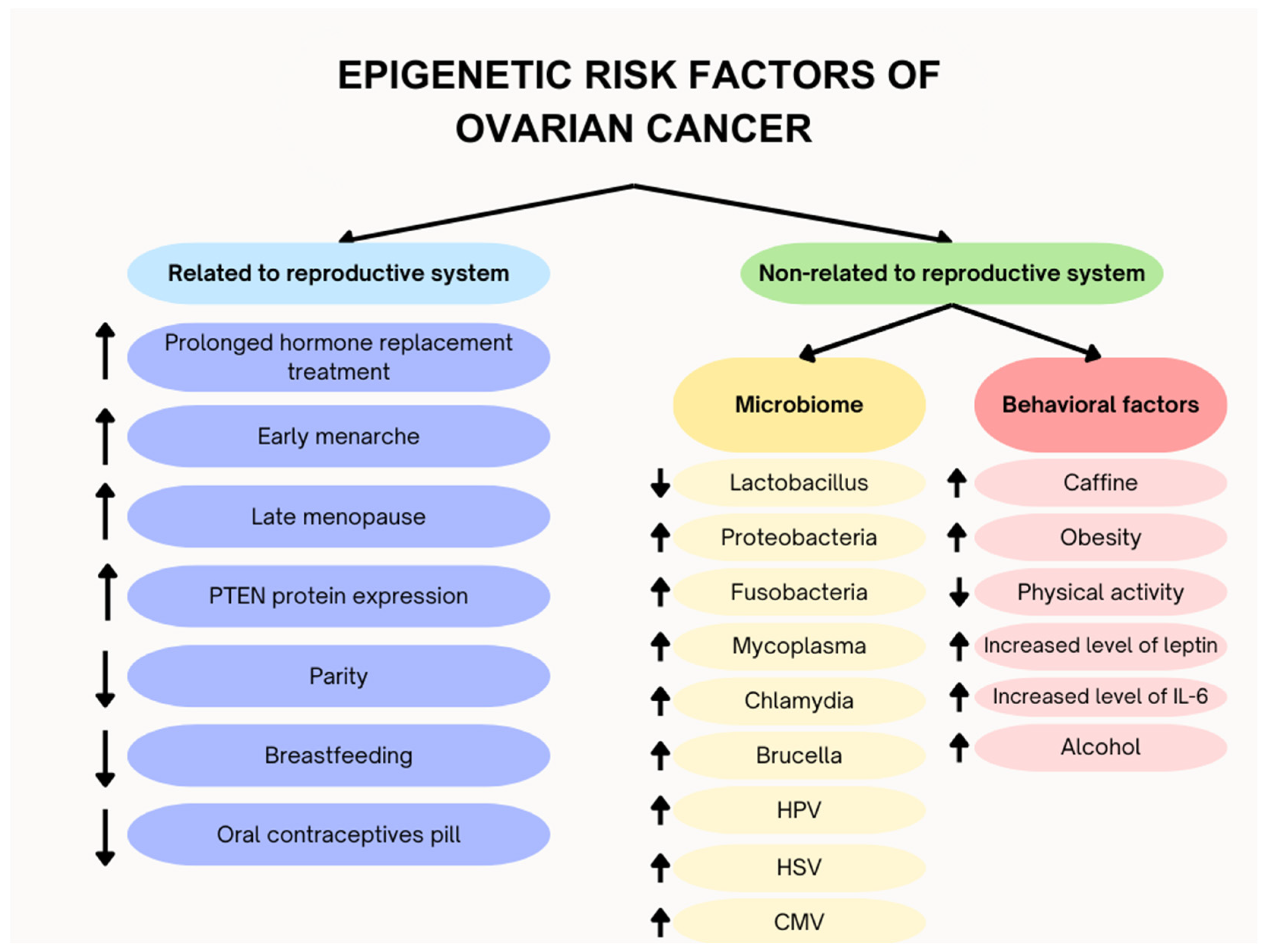
3.3.4. Role of Microbiome
3.3.5. Role of Viruses
3.3.6. Obesity, Physical Activity, and Metabolic Basis of Ovarian Cancer
3.3.7. Alcohol
3.3.8. Caffeine
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- S. Gupta et al., “Prevalence of BRCA1 and BRCA2 Mutations Among Patients With Ovarian, Primary Peritoneal, and Fallopian Tube Cancer in India: A Multicenter Cross-Sectional Study,” JCO Glob Oncol, vol. 7, no. 7, pp. 849–861, Dec. 2021. [CrossRef]
- “Ovarian Cancer By The Numbers | OCRA.” Accessed: Mar. 03, 2024. [Online]. Available online: https://ocrahope.org/get-the-facts/statistics/.
- C. J. Cabasag et al., “Ovarian cancer today and tomorrow: A global assessment by world region and Human Development Index using GLOBOCAN 2020,” Int J Cancer, vol. 151, no. 9, pp. 1535–1541, Nov. 2022. [CrossRef]
- “Ovarian Cancer Statistics | How Common is Ovarian Cancer | American Cancer Society.” Accessed: Feb. 28, 2024. [Online]. Available online: https://www.cancer.org/cancer/types/ovarian-cancer/about/key-statistics.html.
- A. Toss et al., “Hereditary ovarian cancer: Not only BRCA 1 and 2 Genes,” Biomed Res Int, vol. 2015, 2015. [CrossRef]
- A. S. Avramenko and J. M. Flanagan, “An epigenetic hypothesis for ovarian cancer prevention by oral contraceptive pill use,” Clin Epigenetics, vol. 15, no. 1, Dec. 2023. [CrossRef]
- K. Xu, S. Yang, and Y. Zhao, “Prognostic significance of BRCA mutations in ovarian cancer: an updated systematic review with meta-analysis,” Oncotarget, vol. 8, no. 1, p. 285, Jan. 2017. [CrossRef]
- J. T. Casaubon, S. Kashyap, and J.-P. Regan, “BRCA1 and BRCA2 Mutations,” StatPearls, Jul. 2023, Accessed: Feb. 28, 2024. [Online]. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470239/.
- A. Alacacioglu et al., “BRCA genes: BRCA 1 and BRCA 2,” JBUON, vol. 23, no. 4, pp. 862–866, 2018.
- A. R. Venkitaraman, “Cancer susceptibility and the functions of BRCA1 and BRCA2,” Cell, vol. 108, no. 2, pp. 171–182, Jan. 2002. [CrossRef]
- I. Vergote et al., “European experts consensus: BRCA/homologous recombination deficiency testing in first-line ovarian cancer,” Annals of Oncology, vol. 33, no. 3, pp. 276–287, Mar. 2022. [CrossRef]
- A. N. J. Tutt et al., “Exploiting the DNA repair defect in BRCA mutant cells in the design of new therapeutic strategies for cancer,” Cold Spring Harb Symp Quant Biol, vol. 70, pp. 139–148, 2005. [CrossRef]
- X. Li and W. D. Heyer, “Homologous recombination in DNA repair and DNA damage tolerance,” Cell Research 2008 18:1, vol. 18, no. 1, pp. 99–113, Jan. 2008. [CrossRef]
- J. Baselga et al., “A positive defect,” Nature Reviews Cancer 2005 5:5, vol. 5, no. 5, pp. 333–333, Apr. 2005. [CrossRef]
- K. D. Doig, A. P. Fellowes, and S. B. Fox, “Homologous Recombination Repair Deficiency: An Overview for Pathologists,” Modern Pathology, vol. 36, no. 3, p. 100049, Mar. 2023. [CrossRef]
- A. Villani, T. Frebourg, and D. Malkin, “Li-Fraumeni Syndrome,” The Hereditary Basis of Childhood Cancer, pp. 1–21, Aug. 2023. [CrossRef]
- “Li-Fraumeni Syndrome | Cancer.Net.” Accessed: May 06, 2024. [Online]. Available online: https://www.cancer.net/cancer-types/li-fraumeni-syndrome.
- T. Guha and D. Malkin, “Inherited TP53 Mutations and the Li-Fraumeni Syndrome,” Cold Spring Harb Perspect Med, vol. 7, no. 4, Apr. 2017. [CrossRef]
- A. D. Sorrell, C. R. Espenschied, J. O. Culver, and J. N. Weitzel, “TP53 Testing and Li-Fraumeni Syndrome: Current Status of Clinical Applications and Future Directions,” Mol Diagn Ther, vol. 17, no. 1, p. 31, Feb. 2013. [CrossRef]
- T. Kumamoto et al., “Medical guidelines for Li–Fraumeni syndrome 2019, version 1.1,” International Journal of Clinical Oncology 2021 26:12, vol. 26, no. 12, pp. 2161–2178, Oct. 2021. [CrossRef]
- K. A. Underkofler and K. L. Ring, “Updates in gynecologic care for individuals with lynch syndrome,” Front Oncol, vol. 13, 2023. [CrossRef]
- B. J. Bansidhar and J. Silinsky, “History and Pathogenesis of Lynch Syndrome,” Clin Colon Rectal Surg, vol. 25, no. 2, p. 63, 2012. [CrossRef]
- N. A. Ryan, R. F. McMahon, N. C. Ramchander, M. W. Seif, D. G. Evans, and E. J. Crosbie, “Lynch syndrome for the gynaecologist,” The Obstetrician & Gynaecologist, vol. 23, no. 1, p. 9, Jan. 2021. [CrossRef]
- B. Liu et al., “Analysis of mismatch repair genes in hereditary non-polyposis colorectal cancer patients,” Nat Med, vol. 2, no. 2, pp. 169–174, 1996. [CrossRef]
- F. E. McRonald et al., “Identification of people with Lynch syndrome from those presenting with colorectal cancer in England: baseline analysis of the diagnostic pathway,” European Journal of Human Genetics 2024 32:5, vol. 32, no. 5, pp. 529–538, Feb. 2024. [CrossRef]
- H. Hampel and A. De La Chapelle, “The search for unaffected individuals with Lynch syndrome: do the ends justify the means?,” Cancer Prev Res (Phila), vol. 4, no. 1, pp. 1–5, Jan. 2011. [CrossRef]
- E. M. Grindedal et al., “Survival in women with MMR mutations and ovarian cancer: a multicentre study in Lynch syndrome kindreds,” J Med Genet, vol. 47, no. 2, pp. 99–102, Feb. 2010. [CrossRef]
- R. R. Broaddus et al., “Pathologic features of endometrial carcinoma associated with HNPCC: a comparison with sporadic endometrial carcinoma,” Cancer, vol. 106, no. 1, pp. 87–94, Jan. 2006. [CrossRef]
- C. A. Christianson, K. P. Powell, S. E. Hahn, S. H. Blanton, J. Bogacik, and V. C. Henrich, “The use of a family history risk assessment tool within a community health care system: views of primary care providers,” J Genet Couns, vol. 21, no. 5, pp. 652–661, 2012. [CrossRef]
- E. J. Crosbie et al., “The Manchester International Consensus Group recommendations for the management of gynecological cancers in Lynch syndrome,” Genet Med, vol. 21, no. 10, pp. 2390–2400, Oct. 2019. [CrossRef]
- X. Li et al., “Estrogen promotes Epithelial ovarian cancer cells proliferation via down-regulating expression and activating phosphorylation of PTEN,” Arch Biochem Biophys, vol. 743, p. 109662, Jul. 2023. [CrossRef]
- S. Sarwar, A. Alamro, F. Huq, and A. Alghamdi, “Insights Into the Role of Epigenetic Factors Determining the Estrogen Response in Estrogen-Positive Ovarian Cancer and Prospects of Combining Epi-Drugs With Endocrine Therapy,” Front Genet, vol. 13, Jul. 2022. [CrossRef]
- B. Zhou et al., “Hormone replacement therapy and ovarian cancer risk: A meta-analysis,” Gynecol Oncol, vol. 108, no. 3, pp. 641–651, Mar. 2008. [CrossRef]
- F. Mungenast and T. Thalhammer, “Estrogen Biosynthesis and Action in Ovarian Cancer,” Front Endocrinol (Lausanne), vol. 5, no. NOV, 2014. [CrossRef]
- J. H. Choi, A. S. T. Wong, H. F. Huang, and P. C. K. Leung, “Gonadotropins and Ovarian Cancer,” Endocr Rev, vol. 28, no. 4, pp. 440–461, Jun. 2007. [CrossRef]
- H. Yang et al., “Age at menarche and epithelial ovarian cancer risk: A meta-analysis and Mendelian randomization study,” Cancer Med, vol. 8, no. 8, p. 4012, Jul. 2019. [CrossRef]
- N.-M. Ewa et al., “Ovarian cancer – modern approach to its origin and histogenesis,” Ginekol Pol, vol. 83, no. 6, pp. 454–457, 2012, Accessed: Apr. 28, 2024. [Online]. Available online: https://journals.viamedica.pl/ginekologia_polska/article/view/46183.
- K. A. Kujawa and K. M. Lisowska, “Rak jajnika – od biologii do kliniki,” Postepy Hig Med Dosw, vol. 69, pp. 1275–1290, Dec. 2015. [CrossRef]
- Maciej Serda et al., “Utrata heterozygotyczności loci sprzężonych z genami BRCA1 i BRCA2 w rakach jajnika,” Uniwersytet śląski, vol. 7, no. 1, pp. 343–354, 2006.
- P. G. Moorman et al., “Oral contraceptives and risk of ovarian cancer and breast cancer among high-risk women: a systematic review and meta-analysis,” J Clin Oncol, vol. 31, no. 33, pp. 4188–4198, Nov. 2013. [CrossRef]
- M. A. Merritt et al., “Reproductive factors and risk of mortality in the European Prospective Investigation into Cancer and Nutrition; a cohort study,” BMC Med, vol. 13, no. 1, pp. 1–15, Oct. 2015. [CrossRef]
- “Expression of Gonadotropin Receptor and Growth Responses to Key Reproductive Hormones in Normal and Malignant Human Ovarian Surface Epithelial Cells1 | Cancer Research | American Association for Cancer Research.” Accessed: Apr. 16, 2024. [Online]. Available online: https://aacrjournals.org/cancerres/article/61/18/6768/507837/Expression-of-Gonadotropin-Receptor-and-Growth.
- V. Syed, G. Ulinski, S. C. Mok, and S. M. Ho, “Reproductive Hormone-Induced, STAT3-Mediated Interleukin 6 Action in Normal and Malignant Human Ovarian Surface Epithelial Cells,” JNCI: Journal of the National Cancer Institute, vol. 94, no. 8, pp. 617–629, Apr. 2002. [CrossRef]
- V. Syed and S. M. Ho, “Progesterone-induced apoptosis in immortalized normal and malignant human ovarian surface epithelial cells involves enhanced expression of FasL,” Oncogene 2003 22:44, vol. 22, no. 44, pp. 6883–6890, Oct. 2003. [CrossRef]
- Q. Ye, W. Cai, Y. Zheng, B. M. Evers, and Q. B. She, “ERK and AKT signaling cooperate to translationally regulate survivin expression for metastatic progression of colorectal cancer,” Oncogene, vol. 33, no. 14, pp. 1828–1839, Apr. 2014. [CrossRef]
- F. Modugno, “Ovarian Cancer and Polymorphisms in the Androgen and Progesterone Receptor Genes: A HuGE Review,” Am J Epidemiol, vol. 159, no. 4, pp. 319–335, Feb. 2004. [CrossRef]
- C. L. Pearce et al., “Progesterone receptor variation and risk of ovarian cancer is limited to the invasive endometrioid subtype: results from the ovarian cancer association consortium pooled analysis,” Br J Cancer, vol. 98, no. 2, p. 282, Jan. 2008. [CrossRef]
- K. L. Terry, I. De Vivo, L. Titus-Ernstoff, P. M. Sluss, and D. W. Cramer, “Genetic Variation in the Progesterone Receptor Gene and Ovarian Cancer Risk,” Am J Epidemiol, vol. 161, no. 5, pp. 442–451, Mar. 2005. [CrossRef]
- D. B. Leite et al., “Progesterone receptor (PROGINS) polymorphism and the risk of ovarian cancer,” Steroids, vol. 73, no. 6, pp. 676–680, Jul. 2008. [CrossRef]
- I. B. Runnebaum et al., “Progesterone receptor variant increases ovarian cancer risk in BRCA1 and BRCA2 mutation carriers who were never exposed to oral contraceptives,” Pharmacogenetics, vol. 11, no. 7, pp. 635–638, 2001. [CrossRef]
- H. P. K. Throwba et al., “The epigenetic correlation among ovarian cancer, endometriosis and PCOS: A review,” Crit Rev Oncol Hematol, vol. 180, p. 103852, Dec. 2022. [CrossRef]
- B. M. Reid, J. B. Permuth, and T. A. Sellers, “Epidemiology of ovarian cancer: a review,” Cancer Biol Med, vol. 14, no. 1, p. 9, Mar. 2017. [CrossRef]
- R. Troisi et al., “The Role of Pregnancy, Perinatal Factors, and Hormones in Maternal Cancer Risk: A review of the evidence,” J Intern Med, vol. 283, no. 5, p. 430, May 2018. [CrossRef]
- H. O. Adami et al., “Parity, age at first childbirth, and risk of ovarian cancer,” The Lancet, vol. 344, no. 8932, pp. 1250–1254, Nov. 1994. [CrossRef]
- K. K. Tsilidis et al., “Oral contraceptive use and reproductive factors and risk of ovarian cancer in the European Prospective Investigation into Cancer and Nutrition,” British Journal of Cancer 2011 105:9, vol. 105, no. 9, pp. 1436–1442, Sep. 2011. [CrossRef]
- V. Toufakis, S. Katuwal, E. Pukkala, and J. S. Tapanainen, “Impact of parity on the incidence of ovarian cancer subtypes: a population-based case-control study,” Acta Oncol, vol. 60, no. 7, pp. 850–855, 2021. [CrossRef]
- A. Ramírez-de-Arellano, J. C. Villegas-Pineda, C. D. Hernández-Silva, and A. L. Pereira-Suárez, “The Relevant Participation of Prolactin in the Genesis and Progression of Gynecological Cancers,” Front Endocrinol (Lausanne), vol. 12, Oct. 2021. [CrossRef]
- G. Lurie et al., “Combined oral contraceptive use and epithelial ovarian cancer risk: time-related effects,” Epidemiology, vol. 19, no. 2, pp. 237–243, Mar. 2008. [CrossRef]
- J. S. Ferris, M. B. Daly, S. S. Buys, J. M. Genkinger, Y. Liao, and M. B. Terry, “Oral contraceptive and reproductive risk factors for ovarian cancer within sisters in the breast cancer family registry,” Br J Cancer, vol. 110, no. 4, p. 1074, Feb. 2014. [CrossRef]
- A. A. Samkari et al., “Body Microbiota and Its Relationship With Benign and Malignant Breast Tumors: A Systematic Review,” Cureus, vol. 14, no. 5, May 2022. [CrossRef]
- E. Reitano et al., “Oral Bacterial Microbiota in Digestive Cancer Patients: A Systematic Review,” Microorganisms, vol. 9, no. 12, Dec. 2021. [CrossRef]
- X. Yu et al., “Microbial dysbiosis in oral squamous cell carcinoma: A systematic review and meta-analysis,” Heliyon, vol. 9, no. 2, Feb. 2023. [CrossRef]
- L. Su Mun et al., “Association of Microbiome with Oral Squamous Cell Carcinoma: A Systematic Review of the Metagenomic Studies,” Int J Environ Res Public Health, vol. 18, no. 14, Jul. 2021. [CrossRef]
- S. Banerjee et al., “The ovarian cancer oncobiome,” Oncotarget, vol. 8, no. 22, p. 36225, May 2017. [CrossRef]
- X. Hu et al., “Gut microbiota dysbiosis promotes the development of epithelial ovarian cancer via regulating Hedgehog signaling pathway,” Gut Microbes, vol. 15, no. 1, 2023. [CrossRef]
- R. Siddiqui, Z. Makhlouf, A. M. Alharbi, H. Alfahemi, and N. A. Khan, “The Gut Microbiome and Female Health,” Biology (Basel), vol. 11, no. 11, p. 1683, Nov. 2022. [CrossRef]
- A. Sipos et al., “The role of the microbiome in ovarian cancer: mechanistic insights into oncobiosis and to bacterial metabolite signaling,” Molecular Medicine, vol. 27, no. 1, p. 33, Dec. 2021. [CrossRef]
- N. R. Nené et al., “Association between the cervicovaginal microbiome, BRCA1 mutation status, and risk of ovarian cancer: a case-control study,” Lancet Oncol, vol. 20, no. 8, pp. 1171–1182, Aug. 2019. [CrossRef]
- P. Łaniewski, Z. E. Ilhan, and M. M. Herbst-Kralovetz, “The microbiome and gynaecological cancer development, prevention and therapy,” Nat Rev Urol, vol. 17, no. 4, p. 232, 2020. [CrossRef]
- Y. C. Lu, W. C. Yeh, and P. S. Ohashi, “LPS/TLR4 signal transduction pathway,” Cytokine, vol. 42, no. 2, pp. 145–151, May 2008. [CrossRef]
- B. Bertani and N. Ruiz, “Function and Biogenesis of Lipopolysaccharides,” EcoSal Plus, vol. 8, no. 1, Dec. 2018. [CrossRef]
- M. Cazzaniga, M. Cardinali, F. Di Pierro, and A. Bertuccioli, “Ovarian Microbiota, Ovarian Cancer and the Underestimated Role of HPV,” International Journal of Molecular Sciences 2022, Vol. 23, Page 16019, vol. 23, no. 24, p. 16019, Dec. 2022. [CrossRef]
- N. Gulve and T. Rudel, “Chlamydia trachomatis and human herpesvirus 6 infections in ovarian cancer—Casual or causal?,” PLoS Pathog, vol. 15, no. 11, 2019. [CrossRef]
- S. Pathak, J. R. Wilczyński, and E. Paradowska, “Factors in Oncogenesis: Viral Infections in Ovarian Cancer,” Cancers 2020, Vol. 12, Page 561, vol. 12, no. 3, p. 561, Feb. 2020. [CrossRef]
- E. Paradowska, A. Jabłońska, M. Studzińska, M. Wilczyński, and J. R. Wilczyński, “Detection and genotyping of CMV and HPV in tumors and fallopian tubes from epithelial ovarian cancer patients,” Sci Rep, vol. 9, no. 1, Dec. 2019. [CrossRef]
- E. Malisic, R. Jankovic, and K. Jakovljevic, “Detection and genotyping of human papillomaviruses and their role in the development of ovarian carcinomas,” Arch Gynecol Obstet, vol. 286, no. 3, pp. 723–728, Sep. 2012. [CrossRef]
- S. Banerjee et al., “The ovarian cancer oncobiome,” Oncotarget, vol. 8, no. 22, pp. 36225–36245, May 2017. [CrossRef]
- M. F. Svahn, M. T. Faber, J. Christensen, B. Norrild, and S. K. Kjaer, “Prevalence of human papillomavirus in epithelial ovarian cancer tissue. A meta-analysis of observational studies,” Acta Obstet Gynecol Scand, vol. 93, no. 1, pp. 6–19, Jan. 2014. [CrossRef]
- C. M. Olsen, A. C. Green, D. C. Whiteman, S. Sadeghi, F. Kolahdooz, and P. M. Webb, “Obesity and the risk of epithelial ovarian cancer: A systematic review and meta-analysis,” Eur J Cancer, vol. 43, no. 4, pp. 690–709, Mar. 2007. [CrossRef]
- A. Urbute, K. Frederiksen, and S. K. Kjaer, “Early adulthood overweight and obesity and risk of premenopausal ovarian cancer, and premenopausal breast cancer including receptor status: prospective cohort study of nearly 500,000 Danish women,” Ann Epidemiol, vol. 70, pp. 61–67, Jun. 2022. [CrossRef]
- Z. Y. Liu et al., “Dietary inflammatory index and risk of gynecological cancers: a systematic review and meta-analysis of observational studies,” J Gynecol Oncol, vol. 30, no. 3, May 2019. [CrossRef]
- N. Ding, J. Zhan, Y. Shi, T. Qiao, P. Li, and T. Zhang, “Obesity in children and adolescents and the risk of ovarian cancer: A systematic review and dose–response meta-analysis,” PLoS One, vol. 17, no. 12, Dec. 2022. [CrossRef]
- C. M. Friedenreich, C. Ryder-Burbidge, and J. McNeil, “Physical activity, obesity and sedentary behavior in cancer etiology: epidemiologic evidence and biologic mechanisms,” Mol Oncol, vol. 15, no. 3, pp. 790–800, Mar. 2021. [CrossRef]
- M. Baczewska, K. Bojczuk, A. Kołakowski, J. Dobroch, P. Guzik, and P. Knapp, “Obesity and Energy Substrate Transporters in Ovarian Cancer—Review,” Molecules, vol. 26, no. 6, Mar. 2021. [CrossRef]
- N. Khanlarkhani et al., “Metabolic risk factors of ovarian cancer: a review,” JBRA Assist Reprod, vol. 26, no. 2, p. 335, 2022. [CrossRef]
- N. Kolomeyevskaya et al., “Cytokine profiling of ascites at primary surgery identifies an interaction of tumor necrosis factor-α and interleukin-6 in predicting reduced progression-free survival in epithelial ovarian cancer,” Gynecol Oncol, vol. 138, no. 2, pp. 352–357, Aug. 2015. [CrossRef]
- A. Ray, J. Fornsaglio, S. Dogan, S. Hedau, S. D. Naik, and A. De, “Gynaecological cancers and leptin: A focus on the endometrium and ovary,” Facts Views Vis Obgyn, vol. 10, no. 1, p. 5, Mar. 2018, Accessed: Mar. 04, 2024. [Online]. Available: /pmc/articles/PMC6260667/.
- S. C. Larsson, N. Spyrou, and C. S. Mantzoros, “Body fatness associations with cancer: evidence from recent epidemiological studies and future directions,” Metabolism, vol. 137, Dec. 2022. [CrossRef]
- A. Urbute, K. Frederiksen, and S. K. Kjaer, “Early adulthood overweight and obesity and risk of premenopausal ovarian cancer, and premenopausal breast cancer including receptor status: prospective cohort study of nearly 500,000 Danish women,” Ann Epidemiol, vol. 70, pp. 61–67, Jun. 2022. [CrossRef]
- D. Wu et al., “Mediation analysis of alcohol consumption, DNA methylation, and epithelial ovarian cancer,” J Hum Genet, vol. 63, no. 3, p. 339, Mar. 2018. [CrossRef]
- D. Cvetkovic, “Early events in ovarian oncogenesis,” Reprod Biol Endocrinol, vol. 1, no. 1, p. 68, 2003. [CrossRef]
- S. Liu, S. Feng, F. Du, K. Zhang, and Y. Shen, “Association of smoking, alcohol, and coffee consumption with the risk of ovarian cancer and prognosis: a mendelian randomization study,” BMC Cancer, vol. 23, no. 1, Dec. 2023. [CrossRef]
- L. S. Cook et al., “Adult lifetime alcohol consumption and invasive epithelial ovarian cancer risk in a population-based case-control study,” Gynecol Oncol, vol. 140, no. 2, pp. 277–284, Feb. 2016. [CrossRef]
- K. Tanha et al., “Investigation on factors associated with ovarian cancer: an umbrella review of systematic review and meta-analyses,” J Ovarian Res, vol. 14, no. 1, p. 153, Dec. 2021. [CrossRef]
- 95. C. Pelucchi, C. Bosetti, C. Galeone, and C. La Vecchia, “Dietary acrylamide and cancer risk: An updated meta-analysis,” Int J Cancer, vol. 136, no. 12, pp. 2912–2922, Jun. 2015. [CrossRef]
- J. Kotsopoulos et al., “Coffee Intake, Variants in Genes Involved in Caffeine Metabolism and the Risk of Epithelial Ovarian Cancer,” Cancer Causes Control, vol. 20, no. 3, p. 335, Apr. 2009. [CrossRef]
- M. C. Vos, A. A. M. van der Wurff, T. H. van Kuppevelt, and L. F. A. G. Massuger, “The role of MMP-14 in ovarian cancer: a systematic review,” J Ovarian Res, vol. 14, no. 1, p. 101, Dec. 2021. [CrossRef]
- X. M. Zhu and W. F. Sun, “Association between matrix metalloproteinases polymorphisms and ovarian cancer risk: A meta-analysis and systematic review,” PLoS One, vol. 12, no. 9, Sep. 2017. [CrossRef]
- H. Liu, L. Sun, X. Liu, R. Wang, and Q. Luo, “Associations between non-coding RNAs genetic polymorphisms with ovarian cancer risk: A systematic review and meta-analysis update with trial sequential analysis,” Medicine, vol. 102, no. 39, p. E35257, Sep. 2023. [CrossRef]
- M. vahidi, M. Houshmand, M. Banoei, and F. Heidari, “The association between TIPARP gene polymorphisms rs2665390 and ovarian cancer susceptibility,” Gynecol Oncol Rep, vol. 47, p. 101175, Jun. 2023. [CrossRef]
- M. Kamali et al., “Association of XRCC2 rs3218536 Polymorphism with Susceptibility of Breast and Ovarian Cancer: A Systematic Review and Meta-Analysis,” Asian Pac J Cancer Prev, vol. 18, no. 7, p. 1743, Jul. 2017. [CrossRef]
- M. Karimi-Zarchi et al., “Association of XRCC3 18067 C>T (Thr241Met) polymorphism with risk of cervical and ovarian cancers: A systematic review and meta-analysis,” Interv Med Appl Sci, vol. 11, no. 3, p. 172, Sep. 2019. [CrossRef]
- E. Mikó et al., “Microbiome—Microbial Metabolome—Cancer Cell Interactions in Breast Cancer—Familiar, but Unexplored,” Cells, vol. 8, no. 4, Apr. 2019. [CrossRef]
- Y. Wu et al., “KLF5 Promotes Tumor Progression and Parp Inhibitor Resistance in Ovarian Cancer,” Advanced Science, vol. 10, no. 31, Nov. 2023. [CrossRef]
- A. Dovnik and N. F. Dovnik, “Vitamin D and Ovarian Cancer: Systematic Review of the Literature with a Focus on Molecular Mechanisms,” Cells, vol. 9, no. 2, Feb. 2020. [CrossRef]
- E. Deuster, U. Jeschke, Y. Ye, S. Mahner, and B. Czogalla, “Vitamin D and VDR in Gynecological Cancers—A Systematic Review,” Int J Mol Sci, vol. 18, no. 11, Nov. 2017. [CrossRef]
- T. Lawler and S. Warren Andersen, “Serum 25-Hydroxyvitamin D and Cancer Risk: A Systematic Review of Mendelian Randomization Studies,” Nutrients, vol. 15, no. 2, Jan. 2023. [CrossRef]
- X. Jiang, X. Li, W. Li, H. Bai, and Z. Zhang, “PARP inhibitors in ovarian cancer: Sensitivity prediction and resistance mechanisms,” J Cell Mol Med, vol. 23, no. 4, pp. 2303–2313, Apr. 2019. [CrossRef]
- D. M. O’Malley, T. C. Krivak, N. Kabil, J. Munley, and K. N. Moore, “PARP Inhibitors in Ovarian Cancer: A Review,” Target Oncol, vol. 18, no. 4, p. 471, Jul. 2023. [CrossRef]
- T. Kumamoto et al., “Medical guidelines for Li–Fraumeni syndrome 2019, version 1.1,” International Journal of Clinical Oncology 2021 26:12, vol. 26, no. 12, pp. 2161–2178, Oct. 2021. [CrossRef]
- J. Y. Pan et al., “Worldwide Practice Patterns in Lynch Syndrome Diagnosis and Management, Based on Data From the International Mismatch Repair Consortium.,” Clin Gastroenterol Hepatol, vol. 16, no. 12, pp. 1901-1910.e11, Dec. 2018. [CrossRef]
- D. P. Li et al., “Breastfeeding and ovarian cancer risk: a systematic review and meta-analysis of 40 epidemiological studies,” Asian Pac J Cancer Prev, vol. 15, no. 12, pp. 4829–4837, 2014. [CrossRef]
- A. Babic et al., “Association Between Breastfeeding and Ovarian Cancer Risk,” JAMA Oncol, vol. 6, no. 6, Jun. 2020. [CrossRef]
- J. M. Genkinger et al., “Alcohol intake and ovarian cancer risk: a pooled analysis of 10 cohort studies,” Br J Cancer, vol. 94, no. 5, pp. 757–762, Mar. 2006. [CrossRef]
- H. Yan-Hong, L. Jing, L. Hong, H. Shan-Shan, L. Yan, and L. Ju, “Association between alcohol consumption and the risk of ovarian cancer: a meta-analysis of prospective observational studies,” BMC Public Health, vol. 15, no. 1, Mar. 2015. [CrossRef]
- D. Huber, S. Seitz, K. Kast, G. Emons, and O. Ortmann, “Use of oral contraceptives in BRCA mutation carriers and risk for ovarian and breast cancer: a systematic review,” Arch Gynecol Obstet, vol. 301, no. 4, pp. 875–884, Apr. 2020. [CrossRef]
- Y. Y. Xia and J. Kotsopoulos, “Beyond the pill: contraception and the prevention of hereditary ovarian cancer,” Hered Cancer Clin Pract, vol. 20, no. 1, Dec. 2022. [CrossRef]
- J. Steevens, L. J. Schouten, B. A. J. Verhage, R. A. Goldbohm, and P. A. Van Den Brandt, “Tea and coffee drinking and ovarian cancer risk: results from the Netherlands Cohort Study and a meta-analysis,” Br J Cancer, vol. 97, no. 9, p. 1291, Nov. 2007. [CrossRef]
- J. M. Genkinger et al., “Alcohol intake and ovarian cancer risk: a pooled analysis of 10 cohort studies,” Br J Cancer, vol. 94, no. 5, pp. 757–762, Mar. 2006. [CrossRef]
- H. Yan-Hong, L. Jing, L. Hong, H. Shan-Shan, L. Yan, and L. Ju, “Association between alcohol consumption and the risk of ovarian cancer: a meta-analysis of prospective observational studies,” BMC Public Health, vol. 15, no. 1, Mar. 2015. [CrossRef]
- H. R. Harris and K. L. Terry, “Polycystic ovary syndrome and risk of endometrial, ovarian, and breast cancer: a systematic review,” Fertil Res Pract, vol. 2, no. 1, Dec. 2016. [CrossRef]
- H. P. K. Throwba et al., “The epigenetic correlation among ovarian cancer, endometriosis and PCOS: A review,” Crit Rev Oncol Hematol, vol. 180, p. 103852, Dec. 2022. [CrossRef]
- A. N. Rosenthal et al., “Evidence of Stage Shift in Women Diagnosed With Ovarian Cancer During Phase II of the United Kingdom Familial Ovarian Cancer Screening Study,” J Clin Oncol, vol. 35, no. 13, pp. 1411–1420, May 2017. [CrossRef]
- J. Domański, Z. Domagala, J. E. Simmons, and M. Wanat, “Terra Incognita in anatomical museology – A literature review from the perspective of evidence-based care,” Annals of Anatomy - Anatomischer Anzeiger, vol. 245, p. 152013, Jan. 2023. [CrossRef]
- M. Mateusz et al., “Decoding the Mysteries of the Obturator Nerve,” J Anat Soc India, vol. 73, no. 1, pp. 64–69, 2024. [CrossRef]
- D. Domagała et al., “Cellular, Molecular and Clinical Aspects of Aortic Aneurysm—Vascular Physiology and Pathophysiology,” Cells 2024, Vol. 13, Page 274, vol. 13, no. 3, p. 274, Feb. 2024. [CrossRef]

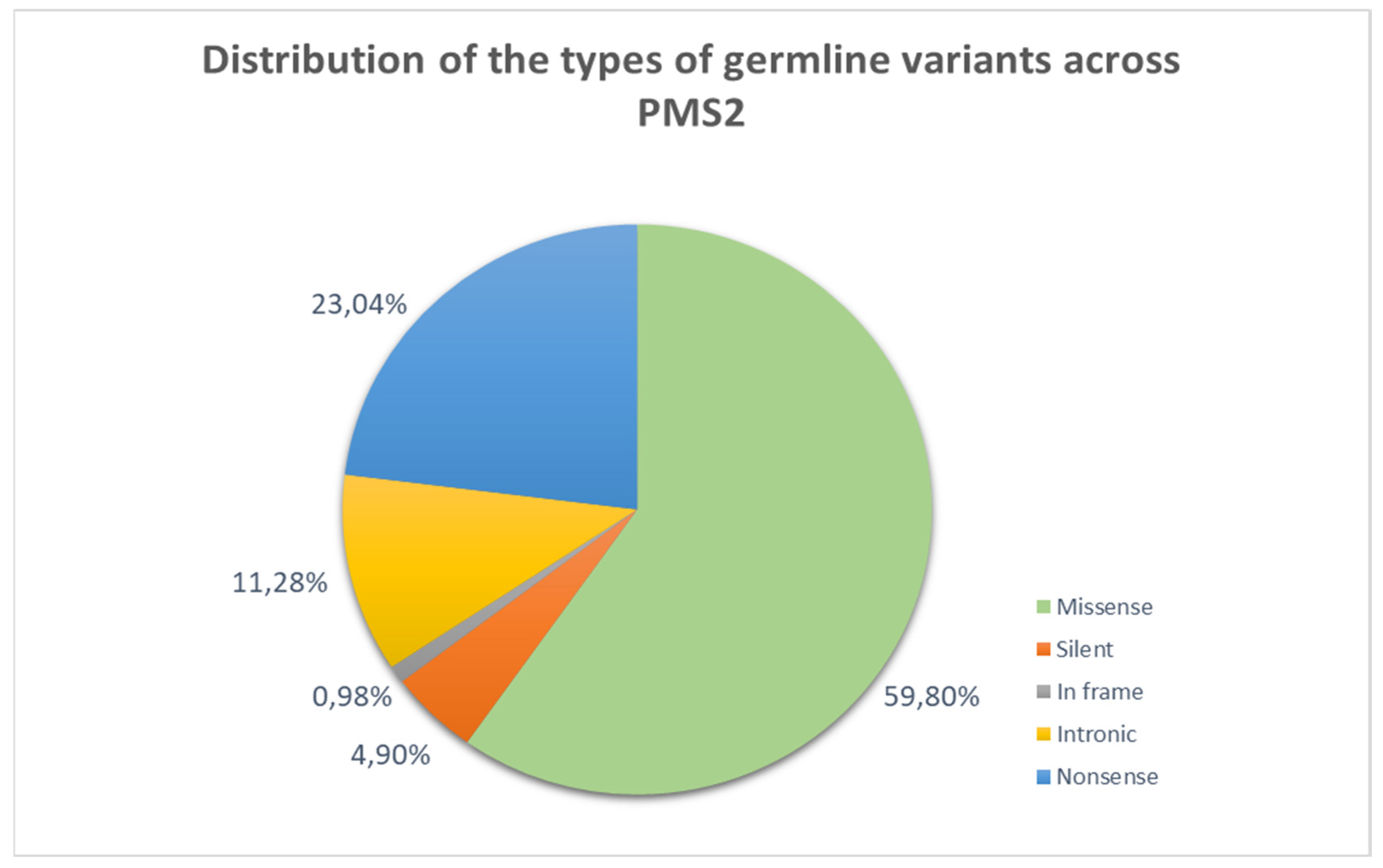
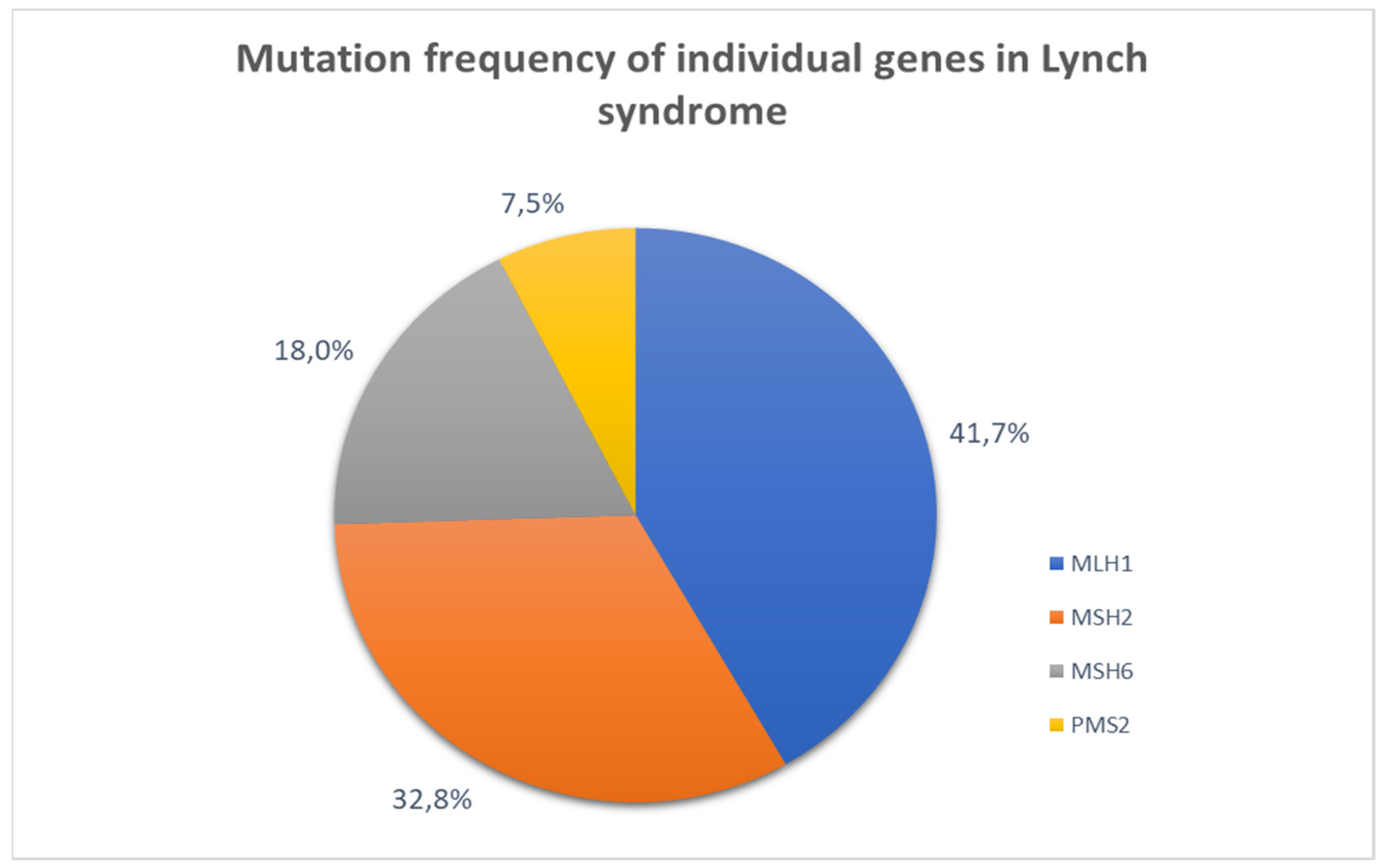
| Cancer Type | Lifetime Risk in general population | Lifetime Risk with MLH1 mutation | Average age of diagnosis in general population | Average age of diagnosis with MLH1 mutation |
|---|---|---|---|---|
| Ovarian | 4-20% | 1,30% | 46 | 63 |
| Colorectal | 46-61% | 4,20% | 44 | 68-72 |
| Endometrial | 34-54% | 3,10% | 49 | 60 |
| Cancer Type | Lifetime Risk in general population | Lifetime Risk with MSH2 mutation | Average age of diagnosis in general population | Average age of diagnosis with MSH2 mutation |
|---|---|---|---|---|
| Ovarian | 8-38% | 1,30% | 43 | 63 |
| Colorectal | 33-52% | 4,20% | 44 | 68-72 |
| Endometrial | 21-57% | 3,10% | 47-48 | 60 |
| Cancer Type | Lifetime Risk in general population | Lifetime Risk with MSH6 mutation | Average age of diagnosis in general population | Average age of diagnosis with MSH6 mutation |
|---|---|---|---|---|
| Ovarian | <1-13% | 1,30% | 46 | 63 |
| Colorectal | 10-44% | 4,20% | 42-69 | 68-72 |
| Endometrial | 16-49% | 3,10% | 53-55 | 60 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




