Submitted:
18 May 2024
Posted:
20 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Patients and Method
Statistical evaluation
Results
Discussion
Author Contributions
Funding
Ethics Committee Approval
Compliance with Ethical Standards
Data Access Statement
Acknowledgments
Conflicts of Interest
References
- Martinez, A.F.; Galindo, D.B.; Sanchez, J.R. Management of hyponatraemia and hypernatraemia during the Covid-19 pandemic: a consensus statement of the Spanish Society for Endocrinology (Acqua Neuroendocrinology Group). Rev. Endocr. Metab. Disord. 2021, 22, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Richardson S, Hirsch JS, Narasimhan M, Crawford, J. M., McGinn, T., et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052–59.
- Lippi, G.; South, A.M.; Henry, B.M. Electrolyte imbalances in patients with severe coronavirus disease 2019 (COVID-19). Ann. Clin. Biochem. 2020, 57, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Bhave, G. , & Neilson, E. G. Body fluid dynamics: back to the future. Journal of the American Society of Nephrology. 2011; 22(12): 2166-81.
- Gupta, A.; Madhavan, M.V.; Sehgal, K.; Nair, N.; Mahajan, S.; Sehrawat, T.S.; Bikdeli, B.; Ahluwalia, N.; Ausiello, J.C.; Wan, E.Y.; et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020, 26, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Nzerue, C.M.; Baffoe-Bonnie, H.; You, W.; Falana, B.; Dai, S. Predictors of outcome in hospitalized patients with severe hyponatremia. J Natl Med Assoc. 2003, 95, 335–43. [Google Scholar] [PubMed]
- Ruiz-Sanchez, J.G.; Nunez-Gil, I.J.; Cuesta, M.; Rubio, M.A.; Maroun-Eid, C.; Arroyo-Espliguero, R.; Romero, R.; Becerra-Munoz, V.M.; Uribarri, A.; Feltes, G.; et al. Prognostic Impact of Hyponatremia and Hypernatremia in COVID-19 Pneumonia. A HOPE-COVID-19 (Health Outcome Predictive Evaluation for COVID-19) Registry Analysis. Front. Endocrinol. 2020, 11, 599255. [Google Scholar] [CrossRef] [PubMed]
- Swart, R.M.; Hoorn, E.J.; Betjes, M.G.; Zietse, R. Hyponatremia and Inflammation: The Emerging Role of Interleukin-6 in Osmoregulation. Nephron Physiol. 2010, 118, p45–p51. [Google Scholar] [CrossRef] [PubMed]
- Landgraf, R.; Neumann, I.; Holsboer, F.; Pittman, Q.J. Interleukin-1β Stimulates both Central and Peripheral Release of Vasopressin and Oxytocin in the Rat. Eur. J. Neurosci. 1995, 7, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Mastorakos, G.; Weber, J.S.; A Magiakou, M.; Gunn, H.; Chrousos, G.P. Hypothalamic-pituitary-adrenal axis activation and stimulation of systemic vasopressin secretion by recombinant interleukin-6 in humans: potential implications for the syndrome of inappropriate vasopressin secretion. J. Clin. Endocrinol. Metab. 1994, 79, 934–939. [Google Scholar] [CrossRef] [PubMed]
- Berni A, Malandrino D, Parenti G, Maggi M, Poggesi L, Peri A. Hyponatremia, IL-6, and SARS-CoV-2 (COVID-19) infection: may all fit together? J Endocrinol Invest, 2020;43:1137–9. [CrossRef]
- Aggarwal S, Garcia-Telles N, Aggarwal G, Lavie C, Lippi G, Henry BM. Clinical features, laboratory characteristics, and out comes of patients hospitalized with coronavirus disease 2019 (COVID-19): early report from the United States. Diagn Berl Ger. 2020;7(2):91–6.
- Zimmer, M.A.; Zink, A.K.; Weißer, C.W.; Vogt, U.; Michelsen, A.; Priebe, H.-J.; Mols, G. Hypernatremia—A Manifestation of COVID-19: A Case Series. A&A Pr. 2020, 14, e01295. [Google Scholar] [CrossRef] [PubMed]
- Cuesta, M.; Slattery, D.; Goulden, E.L.; Gupta, S.; Tatro, E.; Sherlock, M.; Tormey, W.; O'Neill, S.; Thompson, C.J. Hyponatraemia in patients with community-acquired pneumonia; prevalence and aetiology, and natural history of SIAD. Clin. Endocrinol. 2019, 90, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Rong, L.; Nian, W.; He, Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment. Pharmacol. Ther. 2020, 51, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Ata, F.; Munir, W.; Yousaf, Z. Fluid Replacement Versus Fluid Restriction in COVID-19 Associated Hyponatremia. Cureus 2020, 12, e9059. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, Z.; Al-Shokri, S.D.; Al-Soub, H.; Mohamed, M.F.H. COVID-19-associated SIADH: a clue in the times of pandemic! Am. J. Physiol. Endocrinol. Metab. 2020, 318, E882–E885. [Google Scholar] [CrossRef] [PubMed]
| Demographic Parameters and Treatment Duration | No of Patients (n=209) | |
|---|---|---|
| Gender | Male (%) | 114 (54.5) |
| Female (%) | 95 (45.5) | |
| Age (years, SD) | 68.15 (±13.81) | |
| Body Mass Index (SD) | 26.66 (±2.66) | |
| RT-PCR Positivity to Admission (Days, 25th-75th) | 5 (2-10) | |
| Mechanical Ventilation Duration (Days, 25th-75th) | 1 (0-6) | |
| Non-invasive Mechanical Ventilation Duration (Days, 25th-75th) | 2 (1-5) | |
| LOS ICU (Days, 25th-75th) | 6 (2-12) | |
| Total Hospitalization Duration (Days, 25th-75th) | 11 (5-18) | |
| Culture Positivity (%) | 78 (37.3) | |
| Comorbidities | N, % | |
| Diabetes Mellitus | 66 (31.6) | |
| Hypertension | 86 (41.1) | |
| Coronary Arterial Disease | 24 (11.5) | |
| Congestive Heart Failure | 14 (6.7) | |
| Pulmonary Thromboembolism | 10 (4.8) | |
| Cerebrovascular Event | 7 (3.3) | |
| Chronic Renal Failure | 3 (1.4) | |
| Nutritional and Respiratory Support | N, % | |
| Total Parenteral Support Requirement | 20 (9.6) | |
| Enteral Support Requirement | 110 (52.6) | |
| Treatment Modalities and Overall Mortality | N, % | |
| Favipiravir | 102 (48.8) | |
| Tocilizumab | 18 (8.6) | |
| Nephrotoxic Antibiotic Therapy | 47 (22.5) | |
| Inotropic Support Requirement | 124 (59.3) | |
| Intravenous Glucocorticoid Requirement (Low) | 81 (38.8) | |
| Intravenous Glucocorticoid Requirement (High) | 112 (53.6) | |
| Mortality in ICU | 124 (59.3) | |
|
SD: Standard deviation, RT-PCR: Reverse transcription polymerase chain reaction The definition of non-invasive mechanical ventilation includes high flow oxygenation requirements and continuous and bilevel pressure-support ventilation modes. The definition of culture positivity includes any positive bacterial and viral result taken from a patient upon intensive care unit admission, regardless of sampling origin. Any glucocorticoid regimen that includes a dosage of more than 1 mg/kg equivalent of methylprednisolone was defined as a high-dosage regimen. | ||
| Paired Samples T-Test | Sampling Time | Mean | SD | t | dF | p |
|---|---|---|---|---|---|---|
| Sodium (mEq/L) | Admission | 139.44 | 6.80 | -5.057 | 208 | 0.001 |
| Last Evaluation | 142.41 | 8.69 | ||||
| Hemoglobin (g/dL) | Admission | 13.03 | 2.81 | 7.669 | 176 | 0.001 |
| Last Evaluation | 12.05 | 2.92 | ||||
| White Blood Cell (109/L) | Admission | 13.33 | 7.09 | -3.125 | 176 | 0.002 |
| Last Evaluation | 15.06 | 7.86 | ||||
| Lymphocyte (109/L) | Admission | 0.96 | 1.71 | -3.906 | 176 | 0.001 |
| Last Evaluation | 1.25 | 1.91 | ||||
| Neutrophil (109/L) | Admission | 12.51 | 9.19 | -1.229 | 176 | 0.221 |
| Last Evaluation | 13.38 | 7.39 | ||||
| Platelets (109/L) | Admission | 258.23 | 119.17 | 4.618 | 176 | 0.001 |
| Last Evaluation | 218.51 | 128.36 | ||||
| Ferritin (ng/ml) | Admission | 844.93 | 569.92 | -1.516 | 176 | 0.131 |
| Last Evaluation | 898.79 | 588.70 | ||||
| D-Dimer (mg/L) | Admission | 8.793 | 24.663 | 0.635 | 172 | 0.526 |
| Last Evaluation | 7.680 | 11.169 | ||||
| Procalcitonin (ng/ml) | Admission | 3.8 | 13.5 | -0.767 | 170 | 0.444 |
| Last Evaluation | 4.8 | 15.6 | ||||
| Creatinine Kinase (U/L) | Admission | 196.07 | 287.86 | -1.427 | 166 | 0.155 |
| Last Evaluation | 349.61 | 1410.65 | ||||
| LDH (U/L) | Admission | 612.72 | 677.78 | -2.597 | 174 | 0.010 |
| Last Evaluation | 877.96 | 1496.24 | ||||
| Glomerular Filtration Rate | Admission | 70.48 | 29.25 | 1.128 | 175 | 0.261 |
| Last Evaluation | 68.33 | 35.19 | ||||
| Creatinine (mg/dL) | Admission | 1.17 | 0.83 | -3.257 | 174 | 0.001 |
| Last Evaluation | 1.43 | 1.27 | ||||
| Potassium (mEq/L) | Admission | 4.34 | 0.71 | 0.703 | 175 | 0.483 |
| Last Evaluation | 4.29 | 0.99 | ||||
| AST (U/L) | Admission | 141.68 | 671.52 | -2.061 | 175 | 0.041 |
| Last Evaluation | 308.71 | 943.93 | ||||
| ALT (U/L) | Admission | 109.11 | 567.28 | -2.086 | 173 | 0.038 |
| Last Evaluation | 189.24 | 641.40 | ||||
| C-Reactive Protein (mg/L) | Admission | 127.22 | 86.89 | 5.112 | 174 | 0.001 |
| Last Evaluation | 92.30 | 82.57 | ||||
| Albumin (g/L) | Admission | 28.30 | 5.23 | 1.600 | 169 | 0.112 |
| Last Evaluation | 26.74 | 13.09 | ||||
| Sedimentation | Admission | 60.44 | 29.04 | 3.773 | 155 | 0.001 |
| Last Evaluation | 50.47 | 33.00 | ||||
|
SD: Standard deviation, LDH: Lactate dehydrogenase, AST: Aspartate aminotransferase, ALT: Alanine transaminase The last evaluation time period includes the final testing performed before intensive care discharge or the last testing performed before exit. | ||||||
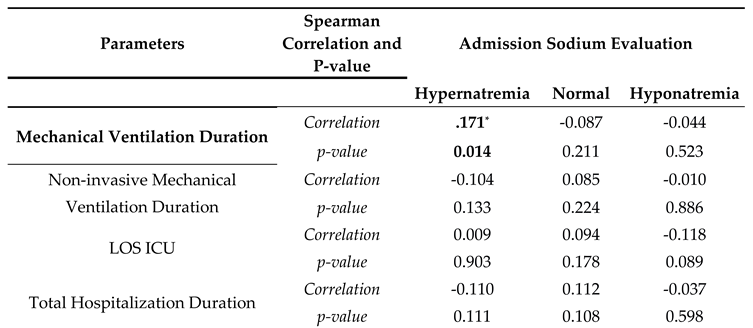
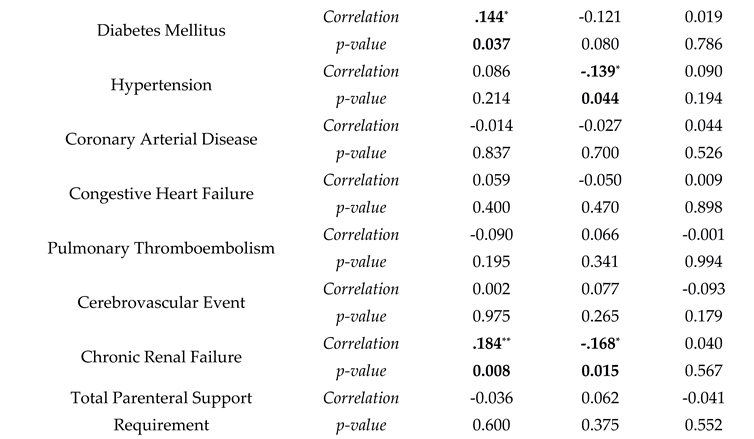
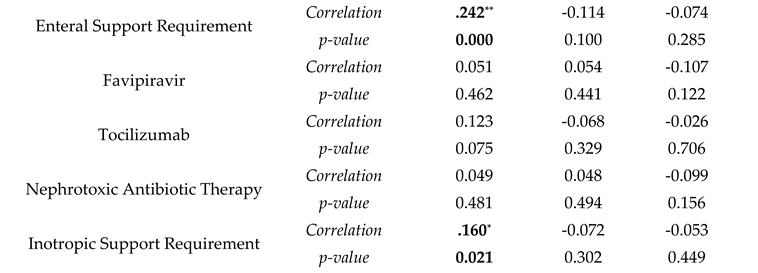
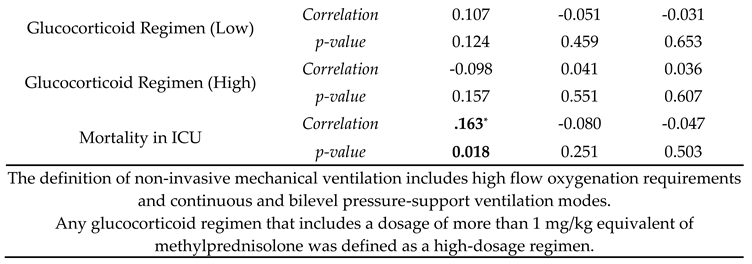
| Mortality in ICU | B | SE | Wald | Odds Ratio | p-value |
|---|---|---|---|---|---|
| Constant | -7.213 | 5.885 | 1.502 | ||
| Age | 0.052 | 0.026 | 3.959 | 1.053 | 0.047 |
| Body Mass Index | 0.211 | 0.093 | 5.214 | 1.235 | 0.022 |
| Gender | -1.375 | 0.643 | 4.581 | 0.253 | 0.032 |
| Total Parenteral Support Requirement | 2.222 | 1.292 | 2.960 | 9.226 | 0.085 |
| Enteral Support Requirement | 2.293 | 0.694 | 10.923 | 9.906 | 0.001 |
| MV Duration | 0.135 | 0.233 | 0.334 | 1.144 | 0.563 |
| NIMV Duration | 0.040 | 0.236 | 0.028 | 1.040 | 0.867 |
| Intensive Care Admission Duration | -0.128 | 0.229 | 0.311 | 0.880 | 0.577 |
| Diabetes Mellitus | 0.168 | 0.717 | 0.055 | 1.183 | 0.815 |
| Hypertension | 0.036 | 0.608 | 0.003 | 1.036 | 0.953 |
| Coronary Arterial Disease | 1.312 | 0.897 | 2.142 | 3.714 | 0.143 |
| Congestive Heart Failure | 0.354 | 1.061 | 0.111 | 1.424 | 0.739 |
| Pulmonary Thromboembolism | 0.280 | 1.431 | 0.038 | 1.322 | 0.845 |
| Cerebrovascular Event | -2.934 | 1.976 | 2.204 | 0.053 | 0.138 |
| Glucocorticoid Requirement (Low) | -2.080 | 1.230 | 2.860 | 0.125 | 0.091 |
| Glucocorticoid Requirement (High) | -1.454 | 1.231 | 1.395 | 0.234 | 0.238 |
| Favipiravir | 0.382 | 0.581 | 0.431 | 1.465 | 0.512 |
| Hypernatremia | 0.882 | 0.923 | 0.914 | 2.416 | 0.339 |
| Hemoglobin | -0.129 | 0.092 | 1.986 | 0.879 | 0.159 |
| White Blood Cell | -0.020 | 0.051 | 0.160 | 0.980 | 0.689 |
| Platelets | -0.001 | 0.003 | 0.275 | 0.999 | 0.600 |
| Ferritin | 0.001 | 0.001 | 4.746 | 1.001 | 0.029 |
| D-Dimer | 0.031 | 0.021 | 2.175 | 1.032 | 0.140 |
| Procalcitonin | -0.002 | 0.025 | 0.006 | 0.998 | 0.938 |
| C-Reactive Protein | 0.009 | 0.004 | 6.422 | 1.009 | 0.011 |
| Sedimentation | -0.011 | 0.010 | 1.202 | 0.989 | 0.273 |
| AST | -0.007 | 0.004 | 2.248 | 0.993 | 0.134 |
| ALT | 0.010 | 0.008 | 1.588 | 1.011 | 0.208 |
|
SE: Standard Error, MV: Mechanical Ventilation, NIMV: Non-invasive mechanical ventilation AST: Aspartate aminotransferase, ALT: Alanine transaminase. | |||||
| Enteral Support Requirement | B | SE | Wald | Odds Ratio | p-value |
|---|---|---|---|---|---|
| Constant | -25.917 | 11.194 | 5.360 | ||
| Age | 0.061 | 0.050 | 1.497 | 1.062 | 0.221 |
| Body Mass Index | 0.283 | 0.156 | 3.293 | 1.328 | 0.070 |
| Gender | -1.005 | 1.132 | 0.789 | 0.366 | 0.375 |
| NIMV Duration | -1.704 | 0.369 | 21.291 | 0.182 | 0.001 |
| Intensive Care Admission Duration | 1.850 | 0.412 | 20.204 | 6.359 | 0.001 |
| Diabetes Mellitus | 0.431 | 1.009 | 0.183 | 1.539 | 0.669 |
| Hypertension | 0.995 | 1.247 | 0.637 | 2.704 | 0.425 |
| Coronary Arterial Disease | 2.442 | 1.497 | 2.660 | 11.499 | 0.103 |
| Congestive Heart Failure | 3.891 | 2.235 | 3.031 | 48.946 | 0.082 |
| Pulmonary Thromboembolism | 0.084 | 2.367 | 0.001 | 1.087 | 0.972 |
| Cerebrovascular Event | -2.799 | 2.456 | 1.299 | 0.061 | 0.254 |
| Glucocorticoid Requirement (Low) | -1.554 | 2.366 | 0.431 | 0.211 | 0.511 |
| Glucocorticoid Requirement (High) | 1.137 | 2.249 | 0.255 | 3.116 | 0.613 |
| Favipiravir | 0.619 | 1.058 | 0.342 | 1.857 | 0.559 |
| Hypernatremia | 4.153 | 1.923 | 4.662 | 63.602 | 0.031 |
| Hemoglobin | -0.017 | 0.165 | 0.010 | 0.984 | 0.920 |
| White Blood Cell | 0.026 | 0.118 | 0.049 | 1.026 | 0.825 |
| Platelets | -0.008 | 0.006 | 1.727 | 0.992 | 0.189 |
| Ferritin | 0.001 | 0.001 | 2.581 | 1.001 | 0.108 |
| D-Dimer | 0.009 | 0.010 | 0.881 | 1.010 | 0.348 |
| Procalcitonin | -0.009 | 0.038 | 0.059 | 0.991 | 0.808 |
| C-Reactive Protein | 0.004 | 0.005 | 0.478 | 1.004 | 0.489 |
| Sedimentation | 0.002 | 0.016 | 0.018 | 1.002 | 0.894 |
| AST | -0.001 | 0.010 | 0.006 | 0.999 | 0.938 |
| ALT | 0.011 | 0.020 | 0.337 | 1.011 | 0.562 |
| SE: Standard Error, MV: Mechanical Ventilation, NIMV: Non-invasive mechanical ventilation AST: Aspartate aminotransferase, ALT: Alanine transaminase. | |||||
| Inotropic Support Requirement | B | SE | Wald | Odds Ratio | p-value |
|---|---|---|---|---|---|
| Constant | -6.640 | 5.954 | 1.244 | ||
| Age | 0.054 | 0.028 | 3.815 | 1.055 | 0.051 |
| Body Mass Index | 0.226 | 0.098 | 5.267 | 1.253 | 0.022 |
| Gender | -1.667 | 0.705 | 5.585 | 0.189 | 0.018 |
| Total Parenteral Support Requirement | 2.710 | 1.404 | 3.727 | 15.033 | 0.054 |
| Enteral Support Requirement | 3.128 | 0.815 | 14.738 | 22.832 | 0.001 |
| MV Duration | 1.232 | 0.575 | 4.593 | 3.429 | 0.032 |
| NIMV Duration | 1.124 | 0.564 | 3.967 | 3.076 | 0.046 |
| Intensive Care Admission Duration | -1.230 | 0.569 | 4.664 | 0.292 | 0.031 |
| Diabetes Mellitus | -0.072 | 0.758 | 0.009 | 0.930 | 0.924 |
| Hypertension | -0.173 | 0.659 | 0.069 | 0.841 | 0.793 |
| Coronary Arterial Disease | 1.154 | 0.935 | 1.525 | 3.172 | 0.217 |
| Congestive Heart Failure | -0.019 | 1.125 | 0.000 | 0.981 | 0.987 |
| Pulmonary Thromboembolism | -0.030 | 1.533 | 0.000 | 0.971 | 0.985 |
| Cerebrovascular Event | -3.651 | 2.086 | 3.062 | 0.026 | 0.080 |
| Glucocorticoid Requirement (Low) | -2.244 | 1.232 | 3.317 | 0.106 | 0.069 |
| Glucocorticoid Requirement (High) | -1.835 | 1.239 | 2.193 | 0.160 | 0.139 |
| Favipiravir | 0.460 | 0.625 | 0.541 | 1.584 | 0.462 |
| Hypernatremia | 0.785 | 0.968 | 0.659 | 2.193 | 0.417 |
| Hemoglobin | -0.114 | 0.095 | 1.427 | 0.892 | 0.232 |
| White Blood Cell | -0.060 | 0.057 | 1.128 | 0.941 | 0.288 |
| Platelets | 0.002 | 0.003 | 0.246 | 1.002 | 0.620 |
| Ferritin | 0.002 | 0.001 | 7.481 | 1.002 | 0.006 |
| D-Dimer | 0.029 | 0.021 | 1.870 | 1.029 | 0.172 |
| Procalcitonin | -0.017 | 0.024 | 0.531 | 0.983 | 0.466 |
| C-Reactive Protein | 0.009 | 0.004 | 5.879 | 1.009 | 0.015 |
| Sedimentation | -0.006 | 0.011 | 0.314 | 0.994 | 0.575 |
| AST | -0.005 | 0.005 | 1.230 | 0.995 | 0.267 |
| ALT | 0.007 | 0.009 | 0.669 | 1.007 | 0.413 |
| SE: Standard Error, MV: Mechanical Ventilation, NIMV: Non-invasive mechanical ventilation AST: Aspartate aminotransferase, ALT: Alanine transaminase. | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




