Submitted:
20 May 2024
Posted:
20 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Understanding the Effects of Covid-19 on CKD, Dialysis and Kidney Transplantation
COVID-19 Mortality in ESKD, Dialysis and Kidney Transplantation
Impact of CKD on COVID-19 Risks and Outcomes
Kidney Complications in CKD and COVID-19 Outcomes
COVID-19 Risks and Considerations for Dialysis Patients
COVID-19 and Arterial and Venous Thromboembolic Complications
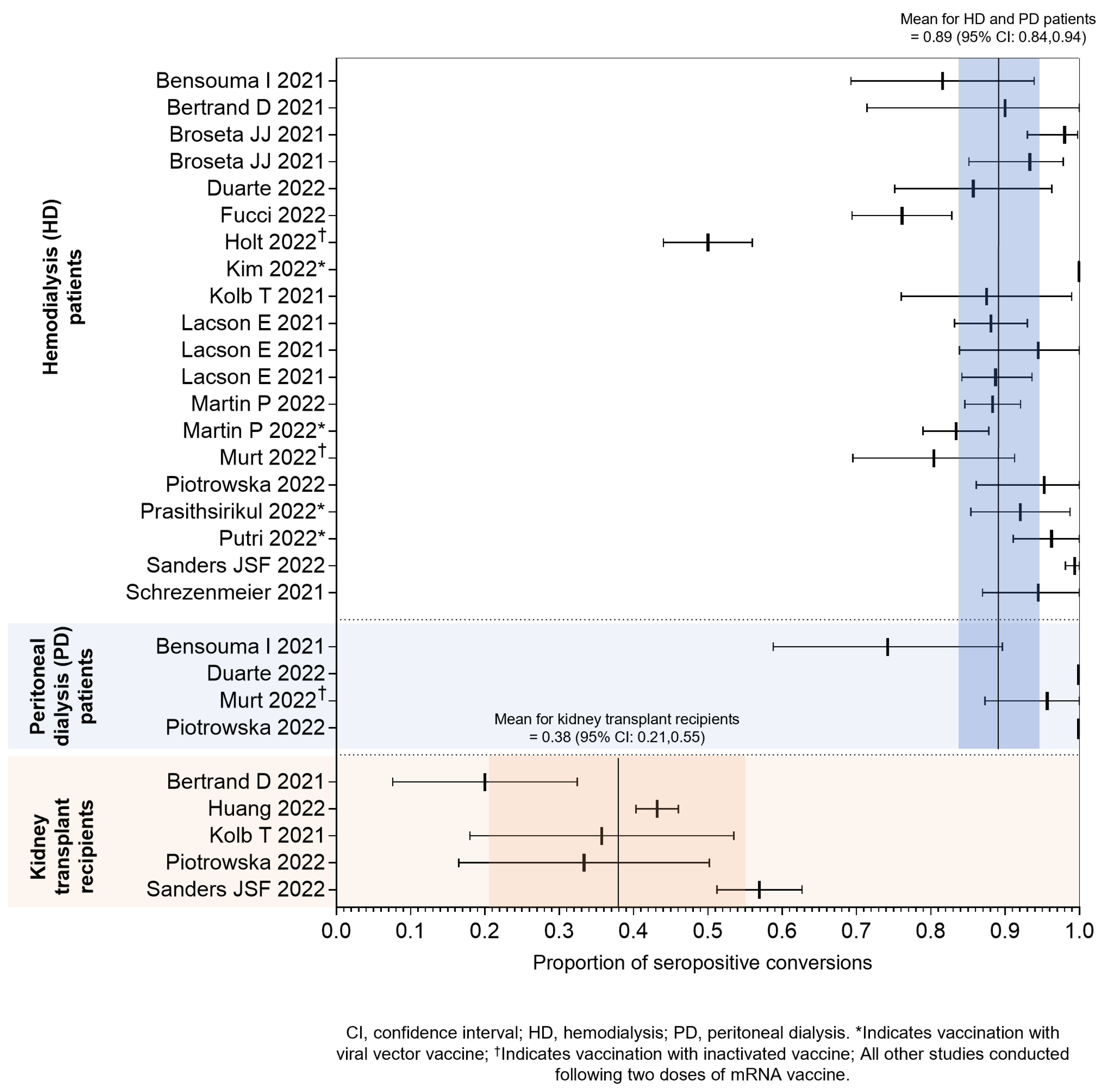
Effectiveness of COVID-19 Vaccination in IC and is Patients
Illustrative Cases
Patient 1
Patient 2
Protecting CKD Patients and KTRS from Covid-19 Infection and Severe Disease
Vaccination and Booster Doses for Effective COVID-19 Prevention
Non-Pharmacological Measures in COVID-19 Prevention
Therapeutic Strategies for COVID-19 Management
Monoclonal Antibodies for COVID-19 Prophylaxis
Monoclonal Antibodies for COVID-19 Treatment
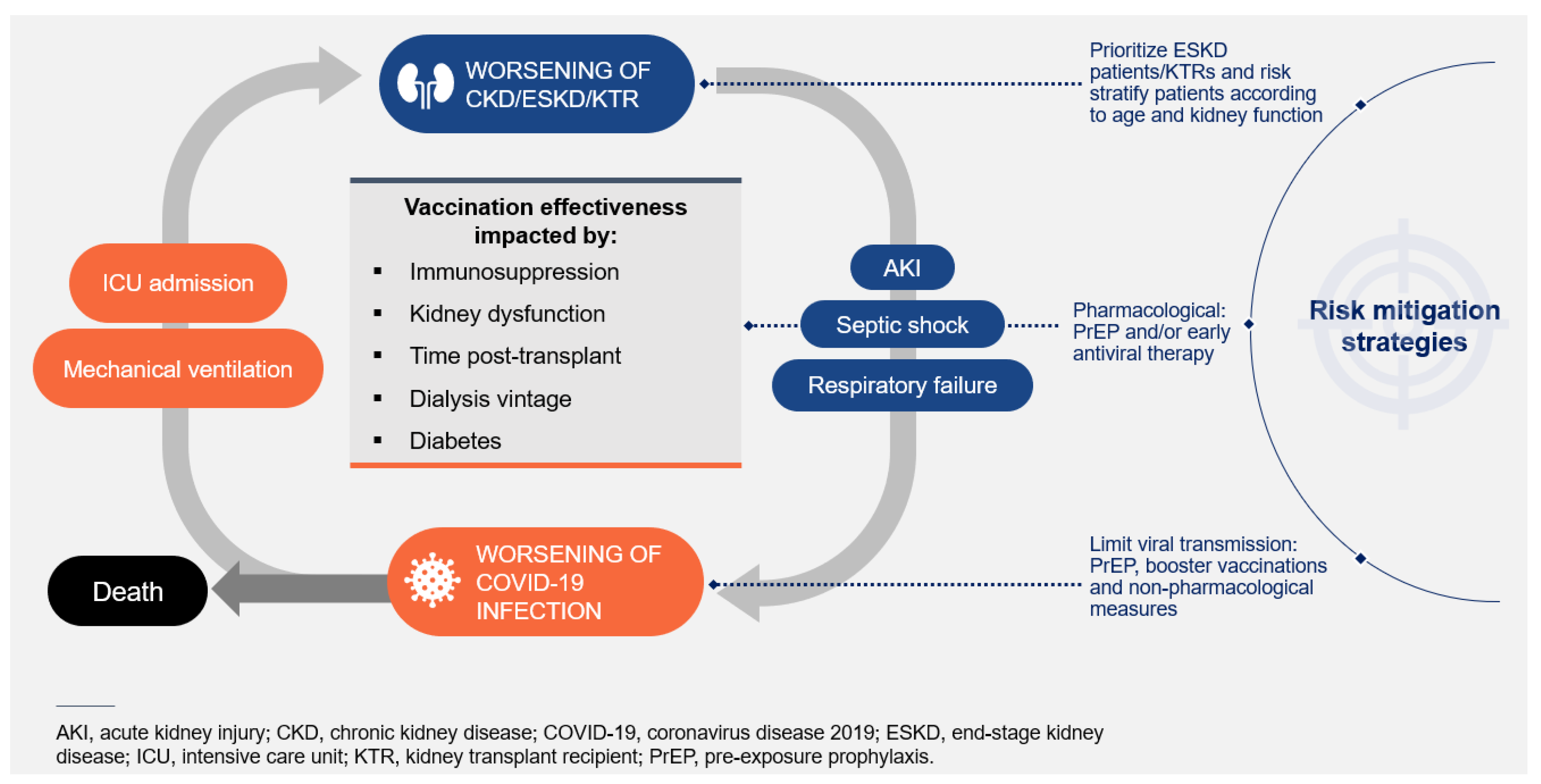
Conclusions and Clinical Implications
Funding
Declaration of Interest
Acknowledgment
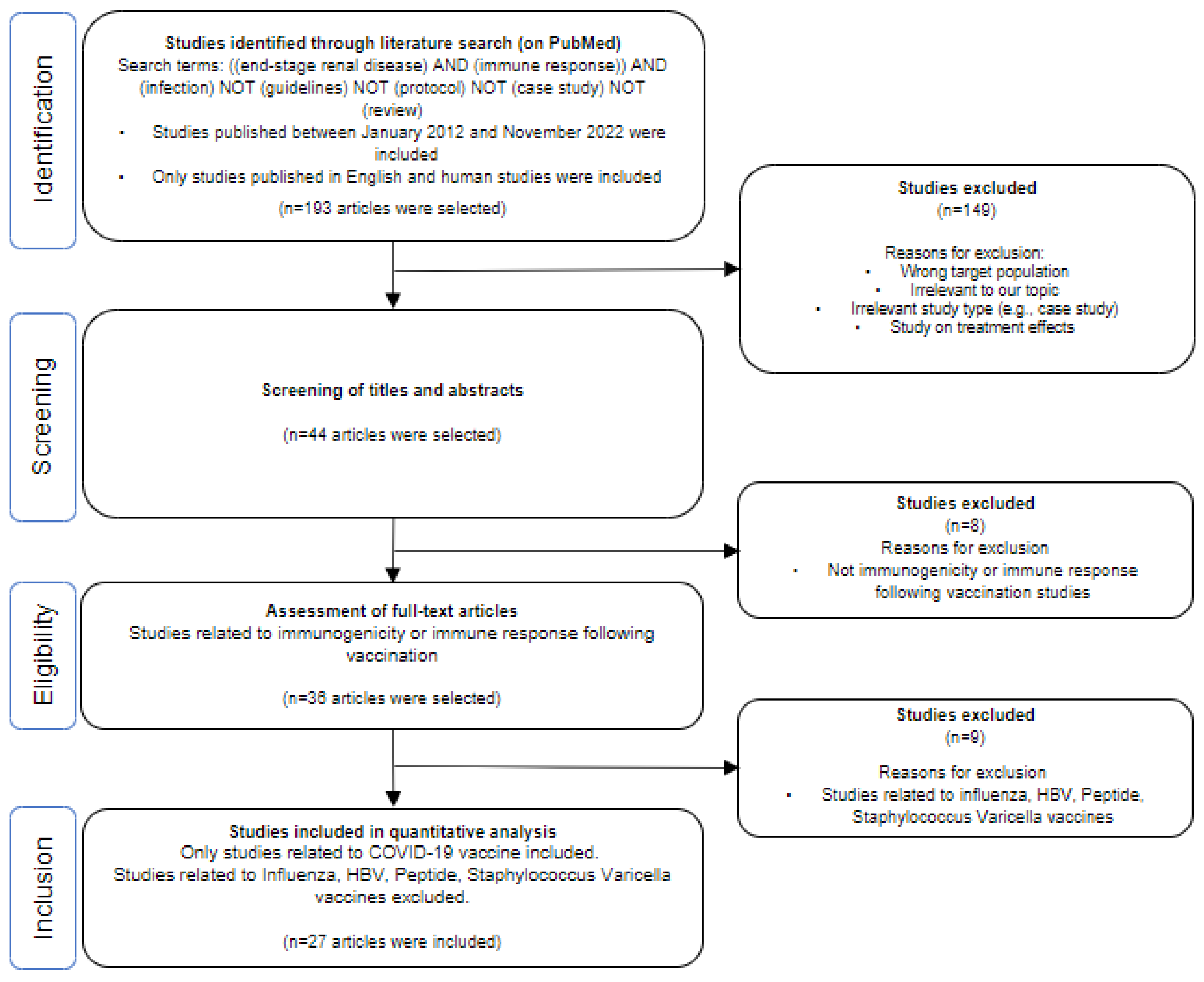
Appendix A: Flow Diagram Describing Systematic Selection of Studies on COVID-19 Vaccination Response for Literature Review
References
- Nejad, M.-M.M.; Moosaie, F.; Dehghanbanadaki, H.; Ghadery, A.H.; Shabani, M.; Tabary, M.; Aryannejad, A.; SeyedAlinaghi, S.; Rezaei, N. Immunogenicity of COVID-19 mRNA vaccines in immunocompromised patients: a systematic review and meta-analysis. Eur. J. Med Res. 2022, 27, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chavarot, N.; Gueguen, J.; Bonnet, G.; Jdidou, M.; Trimaille, A.; Burger, C.; Amrouche, L.; Weizman, O.; Pommier, T.; Aubert, O.; et al. COVID-19 severity in kidney transplant recipients is similar to nontransplant patients with similar comorbidities. Am. J. Transplant. 2020, 21, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Y.; Flahault, A.; Chavarot, N.; Melenotte, C.; Cheminant, M.; Deschamps, P.; Carlier, N.; Lafont, E.; Thomas, M.; Flamarion, E.; et al. Pre-exposure prophylaxis with tixagevimab and cilgavimab (Evusheld) for COVID-19 among 1112 severely immunocompromised patients. Clin. Microbiol. Infect. 2022, 28, 1654.e1–1654.e4. [Google Scholar] [CrossRef]
- Jager, K.J.; Kramer, A.; Chesnaye, N.C.; Couchoud, C.; Sánchez-Álvarez, J.E.; Garneata, L.; Collart, F.; Hemmelder, M.H.; Ambühl, P.; Kerschbaum, J.; et al. Results from the ERA-EDTA Registry indicate a high mortality due to COVID-19 in dialysis patients and kidney transplant recipients across Europe. Kidney Int. 2020, 98, 1540–1548. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Clarke CL, Prendecki M, Dhutia A; et al. Longevity of SARS-CoV-2 immune responses in hemodialysis patients and protection against reinfection. Kidney Int 2021, 99, 1470–1477. [Google Scholar] [CrossRef]
- De Meester, J.; De Bacquer, D.; Naesens, M.; Meijers, B.; Couttenye, M.M.; De Vriese, A.S. ; for the NBVN Kidney Registry Group Incidence, Characteristics, and Outcome of COVID-19 in Adults on Kidney Replacement Therapy: A Regionwide Registry Study. J. Am. Soc. Nephrol. 2020, 32, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Pulliam JRC, van Schalkwyk C, Govender N; et al. Increased risk of SARS-CoV-2 reinfection associated with emergence of Omicron in South Africa. Science 2022, 376, eabn4947. [Google Scholar] [CrossRef] [PubMed]
- SARS-CoV-2 variants of concern as of 19 January 2024. [cited 2024 Jan 24]. Available at: https://www.ecdc.europa.eu/en/covid-19/variants-concern.
- El Karoui, K.; Hourmant, M.; Ayav, C.; Glowacki, F.; Couchoud, C.; Lapidus, N.; Registry, E. Vaccination and COVID-19 Dynamics in Dialysis Patients. Clin. J. Am. Soc. Nephrol. 2022, 17, 395–402. [Google Scholar] [CrossRef]
- Cancarevic, I.; Nassar, M.; Daoud, A.; Ali, H.; Nso, N.; Sanchez, A.; Parikh, A.; Hosna, A.U.; Devanabanda, B.; Ahmed, N.; et al. Mortality rate of COVID-19 infection in end stage kidney disease patients on maintenance hemodialysis: A systematic review and meta-analysis. World J. Virol. 2022, 11, 352–361. [Google Scholar] [CrossRef]
- Sanders, J.-S.F.; Bemelman, F.J.; Messchendorp, A.L.; Baan, C.C.; van Baarle, D.; van Binnendijk, R.; Diavatopoulos, D.A.; Frolke, S.C.; Geers, D.; GeurtsvanKessel, C.H.; et al. The RECOVAC Immune-response Study: The Immunogenicity, Tolerability, and Safety of COVID-19 Vaccination in Patients With Chronic Kidney Disease, on Dialysis, or Living With a Kidney Transplant. Transplantation 2022, 106, 821–834. [Google Scholar] [CrossRef]
- Hilbrands, L.B.; Duivenvoorden, R.; Vart, P.; Franssen, C.F.M.; Hemmelder, M.H.; Jager, K.J.; Kieneker, L.M.; Noordzij, M.; Pena, M.J.; de Vries, H.; et al. COVID-19-related mortality in kidney transplant and dialysis patients: results of the ERACODA collaboration. Nephrol. Dial. Transplant. 2020, 35, 1973–1983. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Garcia, I.; Perales-Fraile, I.; Gonzalez-Garcia, A.; Munoz-Blanco, A.; Manzano, L.; Fabregate, M.; Diez-Manglano, J.; Aizpuru, E.F.; Fernandez, F.A.; Garcia, A.G.; et al. In-hospital mortality among immunosuppressed patients with COVID-19: Analysis from a national cohort in Spain. PLOS ONE 2021, 16, e0255524. [Google Scholar] [CrossRef] [PubMed]
- Flythe, J.E.; Assimon, M.M.; Tugman, M.J.; Chang, E.H.; Gupta, S.; Sosa, M.A.; Renaghan, A.D.; Melamed, M.L.; Wilson, F.P.; Neyra, J.A.; et al. Characteristics and Outcomes of Individuals With Pre-existing Kidney Disease and COVID-19 Admitted to Intensive Care Units in the United States. Am. J. Kidney Dis. 2021, 77, 190–203. [Google Scholar] [CrossRef]
- Valeri, A.M.; Robbins-Juarez, S.Y.; Stevens, J.S.; Ahn, W.; Rao, M.K.; Radhakrishnan, J.; Gharavi, A.G.; Mohan, S.; Husain, S.A. Presentation and Outcomes of Patients with ESKD and COVID-19. J. Am. Soc. Nephrol. 2020, 31, 1409–1415. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.M.; Weiner, D.E.; Aweh, G.; Salenger, P.; Johnson, D.S.; Lacson, E. Epidemiology and Outcomes of COVID-19 in Home Dialysis Patients Compared with In-Center Dialysis Patients. J. Am. Soc. Nephrol. 2021, 32, 1569–1573. [Google Scholar] [CrossRef]
- Alberici F, Delbarba E, Manenti C; et al. A single center observational study of the clinical characteristics and short-term outcome of 20 kidney transplant patients admitted for SARS-CoV2 pneumonia. Kidney Int 2020, 97, 1083–1088. [Google Scholar] [CrossRef]
- Tannor, E.K.; Bajpai, D.; Nlandu, Y.M.; Wijewickrama, E. COVID-19 and Kidney Disease: Progress in Health Inequity From Low-Income Settings. Semin. Nephrol. 2022, 42, 151318. [Google Scholar] [CrossRef]
- Chou C-Y, Wang S-M, Liang C-C; et al. Risk of pneumonia among patients with chronic kidney disease in outpatient and inpatient settings: A nationwide population-based study. Medicine (Baltimore) 2014, 93, e174. [Google Scholar] [CrossRef]
- Sato, R.; Matsuzawa, Y.; Ogawa, H.; Kimura, K.; Tsuboi, N.; Yokoo, T.; Okada, H.; Konishi, M.; Kirigaya, J.; Fukui, K.; et al. Chronic kidney disease and clinical outcomes in patients with COVID-19 in Japan. Clin. Exp. Nephrol. 2022, 26, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Mohamed MMB, Velez JCQ. Proteinuria in COVID-19. Clin Kidney J 2021, 14, i40–i47.
- Mahalingasivam, V.; Su, G.; Iwagami, M.; Davids, M.R.; Wetmore, J.B.; Nitsch, D. COVID-19 and kidney disease: insights from epidemiology to inform clinical practice. Nat. Rev. Nephrol. 2022, 18, 485–498. [Google Scholar] [CrossRef]
- Brogan, M.; Ross, M.J. The Impact of Chronic Kidney Disease on Outcomes of Patients with COVID-19 Admitted to the Intensive Care Unit. Nephron 2021, 146, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Hayek, S.S.; Wang, W.; Chan, L.; Mathews, K.S.; Melamed, M.L.; Brenner, S.K.; Leonberg-Yoo, A.; Schenck, E.J.; Radbel, J.; et al. Factors Associated With Death in Critically Ill Patients With Coronavirus Disease 2019 in the US. JAMA Intern. Med. 2020, 180, 1436–1447. [Google Scholar] [CrossRef] [PubMed]
- Geetha, D.; Kronbichler, A.; Rutter, M.; Bajpai, D.; Menez, S.; Weissenbacher, A.; Anand, S.; Lin, E.; Carlson, N.; Sozio, S.; et al. Impact of the COVID-19 pandemic on the kidney community: lessons learned and future directions. Nat. Rev. Nephrol. 2022, 18, 724–737. [Google Scholar] [CrossRef] [PubMed]
- Behlul, S.; Ozdal, M.A. Risk of COVID-19 and Cost Burden in End-Stage Renal Disease Patients and Policy Implications for Managing Nephrology Services during the COVID-19 Pandemic. Healthcare 2022, 10, 2351. [Google Scholar] [CrossRef] [PubMed]
- Hemmelder, M.H.; Noordzij, M.; Vart, P.; Hilbrands, L.B.; Jager, K.J.; Abrahams, A.C.; Arroyo, D.; Battaglia, Y.; Ekart, R.; Mallamaci, F.; et al. Recovery of dialysis patients with COVID-19: health outcomes 3 months after diagnosis in ERACODA. Nephrol. Dial. Transplant. 2022, 37, 1140–1151. [Google Scholar] [CrossRef] [PubMed]
- Impact of COVID-19 on the mortality of dialysis patients and kidney transplant recipients during the Omicron fifth wave in Hong Kong. HKMJ.. [cited 2024 Jan 25]. Available at: https://www.hkmj.org/abstracts/v29n1/82.htm.
- Pan, X.W.; Xu, D.; Zhang, H.; Zhou, W.; Wang, L.-H.; Cui, X.G. Identification of a potential mechanism of acute kidney injury during the COVID-19 outbreak: a study based on single-cell transcriptome analysis. Intensive Care Med. 2020, 46, 1114–1116. [Google Scholar] [CrossRef]
- Su, H.; Yang, M.; Wan, C.; Yi, L.-X.; Tang, F.; Zhu, H.-Y.; Yi, F.; Yang, H.-C.; Fogo, A.B.; Nie, X.; et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020, 98, 219–227. [Google Scholar] [CrossRef]
- Galmiche, S.; Nguyen, L.B.L.; Tartour, E.; de Lamballerie, X.; Wittkop, L.; Loubet, P.; Launay, O. Immunological and clinical efficacy of COVID-19 vaccines in immunocompromised populations: a systematic review. Clin. Microbiol. Infect. 2022, 28, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Bertrand D, Hamzaoui M, Lemée V; et al. Antibody and T Cell Response to SARS-CoV-2 Messenger RNA BNT162b2 Vaccine in Kidney Transplant Recipients and Hemodialysis Patients. J Am Soc Nephrol 2021, 32, 2147–2152. [Google Scholar] [CrossRef] [PubMed]
- Cucchiari D, Egri N, Bodro M; et al. Cellular and humoral response after MRNA-1273 SARS-CoV-2 vaccine in kidney transplant recipients. Am J Transplant 2021, 21, 2727–2739. [Google Scholar] [CrossRef] [PubMed]
- Rozen-Zvi B, Yahav D, Agur T; et al. Antibody response to SARS-CoV-2 mRNA vaccine among kidney transplant recipients: A prospective cohort study. Clin Microbiol Infect 2021, 27, 1173–e1. [Google Scholar]
- Stumpf J, Siepmann T, Lindner T; et al. Humoral and cellular immunity to SARS-CoV-2 vaccination in renal transplant versus dialysis patients: A prospective, multicenter observational study using mRNA-1273 or BNT162b2 mRNA vaccine. Lancet Reg Health Eur 2021, 9, 100178. [Google Scholar] [CrossRef] [PubMed]
- See, K.C. Vaccination for the Prevention of Infection among Immunocompromised Patients: A Concise Review of Recent Systematic Reviews. Vaccines 2022, 10, 800. [Google Scholar] [CrossRef] [PubMed]
- Tenforde, M.W.; Self, W.H.; Adams, K.; Gaglani, M.; Ginde, A.A.; McNeal, T.; Ghamande, S.; Douin, D.J.; Talbot, H.K.; Casey, J.D.; et al. Association Between mRNA Vaccination and COVID-19 Hospitalization and Disease Severity. Jama-Journal Am. Med Assoc. 2021, 326, 2043–2054. [Google Scholar] [CrossRef]
- Falahi, S.; Sayyadi, H.; Kenarkoohi, A. Immunogenicity of COVID-19 mRNA vaccines in hemodialysis patients: Systematic review and meta-analysis. Heal. Sci. Rep. 2022, 5, e874. [Google Scholar] [CrossRef] [PubMed]
- Intawong, K.; Chariyalertsak, S.; Chalom, K.; Wonghirundecha, T.; Kowatcharakul, W.; Thongprachum, A.; Chotirosniramit, N.; Noppakun, K.; Khwanngern, K.; Teacharak, W.; et al. Waning vaccine response to severe COVID-19 outcomes during omicron predominance in Thailand. PLOS ONE 2023, 18, e0284130. [Google Scholar] [CrossRef]
- COVID-19 Related Infection Prevention for the Immunocompromised Host - ISID. [cited 2024 Jan 25]. Available at: https://isid.org/guide/pathogens/covid-19-infection-prevention/. https://isid.org/guide/pathogens/covid-19-infection-prevention/.
- Gruell H, Vanshylla K, Tober-Lau P; et al. mRNA booster immunization elicits potent neutralizing serum activity against the SARS-CoV-2 Omicron variant. Nat Med 2022, 28, 477–480. [Google Scholar] [CrossRef]
- Pérez-Then E, Lucas C, Monteiro VS; et al. Neutralizing antibodies against the SARS-CoV-2 Delta and Omicron variants following heterologous CoronaVac plus BNT162b2 booster vaccination. Nat Med 2022, 28, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Thammathiwat, T.; Tungsanga, S.; Tiankanon, K.; Torvorapanit, P.; Chumpangern, W.; Udomkarnjananun, S.; Avihingsanon, Y.; Sriprasart, T.; Srisawat, N.; Jutivorakool, K.; et al. A case of successful treatment of severe COVID-19 pneumonia with favipiravir and tocilizumab in post–kidney transplant recipient. Transpl. Infect. Dis. 2020, 23, e13388. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lin, H.; Wu, Y.; Fang, Y.; Kumar, R.; Chen, G.; Lin, S. COVID-19 in posttransplant patients—report of 2 cases. Am. J. Transplant. 2020, 20, 1879–1881. [Google Scholar] [CrossRef] [PubMed]
- Hilhorst, M.; Bemelman, F.J.; Bruchfeld, A.; Fernandez-Juarez, G.M.; Floege, J.; Frangou, E.; Goumenos, D.; van Kooten, C.; Kronbichler, A.; I Stevens, K.; et al. Prophylactic and early outpatient treatment of COVID-19 in patients with kidney disease: considerations from the Immunonephrology Working Group of the European Renal Association (ERA-IWG). Nephrol. Dial. Transplant. 2023, 38, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Hus, I.; Szymczyk, A.; Mańko, J.; Drozd-Sokołowska, J. COVID-19 in Adult Patients with Hematological Malignancies—Lessons Learned after Three Years of Pandemic. Biology 2023, 12, 545. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, MW. Outpatient treatment options to address the SARS-CoV-2 variant Omicron. Expert Rev Anti Infect Ther 2022, 20, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.J.; Ustianowski, A.; De Wit, S.; Launay, O.; Avila, M.; Templeton, A.; Yuan, Y.; Seegobin, S.; Ellery, A.; Levinson, D.J.; et al. Intramuscular AZD7442 (Tixagevimab–Cilgavimab) for Prevention of Covid-19. New Engl. J. Med. 2022, 386, 2188–2200. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, A.; Akerman, A.; Milogiannakis, V.; Silva, M.R.; Walker, G.; Stella, A.O.; Kindinger, A.; Angelovich, T.; Waring, E.; Amatayakul-Chantler, S.; et al. SARS-CoV-2 Omicron BA.5: Evolving tropism and evasion of potent humoral responses and resistance to clinical immunotherapeutics relative to viral variants of concern. EBioMedicine 2022, 84, 104270. [Google Scholar] [CrossRef] [PubMed]
- Hirsch C, Park YS, Piechotta V; et al. SARS-CoV-2-neutralising monoclonal antibodies to prevent COVID-19. Cochrane Database Syst Rev 2022, 6, CD014945. [Google Scholar]
- Focosi, D.; Casadevall, A. A Critical Analysis of the Use of Cilgavimab plus Tixagevimab Monoclonal Antibody Cocktail (Evusheld™) for COVID-19 Prophylaxis and Treatment. Viruses 2022, 14, 1999. [Google Scholar] [CrossRef]
- Takashita, E.; Yamayoshi, S.; Simon, V.; Van Bakel, H.; Sordillo, E.M.; Pekosz, A.; Fukushi, S.; Suzuki, T.; Maeda, K.; Halfmann, P.; et al. Efficacy of Antibodies and Antiviral Drugs against Omicron BA.2.12.1, BA.4, and BA.5 Subvariants. New Engl. J. Med. 2022, 387, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Huygens, S.; Munnink, B.O.; Gharbharan, A.; Koopmans, M.; Rijnders, B. Sotrovimab Resistance and Viral Persistence After Treatment of Immunocompromised Patients Infected With the Severe Acute Respiratory Syndrome Coronavirus 2 Omicron Variant. Clin. Infect. Dis. 2022, 76, E507–E509. [Google Scholar] [CrossRef] [PubMed]
- Destras G, Bal A, Simon B, Lina B, Josset L. Sotrovimab drives SARS-CoV-2 omicron variant evolution in immunocompromised patients. Lancet Microbe 2022, 3, e559. [Google Scholar] [CrossRef] [PubMed]
- Tao K, Tzou PL, Kosakovsky Pond SL, Ioannidis JPA, Shafer RW. Susceptibility of SARS-CoV-2 Omicron Variants to Therapeutic Monoclonal Antibodies: Systematic Review and Meta-analysis. Microbiol Spectr 2022, 10, e0092622. [Google Scholar]
- Khan BA, Pagsinohin M, Lu LM, Tan P, Teo R. Tixagevimab and Cilgavimab Administration for Hemodialysis Patients at Community-Based Dialysis Centers in Singapore as Pre-Exposure Prophylaxis for SARS-CoV-2 Infection. Cureus 2023, 15, e41297. [Google Scholar]
- Yetmar, Z.A.; Beam, E.; O'Horo, J.C.; Seville, M.T.; Brumble, L.; Ganesh, R.; Razonable, R.R. Outcomes of bebtelovimab and sotrovimab treatment of solid organ transplant recipients with mild-to-moderate coronavirus disease 2019 during the Omicron epoch. Transpl. Infect. Dis. 2022, 24, e13901. [Google Scholar] [CrossRef] [PubMed]
- Bruel T, Hadjadj J, Maes P; et al. Serum neutralization of SARS-CoV-2 Omicron sublineages BA.1 and BA.2 in patients receiving monoclonal antibodies. Nat Med 2022, 28, 1297–1302. [Google Scholar] [CrossRef]
- Tai, Y.-L.; Lee, M.-D.; Chi, H.; Chiu, N.-C.; Lei, W.-T.; Weng, S.-L.; Liu, L.Y.-M.; Chen, C.-C.; Huang, S.-Y.; Huang, Y.-N.; et al. Effects of bamlanivimab alone or in combination with etesevimab on subsequent hospitalization and mortality in outpatients with COVID-19: a systematic review and meta-analysis. PeerJ 2023, 11, e15344. [Google Scholar] [CrossRef] [PubMed]
- Kijima, Y.; Shimizu, T.; Kato, S.; Kano, K.; Horiuchi, T.; Nozaki, T.; Omoto, K.; Inui, M.; Toma, H.; Iida, S.; et al. Case Report of COVID-19 Infection After Kidney Transplant Treated With Casirivimab-Imdevimab and Mycophenolate Mofetil Changed to Everolimus. Transplant. Proc. 2021, 54, 1561–1563. [Google Scholar] [CrossRef]
- Arbel, R.; Sagy, Y.W.; Hoshen, M.; Battat, E.; Lavie, G.; Sergienko, R.; Friger, M.; Waxman, J.G.; Dagan, N.; Balicer, R.; et al. Nirmatrelvir Use and Severe Covid-19 Outcomes during the Omicron Surge. New Engl. J. Med. 2022, 387, 790–798. [Google Scholar] [CrossRef]
- Zhou S, Hill CS, Sarkar S; et al. β-d-N4-hydroxycytidine Inhibits SARS-CoV-2 Through Lethal Mutagenesis But Is Also Mutagenic To Mammalian Cells. J Infect Dis 2021, 224, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Kabinger F, Stiller C, Schmitzová J; et al. Mechanism of molnupiravir-induced SARS-CoV-2 mutagenesis. Nat Struct Mol Biol 2021, 28, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Caraco, Y.; Crofoot, G.E.; Moncada, P.A.; Galustyan, A.N.; Musungaie, D.B.; Payne, B.; Kovalchuk, E.; Gonzalez, A.; Brown, M.L.; Williams-Diaz, A.; et al. Phase 2/3 Trial of Molnupiravir for Treatment of Covid-19 in Nonhospitalized Adults | NEJM Evidence. [cited 2024 Jan 25]. Available at: https://evidence.nejm.org/doi/full/10.1056/EVIDoa2100043. [CrossRef] [PubMed]
- Jayk Bernal A, Gomes da Silva MM, Musungaie DB; et al. Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients. N Engl J Med 2022, 386, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Butler, C.C.; Hobbs, F.D.R.; A Gbinigie, O.; Rahman, N.M.; Hayward, G.; Richards, D.B.; Dorward, J.; Lowe, D.M.; Standing, J.F.; Breuer, J.; et al. Molnupiravir plus usual care versus usual care alone as early treatment for adults with COVID-19 at increased risk of adverse outcomes (PANORAMIC): an open-label, platform-adaptive randomised controlled trial. Lancet 2023, 401, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Ader, F.; Bouscambert-Duchamp, M.; Hites, M.; Peiffer-Smadja, N.; Poissy, J.; Belhadi, D.; Diallo, A.; Lê, M.-P.; Peytavin, G.; Staub, T.; et al. Remdesivir plus standard of care versus standard of care alone for the treatment of patients admitted to hospital with COVID-19 (DisCoVeRy): a phase 3, randomised, controlled, open-label trial. Lancet Infect. Dis. 2022, 22, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, R.L.; Vaca, C.E.; Paredes, R.; Mera, J.; Webb, B.J.; Perez, G.; Oguchi, G.; Ryan, P.; Nielsen, B.U.; Brown, M.; et al. Early Remdesivir to Prevent Progression to Severe Covid-19 in Outpatients. New Engl. J. Med. 2021, 386, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Pettit NN, Pisano J, Nguyen CT; et al. Remdesivir Use in the Setting of Severe Renal Impairment: A Theoretical Concern or Real Risk? Clin Infect Dis 2021, 73, e3990–e3995. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.; Leister-Tebbe, H.; Gardner, A.; Abreu, P.; Bao, W.; Wisemandle, W.; Baniecki, M.; Hendrick, V.M.; Damle, B.; Simón-Campos, A.; et al. Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19. New Engl. J. Med. 2022, 386, 1397–1408. [Google Scholar] [CrossRef]
- Shah S, Ackley TW, Topal JE. Renal and Hepatic Toxicity Analysis of Remdesivir Formulations: Does What Is on the Inside Really Count? Antimicrob Agents Chemother 2021, 65, e0104521. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



