Submitted:
11 May 2024
Posted:
13 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Origins and Mechanism of the Julia-Kocienski Olefination Reaction
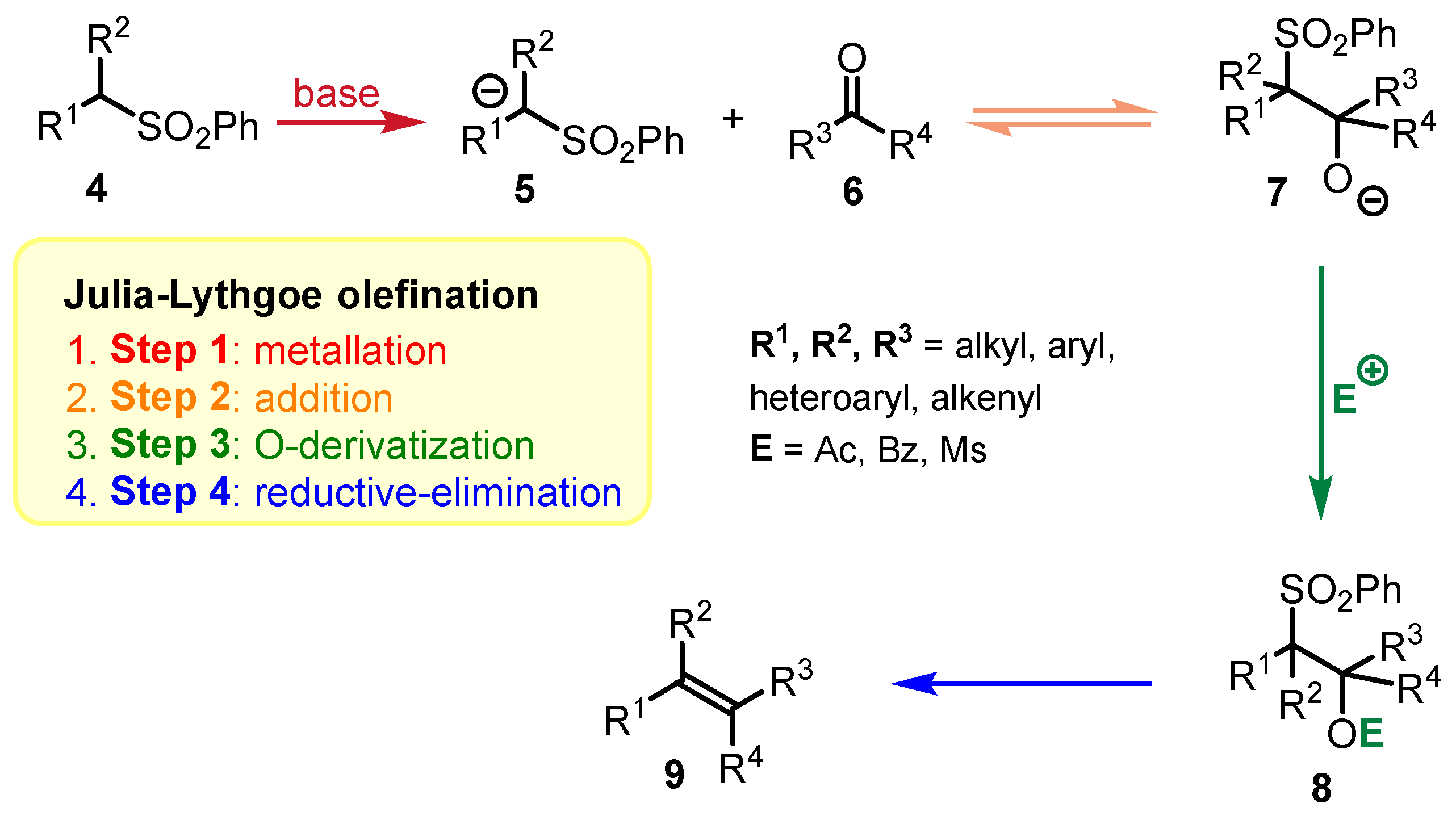
2.1. Julia-Lythgoe Olefination vs. Julia-Kocienski Olefination: A Comparison
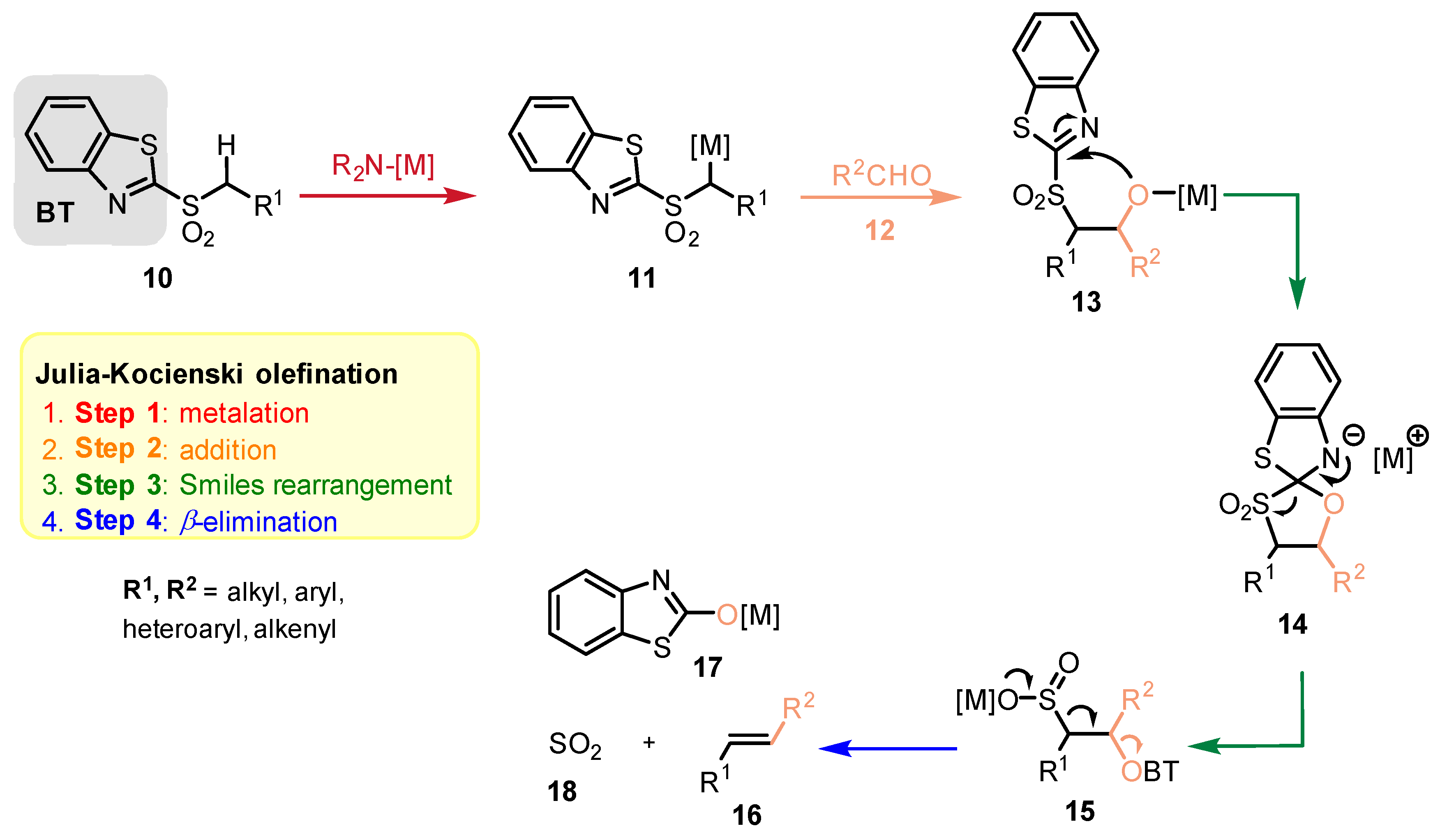
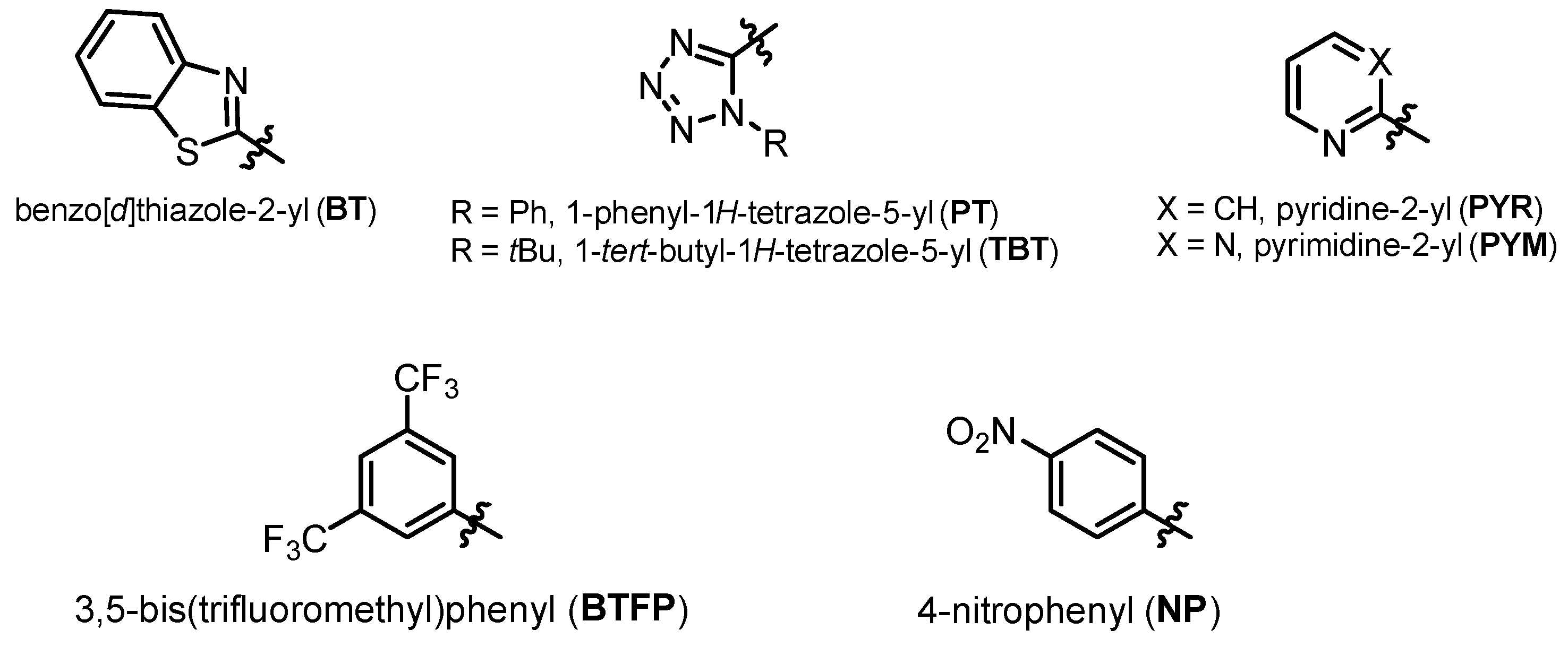
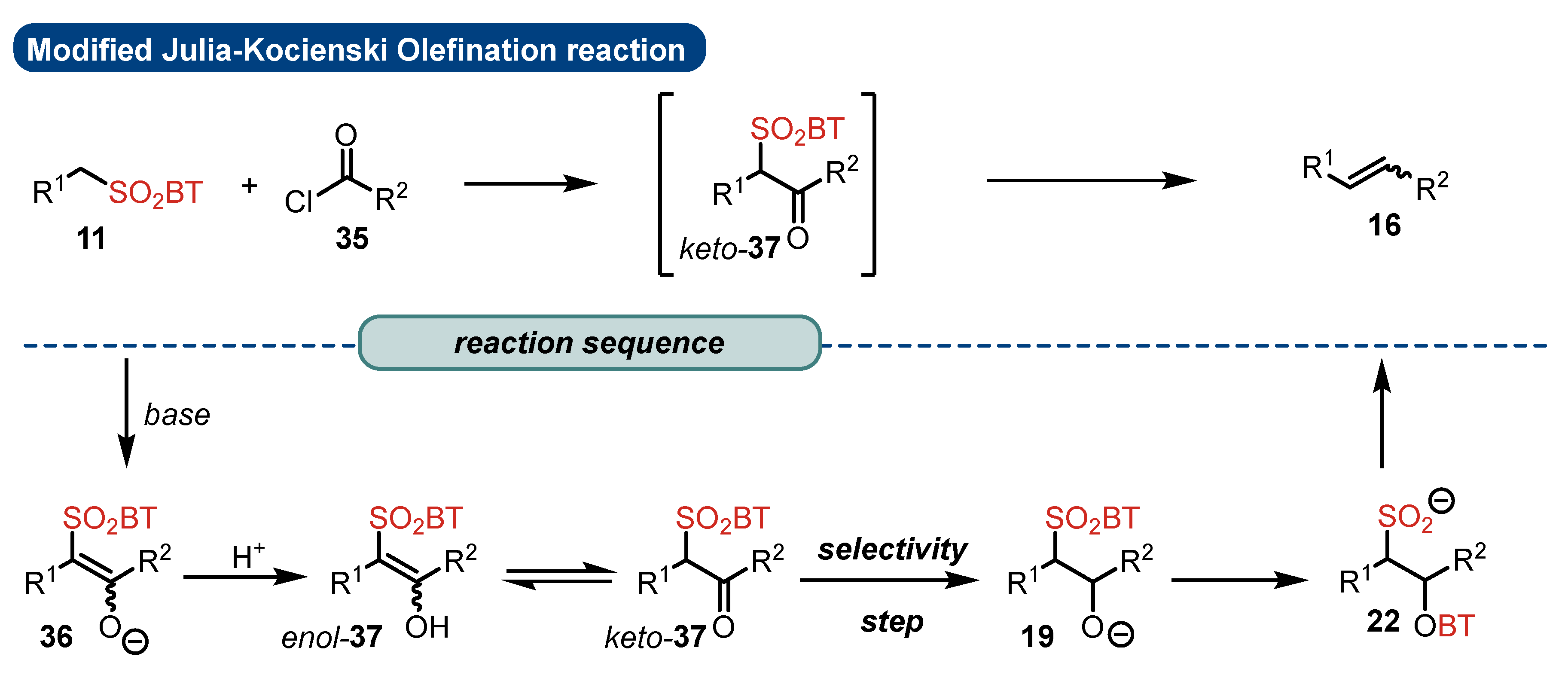
2.2. Reaction Mechanism and Its Impact on the Selectivity of Julia-Kocienski Olefination
- (1)
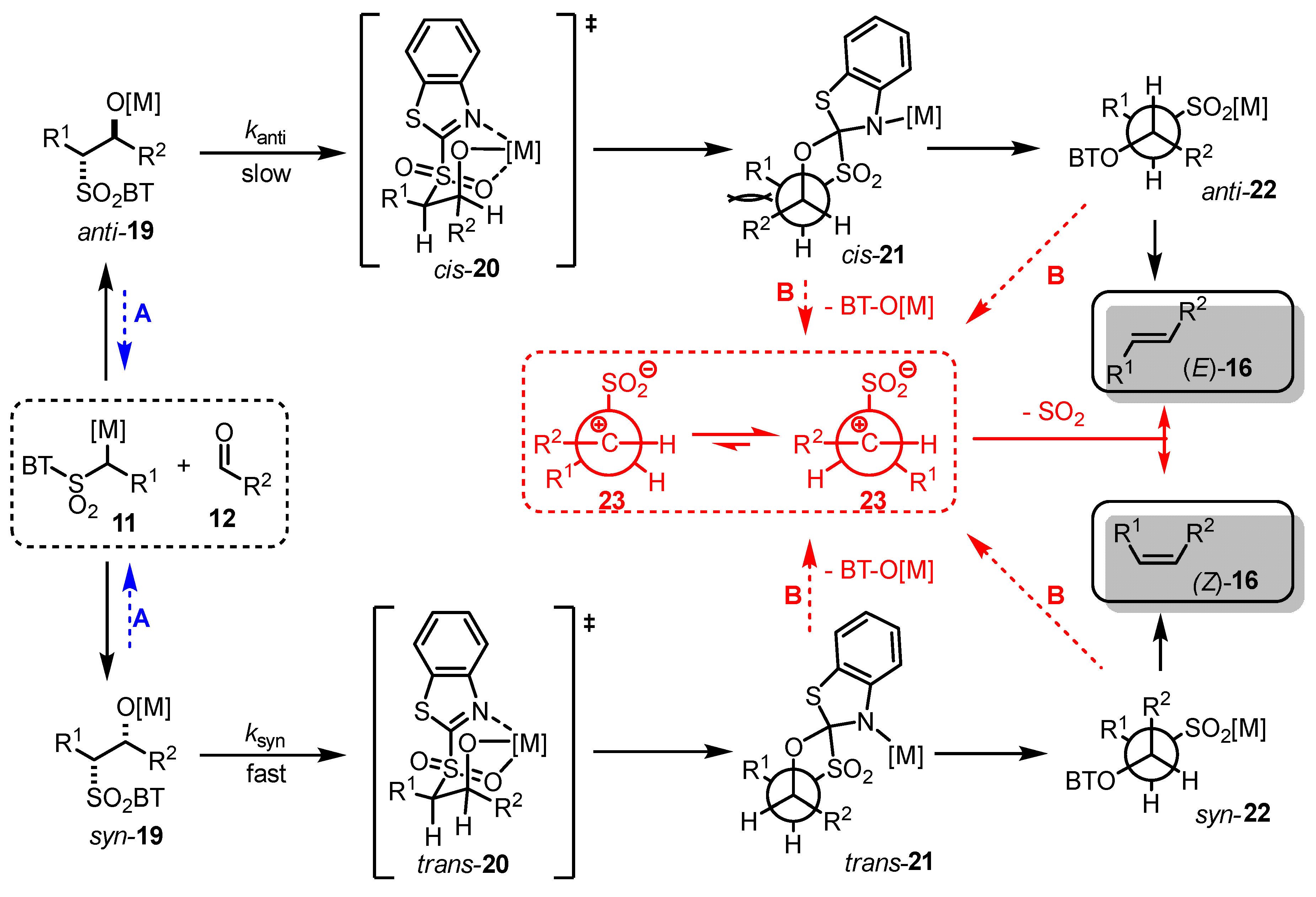
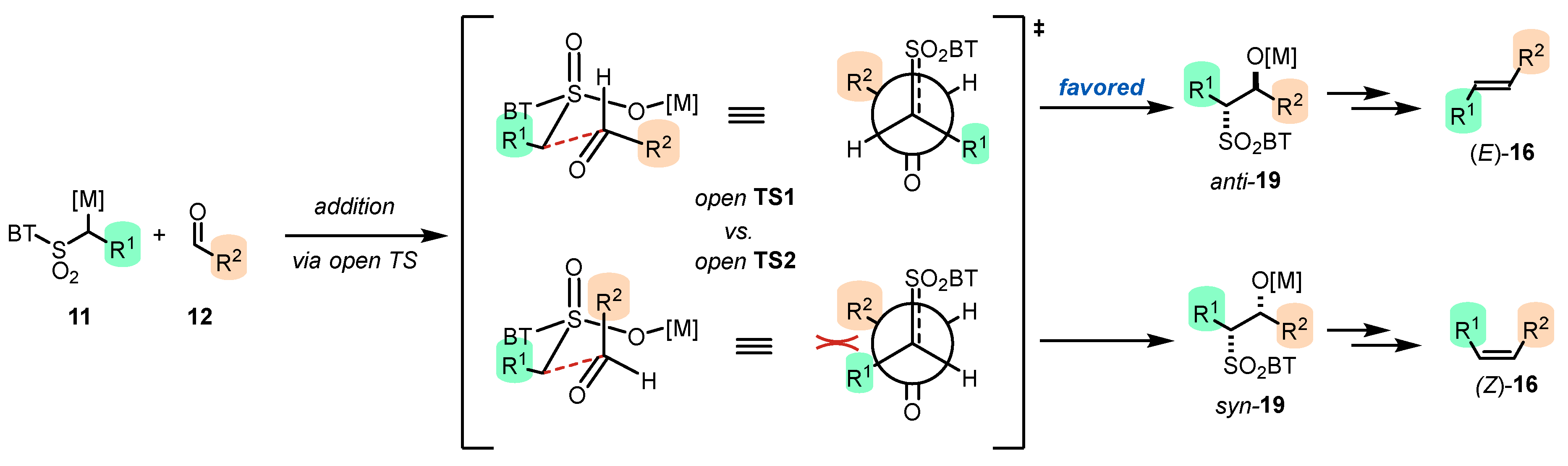
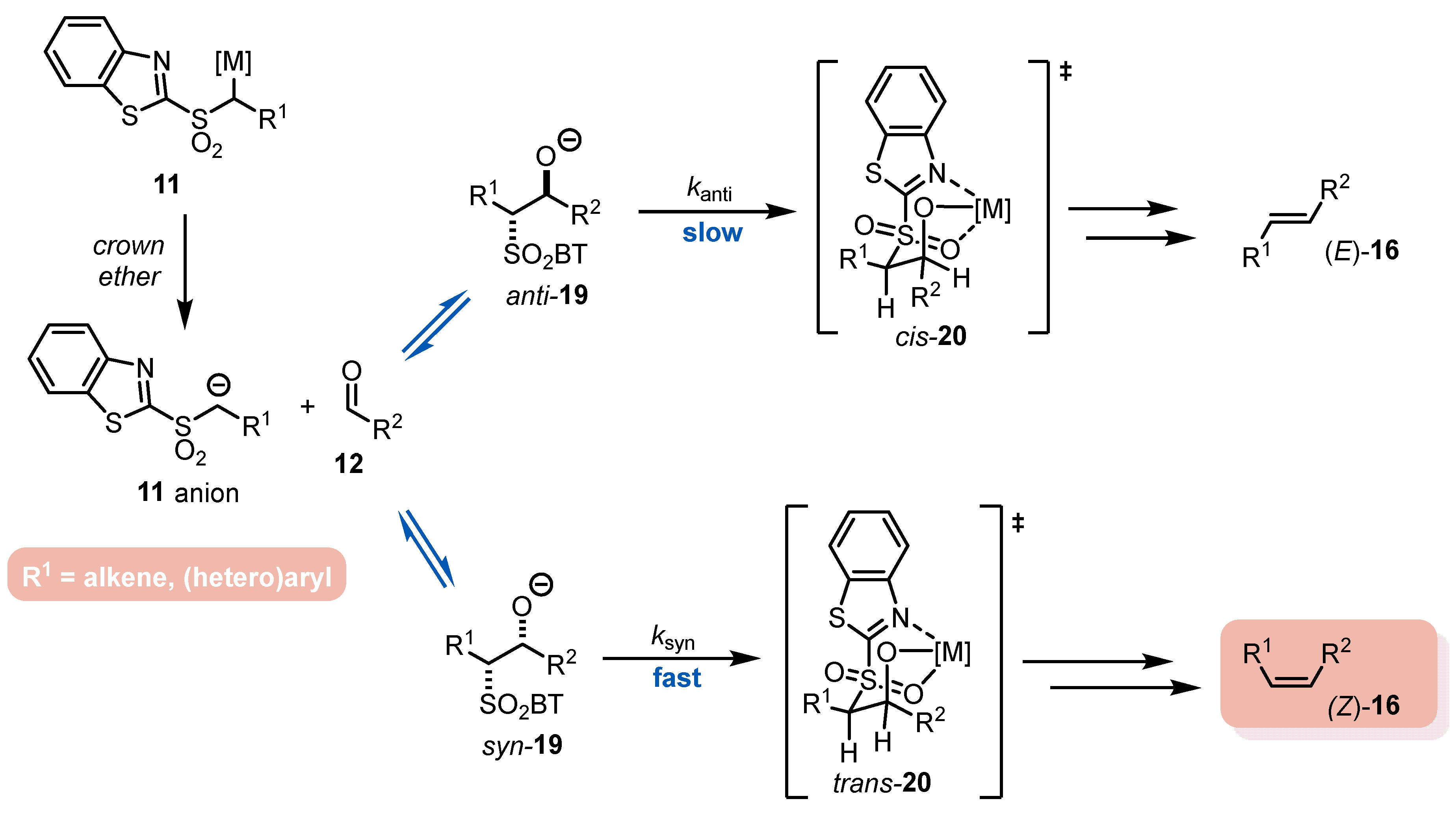
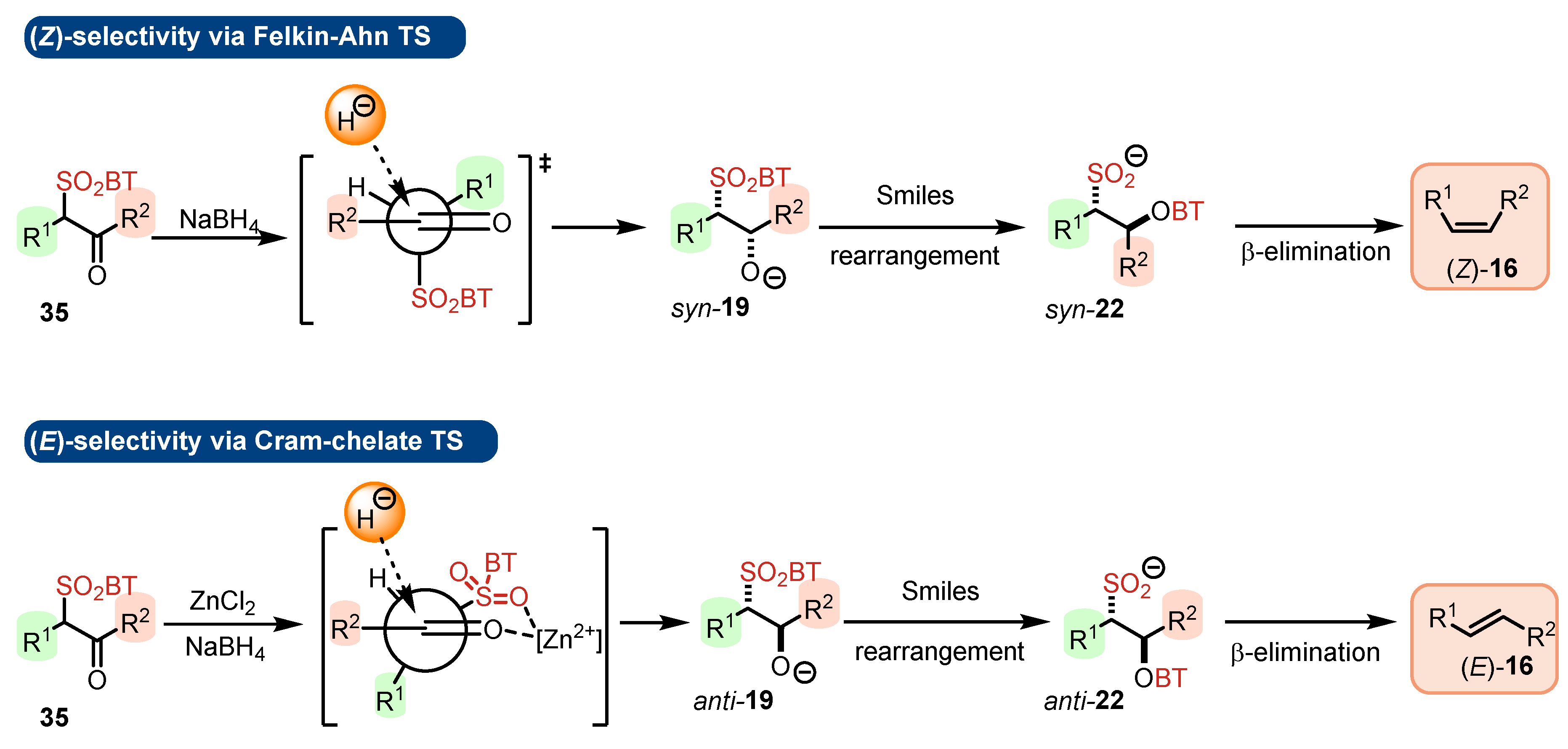
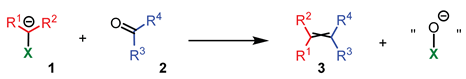
- The addition step of metalated sulfone 11 to aldehyde 12 can provide anti-adduct anti-19 via TS1 or syn-adduct syn-19 via TS2 (Figure 2). The selectivity in this step is extremely important since all subsequent transformations of intermediate 19, Smiles rearrangement and β-elimination, are stereospecific. Thus, the syn/anti-selectivity of the addition step determines the final (E/Z)-olefin ratio. Therefore, in theory, the (E/Z)-selectivity of the reaction could be swapped from (E) to (Z) if proper reaction conditions are applied.
- (2)
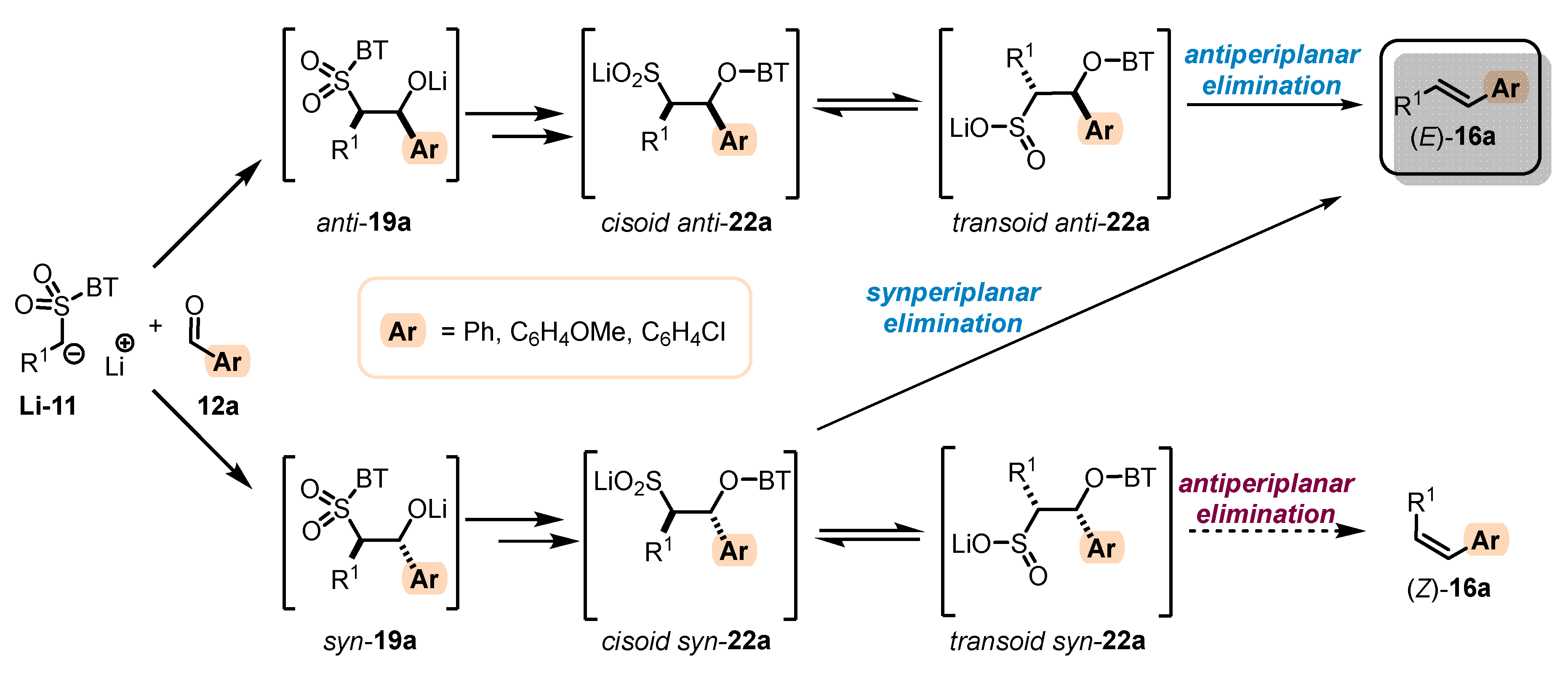
- When stabilized metalated sulfonyl anions 11 (R1 = Ph, alkenyl, etc.) are used, the addition step of 11 to 12 becomes reversible (Scheme 3, path A). In this case, the original kinetically driven syn/anti-ratio of adduct 19 becomes less important in comparison with the Smiles rearrangement reaction rates (transformation of 19 to 22). In such cases, the rearrangement of anti-19 adduct leading to (E)-olefin 16 is slower compared to the rearrangement of syn-19 to olefin (Z)-16 due to repulsive 1,2-interactions in the transition state (see cis-20).
- (3)
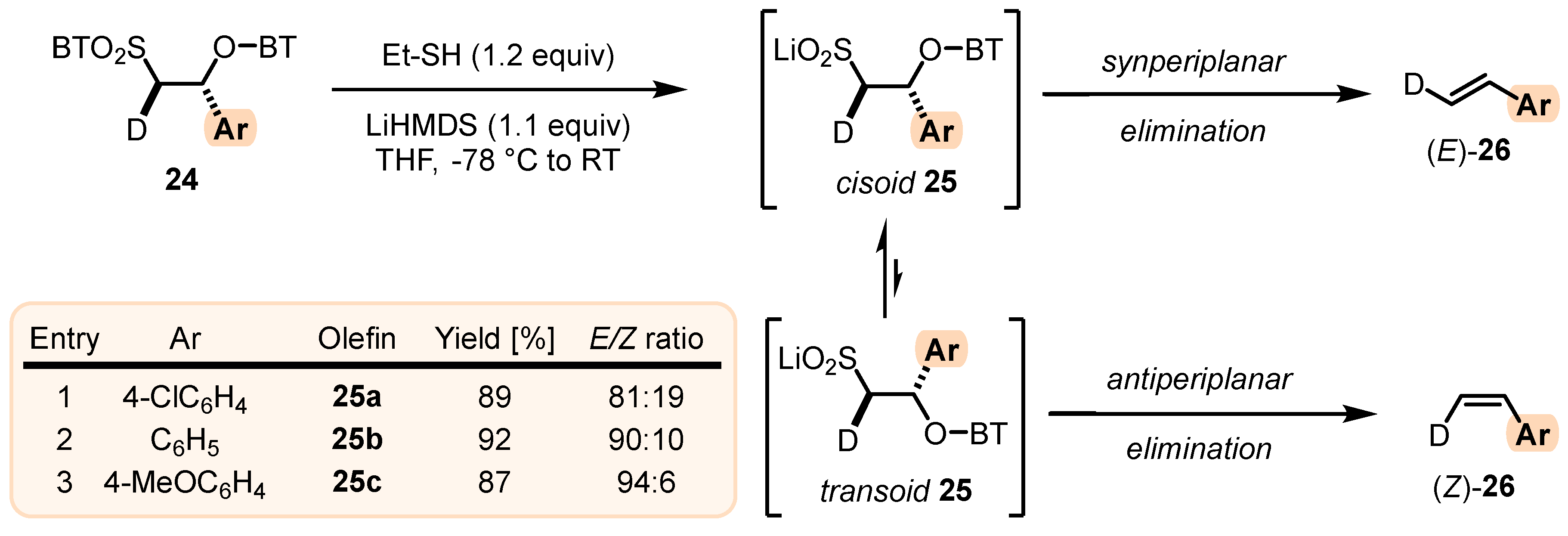
- For the elimination step, two borderline mechanisms are generally accepted. In the first, which is the most common, the rearranged intermediate 22 undergoes β-elimination. The elimination is stereospecific, and the syn-19 adduct rearranged intermediate syn-22 furnishes (Z)-olefin and the anti-19 adduct rearranged intermediate, compound trans-22 (trans refers to the arrangement of R1 and R2 within the intermediate cycle), yields (E)-olefin. Alternatively, when (hetero)aryl aldehydes 12 (R2 = (hetero)aryl) an alternative elimination pathway (path B) was postulated to occur. In this case, the elimination pathway should proceed through the formation of intermediate carbocation 23. The steric requirements of R1 and R2 then play a crucial role in the final (E/Z)-selectivity of the reaction. Path B was used to explain the unexpected (E)-selectivity of the coupling reactions carried out using (hetero)aryl aldehydes 12 as substrates.
2.3. Recent Achievements in the Reaction Selectivity Improvements
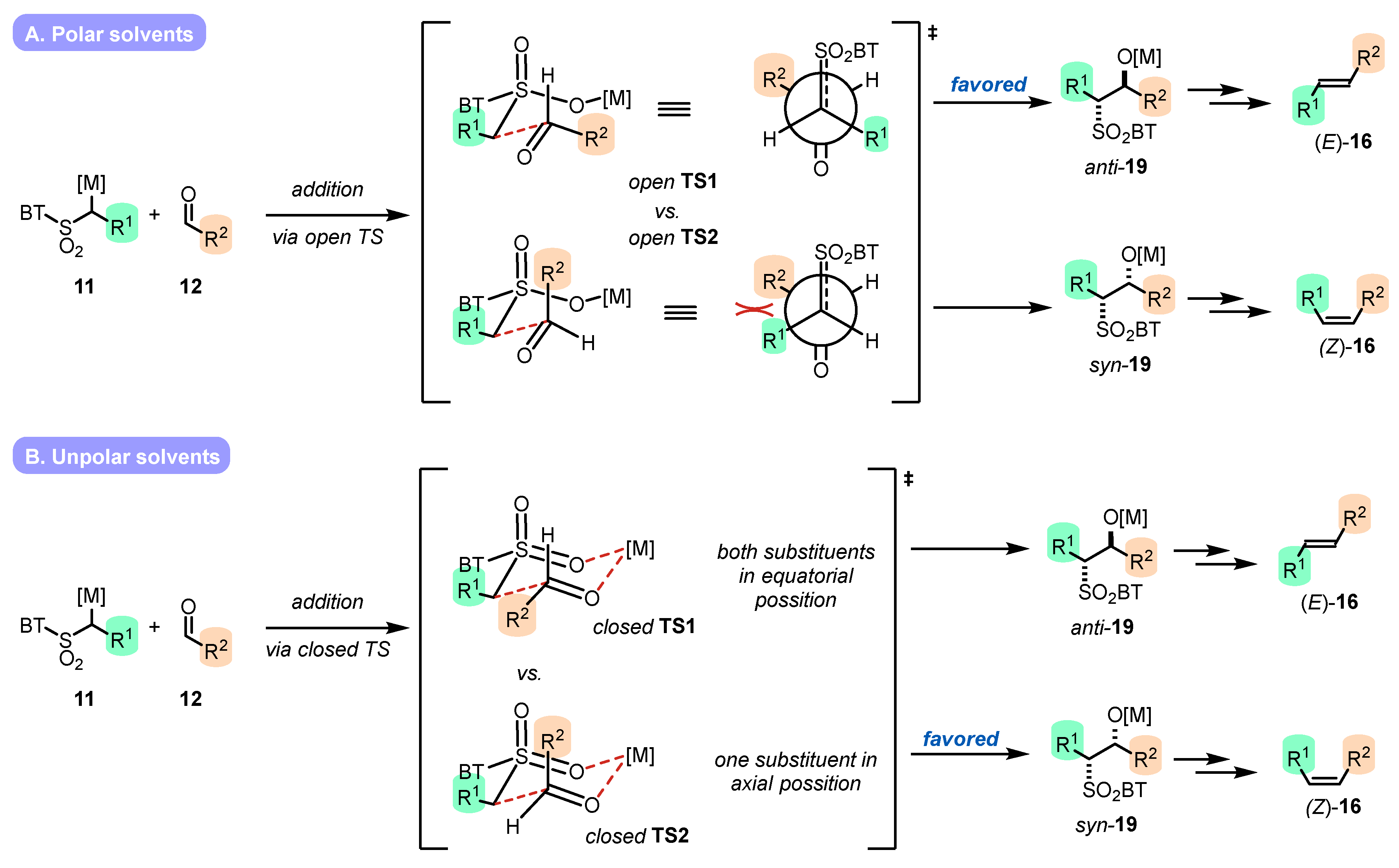
2.3.1. Solvent Effect
- Metal cation
- Co-solvents
2.3.2. Additives
- Crown ethers
- Ammonium salts
- Chelating Metals
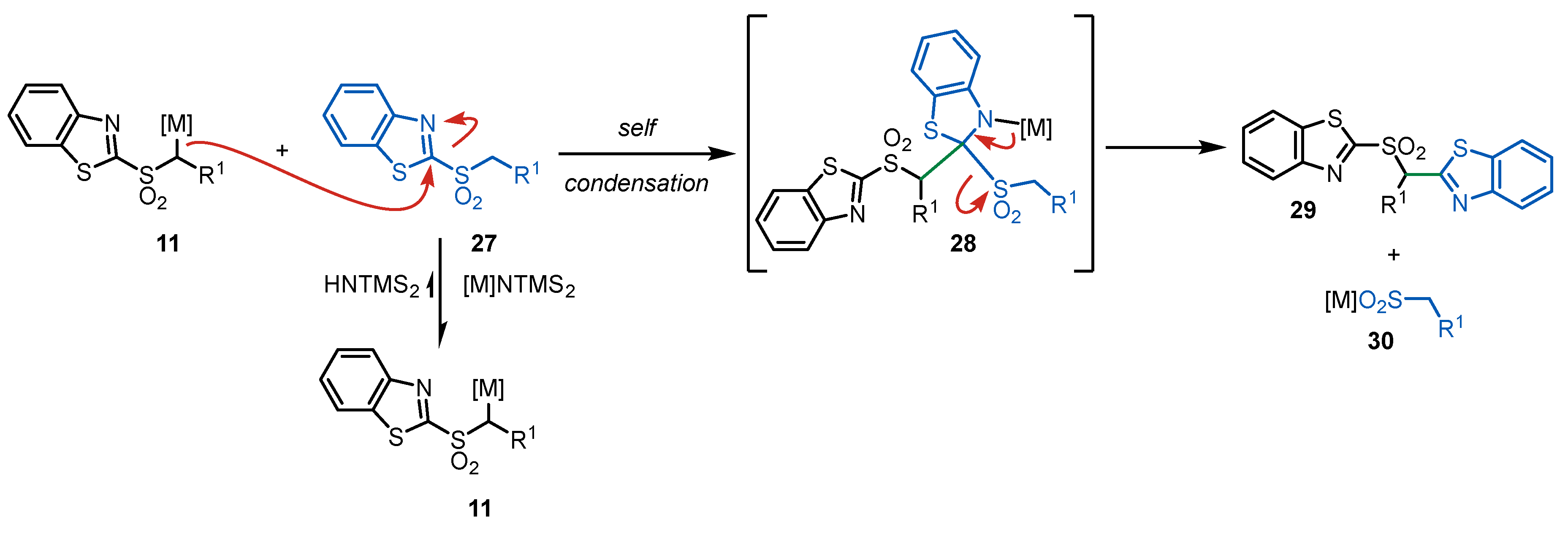
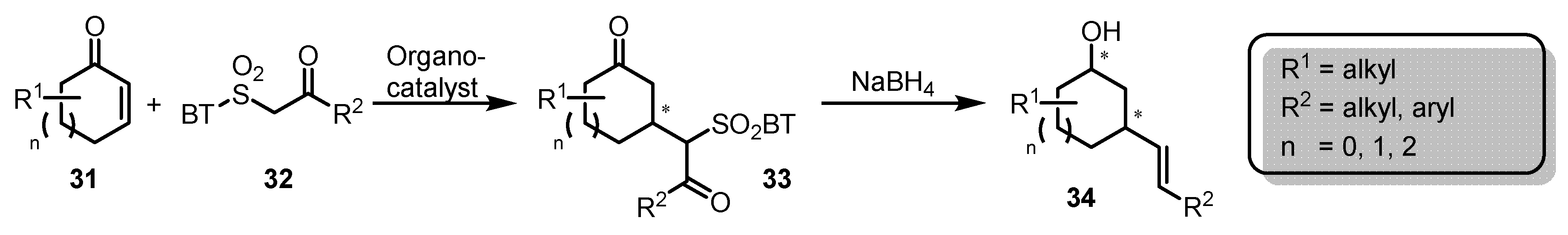
3. Julia-Kocienski Olefination – Extension to Carboxylic Acid Derivatives
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Markó, I.E.; Pospíšil, J. Julia, Julia–Kocienski, and Related Sulfur-Based Alkenations. In Science of Synthesis; de Meijere, Ed.; Georg Thieme Verlag: Stuttgart, 2010; Vol. 47a, pp. 105–160. [Google Scholar]
- Johnson, C.R.; Shanklin, J.R.; Kirchhoff, R.A. Olefin Synthesis by Reductive Elimination of.Beta.-Hydroxysulfoximines. Methylenation of Carbonyl Compounds. J Am Chem Soc 1973, 95, 6462–6463. [Google Scholar] [CrossRef]
- Maryanoff, B.E.; Reitz, A.B. The Wittig Olefination Reaction and Modifications Involving Phosphoryl-Stabilized Carbanions. Stereochemistry, Mechanism, and Selected Synthetic Aspects. Chem Rev 1989, 89, 863–927. [Google Scholar] [CrossRef]
- Bisceglia, J.A.; Orelli, L.R. Recent Progress in the Horner-Wadsworth-Emmons Reaction. Curr Org Chem 2015, 19, 744–775. [Google Scholar] [CrossRef]
- Staden, L.F. van; Gravestock, D.; Ager, D.J. New Developments in the Peterson Olefination Reaction. Chem Soc Rev 2002, 31, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Coombs, J.R.; Zhang, L.; Morken, J.P. Synthesis of Vinyl Boronates from Aldehydes by a Practical Boron-Wittig Reaction. Org Lett 2015, 17, 1708–1711. [Google Scholar] [CrossRef]
- Wittig, G.; Geissler, G. Zur Reaktionsweise Des Pentaphenyl-phosphors Und Einiger Derivate. Justus Liebigs Ann Chem 1953, 580, 44–57. [Google Scholar] [CrossRef]
- Wittig, G.; Schöllkopf, U. Über Triphenyl-phosphin-methylene Als Olefinbildende Reagenzien (I. Mitteil. Chem Ber 1954, 87, 1318–1330. [Google Scholar] [CrossRef]
- Chatterjee, B.; Bera, S.; Mondal, D. Julia-Kocienski Olefination: A Key Reaction for the Synthesis of Macrolides. Tetrahedron Asymm 2014, 25, 1–55. [Google Scholar] [CrossRef]
- Legnani, L.; Porta, A.; Caramella, P.; Toma, L.; Zanoni, G.; Vidari, G. Computational Mechanistic Study of the Julia-Kocieński Reaction. J Org Chem 2015, 80, 3092–3100. [Google Scholar] [CrossRef]
- Aïssa, C. Mechanistic Manifold and New Developments of the Julia-Kocienski Reaction. European J Org Chem 2009, 1831–1844. [Google Scholar] [CrossRef]
- Blakemore, P.R. The Modified Julia Olefination: Alkene Synthesis via the Condensation of Metallated Heteroarylalkylsulfones with Carbonyl Compounds. J Chem Soc Perkin 1 2002, 2, 2563–2585. [Google Scholar] [CrossRef]
- Blakemore, P.R.; Cole, W.J.; Kocieński, P.J.; Morley, A. A Stereoselective Synthesis of Trans-1,2-Disubstituted Alkenes Based on the Condensation of Aldehydes with Metallated 1-Phenyl-1 H -Tetrazol-5-Yl Sulfones. Synlett 1998, 1998, 26–28. [Google Scholar] [CrossRef]
- Robiette, R.; Pospíšil, J. On the Origin of E/Z Selectivity in the Modified Julia Olefination - Importance of the Elimination Step. European J Org Chem 2013, 836–840. [Google Scholar] [CrossRef]
- Baudin, J.B.; Hareau, G.; Julia, S.A.; Ruel, O. A Direct Synthesis of Olefins by Reaction of Carbonyl Compounds with Lithio Derivatives of 2-[Alkyl- or (2′-Alkenyl)- or Benzyl-Sulfonyl]-Benzothiazoles. Tetrahedron Lett 1991, 32, 1175–1178. [Google Scholar] [CrossRef]
- Gueyrard, D. Extension of the Modified Julia Olefination on Carboxylic Acid Derivatives: Scope and Applications. Synlett 2018, 29, 34–45. [Google Scholar] [CrossRef]
- Julia, M.; Paris, J.M. Syntheses a l’aide de Sulfones v(+)- Methode de Synthese Generale de Doubles Liaisons. Tetrahedron Lett 1973, 14, 4833–4836. [Google Scholar] [CrossRef]
- Kocienski, P.J.; Lythgoe, B.; Ruston, S. Scope and Stereochemistry of an Olefin Synthesis from β-Hydroxysulphones. J Chem Soc Perkin 1 1978, 829–834. [Google Scholar] [CrossRef]
- Keck, G.E.; Savin, K.A.; Weglarz, M.A. Use of Samarium Diiodide as an Alternative to Sodium/Mercury Amalgam in the Julia-Lythgoe Olefination. J Org Chem 1995, 60, 3194–3204. [Google Scholar] [CrossRef]
- Baudin, J.B.; Hareau, G.; Julia, S.A.; Lorne, R.; Ruel, O. Stereochemistry of Direct Olefin Formation from Carbonyl Compounds and Lithiated Heterocyclic Sulfones. Bull Soc Chim Fr 1993, 130, 856–878. [Google Scholar]
- Baudin, J.B.; Hareau, G.; Julia, S.A.; Ruel, O. Stereochemistry of the Olefin Formation from Anti and Syn Heterocyclic β-Hydroxy-Sulfones. Bull Soc Chim Fr 1993, 130, 336–357. [Google Scholar]
- Charette, A.B.; Berthelette, C.; St-Martin, D. An Expedient Approach to E, Z-Dienes Using the Julia Olefination. Tetrahedron Lett 2001, 42, 5149–5153. [Google Scholar] [CrossRef]
- Blakemore, P.R.; Cole, W.J.; Kocieński, P.J.; Morley, A. A Stereoselective Synthesis of Trans -1,2-Disubstituted Alkenes Based on the Condensation of Aldehydes with Metallated 1-Phenyl-1 H -Tetrazol-5-Yl Sulfones. Synlett 1998, 1998, 26–28. [Google Scholar] [CrossRef]
- Kocienski, P.J.; Bell, A.; Blakemore, P.R. 1- Tert -Butyl-1 H -Tetrazol-5-Yl Sulfones in the Modified Julia Olefination. Synlett 2000, 2000, 365–366. [Google Scholar] [CrossRef]
- Alonso, D.A.; Fuensanta, M.; Nájera, C.; Varea, M. 3,5-Bis(Trifluoromethyl)Phenyl Sulfones in the Direct Julia−Kocienski Olefination. J Org Chem 2005, 70, 6404–6416. [Google Scholar] [CrossRef] [PubMed]
- Mąkosza, M.; Bujok, R. Synthesis of Benzylidenecyclopropanes from γ-Halopropyl Pentachlorophenyl Sulfones Using a Julia-Kocienski Olefination. Synlett 2008, 2008, 586–588. [Google Scholar] [CrossRef]
- Pospíšil, J. Simple Protocol for Enhanced (E)-Selectivity in Julia–Kocienski Reaction. Tetrahedron Lett 2011, 52, 2348–2352. [Google Scholar] [CrossRef]
- Jana, N.; Nanda, S. Asymmetric Total Syntheses of Cochliomycin A and Zeaenol. European J Org Chem 2012, 4313–4320. [Google Scholar] [CrossRef]
- Mohapatra, D.K.; Reddy, D.S.; Mallampudi, N.A.; Yadav, J.S. Stereoselective Total Syntheses of Paecilomycins e and F through a Protecting Group Directed Diastereoselective Intermolecular Nozaki-Hiyama-Kishi (NHK) Reaction. European J Org Chem 2014, 2014, 5023–5032. [Google Scholar] [CrossRef]
- Sánchez, D.; Andreou, T.; Costa, A.M.; Meyer, K.G.; Williams, D.R.; Barasoain, I.; Díaz, J.F.; Lucena-Agell, D.; Vilarrasa, J. Total Synthesis of Amphidinolide K, a Macrolide That Stabilizes F-Actin. J Org Chem 2016, 80, 8511–8519. [Google Scholar] [CrossRef]
- Wilson, D.M.; Britton, R. Enantioselective Total Synthesis of the Marine Macrolides Salarins A and C. J Am Chem Soc 2024, 146, 8456–8463. [Google Scholar] [CrossRef]
- Billard, F.; Robiette, R.; Pospíšil, J. Julia-Kocienski Reaction-Based 1,3-Diene Synthesis: Aldehyde-Dependent (E, E/E, Z)-Selectivity. J Org Chem 2012, 77, 6358–6364. [Google Scholar] [CrossRef]
- Rehman, M.; Surendran, S.; Siddavatam, N.; Rajendar, G. The Influence of α-Coordinating Groups of Aldehydes on E/Z-Selectivity and the Use of Quaternary Ammonium Counter Ions for Enhanced E-Selectivity in the Julia–Kocienski Reaction. Org Biomol Chem 2022, 20, 329–333. [Google Scholar] [CrossRef]
- Rajendar, G.; Corey, E.J. A Systematic Study of Functionalized Oxiranes as Initiating Groups for Cationic Polycyclization Reactions. J Am Chem Soc 2015, 137, 5837–5844. [Google Scholar] [CrossRef]
- Tsubone, K.; Hashizume, K.; Fuwa, H.; Sasaki, M. Studies toward the Total Synthesis of Gambieric Acids: Convergent Synthesis of the GHIJ-Ring Fragment Having a Side Chain. Tetrahedron Lett 2011, 52, 548–551. [Google Scholar] [CrossRef]
- Tsubone, K.; Hashizume, K.; Fuwa, H.; Sasaki, M. Studies toward the Total Synthesis of Gambieric Acids, Potent Antifungal Polycyclic Ethers: Convergent Synthesis of a Fully Elaborated GHIJ-Ring Fragment. Tetrahedron 2011, 67, 6600–6615. [Google Scholar] [CrossRef]
- Rej, R.K.; Kumar, R.; Nanda, S. Asymmetric Synthesis of Cytospolides C and D through Successful Exploration of Stereoselective Julia-Kocienski Olefination and Suzuki Reaction Followed by Macrolactonization. Tetrahedron 2015, 71, 3185–3194. [Google Scholar] [CrossRef]
- Eliel, E.L.; Frye, S.V.; Hortelano, E.R.; Chen, X.; Bai, X. Asymmetric Synthesis and Cram’s (Chelate) Rule. Pure & App Chem 1991, 63, 1591–1598. [Google Scholar] [CrossRef]
- Bon, D.J. -Y. D.; Chrenko, D.; Kováč, O.; Ferugová, V.; Lasák, P.; Fuksová, M.; Zálešák, F.; Pospíšil, J. Julia-Kocienski-Like Connective C−C and C=C Bond-Forming Reaction. Adv Synth Catal 2024, 366, 480–487. [Google Scholar] [CrossRef]
- Nielsen, M.; Jacobsen, C.B.; Paixão, M.W.; Holub, N.; Jørgensen, K.A. Asymmetric Organocatalytic Formal Alkynylation and Alkenylation of α,β-Unsaturated Aldehydes. J Am Chem Soc 2009, 131, 10581–10586. [Google Scholar] [CrossRef]
- Jacobsen, C.B.; Nielsen, M.; Worgull, D.; Zweifel, T.; Fisker, E.; Jørgensen, K.A. Asymmetric Organocatalytic Monofluorovinylations. J Am Chem Soc 2011, 133, 7398–7404. [Google Scholar] [CrossRef]
- Pospíšil, J.; Sato, H. Practical Synthesis of β-Acyl and β-Alkoxycarbonyl Heterocyclic Sulfones. J Org Chem 2011, 76, 2269–2272. [Google Scholar] [CrossRef] [PubMed]
- Pospíšil, J.; Robiette, R.; Sato, H.; Debrus, K. Practical Synthesis of β-Oxo Benzo[d]Thiazolyl Sulfones: Scope and Limitations. Org Biomol Chem 2012, 10, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Bettens, T.; Alonso, M.; Geerlings, P.; De Proft, F. Mechanochemical Felkin–Anh Model: Achieving Forbidden Reaction Outcomes with Mechanical Force. J Org Chem 2023, 88, 2046–2056. [Google Scholar] [CrossRef] [PubMed]
 | ||
| Activating unit X | Olefination Method | Litt. reference |
| PhSO2 | Julia-Lythgoe | Ref.[1] |
| ActSO2 | Julia-Kocienski | Ref.[1] |
| PhSO(NMe) | Johnson | Ref.[2] |
| R3P+ | Wittig | Ref.[3] |
| R2P(=O) | Wittig-Horner | Ref.[3] |
| (RO)2P(=O) | Horner-Wodsworth-Emmons (HWE) | Ref.[4] |
| R3Si | Peterson | Ref.[5] |
| R2B | Boron-Wittig | Ref.[6] |
| Key Features | Julia-Lythgoe | Julia-Kocienski |
| Practical Difference | Two-pot protocol | One-pot protocol |
| Origin of Stereoselectivity | Reductive Elimination Step | The addition step |
| Scope of olefin formation | ||
| Terminal |  |
 |
| 1,2-disubstituted |  |
 |
| Trisubstituted |  |
 |
| Tetrasubstituted |  |
 |
| Scope of(E)-Stereoselectivity | ||
| 1,2-disubstituted |  |
 |
| Trisubstituted |  |
 |
| Tetrasubstituted |  |
 |
| Scope of(Z)-Stereoselectivity | ||
| 1,2-disubstituted |  |
 if theTBT-activatinggroup is used; if theTBT-activatinggroup is used;
|
| Trisubstituted |  |
 |
| Tetrasubstituted |  |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




