Submitted:
08 May 2024
Posted:
08 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
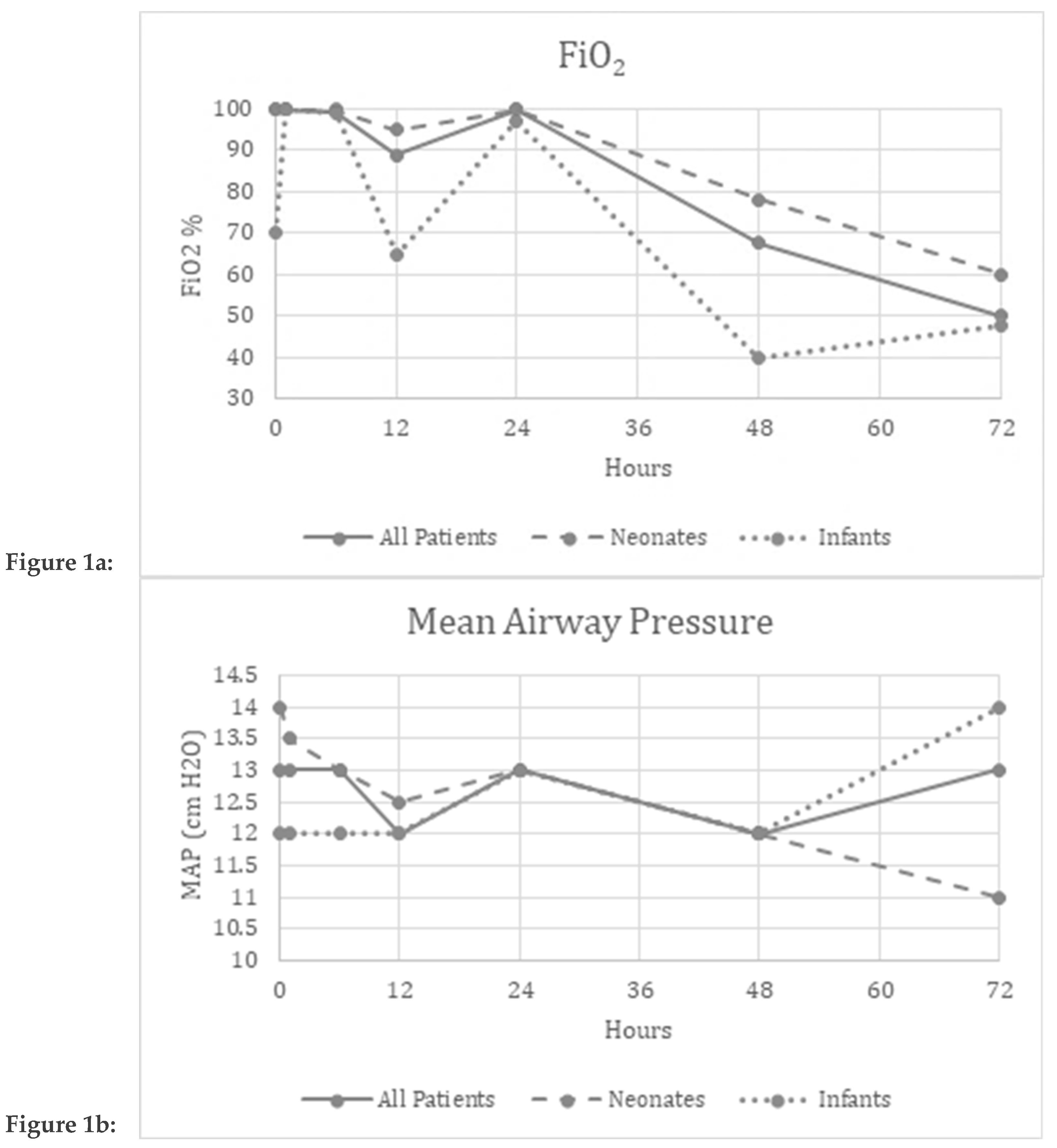
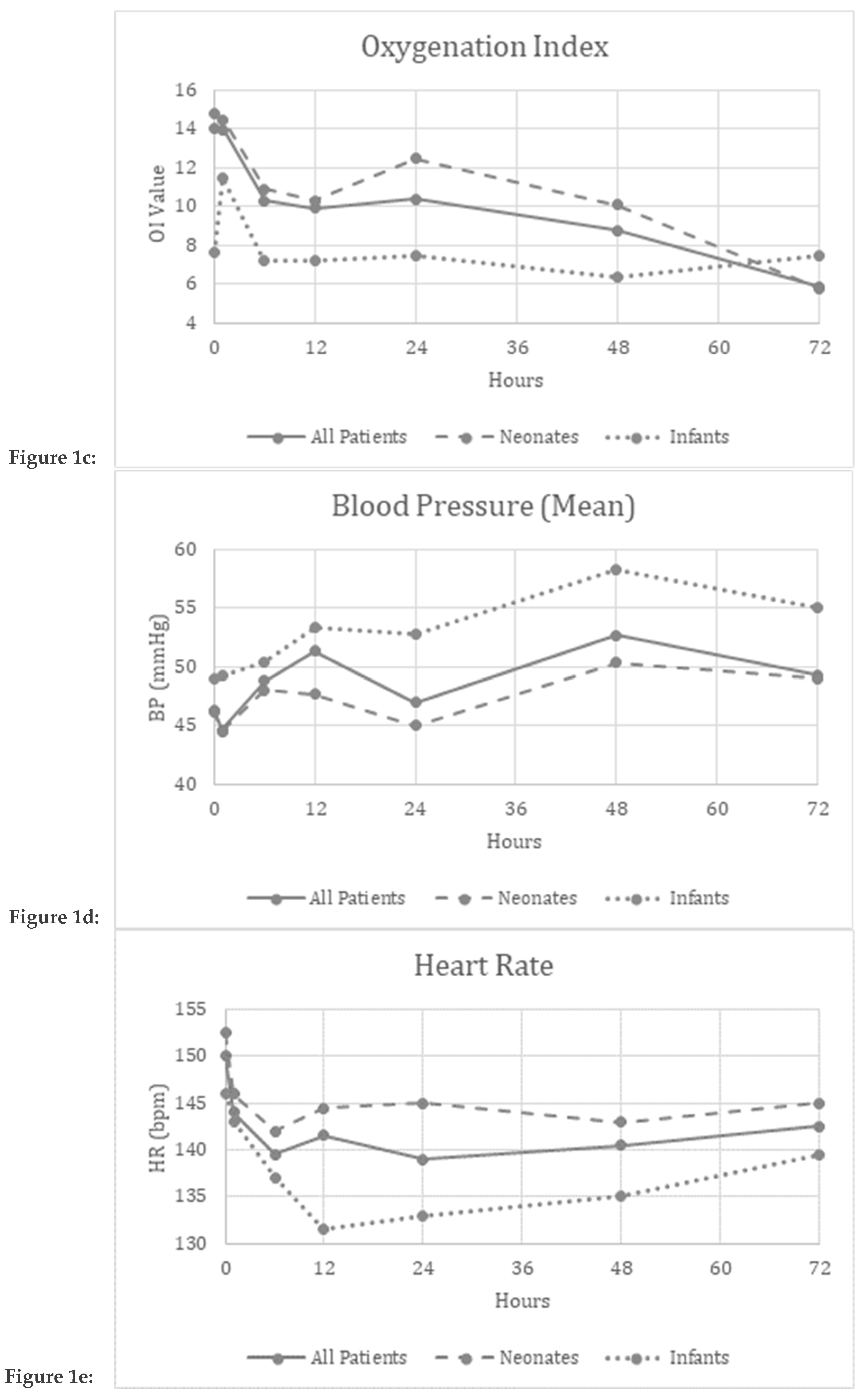
3. Results
3.1. Demographics
4. Discussion
5. Conclusions:
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- del Cerro, M.J.; Abman, S.; Diaz, G.; Freudenthal, A.H.; Freudenthal, F.; Harikrishnan, S.; Haworth, S.G.; Ivy, D.; Lopes, A.A.; Raj, J.U.; et al. A Consensus Approach to the Classification of Pediatric Pulmonary Hypertensive Vascular Disease: Report from the PVRI Pediatric Taskforce, Panama 2011. Pulm. Circ. 2011, 1, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Singh, Y.; Lakshminrusimha, S. Pathophysiology and Management of Persistent Pulmonary Hypertension of the Newborn. Clin. Perinatol. 2021, 48, 595–618. [Google Scholar] [CrossRef]
- Nandula, P.S.; Shah, S.D. Persistent Pulmonary Hypertension of the Newborn. [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK585100/.
- Takatsuki, S.; Ivy, D.D. Current challenges in pediatric pulmonary hypertension. Semin Respir Crit Care Med. 2013, 34, 627–644. [Google Scholar] [CrossRef] [PubMed]
- Ivy, D. Pulmonary hypertension in children. Cardiol Clin. 2016, 34, 451–472. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, H.-M.; Gu, L.; Li, Q.-W.; Zhu, L. Prostacyclins and pulmonary arterial hypertension in children. European Review for Medical & Pharmacological Sciences 2022, 26, 37–45. [Google Scholar] [CrossRef]
- Barrington, K.J.; Finer, N.; Pennaforte, T.; Altit, G. Nitric oxide for respiratory failure in infants born at or near term. Emergencias 2017, 2017, CD000399. [Google Scholar] [CrossRef] [PubMed]
- Olschewski, H.; Simonneau, G.; Galiè, N.; Higenbottam, T.; Naeije, R.; Rubin, L.J.; Nikkho, S.; Speich, R.; Hoeper, M.M.; Behr, J.; et al. Inhaled Iloprost for Severe Pulmonary Hypertension. New Engl. J. Med. 2002, 347, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Lakshminrusimha, S. Persistent Pulmonary Hypertension of the Newborn. NeoReviews 2015, 16, e680–e692. [Google Scholar] [CrossRef]
- Colglazier, E.; Ng, A.J.; Parker, C.; Woolsey, D.; Holmes, R.; Dsouza, A.; Becerra, J.; Stevens, L.; Nawaytou, H.; Keller, R.L.; et al. Safety and tolerability of continuous inhaled iloprost in critically ill pediatric pulmonary hypertension patients: A retrospective case series. Pulm. Circ. 2023, 13, e12289. [Google Scholar] [CrossRef]
- Walmrath, D.; Schermuly, R.; Pilch, J.; Grimminger, F.; Seeger, W. Effects of inhaled versus intravenous vasodilators in experimental pulmonary hypertension. Eur. Respir. J. 1997, 10, 1084–1092. [Google Scholar] [CrossRef]
- Limsuwan, A.; Wanitkul, S.; Khosithset, A.; Attanavanich, S.; Samankatiwat, P. Aerosolized iloprost for postoperative pulmonary hypertensive crisis in children with congenital heart disease. Int. J. Cardiol. 2008, 129, 333–338. [Google Scholar] [CrossRef]
- Verma, S.; Lumba, R.; Kazmi, S.H.; Vaz, M.J.; Prakash, S.S.; Bailey, S.M.; Mally, P.V.; Randis, T.M. Effects of Inhaled Iloprost for the Management of Persistent Pulmonary Hypertension of the Newborn. Am. J. Perinatol. 2021, 39, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Ivy, D.D.; Doran, A.K.; Smith, K.J.; Mallory, G.B.; Beghetti, M.; Barst, R.J.; Brady, D.; Law, Y.; Parker, D.; Claussen, L.; et al. Short- and Long-Term Effects of Inhaled Iloprost Therapy in Children With Pulmonary Arterial Hypertension. Circ. 2008, 51, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Calcaterra, G.; Bassareo, P.P.; Barilla, F.; Martino, F.; Fanos, V.; Fedele, F.; Romeo, F. Pulmonary hypertension in pediatrics. A feasible approach to bridge the gap between real world and guidelines. J. Matern. Neonatal Med. 2019, 34, 3820–3826. [Google Scholar] [CrossRef] [PubMed]
- Rimensberger, P.C.; Spahr-Schopfer, I.; Berner, M.; Jaeggi, E.; Kalangos, A.; Friedli, B. Inhaled nitric oxide versus aerosolized iloprost in secondary pulmonary hypertension in children with congenital heart disease: vasodilator capacity and cellular mechanisms. Circulation 2001, 103, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Gopal, S.H.; Toy, C.L.; Hanna, M.; Furtun, B.Y.; Hagan, J.L.; Nassr, A.A.; Fernandes, C.J.; Keswani, S.; Gowda, S.H. Inotropic score and vasoactive inotropic score as predictors of outcomes in congenital diaphragmatic hernia: A single center retrospective study. Front. Pediatr. 2023, 11, 1101546. [Google Scholar] [CrossRef] [PubMed]
- Belletti, A.; Lerose, C.C.; Zangrillo, A.; Landoni, G. Vasoactive-Inotropic Score: Evolution, Clinical Utility, and Pitfalls. J. Cardiothorac. Vasc. Anesthesia 2021, 35, 3067–3077. [Google Scholar] [CrossRef] [PubMed]
- Zaccolo, M.; Movsesian, M.A. cAMP and cGMP Signaling Cross-Talk. Circ. Res. 2007, 100, 1569–1578. [Google Scholar] [CrossRef]
- Kelly, L.K.; Porta, N.F.; Goodman, D.M.; Carroll, C.L.; Steinhorn, R.H. Inhaled prostacyclin for term infants with persistent pulmonary hypertension refractory to inhaled nitric oxide. J. Pediatr. 2002, 141, 830–832. [Google Scholar] [CrossRef]
- De Luca, D.; Zecca, E.; Piastra, M.; Romagnoli, C. Iloprost as ‘rescue’ therapy for pulmonary hypertension of the neonate. Paediatr Anaesth 2007, 17, 394–395. [Google Scholar] [CrossRef]
- Berger-Caron, F.; Piedboeuf, B.; Morissette, G.; Simonyan, D.; Chétaille, P.; Pellerin, A.; Hébert, A. Inhaled Epoprostenol for Pulmonary Hypertension Treatment in Neonates: A 12-Year Experience. Am. J. Perinatol. 2018, 36, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Debillon, T.; Sentilhes, L.; Kayem, G.; Chevallier, M.; Zeitlin, J.; Baud, O.; Vilotitch, A.; Pierrat, V.; Guellec, I.; Ancel, P.Y.; et al. Risk factors for unfavorable outcome at discharge of newborns with hypoxic-ischemic encephalopathy in the era of hypothermia. Pediatr. Res. 2022, 93, 1975–1982. [Google Scholar] [CrossRef] [PubMed]
- Inkster, A.M.; Fernández-Boyano, I.; Robinson, W.P. Sex Differences Are Here to Stay: Relevance to Prenatal Care. J. Clin. Med. 2021, 10, 3000. [Google Scholar] [CrossRef] [PubMed]
- Albert, R.; Lee, A.; Lingappan, K. Response to Therapeutic Interventions in the NICU: Role of Sex as a Biological Variable. NeoReviews 2023, 24, e797–e805. [Google Scholar] [CrossRef] [PubMed]
| Number of Patients (%) | Male (%) | Female (%) | p* | |
| Total | 51 (100%) | 30 | 21 | |
| Neonate | 32 (62.7%) | 17 (56.7%) | 15 (71.4%) | 0.28 |
| Need for ECMO | 15 (29.4%) | 8 (26.7%) | 7 (33.3%) | 0.61 |
| Mortality | 20 (39.2%) | 14 (46.7%) | 6 (28.6%) | 0.19 |
| Total (range) | Male (IQR) | Female (IQR) | p** | |
| Median Birth Weight (Kg) | 3.080 (0.375-4.420) | 3.13 (2.320-3.380) | 3.08 (2.550-3.300) | 0.69 |
| Median Gest. Age (Weeks) | 38.57 (23-41) | 37.57 (35.71-39.00) | 38.71 (37.14-39.14) | 0.28 |
| Median Weight at Iloprost Admin. (kg) | 3.37 (2.06-7.3) | 3.43 (2.99-4.38) | 3.30 (2.97-3.71) | 0.70 |
| VIS | 13.00 (0-19.00) | 13.00 (3.50-16.80) | 13.00 (6.00-20.00) | 0.62 |
| Iloprost # Days | 3.00 (0.25-37) | 3.00 (1.00-8.80) | 3.00 (1.00-7.00) | 0.76 |
| Neonate (%) | Infant (%) | p* | |
| Total | 32 (62.7%) | 19 (37.3%) | 0.01 |
| Male | 17 (53.1%) | 13 (68.4%) | 0.28 |
| ECMO | 12 (37.5%) | 3 (15.8%) | 0.12** |
| Deceased | 13 (40.6%) | 7 (36.8%) | 0.79 |
| median (IQR) | Median (IQR) | p*** | |
| Birth Wt (kg) (range) | 3.195 (0.540-4.29) | 3.05 (0.375-4.42) | 0.52 |
| Gest. Age (Weeks) | 39.00 (37.14-39.14) | 37.00 (30.14-38.79) | 0.02 |
| Weight at iloprost administration (kg) | 3.21 (2.75-3.38) | 4.53 (3.77-5.56) | <0.01 |
| VIS | 14.50 (6.75-23.13) | 5.00 (0.00-14.50) | 0.03 |
| Iloprost # Days | 1.00 (1.00-5.50) | 3.00 (1.00-10.00) | 0.07 |
| Group I (%) | Group III (%) | p* | |
| Total | 24 (47%) | 27 (53%) | |
| Male | 13 (54%) | 17 (63%) | 0.52 |
| ECMO | 7 (29.1%) | 8 (29.6%) | 0.97 |
| Deceased | 11 (45.8%) | 9 (33.3%) | 0.36 |
| Median (IQR) | Median (IQR) | p** | |
| Birth Wt (kg) | 3.21 (2.96-3.4) | 3.07 (2.17-3.26) | 0.057 |
| Gest. Age (Weeks) | 38.5 (37-39) | 38.2 (34.1-39.1) | 0.14 |
| Iloprost dosing wt (kg) | 3.21 (2.96-3.4) | 3.38 (2.78-3.76) | 0.211 |
| VIS | 10(5.75-20.63) | 13.00 (5.00-5.25)* | 0.77 |
| Iloprost # Days | 2.00 (1.00-5.00) | 3.00 (1.00-15.5) | 0.11 |
| ECMO (%) | No ECMO (%) | p * | |
| Total | 15 (29.4%) | 36 (70.6%) | <0.01 |
| Male | 8 (53.3%) | 22 (61.1%) | 0.61 |
| Neonate | 12 (80.0%) | 20 (55.6%) | 0.12 ** |
| Deceased | 7 (46.7%) | 13 (36.1%) | 0.48 |
| ECMO (IQR) | No ECMO (IQR) | p *** | |
| Birth Wt (kg) | 3.08 (2.49-3.28) | 3.13 (2.50-3.39) | 0.88 |
| Gest. Age (Weeks) | 38.57 (37.14-39.00) | 38.21 (35.67-39.00) | 0.88 |
| Dosing Wt (kg) | 3.22 (2.72-3.71) | 3.51 (3.20-4.13) | 0.10 |
| VIS | 15.00 (4.50-22.50) | 10.50 (3.75-15.63) | 0.22 |
| Iloprost # Days | 2.00 (1.00-14.50) | 3.00 (1.00-7.25) | 0.55 |
| Survived (%) | Deceased (%) | p* | |
| Total | 31 (60.8%) | 20 (39.2%) | 0.03 |
| Male | 16 (51.6%) | 14 (70.0%) | 0.19 |
| Neonate [<4 wk age] | 19 (61.3%) | 13 (65.0%) | 0.79 |
| ECMO | 8 (25.8%) | 7 (35.0%) | 0.48 |
| Survived (IQR) | Deceased (IQR) | p** | |
| Birth Wt (kg) | 3.20 (2.50-3.58) | 3.06 (2.41-3.28) | 0.32 |
| Gest. Age (Weeks) | 39.00 (37.00-39.14) | 37.29 (35.71-38.89) | 0.23 |
| Dose Wt (kg) | 3.70 (3.21-4.41) | 3.22 (2.75-3.61) | 0.06 |
| VIS | 11.00 (3.00-16.50) | 14.00 (6.00-21.25) | 0.40 |
| Iloprost # Days | 3.00 (1.00-10.00) | 1.50 (0.94-6.75) | 0.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




