Submitted:
28 April 2024
Posted:
29 April 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Materials
Preparation of Chiral Nanomaterials
Immunofluorescence
Fluorescence Imaging and Graph Processing
Treatments
Time-Lapse Study and FRAP
Photo-Proximity Labeling and Co-IP Experiments
Proteomics Study
Statistical Analysis
Results and Discussion
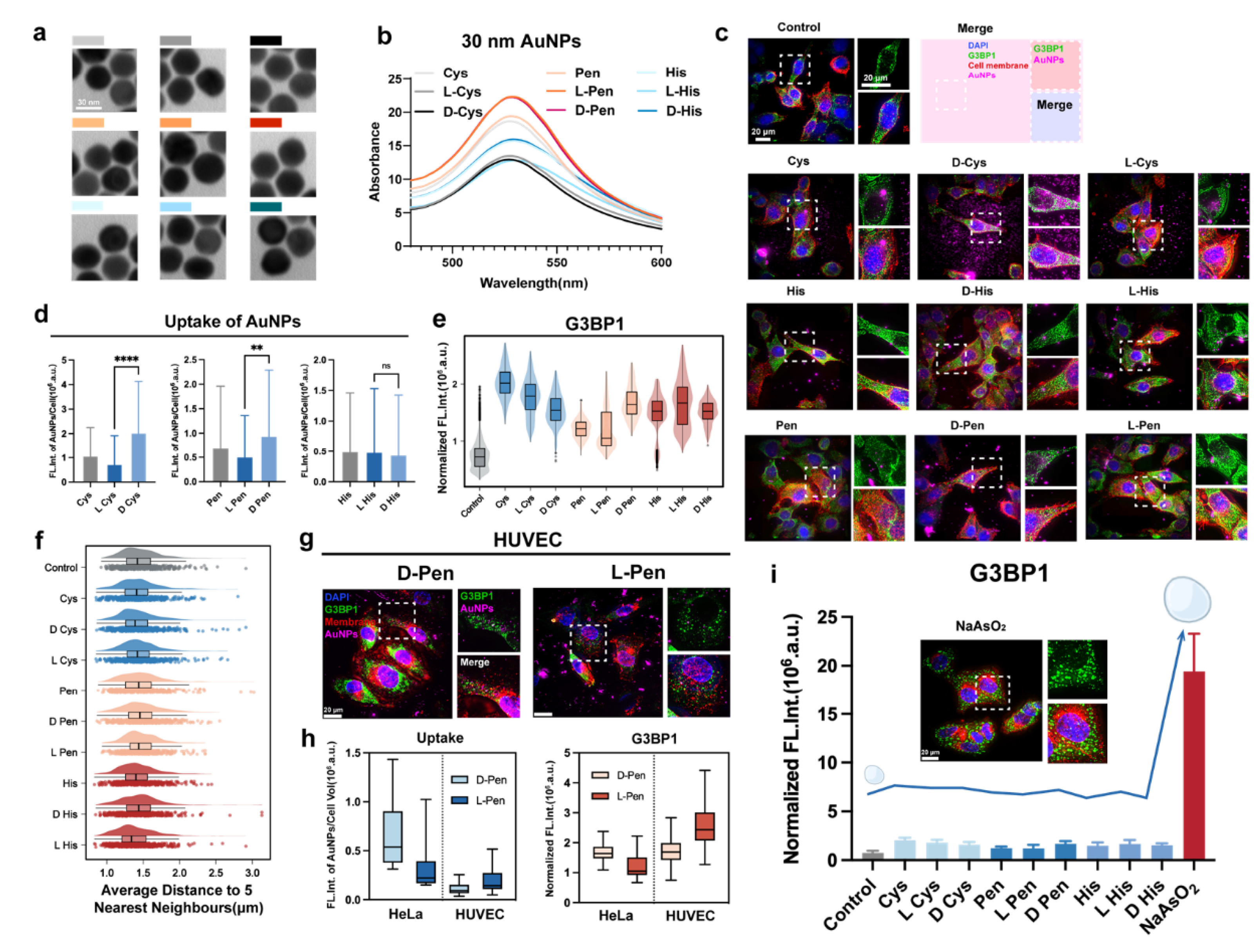
Stress Conditions Induced by Chiral Nanomaterials
Modulation of Stress Granule with Chiral Nanomedicines
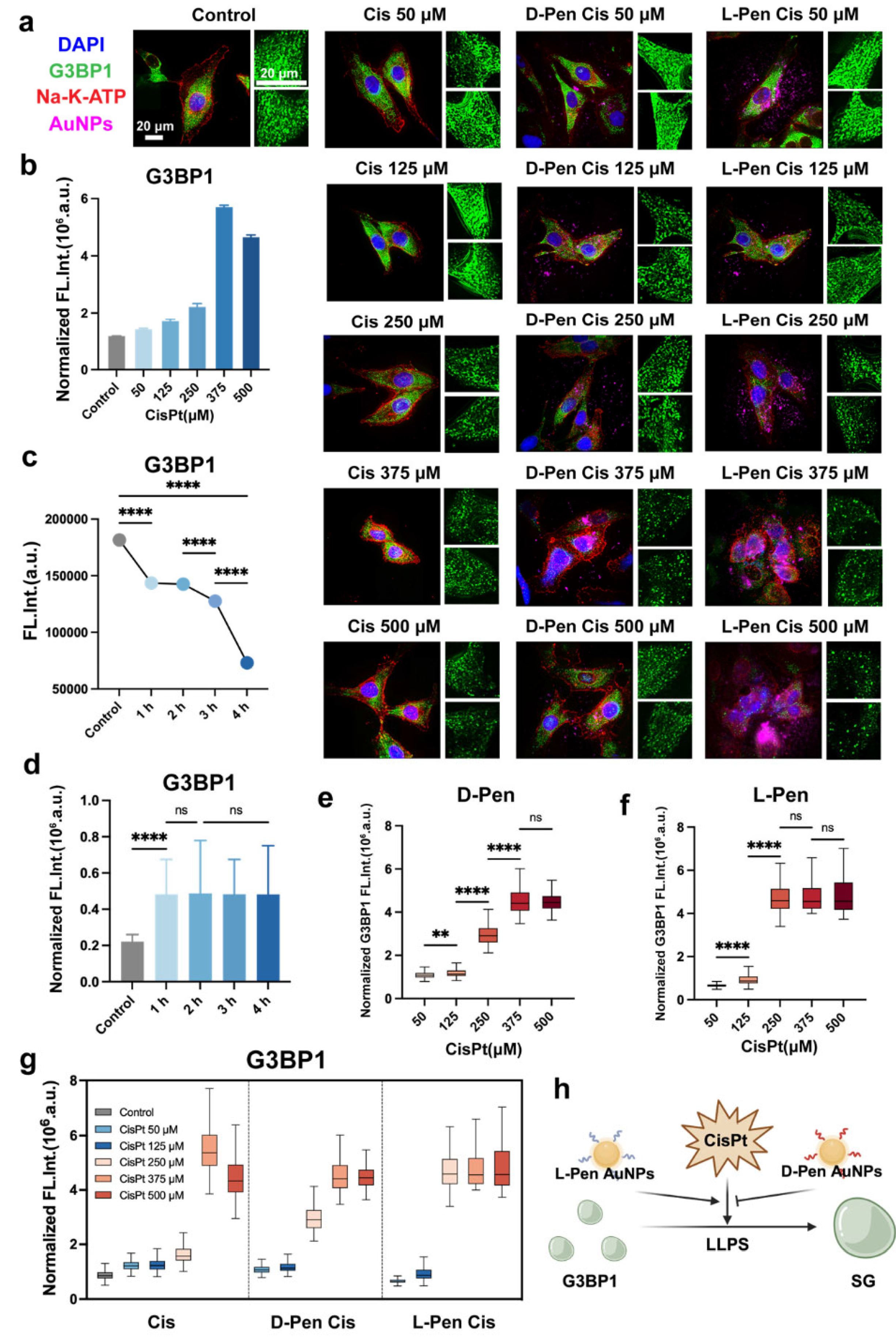
Synergy Action on Stress Granule Formation
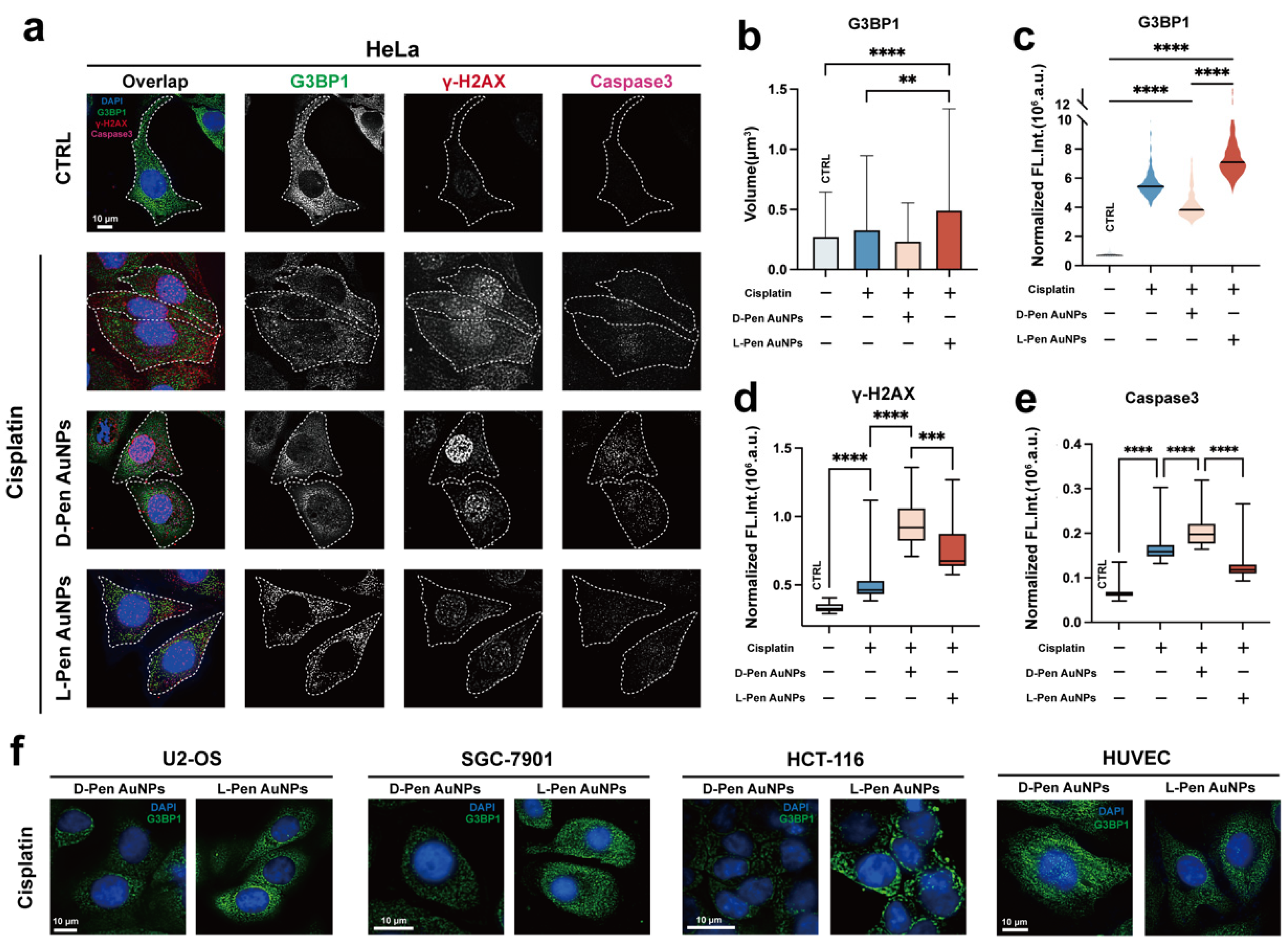
Characterization of Multiple Treatments-Induced Stress Granules
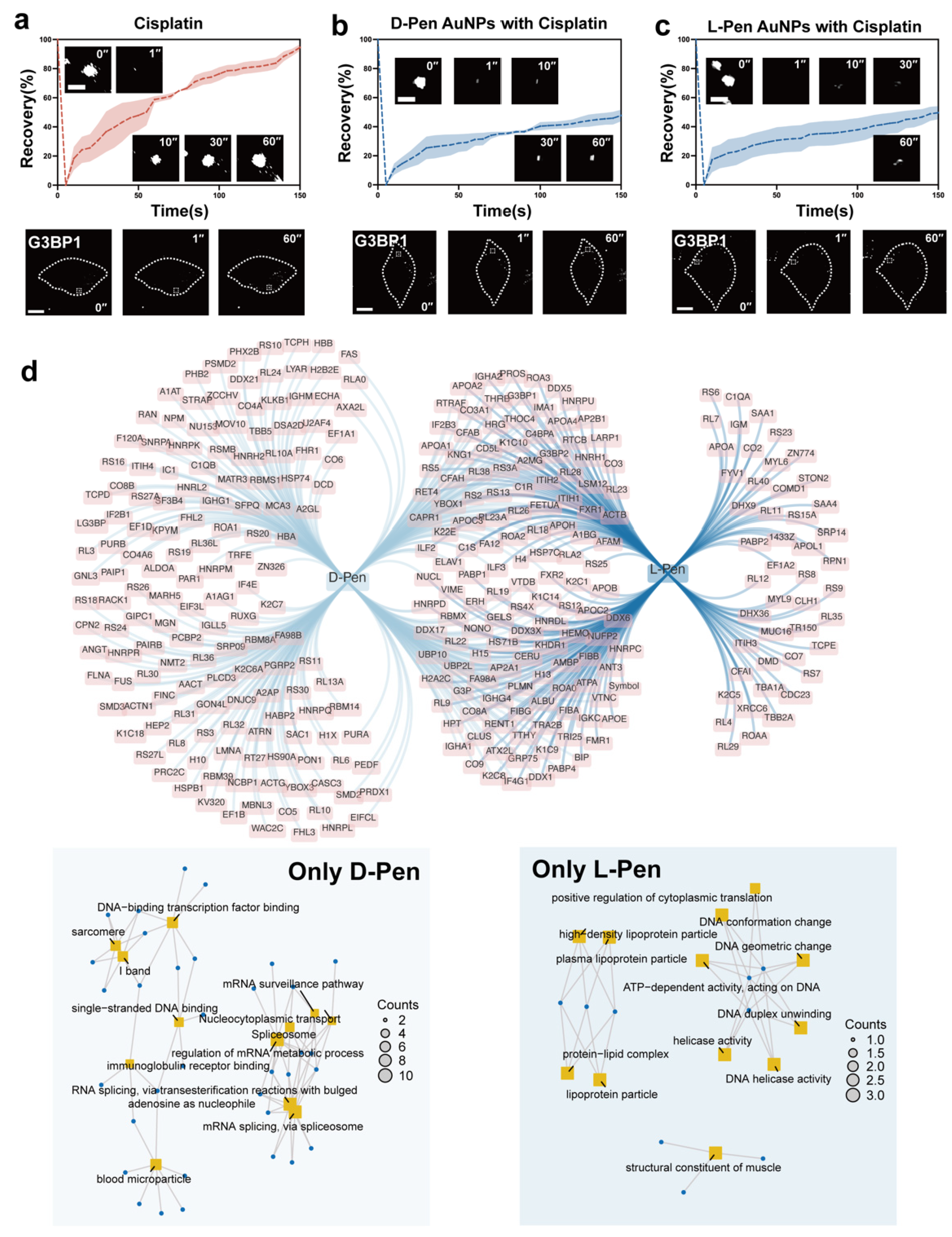
Mechanism Study of Chiral Nanomaterials-Modulated Stress Granules Formation
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Milton FP, Govan J, Mukhina MV, Gun’ko YK. The chiral nano-world: chiroptically active quantum nanostructures. Nanoscale Horiz. 2016;18;1(1):14-26. [CrossRef]
- Bird GH, Mazzola E, Opoku-Nsiah K, et al. Biophysical determinants for cellular uptake of hydrocarbon-stapled peptide helices. Nat Chem Biol. 2016;12(10):845-52. [CrossRef]
- Speranza L, di Porzio U, Viggiano D, de Donato A, Volpicelli F. Dopamine: The neuromodulator of long-term synaptic plasticity, reward and movement control. Cells. 2021;26;10(4):735. [CrossRef]
- Yanagisawa N, Ikeda S, Hashimoto T, et al. Effects of L-threo-Dops on orthostatic hypotension in Parkinson’s disease. No To Shinkei. 1998;50(2):157-63. Japanese.
- Sun M, Wang X, Guo X, et al. Chirality at nanoscale for bioscience. Chem Sci. 2022;8;13(11):3069-3081. [CrossRef]
- Ma Y, Shi L, Yue H, Gao X. Recognition at chiral interfaces: from molecules to cells. Colloids Surf B Biointerfaces. 2020;195:111268. [CrossRef]
- John M, Kalvala PR, Misra M, Menezes PL. Peening techniques for surface modification: processes, properties, and applications. Materials (Basel). 2021;9;14(14):3841. [CrossRef]
- hao Y, Yang G, Lin J, et al. Shining light on chiral inorganic nanomaterials for biological issues. Theranostics. 2021;7;11(19):9262-9295. [CrossRef]
- Zhang H, Li S, Qu A, et al. Engineering of chiral nanomaterials for biomimetic catalysis. Chem Sci. 2020;21;11(48):12937-12954. [CrossRef]
- Tee YH, Goh WJ, Yong X, et al. Actin polymerisation and crosslinking drive left-right asymmetry in single cell and cell collectives. Nat Commun. 2023;11;14(1):776. [CrossRef]
- Purcell-Milton F, Visheratina AK, Kuznetsova VA, et al. Impact of shell thickness on photoluminescence and optical activity in chiral CdSe/CdS core/shell quantum dots. ACS Nano. 2017;26;11(9):9207-9214. [CrossRef]
- Li Y, Zhou Y, Wang HY, et al. Chirality of glutathione surface coating affects the cytotoxicity of quantum dots. Angew Chem Int Ed Engl. 2011;20;50(26):5860-4. [CrossRef]
- Zhang C, Zhou Z, Zhi X, et al. Insights into the distinguishing stress-induced cytotoxicity of chiral gold nanoclusters and the relationship with GSTP1. Theranostics. 2015;1;5(2):134-49. [CrossRef]
- Zhang H, Hao C, Qu A, et al. Light-induced chiral iron copper selenide nanoparticles prevent β-amyloidopathy in vivo. Angew Chem Int Ed Engl. 2020;27;59(18):7131-7138. [CrossRef]
- Anderson P, Kedersha N. Stressful initiations. J Cell Sci. 2002;15;115(Pt 16):3227-34. [CrossRef]
- Anderson P, Kedersha N, Ivanov P. Stress granules, P-bodies and cancer. Biochim Biophys Acta. 2015;1849(7):861-70. [CrossRef]
- Kang W, Wang Y, Yang W, Zhang J, Zheng H, Li D. Research progress on the structure and function of G3BP. Front Immunol. 2021;30;12:718548. [CrossRef]
- Yang P, Mathieu C, Kolaitis RM, et al. G3BP1 is a tunable switch that triggers phase separation to assemble stress granules. Cell. 2020;16;181(2):325-345.e28. [CrossRef]
- Ge Y, Jin J, Li J, Ye M, Jin X. The roles of G3BP1 in human diseases (review). Gene. 2022;5;821:146294. [CrossRef]
- Fujikawa D, Nakamura T, Yoshioka D, et al. Stress granule formation inhibits stress-induced apoptosis by selectively sequestering executioner caspases. Curr Biol. 2023;22;33(10):1967-1981.e8. [CrossRef]
- Reits EA, Neefjes JJ. From fixed to FRAP: measuring protein mobility and activity in living cells. Nat Cell Biol. 2001;3(6):E145-7. [CrossRef]
- Bersuker K, Peterson CWH, To M, et al. A proximity labeling strategy provides insights into the composition and dynamics of lipid droplet proteomes. Dev Cell. 2018;8;44(1):97-112.e7. [CrossRef]
- Zheng Y, Zhong X, Li Z, et al. Successive, seed-mediated growth for the synthesis of single-crystal gold nanospheres with uniform diameters controlled in the range of 5–150 nm. Particle & Particle Systems Characterization. 2014;31(2):266-273. [CrossRef]
- Zhu M, Nie G, Meng H, Xia T, Nel A, Zhao Y. Physicochemical properties determine nanomaterial cellular uptake, transport, and fate. Acc Chem Res. 2013;19;46(3):622-31. [CrossRef]
- Zhao B, Yang S, Deng J, Pan K. Chiral graphene hybrid materials: structures, properties, and chiral applications. Adv Sci (Weinh). 2021;12;8(7):2003681. [CrossRef]
- Nguyen VH, Lee BJ. Protein corona: a new approach for nanomedicine design. Int J Nanomedicine. 2017;18;12:3137-3151. [CrossRef]
- Mahmoudi M, Landry MP, Moore A, Coreas R. The protein corona from nanomedicine to environmental science. Nat Rev Mater. 2023;24:1-17. [CrossRef]
- Yeom J, Guimaraes PPG, Ahn HM, et al. Chiral supraparticles for controllable nanomedicine. Adv Mater. 2020;32(1):e1903878. [CrossRef]
- Lee JI, Namkoong S. Stress granules dynamics: benefits in cancer. BMB Rep. 2022;55(12):577-586. [CrossRef]
- Song MS, Grabocka E. Stress granules in cancer. Rev Physiol Biochem Pharmacol. 2023;185:25-52. [CrossRef]
- Guillén-Boixet J, Kopach A, Holehouse AS, et al. RNA-induced conformational switching and clustering of G3BP drive stress granule assembly by condensation. Cell. 2020;16;181(2):346-361.e17. [CrossRef]
- Begovich K, Wilhelm JE. An in vitro assembly system identifies roles for RNA nucleation and ATP in yeast stress granule formation. Mol Cell. 2020;17;79(6):991-1007.e4. [CrossRef]
- Gwon Y, Maxwell BA, Kolaitis RM, Zhang P, Kim HJ, Taylor JP. Ubiquitination of G3BP1 mediates stress granule disassembly in a context-specific manner. Science. 2021;25;372(6549):eabf6548. [CrossRef]
- Klein IA, Boija A, Afeyan LK, et al. Partitioning of cancer therapeutics in nuclear condensates. Science. 2020;19;368(6497):1386-1392. [CrossRef]
- Wang X, Chen T, Li C, et al. CircRNA-CREIT inhibits stress granule assembly and overcomes doxorubicin resistance in TNBC by destabilizing PKR. J Hematol Oncol. 2022;29;15(1):122. [CrossRef]
- Dasari S, Tchounwou PB. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 2014;5;740:364-78. [CrossRef]
- Zhang J, Ye ZW, Tew KD, Townsend DM. Cisplatin chemotherapy and renal function. Adv Cancer Res. 2021;152:305-327. [CrossRef]
- Kedersha N, Panas MD, Achorn CA, et al. G3BP-Caprin1-USP10 complexes mediate stress granule condensation and associate with 40S subunits. J Cell Biol. 2016;28;212(7):845-60. [CrossRef]
- Peng PH, Hsu KW, Wu KJ. Liquid-liquid phase separation (LLPS) in cellular physiology and tumor biology. Am J Cancer Res. 2021;15;11(8):3766-3776.
- Tong X, Tang R, Xu J, et al. Liquid-liquid phase separation in tumor biology. Signal Transduct Target Ther. 2022;8;7(1):221. [CrossRef]
- Díaz-Moreno I, De la Rosa MA. Membraneless organelles: a smart design for metabolic control. FEBS Open Bio. 2021;11(9):2388-2389. [CrossRef]
- Koppers M, Özkan N, Farías GG. Complex interactions between membrane-bound organelles, biomolecular condensates and the cytoskeleton. Front Cell Dev Biol. 2020;21;8:618733. [CrossRef]
- Li J, Zhang M, Ma W, et al. Post-translational modifications in liquid-liquid phase separation: a comprehensive review. Mol Biomed. 2022;11;3(1):13. [CrossRef]
- Wang B, Zhang L, Dai T, et al. Liquid-liquid phase separation in human health and diseases. Signal Transduct Target Ther. 2021;2;6(1):290. [CrossRef]
- Feng Q, Wilhelm J, Gao J. Transistor-like ultra-pH-sensitive polymeric nanoparticles. Acc Chem Res. 2019;18;52(6):1485-1495. [CrossRef]
- Hofmann S, Kedersha N, Anderson P, Ivanov P. Molecular mechanisms of stress granule assembly and disassembly. Biochim Biophys Acta Mol Cell Res. 2021;1868(1):118876. [CrossRef]
- Ivanov P, Kedersha N, Anderson P. Stress granules and processing bodies in translational control. Cold Spring Harb Perspect Biol. 2019;1;11(5):a032813. [CrossRef]
- Riggs CL, Kedersha N, Ivanov P, Anderson P. Mammalian stress granules and P bodies at a glance. J Cell Sci. 20201;133(16):jcs242487. [CrossRef]
- Kopac T. Protein corona, understanding the nanoparticle-protein interactions and future perspectives: A critical review. Int J Biol Macromol. 2021;1;169:290-301. [CrossRef]
- Park SJ. Protein-nanoparticle interaction: corona formation and conformational changes in proteins on nanoparticles. Int J Nanomedicine. 2020;6;15:5783-5802. [CrossRef]
- Porter NJ, Danelius E, Gonen T, Arnold FH. Biocatalytic carbene transfer using diazirines. J Am Chem Soc. 2022;25;144(20):8892-8896. [CrossRef]
- Protter DSW, Parker R. Principles and properties of stress granules. Trends Cell Biol. 2016;26(9):668-679. [CrossRef]
- Jain S, Wheeler JR, Walters RW, et al. ATPase-modulated stress granules contain a diverse proteome and substructure. Cell. 201628;164(3):487-98. [CrossRef]
- Pare JM, Tahbaz N, López-Orozco J, et al. Hsp90 regulates the function of argonaute 2 and its recruitment to stress granules and P-bodies. Mol Biol Cell. 2009;20(14):3273-84. [CrossRef]
- Tolay N, Buchberger A. Role of the ubiquitin system in stress granule metabolism. Int J Mol Sci. 2022;26;23(7):3624. [CrossRef]
- Zheng Y, Zhu G, Tang Y, et al. HDAC6, A novel cargo for autophagic clearance of stress granules, mediates the repression of the type I interferon response during coxsackievirus A16 infection. Front Microbiol. 2020;31;11:78. [CrossRef]
- Peng G, Gu A, Niu H, et al. Amyotrophic lateral sclerosis (ALS) linked mutation in Ubiquilin 2 affects stress granule assembly via TIA-1. CNS Neurosci Ther. 2022;28(1):105-115. [CrossRef]
- Kedersha NL, Gupta M, Li W, Miller I, Anderson P. RNA-binding proteins TIA-1 and TIAR link the phosphorylation of eIF-2 alpha to the assembly of mammalian stress granules. J Cell Biol. 1999;27;147(7):1431-42. [CrossRef]
- Song D, Kuang L, Yang L, et al. Yin and yang regulation of stress granules by Caprin-1. Proc Natl Acad Sci U S A. 2022;119(44):e2207975119. [CrossRef]
- Fefilova AS, Fonin AV, Vishnyakov IE, Kuznetsova IM, Turoverov KK. Stress-induced membraneless organelles in eukaryotes and prokaryotes: bird’s-eye View. Int J Mol Sci. 2022;30;23(9):5010. [CrossRef]
- Millar SR, Huang JQ, Schreiber KJ, et al. A new phase of networking: the molecular composition and regulatory dynamics of mammalian stress granules. Chem Rev. 2023;26;123(14):9036-9064. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



