Submitted:
11 April 2024
Posted:
15 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Structural Characterization of Peptide-Based Drugs via XRPD
1.2. The Pharmaceutical Peptide Octreotide
1.3. In Situ XRPD Measurements upon Controlled Relative Humidity and Temperature Variation
2. Materials and Methods
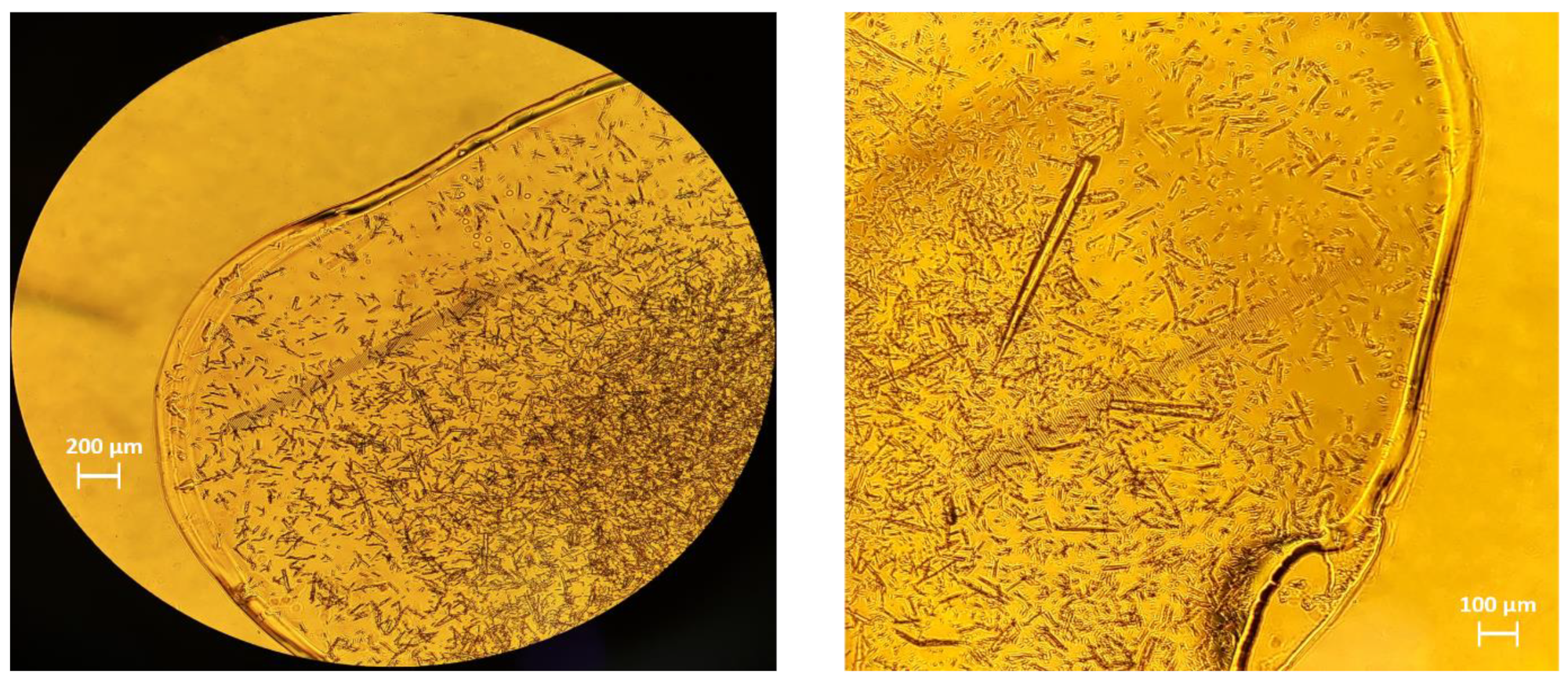
2.1. Crystallization
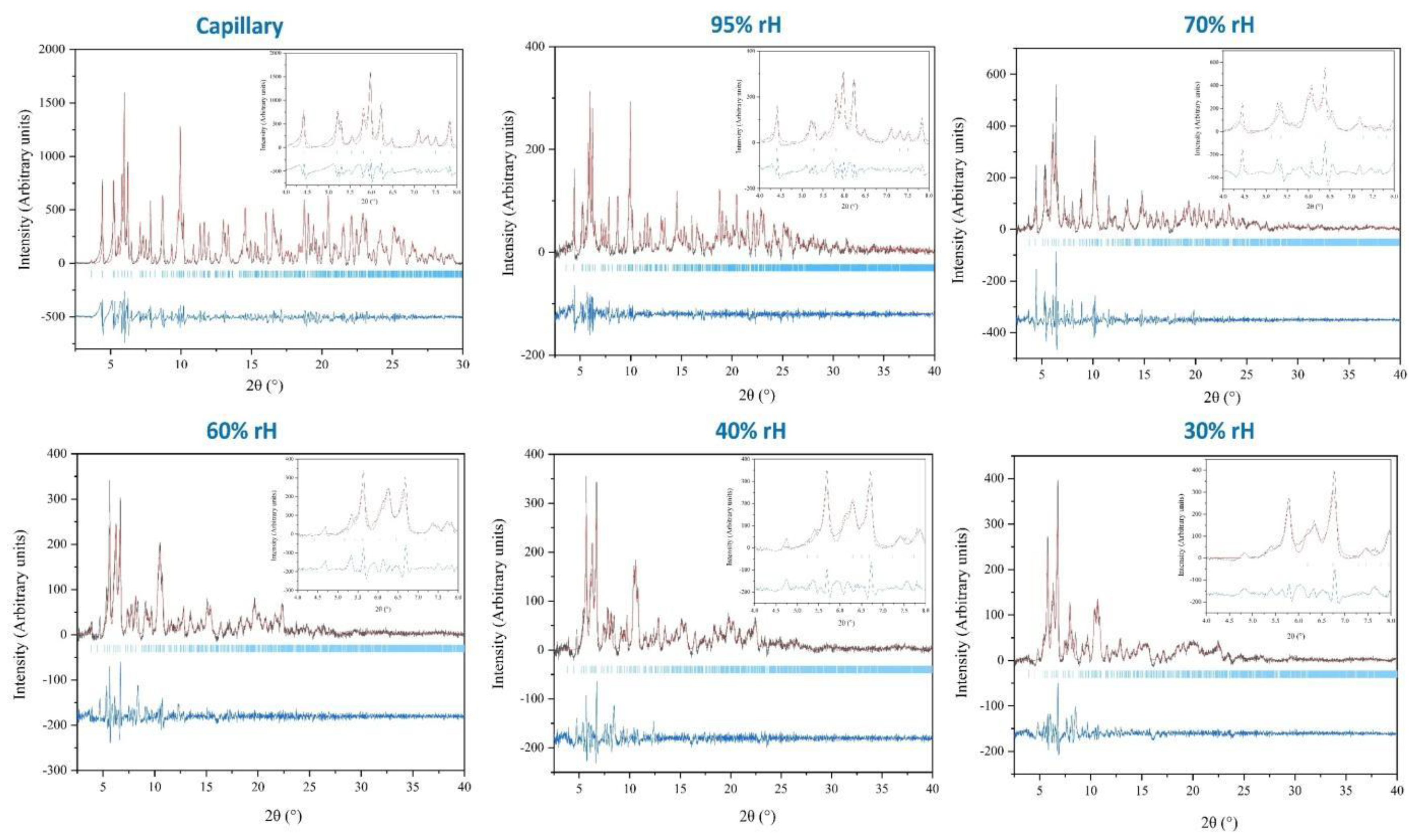
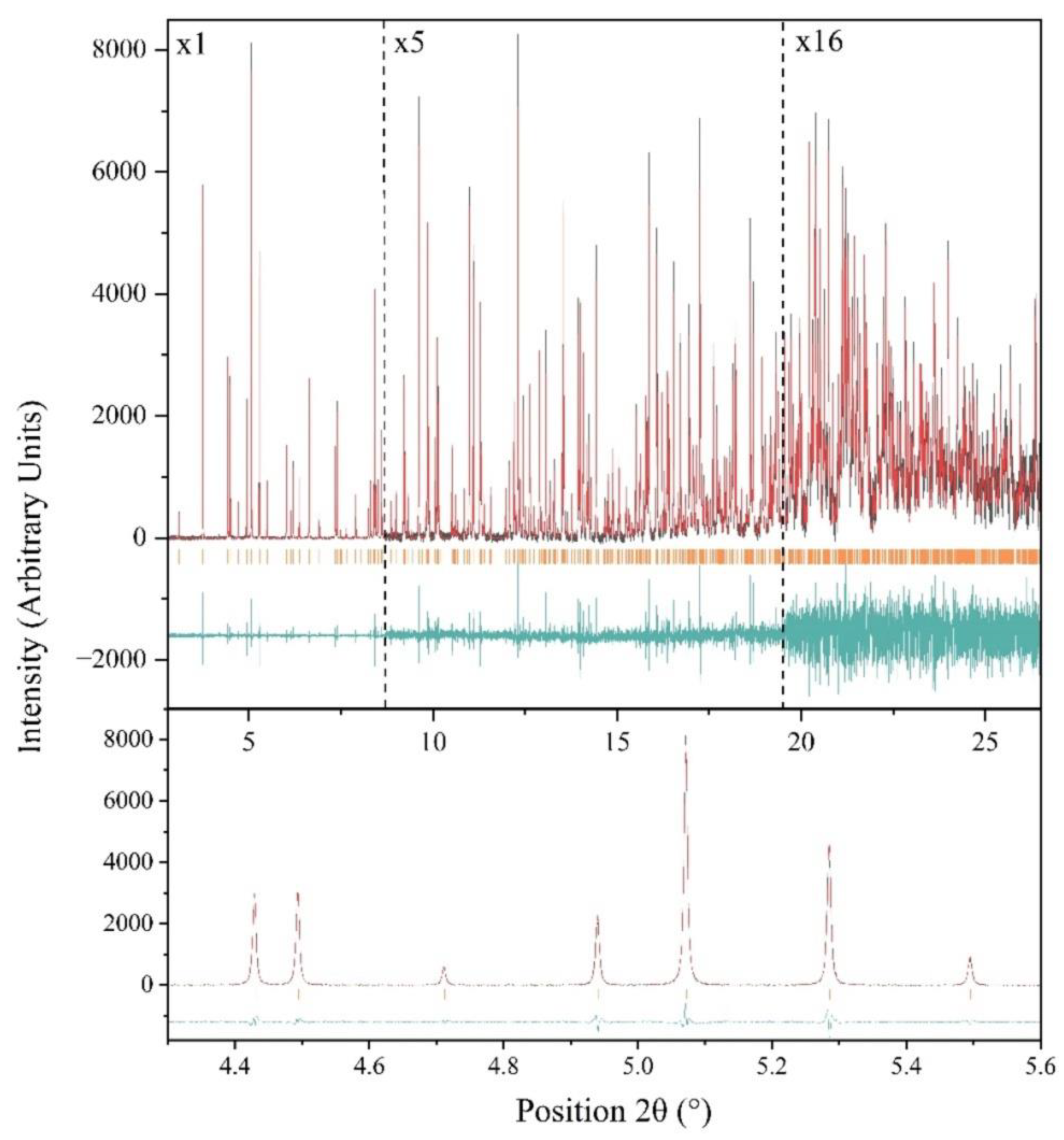
2.2. Laboratory and Synchrotron XRPD Data Collection
3. Results
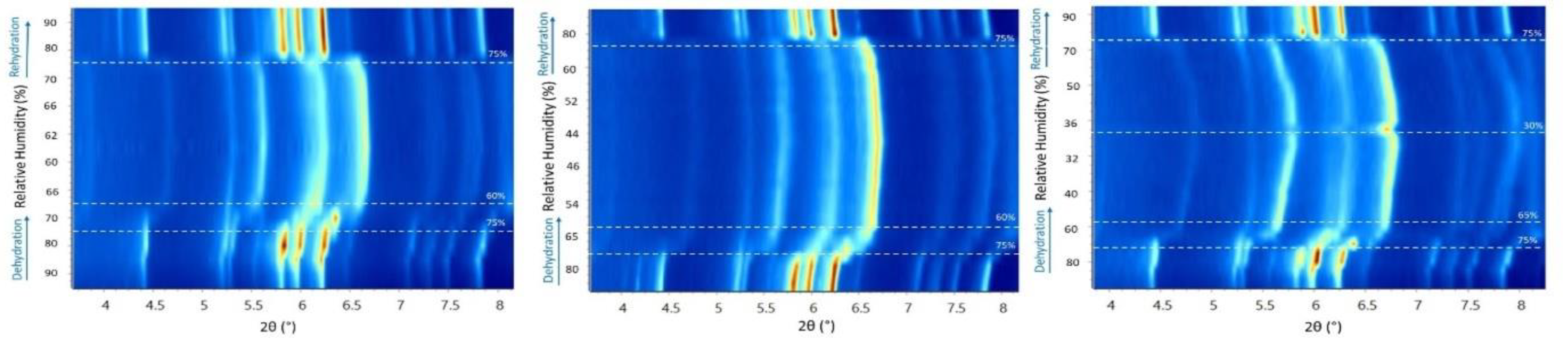
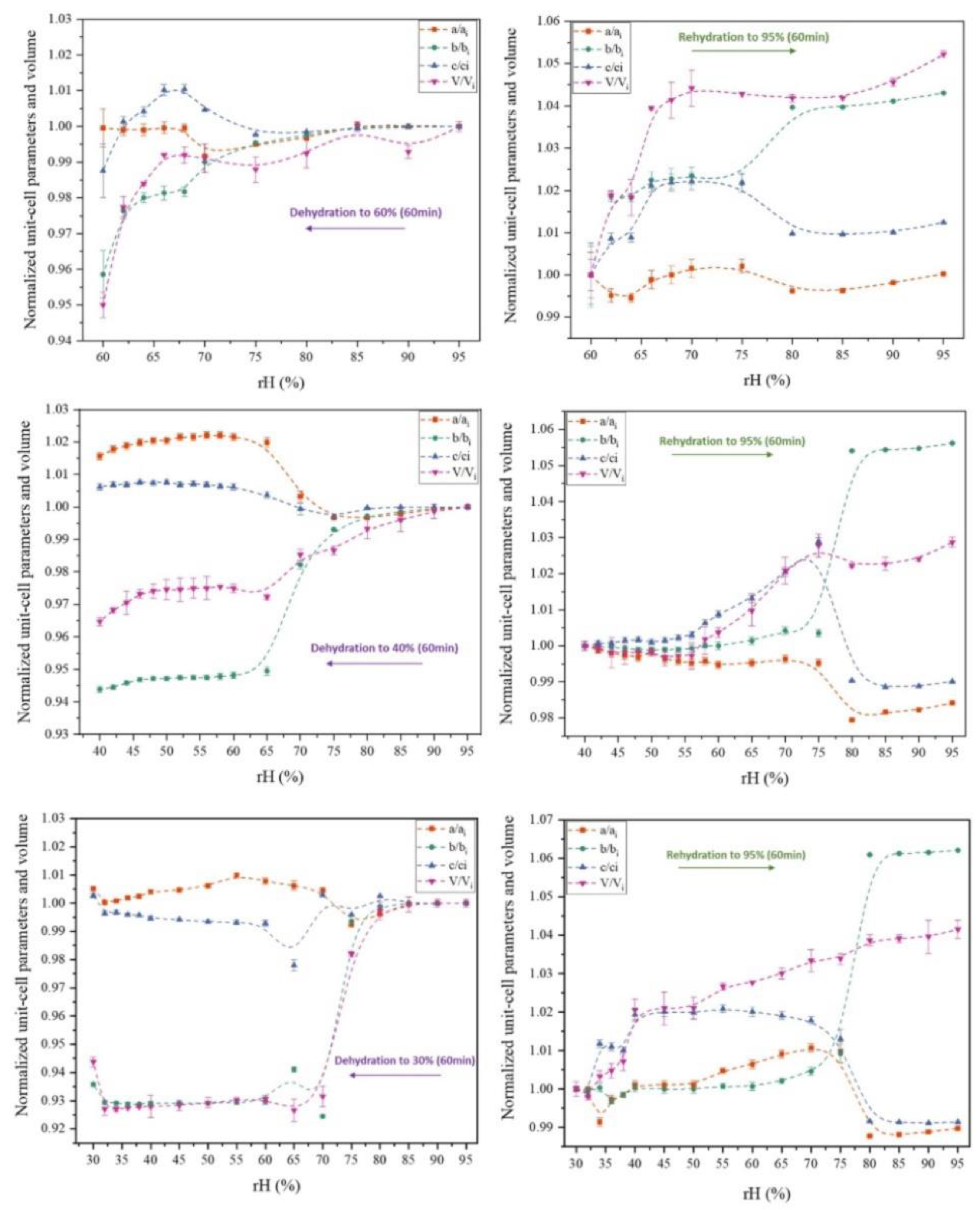
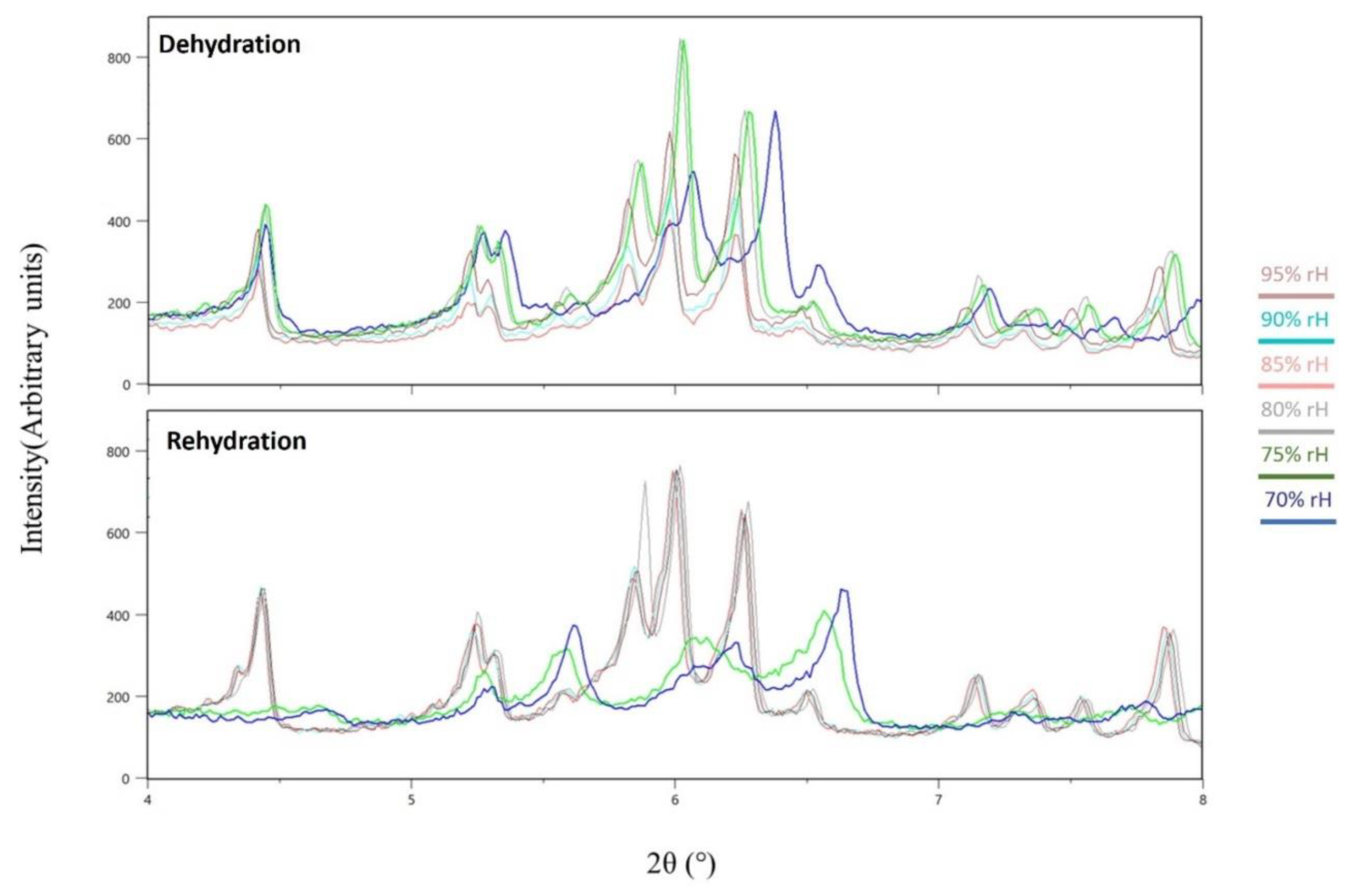
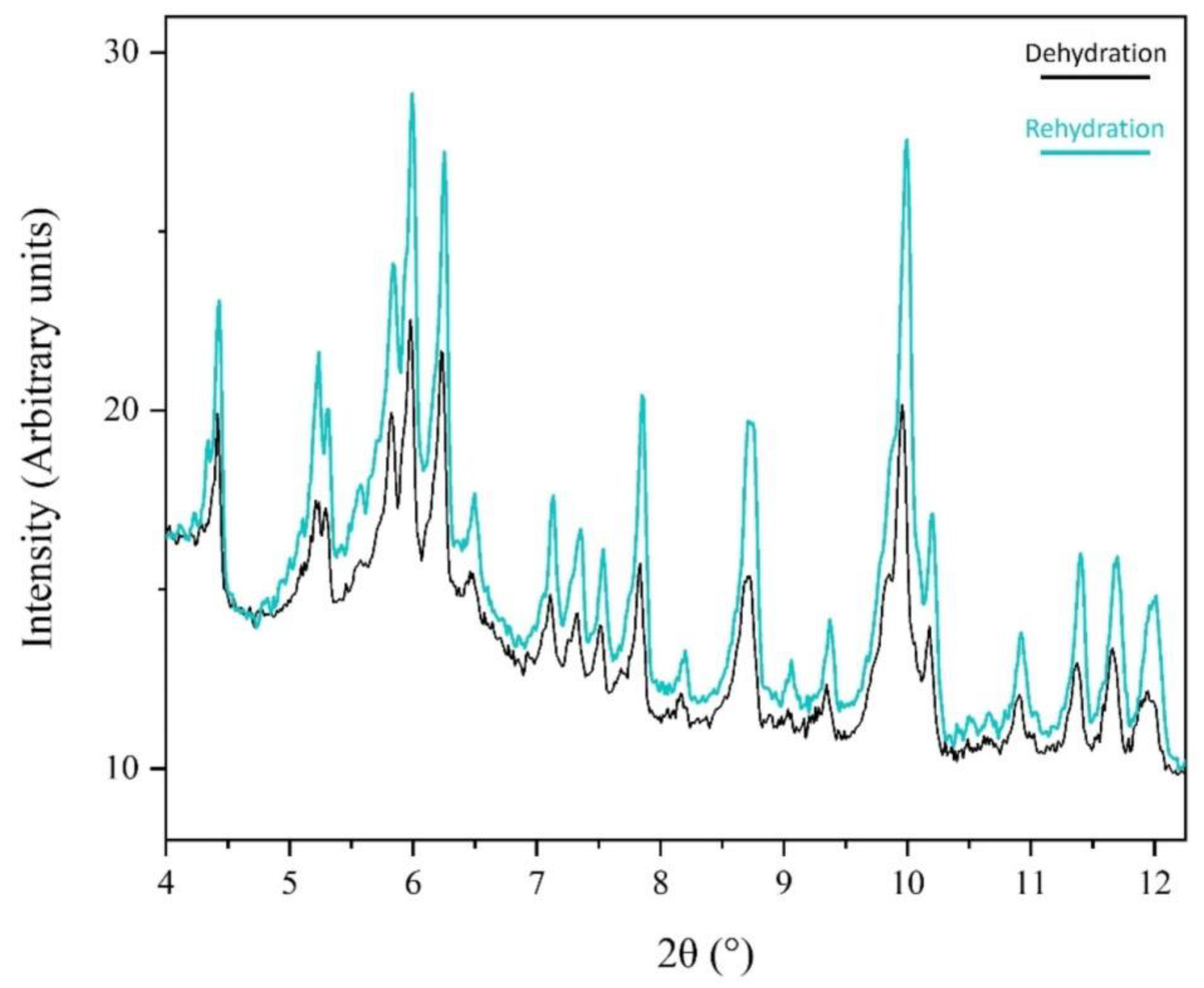
3.1. The Effect of rH Variation on Octreotide at Ambient Temperature
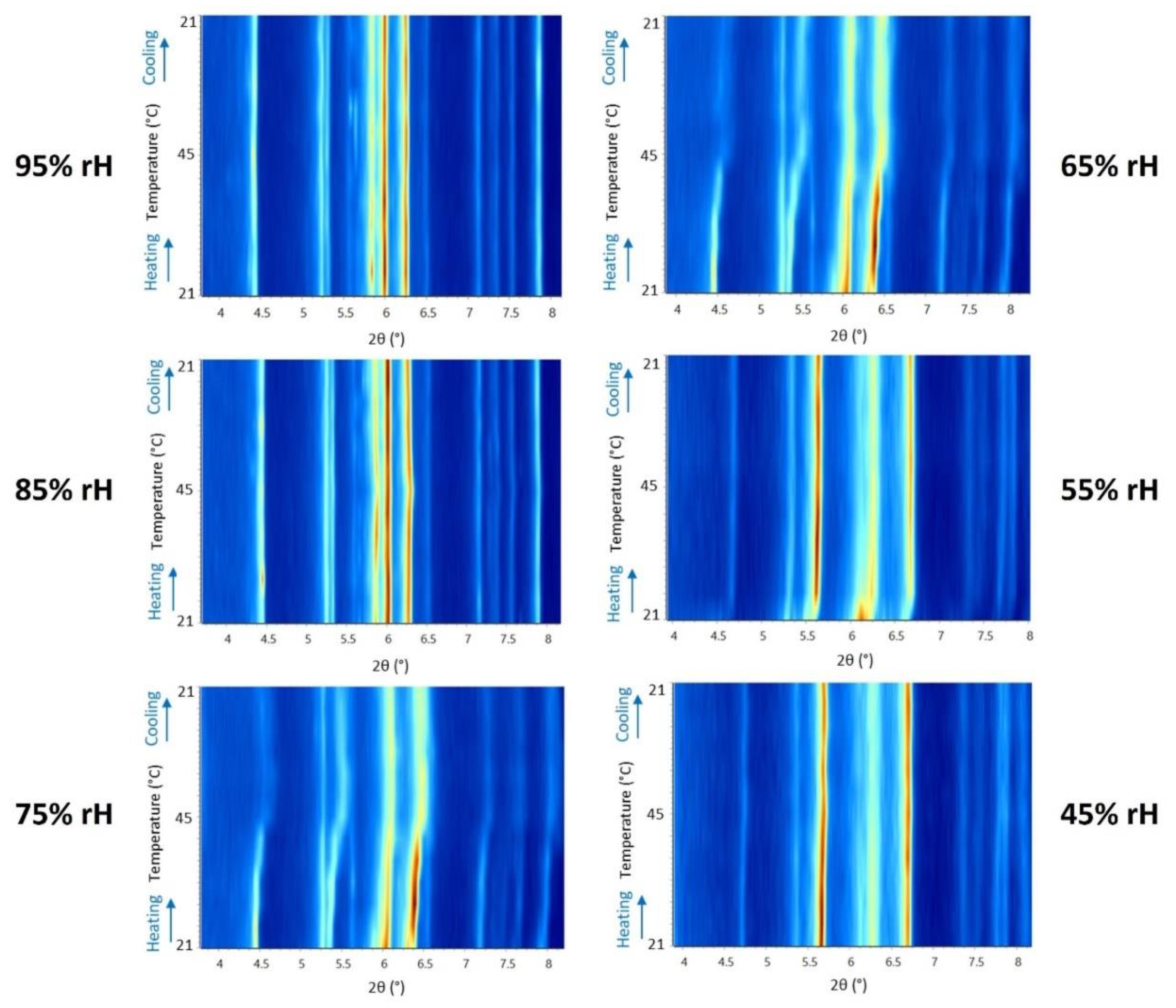
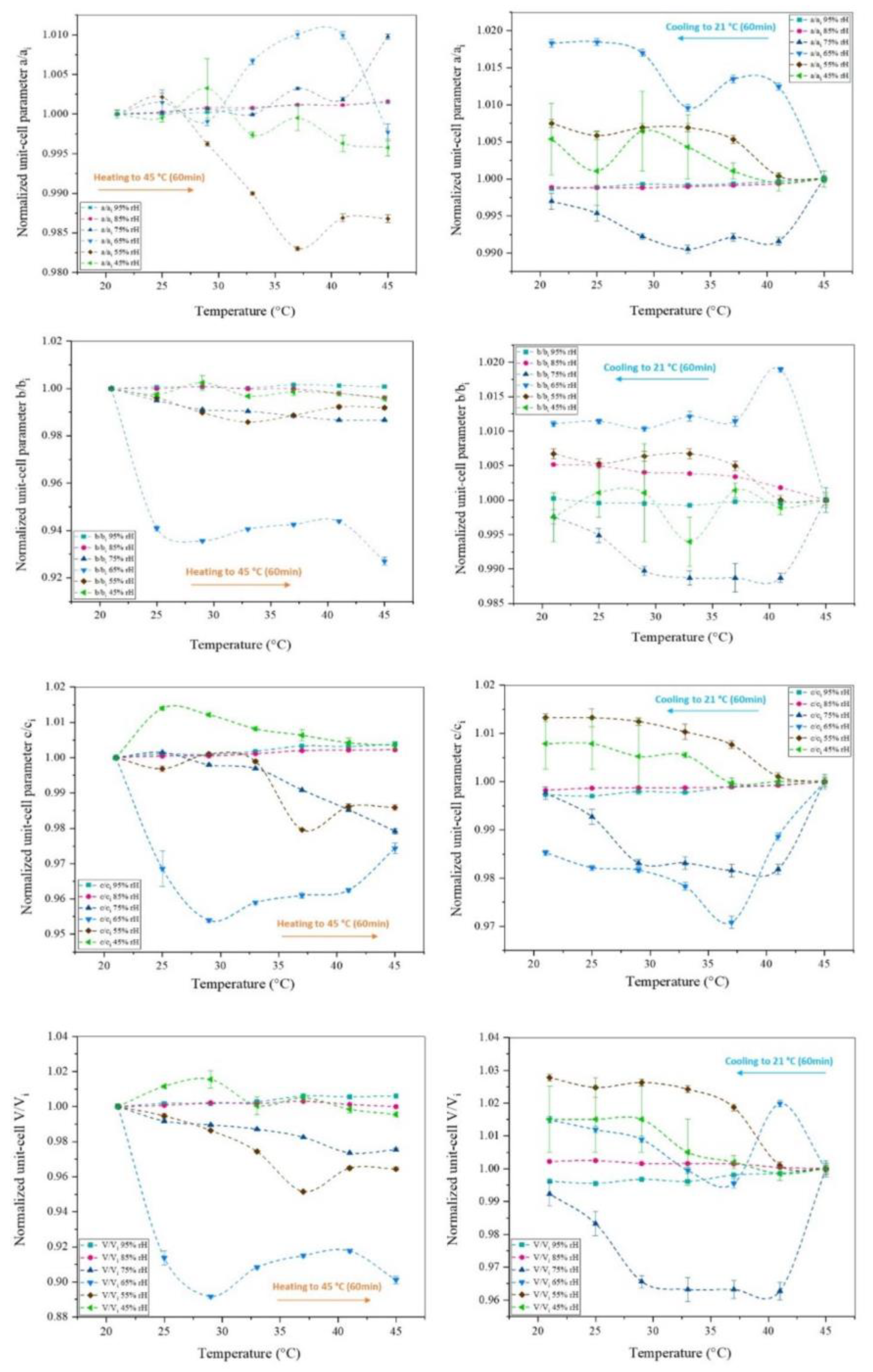
3.2. The Effect of Temperature on Octreotide at Selected rH Levels
4. Discussion
5. Conclusions
Funding
Acknowledgments
References
- Abraham, J. (2010). Journal of Pharmaceutical Sciences, 99, 3, 1500-1515.
- Asherie, N. (2004). Methods 34, 266–272.
- Atakisi, H., Moreau, D. W. & Thorne, R. E. (2018). Acta Crystallogr D Struct Biol 74, 264–278.
- Bashir, S., Fitaihi, R. & Abdelhakim, H. E. (2023). European Journal of Pharmaceutical Sciences 182, 106374.
- Basso, S., Fitch, A. N., Fox, G. C., Margiolaki, I. & Wright, J. P. (2005). Acta Crystallogr D Biol Crystallogr 61, 1612–1625.
- Budayova-Spano, M., Dauvergne, F., Audiffren, M., Bactivelane, T. & Cusack, S. (2007). Acta Crystallogr D Biol Crystallogr 63, 339–347.
- Degen, T., Sadki, M., Bron, E., König, U. & Nénert, G. (2014). Powder Diffr. 29, S13–S18.
- Ducruix, A. & Giegé, R. (1999). Crystallization of Nucleic Acids and Proteins: A Practical Approach Oxford University Press.
- Fenimore, P. W., Frauenfelder, H., McMahon, B. H. & Young, R. D. (2004). Proc. Natl. Acad. Sci. U.S.A. 101, 14408–14413.
- Fili, S., Valmas, A., Spiliopoulou, M., Kontou, P., Fitch, A., Beckers, D., Degen, T., Barlos, K., Barlos, K. K., Karavassili, F. & Margiolaki, I. (2019). Acta Crystallogr B Struct Sci Cryst Eng Mater 75, 611–620.
- Fitch, A., Dejoie, C., Covacci, E., Confalonieri, G., Grendal, O., Claustre, L., Guillou, P., Kieffer, J., De Nolf, W., Petitdemange, S., Ruat, M. & Watier, Y. (2023). J Synchrotron Rad 30, 1003–1012.
- Fosgerau, K. & Hoffmann, T. (2015). Drug Discovery Today 20, 122–128.
- Giegé, R. (2013). The FEBS Journal 280, 6456–6497.
- Govada, L. & Chayen, N. (2019). Crystals 9, 106.
- Hageman, M. J. (1988). Drug Development and Industrial Pharmacy 14, 2047–2070.
- Hudaverdyan, T. G., Kachalova, G. S. & Bartunik, H. D. (2006). Crystallogr. Rep. 51, 519–524.
- Karavassili, F., Valmas, A., Fili, S., Georgiou, C. & Margiolaki, I. (2017). Biomolecules 7, 63.
- Khadka, P., Ro, J., Kim, H., Kim, I., Kim, J. T., Kim, H., Cho, J. M., Yun, G. & Lee, J. (2014). Asian Journal of Pharmaceutical Sciences 9, 304–316.
- Kumar, V., Bansal, V., Madhavan, A., Kumar, M., Sindhu, R., Awasthi, M. K., Binod, P. & Saran, S. (2022). Bioengineered 13, 4309–4327.
- Kuntz, I. D. (1992). Science 257, 1078–1082.
- Lau, J. L. & Dunn, M. K. (2018). Bioorganic & Medicinal Chemistry 26, 2700–2707.
- Lipinski, C. A., Lombardo, F., Dominy, B. W. & Feeney, P. J. (1997). Advanced Drug Delivery Reviews 23, 3–25.
- Logotheti, S., Valmas, A., Trampari, S., Fili, S., Saslis, S., Spiliopoulou, M., Beckers, D., Degen, T., Nénert, G., Fitch, A. N., Karavassili, F. & Margiolaki, I. (2019). J Appl Crystallogr 52, 816–827.
- Manning, M. C., Chou, D. K., Murphy, B. M., Payne, R. W. & Katayama, D. S. (2010). Pharm Res 27, 544–575.
- Margiolaki, I. (2019). International Tables of Crystallography- Volume H: Powder Diffraction, chapter 7.1, 718-736, 2019.
- Margiolaki, I. & Wright, J. P. (2008). Acta Crystallogr A Found Crystallogr 64, 169–180.
- Mirza, S., Miroshnyk, I., Heinämäki, J., Antikainen, O., Rantanen, J., Vuorela, P., Vuorela, H. & Yliruusi, J. (2009). AAPS PharmSciTech 10, 113–119.
- Muheem, A., Shakeel, F., Jahangir, M. A., Anwar, M., Mallick, N., Jain, G. K., Warsi, M. H. & Ahmad, F. J. (2016). Saudi Pharmaceutical Journal 24, 413–428.
- Norrman, M., Ståhl, K., Schluckebier, G. & Al-Karadaghi, S. (2006). J Appl Crystallogr 39, 391–400.
- Nugrahadi, P. P., Hinrichs, W. L. J., Frijlink, H. W., Schöneich, C. & Avanti, C. (2023). Pharmaceutics 15, 935.
- Pawley, G. S. (1981). J. Appl. Crystallogr. 14, 357–361.
- Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C. & Ferrin, T. E. (2004). J. Comput. Chem. 25, 1605–1612.
- Pohl, E., Heine, A., Sheldrick, G. M., Dauter, Z., Schneider, T., Wilson, K. S. & Kallen, J. (1995). Acta Crystallogr. D Biol. Crystallogr. 51, 60–68.
- Prueksaritanont, T. & Tang, C. (2012). AAPS J. 14, 410–419.
- Rosenberger, F., Howard, S. B., Sowers, J. W. & Nyce, T. A. (1993). Journal of Crystal Growth 129, 1–12.
- Rupley, J. A. & Careri, G. (1991). Advances in Protein Chemistry, 41, 37–172.
- Spiliopoulou, M., Karavassili, F., Triandafillidis, D.-P., Valmas, A., Fili, S., Kosinas, C., Barlos, K., Barlos, K. K., Morin, M., Reinle-Schmitt, M. L., Gozzo, F. & Margiolaki, I. (2021). Acta Crystallogr. A Found. Adv. 77, 186–195.
- Spiliopoulou, M., Valmas, A., Triandafillidis, D.-P., Kosinas, C., Fitch, A., Karavassili, F. & Margiolaki, I. (2020). Crystals 10, 54.
- Trampari, S., Valmas, A., Logotheti, S., Saslis, S., Fili, S., Spiliopoulou, M., Beckers, D., Degen, T., Nénert, G., Fitch, A. N., Calamiotou, M., Karavassili, F. & Margiolaki, I. (2018). J Appl Crystallogr 51, 1671–1683.
- Triandafillidis, D. P., Karavassili, F., Spiliopoulou, M., Valmas, A., Athanasiadou, M., Nikolaras, G., Fili, S., Kontou, P., Bowler, M. W., Chasapis, C. T., Von Dreele, R. B., Fitch, A. N. & Margiolaki, I. (2023). Acta Crystallogr. D Biol. Crystallogr. 79, 374–386.
- Wang, L., Wang, N., Zhang, W., Cheng, X., Yan, Z., Shao, G., Wang, X., Wang, R. & Fu, C. (2022). Sig. Transduct. Target Ther. 7, 48.
- Zellnitz, S., Narygina, O., Resch, C., Schroettner, H. & Urbanetz, N. A. (2015). International Journal of Pharmaceutics 489, 170–176.

| Cycle | Initial rH levels (%) | Final rH levels (%) | Step (% rH) | Temperature (K) | Waiting time | Scans /level |
|---|---|---|---|---|---|---|
| 1 | 95 | 70 | 5 | 294.15 | 60 minutes | 10 |
| 70 | 60 | 2 | 294.15 | 60 minutes | 10 | |
| 2 | 95 | 70 | 5 | 294.15 | 60 minutes | 10 |
| 70 | 40 | 2 | 294.15 | 60 minutes | 10 | |
| 3 | 95 | 70 | 5 | 294.15 | 60 minutes | 10 |
| 70 | 30 | 2 | 294.15 | 60 minutes | 10 |
| Cycle | Initial temperature (K) | Final temperature (K) | Step (K) | rH level (%) | Waiting time | Scans /level |
|---|---|---|---|---|---|---|
| 1 | 294.15 | 318.15 | 4 | 95 | 60 minutes | 10 |
| 2 | 294.15 | 318.15 | 4 | 85 | 60 minutes | 10 |
| 3 | 294.15 | 318.15 | 4 | 75 | 60 minutes | 10 |
| 4 | 294.15 | 318.15 | 4 | 65 | 60 minutes | 10 |
| 5 | 294.15 | 318.15 | 4 | 55 | 60 minutes | 10 |
| 6 | 294.15 | 318.15 | 4 | 45 | 60 minutes | 10 |
| 95 - 60% rH | 95 - 40% rH | 95 - 30% rH | |
|---|---|---|---|
| Δa/ai (%) | 0.04 | -1.56 | -0.51 |
| Δb/bi (%) | 4.14 | 5.61 | 6.43 |
| Δc/ci (%) | 1.24 | -0.61 | -0.27 |
| ΔV/Vi (%) | 5.00 | 3.53 | 5.63 |
| 294.15 - 318.15 K | 95% rH | 85% rH | 75% rH | 65% rH | 55% rH | 45% rH |
|---|---|---|---|---|---|---|
| Δa/ai (%) | -0.15 | -0.16 | -0.98 | 0.23 | 1.32 | 0.42 |
| Δb/bi (%) | -0.08 | 0.38 | 1.33 | 7.30 | 0.81 | 0.42 |
| Δc/ci (%) | -0.38 | -0.22 | 2.08 | 2.57 | 1.41 | -0.34 |
| ΔV/Vi (%) | -0.61 | 0.00 | 2.46 | 9.89 | 3.56 | 0.45 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




